Abstract
Anaplasma marginale has recently been shown to infect endothelial cells in vitro, but it remains unknown as to whether endothelial infection also occurs in vivo. In this report, we demonstrate through dual fluorescence microscopy that A marginale, detected by the monoclonal antibody ANAF16C1, co-localizes with the endothelial cell marker, von Willebrand factor, in tissue sections from an experimentally inoculated calf. The results indicate that A marginale infection includes endothelial cells and has implications for both pathogenesis and immune mechanisms.
Anaplasmosis is an arthropod-borne hemoparasitic disease of cattle and other ruminants caused by the gram-negative bacterium, Anaplasma marginale, belonging to the order Rickettsiales and the family Anaplasmataceae. 1 The disease is characterized by severe anemia associated with intraerythrocytic parasitism, resulting in fever, depression, and weakness. 5 Cattle that recover clinically remain persistently infected and serve as a reservoir for transmission to uninfected cattle. 7 Anaplasmosis severely reduces the production of meat, milk, and fiber in tropical and subtropical areas of the world. 8 The family Anaplasmataceae includes the genera Anaplasma, Ehrlichia, Neorickettsia, and Wolbachia, encompassing a group of obligate intracellular bacteria that reside within a cytoplasmic vacuole in eukaryotic cells, including erythrocytes, macrophages, endothelial cells, and cells of insect, helminth, and arthropod reproductive tissues, in addition to other tissues. 1
A marginale enters red blood cells by endocytosis after which it divides by binary fission. 2, 11 Presently, A marginale is documented to infect in vivo only mature, circulating erythrocytes of its mammalian hosts, domestic and wild ruminants. 6 In contrast, the organism invades and replicates in the nucleated cells of the midgut and salivary gland epithelium of the ixodid tick vectors, including Rhipicephalus (Boophilus) microplus, Rhipicephalus annulatus, Dermacentor andersoni, and Dermacentor variabilis.
Recently, Munderloh et al. (2004) 10 have used tick cell cultures of the Virginia isolate of A marginale, Am291, to inoculate, in vitro, the cell line BCE C/D1-b (derived from bovine vascular endothelial cells), primate Vero cells, and RF/6A cells (derived from rhesus monkey microvascular endothelium). The formation of intracellular A marginale colonies was associated with the death of endothelial cells within a week of inoculation and the release of bacteria into the culture supernatant. It is unknown whether or not A marginale can infect endothelial cells in vivo. If endothelial cells can be infected in vivo, these cells might represent a site of initial replication after tick-borne transmission or be a reservoir for the organism during persistent infection. In addition, endothelial cells, due to the expression of MHC class-I, could initiate a cell-mediated immune response, whereas intraerythrocytic infection could not because of the lack of MHC Class-I expression on erythrocytes.
A splenectomized calf (animal No. C1058) was inoculated intravenously with 10 9 organisms of the St. Maries strain of A marginale. 3 The calf was euthanized at 47 days postinoculation when organisms were detected in 44.5% of circulating red blood cells. 3 Samples of kidney (taken along the longitudinal axis of the renal cortex, approximately 1 × 1 × 0.1 cm) were harvested immediately after death and frozen in Neg-50 tissue-freezing compound (Richard Allan Scientific, Kalamazoo, Michigan) using isopentane cooled in liquid nitrogen. Samples were held at -70°C until they were sectioned at 5 μm and applied to Superfrost Plus slides (Fisher Scientific Inc., Hampton, NH). The slides were fixed for 5 minutes in acetone (4°C) before staining.
Tissue sections and RF/6A cells were first blocked with 5% normal rabbit serum (Biomeda, Foster City, CA) for 30 minutes at room temperature, rinsed in phosphate buffered saline (PBS), and then co-incubated with the A marginale monoclonal antibody, anti-ANAF16C1 12 (100 μg/ml) and polyclonal rabbit antihuman von Willebrand factor (vWF, Dako, Carpinteria, CA) at a 1°2 dilution of the concentration, supplied by the manufacturer, for 30 minutes at room temperature, then rinsed for 3 minutes in PBS. Next, the tissue sections were coincubated with 200 μl of Alexa Fluor 488-conjugated goat antimouse IgG and Alexa Fluor 568-conjugated goat antirabbit IgG (Molecular Probes, Carlsbad, CA), both at a concentration of 2 μg/ml for 30 minutes at room temperature. Sections were then rinsed for 3 minutes in 1X PBS. RF/6A cells infected with A marginale were used as a positive control for organism staining. 10 In addition, RF/6A cells and tissue sections were labeled with the lectin, FITC-conjugated Ulex europaeus Agglutinin I (UEAI) (Vector Laboratories, Burlingame, CA), in the same manner as the primary antibodies. UEAI is reported to label bovine endothelial cells. 4 All procedures after the application of fluorochromes were performed in a dark chamber. Tissue sections were then incubated with 200 μl of 4′, 6-diamidino-2-phenylindole, dihydrochloride (DAPI) (Molecular Probes, Carlsbad, CA) at a concentration of 284 nm, for 30 minutes at room temperature. Negative control sections were incubated with secondary antibodies alone and counterstained with DAPI. Tissue sections were coverslipped with Citifluor (Electron Microscopy Sciences, Hatfield, PA) and maintained in the dark at 4°C prior to examination.
Tissue sections and the RF/6A cells were analyzed with a Nikon Eclipse E800 microscope (Nikon Instruments, Inc., Kanagawa, Japan) fitted with 3 Nikon filter cubes for widefield fluorescence capture. The UV2A filter cube (330–380 nm excitation) was used for DAPI capture; the G2A filter cube (510–570 nm), for Alexa 568 capture; and the B3A filter cube (420–495 nm), for Alexa 488 capture. Images were captured using the Spot Advanced software (Diagnostic Instruments, Sterling Heights, MI).
Widefield fluorescence microscopy of kidney sections revealed colocalization of A marginale and vWF along vascular endothelium in a granular pattern of fluorescence (Fig. 1). Similar patterns of fluorescence were identified in the lung and hemal lymph nodes (data not shown). The granular pattern was attributable to the localization of vWF in Weibel-Palade bodies, which are reported to occur at a lower density in endothelial cells of capillaries when compared with endothelial cells of small arteries. 13 As a confirmation of endothelial identification, the kidney sections were also labeled with ANAF16C1 and the lectin, UEAI (Fig. 2). Negative controls, consisting of sections from the same tissue without the application of primary antibody or lectin, were devoid of specific staining. RF/6A cells expressed low levels of vWF, as reported, 9 and therefore UEAI (expressed highly in these cells) was used as an endothelial cell marker. These cells demonstrated co-labeling with the endothelial cell-lectin UEAI-FITC and mAb ANAF16C1 conjugated to Alexa 568 (counterstained with DAPI) (Fig. 3).
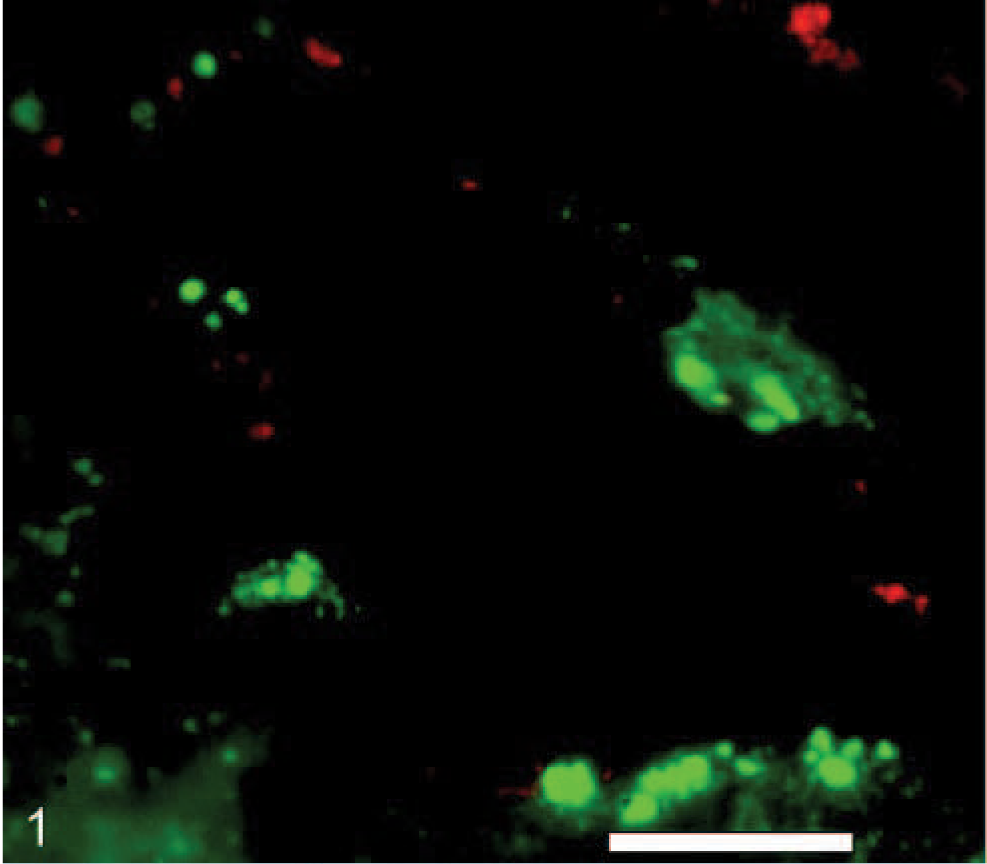
Kidney; calf No. C1058. Green fluorescence localizes A marginale (antibody ANAF16C1) along the endothelial surface of a vein when detected by indirect immunofluorescence method with Alexa 488 fluorochrome. Red fluorescence shows endothelial cells as identified by vWF expression in the same section using indirect immunofluorescence method with Alexa 568 fluorochrome. Bar = 20 μm.
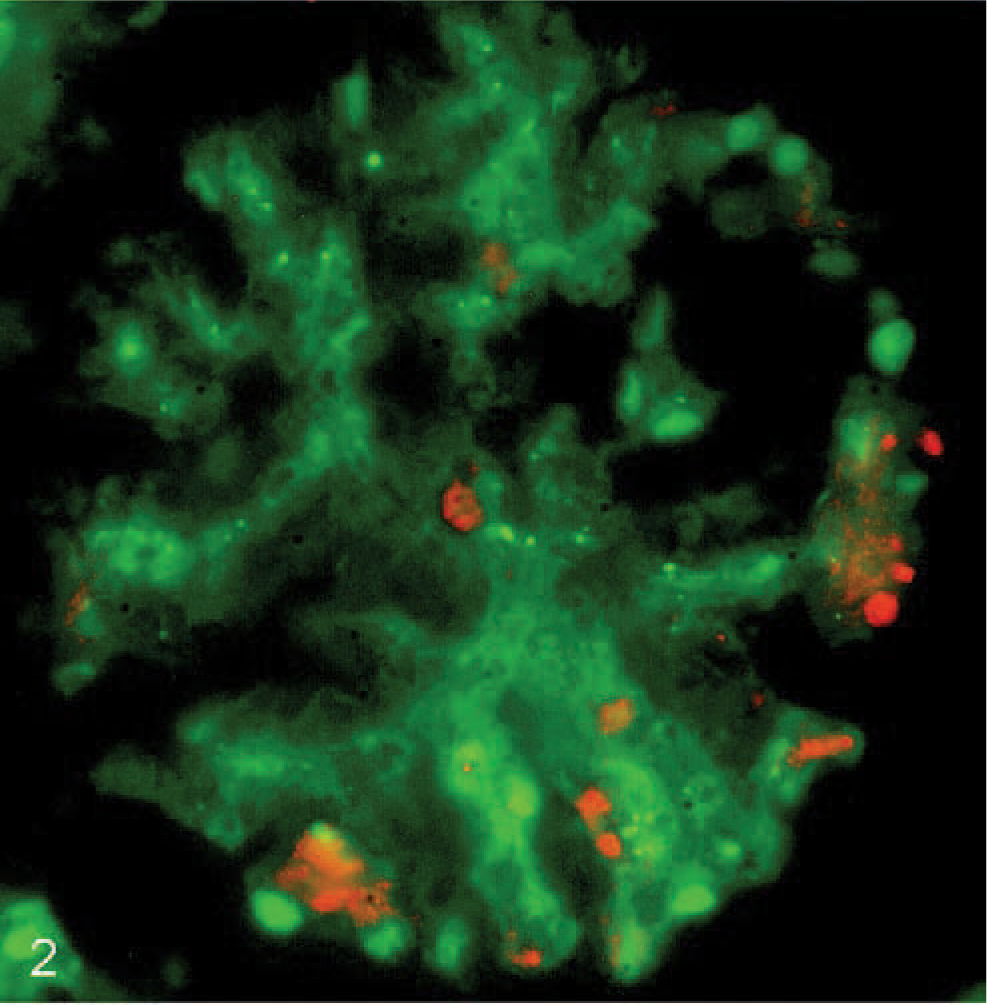
Kidney; calf No. C1058. Green fluorescence with FITC-conjugated Ulex europaeus Agglutinin I (UEAI) shows endothelial labeling within a glomerulus. Red fluorescence identifies A marginale (antibody ANAF16C1) in the same section using an indirect immunofluorescence method with Alexa 568 fluorochrome.
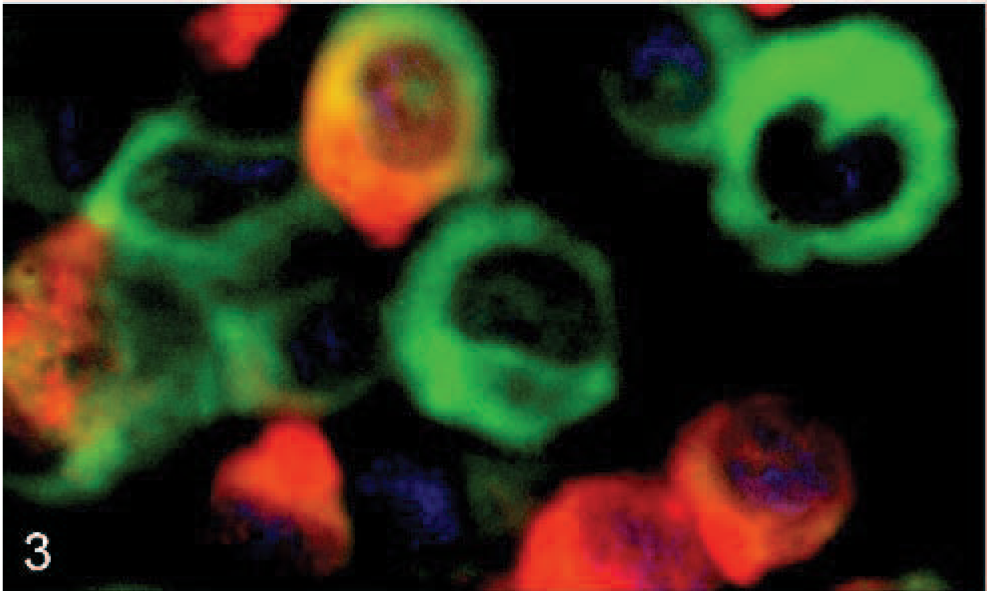
RF/6A cell culture. Green fluorescence with UEAI identifies the endothelial cell line RF/6A by direct lectin-FITC labeling and DAPI counterstaining. Red fluorescence shows A marginale labeling of RF/6A cells incubated with antibody ANAF16C1 and detected by indirect immunofluorescence method using Alexa 568 fluorochrome.
These data demonstrate endothelial cell infection in vivo after experimental infection of a calf with A marginale and extend the finding that endothelial cells are susceptible to infection after in vitro challenge. 10 Endothelial cells may serve as an early reservoir for A marginale infection at the site of tick attachment and, through their expression of MHC Class-I, could be important in the initiation of a cytotoxic T-lymphocyte response during the early stages of infection. Further studies are needed to define the role of endothelial cells in these key components of A marginale infection and pathogenesis.
Footnotes
Acknowledgement
This work was funded by the United States Department of Agriculture, Grant number 9922.
