Abstract
Clostridium difficile is a confirmed pathogen in a wide variety of mammals, but the incidence of disease varies greatly in relation to host species, age, environmental density of spores, administration of antibiotics, and possibly, other factors. Lesions vary as well, in severity and distribution within individuals, and in some instances, age groups, of a given species. The cecum and colon are principally affected in most species, but foals and rabbits develop severe jejunal lesions. Explanations for variable susceptibility of species, and age groups within a species, are largely speculative. Differences in colonization rates and toxin-receptor densities have been proposed. Clostridium difficile-associated disease is most commonly diagnosed in Syrian hamsters, horses, and neonatal pigs, but it is reported sporadically in many other species. The essential virulence factors of C. difficile are large exotoxins, toxin A (TcdA) and toxin B (TcdB). Receptor-mediated endocytosis of the toxins is followed by endosomal acidification, a necessary step for conversion of the toxin to its active form in the cytosol. Cell-surface receptors have been characterized for TcdA, but remain to be identified for TcdB. Both TcdA and TcdB disrupt the actin cytoskeleton by disrupting Rho-subtype, intracellular signaling molecules. Disruption of the actin cytoskeleton is catastrophic for cellular function, but inflammation and neurogenic stimuli are also involved in the pathogenesis of the disease.
Clostridium difficile is a gram-positive, anaerobic, spore-forming bacillus commonly associated with diarrhea and colitis in humans and other mammals. 140 It was first isolated from feces and meconium of asymptomatic newborn infants, and was originally named Bacillus difficilis because of its morphology and the difficulties encountered in cultivating it. 55 The initial report described production of a potent toxin, but the organism was not considered an important pathogen due to the high carrier rate among asymptomatic human infants. In the past 30 years, however, C. difficile has been implicated as the principal infectious cause of antibiotic-associated diarrhea in adult humans, and similar clinical conditions in a variety of other mammals. 65, 80, 140 It is now recognized as one of the most important nosocomial pathogens of humans. Clinically affected humans often experience diarrhea without appreciable lesions, but the disease can progress, resulting in the severe tissue changes associated with pseudomembranous colitis. 65 More serious sequelae, including prolonged ileus (toxic megacolon), intestinal perforation, and peritonitis develop, but are less common.
The relevance of C. difficile to disease in animals has also become more obvious. Lesions in nonhuman mammals are similar to those in humans, but vary widely in severity and distribution within the gastrointestinal tract (Fig. 1). This variation is evident for different species and different age groups within a species. 93, 114
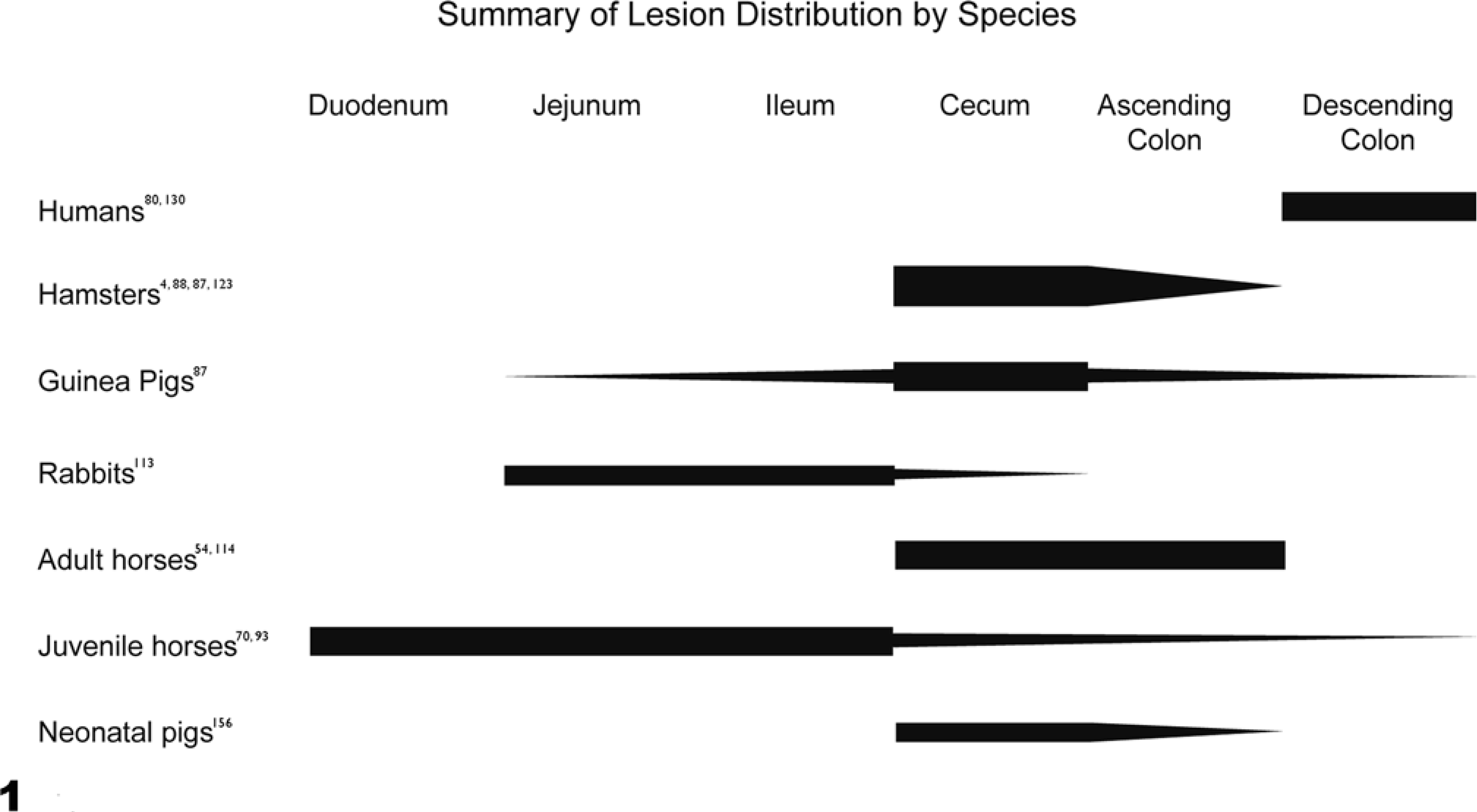
Summary of the species-dependent distribution of intestinal lesions induced by Clostridium difficile. The thickness of the bars represents the severity of lesions typically seen in an infected individual of a given species at a particular site. The figure lists only those species for which the distribution of spontaneous lesions of Clostridium difficile–associated disease (CDAD) is well documented.
Among the animals commonly used for laboratory research, Syrian hamsters (Mesocricetus auratus) are the most sensitive to naturally acquired disease. 84, 137 Lesions attributed to spontaneous or experimentally induced C. difficile infection have also been documented in guinea pigs (Cavia porcellus), 87 mice (Mus musculus), 105 rats (Rattus norvegicus and R. rattus), 31 and rabbits (Oryctolagus cuniculus); 24 all of these species are used as animal models of the human disease. Clostridium difficile–associated disease develops spontaneously in a variety of other species including horses (Equus caballus), 93 hares (Lepus spp.), 32 pigs (Sus scrofa), 156 nonhuman primates (Macaca spp., Pongo pygmaeus, and Saguinus oedipus), 128, 150 domestic dogs (Canis familiaris), 161 domestic cats (Felis catus), 162 ostriches (Struthio camellus) 42 and black-tailed prairie dogs (Cynomus ludovicianus). 104 This review addresses the pathogenesis of C. difficile–associated disease (CDAD) and compares the lesions and clinical conditions among known susceptible species.
Pathogenesis of CDAD
The essential virulence factors of C. difficile are toxin A (TcdA) and toxin B (TcdB). The corresponding genes, tcdA and tcdB, respectively, are located on a large pathogenicity island in the bacterial chromosome. 64 TcdA is a potent enterotoxin; 146 TcdB is a potent cytotoxin in vitro, but reportedly has little activity in vivo unless there is prior damage to mucosal epithelium. 90 The toxins act synergistically; TcdA creates widespread damage to the mucosa, permitting TcdB to affect epithelial cells. 75, 85 Though production of TcdA is more consistently associated with CDAD, there are reports of TcdA-negative, TcdB-positive strains of C. difficile causing clinical disease in humans. 153
TcdA and TcdB consist of single, large polypeptide chains with folds stabilized by disulfide bonds. 134 Aminotermini are highly conserved and are catalytic; both function in the target cell cytoplasm by the same mechanism. 34, 63, 72 The carboxyterminus, however, includes a receptor-binding domain that differs significantly between the two toxins. 155 Carbohydrate receptors for TcdA include Galα1-3β1-4GlcNAc-R (α-Gal epitope), Galβ1-4[Fuα1–3]GlcNAc (Lewis X), Galβ1–4GlcNAcβ1 (Lewis Y), Galβ1–14GlcNAcβ1–3Galβ1–4(Glc) (Lewis I), and sucrose-isomaltase. 83, 118, 151 These receptors are on the brush border of susceptible intestinal epithelial cells and may be found at other sites as well (Figs. 2, 3). A receptor for TcdB has never been identified, but is hypothesized to be situated on the basolateral aspect of epithelial cells so that it is hidden when the mucosa is intact.
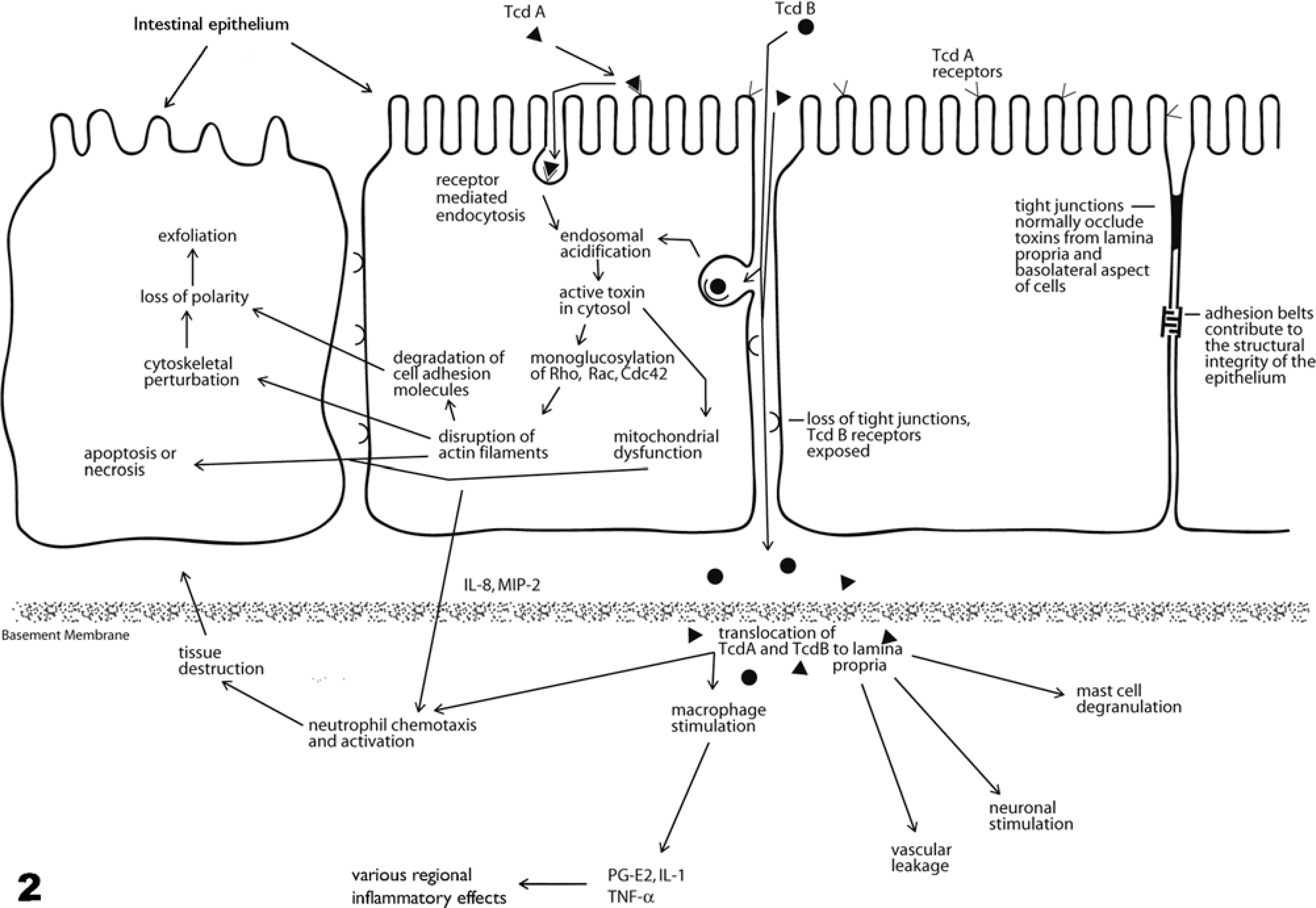
The pathogenesis of CDAD is a multifactorial disease process involving the alteration of cell signaling mechanisms and mitochondrial functions by C. difficile toxins, as well as direct and indirect stimulation of inflammatory and neurogenic pathways. Toxin A (TcdA) activity is dependent on attachment to receptors on the luminal surface of enterocytes followed by its internalization and activation in the endosome. It is hypothesized that toxin B (TcdB) recognizes receptors on the basolateral aspect of enterocytes, not normally accessible from the gut lumen. After loss of tight junctions, TcdB can bind the exposed receptors, contributing to a synergistic response between the two toxins.
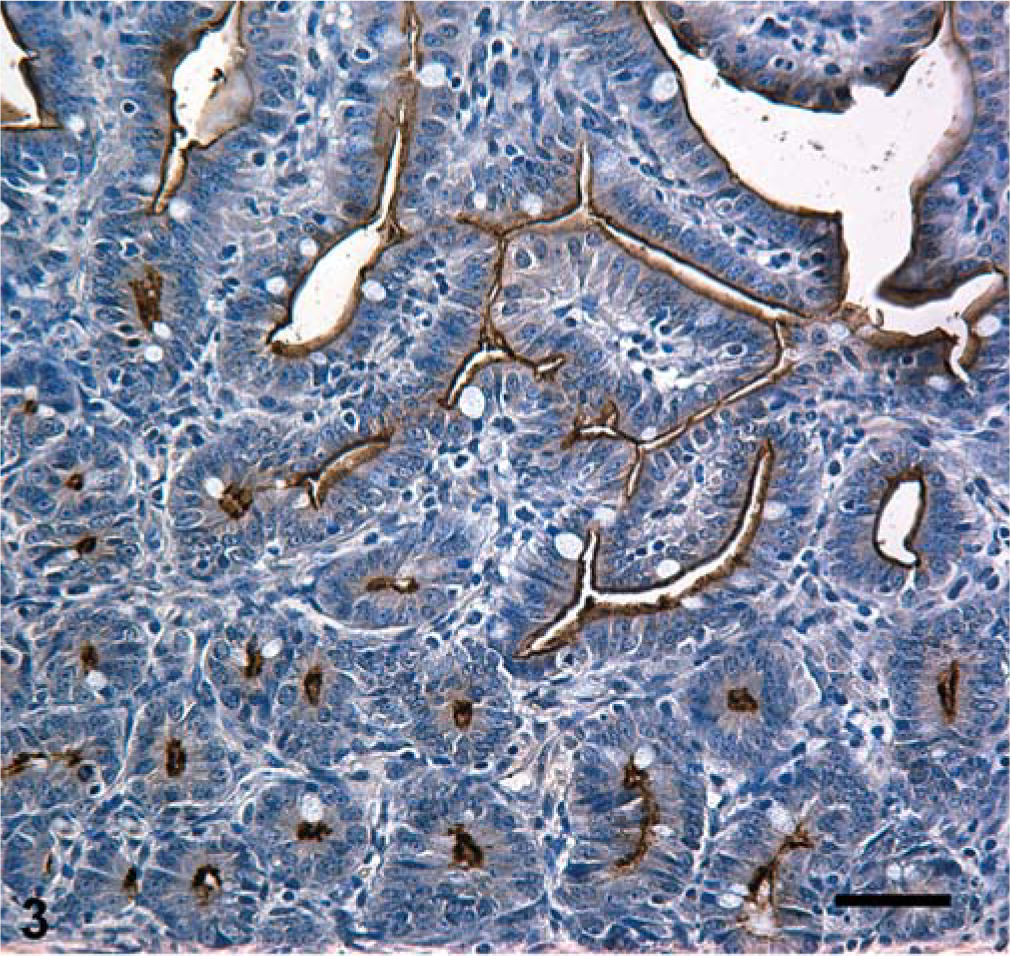
Jejunum; normal neonatal pig. Intestinal sections demonstrating specific adhesion of TcdA to the brush border of enterocytes. Formalin-fixed sections were labeled with biotinylated TcdA. Streptavidin-horseradish peroxidase with hematoxylin. Bar = 25 m.
Receptor binding by TcdA initiates endocytosis by coated pits, and the vesicles formed subsequently fuse with lysosomes. 44, 62, 115 Acidification of the endolysosome produces a conformational change in the toxin, followed by its activation and escape into the cytoplasm. 120 The mechanisms of cell attachment and endocytosis have not been described for TcdB, though it is activated in the endolysosomal compartment. 115 In the cytoplasm, toxin specifically inactivates Rho, and other Rho-subtype GTPases, by glycosylation of threonine at site 37/35. 46, 73, 74 This class of GTPases comprises signal transduction molecules associated with apoptosis and the maintenance and regulation of cytosolic actin filaments. Actin filaments are a necessary part of certain cell adhesion molecules (e.g., zonula adherens, tight junctions, and focal adhesions), and their disruption results in loss of cell-to-cell contacts, increased paracellular permeability of mucosal surfaces, cell rounding, and eventually, cell death. 119
TcdA and TcdB have many additional effects and amplify the disease process beyond their direct activity on mucosal epithelial cells (Fig. 2). Of particular importance is the initiation of an inflammatory cascade that can result in increased damage to host tissues and exudation of fluid. 119 An early event in this cascade is TcdA-induced mitochondrial dysfunction, resulting in depletion of adenosine triphosphate and the intracellular generation of reactive oxygen intermediates. 60 The oxidative burst is rapidly followed by nuclear translocation of nuclear factor-κB, leading to elaboration of interleukin (IL) 8 and, possibly, other proinflammatory mediators. 61 IL-8 is a chemokine associated with the mobilization, activation, and degranulation of neutrophils, as well as the chemotaxis of basophils and T cells. 3, 56, 102 Macrophage inhibitory protein 2 (MIP-2) is another potent chemotaxin of neutrophils elaborated by mucosal epithelial cells in response to TcdA, 25 which can also stimulate the in vitro migration of neutrophils directly. 79 Neutrophils play a prominent role in the pathophysiology of CDAD and contribute to necrosis of host tissues. 149 Inhibition of neutrophil infiltration by blocking the activity of CD18 or MIP-2 reduces the severity of lesions and decreases exudation of fluid into the intestinal lumen. 25, 79
Macrophages and monocytes are also involved in the inflammatory cascade associated with CDAD. Toxin A causes macrophages in the lamina propria to express cyclooxygenase-2 (COX-2), with subsequent production of prostaglandin E2. 1 The effect is probably due to direct action of TcdA on intestinal macrophages, as opposed to the influence of intermediary cytokines released by epithelial cells or some other cell type. Circulating monocytes produce IL-1, IL-6, IL-8, and tumor necrosis factor-α in response to application of TcdA. 40, 86 Prostaglandin E2 inhibits sodium chloride and water absorption in the intestine and induces chloride secretion by enterocytes. 1 In addition, it induces vasodilatation that may contribute to effusion across the intestinal mucosa. The secretory effects of TcdA can be inhibited by administration of a COX-2 inhibitor. 1
Mucosal mast cells may be involved in the development of CDAD. Administration of TcdA causes degranulation of mast cells within 15 to 30 minutes of toxin exposure. 26 Mast cell-deficient mice have reduced infiltration of neutrophils and intestinal secretion in response to TcdA challenge, but reconstitution of the mice fully restores their susceptibility. 163
The disease and lesions of CDAD have a neural component, as well. Release of substance P apparently results from direct stimulation of primary sensory neurons by TcdA; this, in turn, stimulates mast cell degranulation. 99, 117 Substance P is also associated with mast cell-independent pathways of inflammation, through its potent vascular effects and its ability to increase vascular permeability by directing the formation of endothelial gaps in venules. 10 Another neurogenic affect of TcdA is the inhibition of norepinephrine release from sympathetic postganglionic nerve fibers in the small intestine, causing direct excitation of enteric secretomotor neurons in the submucosal plexus. 169 The net result is increased secretion of water and electrolytes into the intestinal lumen. Secretion and inflammation attributable to TcdA can be greatly reduced by the denervation of ileal loops, but the protective effect is overcome by higher dosages of toxin. 94, 141
A small percentage of C. difficile strains produce an actin-specific ADP-ribosyltransferase, named CDT, which is unrelated to TcdA or TcdB. 112 The CDT is a binary toxin formed by separate, unlinked polypeptides, CDTa and CDTb. CDTb binds a cell surface receptor and mediates translocation of the enzymatic component, CDTa. 112 Once in the cytosol, CDTa catalyzes the ADP-ribosylation of monomeric actin, leading to depolymerization of actin filaments and disruption of the cytoskeleton. 52, 112 The binary toxin is directly toxic to eukaryotic cells in culture, and may be an additional virulence factor. 52, 111 However, the vast majority (approximately 95%) of clinical isolates produce TcdA and/or TcdB, but not CDT. 51
Epidemiology and Prevalence
The carrier rate of C. difficile varies among asymptomatic individuals by species and, within a species, by age and other population characteristics. The most detailed information is available for humans, but such data seem to be reflective of the situation in many species. Up to 50% of human infants are culture positive, though disease is almost unheard of in this group. 13 Infants may be refractory to disease because they lack toxin receptors. 18 The next highest prevalence of asymptomatic individuals is among hospital patients, of whom approximately 20% are culture positive. 67, 98 The bacteria are usually acquired from the hospital environment, and culture positivity and incidence of CDAD increase with length of stay. 9, 28, 67 Clostridium difficile is uncommon among healthy adults in the general population. 154
The carrier rate of C. difficile is low in asymptomatic foals and adult horses. The rate of isolation from clinically normal adults is 0–4.3%, whereas normal foals are generally reported to be culture negative. 7, 71, 92, 159 However, greater resolution of age classes may be necessary for meaningful interpretation. One investigator found a 29% culture-positive rate for asymptomatic foals <14 days old and a 0.6% carrier rate for older foals. 6 The rate of C. difficile isolation from horses with diarrhea varies from 12.7 to 90% of adults, and 16.7 to 63% of foals. 71, 92, 159 However, the highest culture rates were associated with CDAD outbreaks and might not accurately reflect the rate in all cases of equine diarrhea. Of all equine diarrhea cases at one veterinary teaching hospital, 20–25% were attributed to C. difficile. 158
Prevalence of C. difficile in normal cats may range from <10% to nearly 40%, although many isolates are nontoxigenic. 16, 124 The situation is much the same in normal dogs, with a culture-positive rate of approximately 20–40% and about 50% of isolates unable to produce toxin. 16, 91, 124, 142 Other surveys, however, failed to isolate C. difficile from clinically normal animals, though some asymptomatic dogs (7%) were toxin positive. 57, 161
Data concerning carrier rates or prevalence of CDAD in other species are lacking. Nonetheless, numerous case reports document CDAD in a variety of other mammal species and rarely among other vertebrates.
Predisposing Factors
The major predisposing factors for most species are antibiotic therapy and, for humans and horses, hospitalization. 7, 68, 92 The increased risk of CDAD in hospitals is due to persistent environmental contamination with C. difficile. Although hospitalization has not been documented as a predisposing factor for CDAD in other species, the environmental burden of C. difficile spores could be related to the incidence of disease in other situations. 123
Disturbance of the normal intestinal flora is the most important condition for development of CDAD, which is predisposed by antibiotic therapy. 122, 166 The indigenous flora of the gastrointestinal tract acts as a colonization barrier for pathogenic bacteria by, as yet, poorly defined mechanisms. The composition of this flora varies significantly by host species, but is always complex and is normally devoid of C. difficile. 96, 133 The inability of nonindigenous microbes to colonize a host with a fully established flora (colonization resistance) has been well documented by controlled experiments with multiple host and pathogen species. 20, 43 The antagonism of normal flora to C. difficile was documented by the suppression of C. difficile infection in vancomycin-treated hamsters when reconstituted with cecal homogenates from conventional animals. 168
A variety of mechanisms may be used by indigenous flora in the exclusion of nonindigenous bacteria, including: elaboration of bacteriocins; depletion of essential nutrients; production of toxic metabolic end products, such as volatile fatty acids (VFAs); competition for adhesion sites; stimulation of peristalsis; induction of immunologic responses; and production of H2S and secondary bile acids. 41, 96, 106, 121, 122 Such redundancy in the control of the microbial ecosystem confers stability among the constituent populations. However, evidence of only a few of these mechanisms has been disclosed to explain the exclusion of C. difficile from the normal intestinal tract.
Competition for essential nutrients is likely to be one factor limiting the growth of C. difficile. Monosaccharide components of mucin are colonic substrates that appear to be important to the growth of C. difficile in vivo, and results of in vitro experiments documented that mucin-degrading bacteria are prominent among the species that inhibit C. difficile growth. 164, 166
The production of VFAs by indigenous bacteria is associated with the exclusion of C. difficile from the large intestine. Of the VFAs produced by the commensal flora of the hamster gastrointestinal tract, butyric acid was the most inhibitory at naturally occurring concentrations. 126 Lactobacillus species are important inhibitors of C. difficile, as a commensal microbe and as an agent of probiotic therapy. This inhibition is strain specific and is correlated with the production of lactic acid and H2O2. 106 Other research failed to confirm the inhibitory effects of VFAs, and C. difficile, itself, can produce VFAs, including butyric acid. 77, 103, 143 Therefore, the role of VFAs in the exclusion of C. difficile by normal flora is still uncertain.
More mechanisms of C. difficile inhibition by indigenous flora may exist. Some bacteria have a protective effect against CDAD due to the partial inhibition of toxin production by C. difficile. 30 Also, adhesion factors may be important for the pathogenesis of CDAD. 17, 76 If the intestine contains bacteria or bacterial products that normally inhibit the adhesion of C. difficile, the pathogen might be more readily flushed from the system, preventing significant colonization. 170
Disruption of indigenous flora, followed by development of CDAD, can occur after administration of virtually any antibiotic. Even antibiotics that are efficacious against C. difficile can predispose individuals to CDAD; the gastrointestinal tract is readily recolonized by C. difficile spores ingested after cessation of therapy. 81 For many species, clindamycin and vancomycin therapy are a greater risk factor than other antibiotics. However, human cases of CDAD are more often associated with ampicillin and cephalosporins due to their more extensive use. 147 Similarly, CDAD in horses has been closely associated with administration of β-lactam antibiotics, presumably due to the prevalence of their use. 7 Erythromycin has also been implicated as a risk factor for acute colitis due to C. difficile in horses. 5, 54
For hamsters and guinea pigs, fatal enteric disease has long been observed in association with antibiotic administration. 82, 87, 137 Hamsters are more likely to develop CDAD as a sequela of antibiotic administration because they are more sensitive to C. difficile than are other species. The median lethal dose (LD50) of subcutaneously administered clindamycin for hamsters is 50 to 100 µg/kg of body weight, and typhlitis develops as soon as 48 hours after treatment. 88 Although CDAD is most commonly associated with antibiotic therapy, it can apparently develop in hamsters without use of antibiotics, if their environment is heavily contaminated by C. difficile. 123
Sporadic case reports of disease in captive exotic species have also indicated association with antibiotic administration. A captive Kodiak bear developed pseudomembranous colitis after initiation of kanamycin therapy, followed by tetracycline therapy. 108 Clostridium difficile–associated disease followed cefoxitin therapy in prairie dogs, and this species was proposed as a potential model for human pseudomembranous colitis. 104
Maturity is another well documented, though species-dependent prerequisite for CDAD; neonates of many species (e.g., humans, hamsters, and rabbits) are unaffected by C. difficile infection. 35, 55, 127 More than half of clinically normal human infants may be carriers of C. difficile, whereas asymptomatic, nonhospitalized adults are routinely culture negative. 84 Human cases of CDAD nearly always develop among adults, predominantly among elderly hospital patients. 9
Newborn rabbits are also resistant to CDAD, and TcdA does not affect ileal explants from 5-day-old rabbits, even at dosages that cause severe lesions in ileal explants from adults. 35 A prominent hypothesis to explain the resistance of such neonates is that they lack the proper toxin receptors until later in life. 18 Binding of TcdA to ileal brush borders is decreased in neonatal rabbits, but maximal binding is observed in 90-day-old rabbits. 35 However, in apparent contradiction to these findings, the binding kinetic variables of TcdA to intestinal brush borders of hamsters were similar for adults and infants and could not account for the age-related susceptibility to CDAD. 125
In contrast to neonatal rabbits, newborn hares are sensitive to the enteric effects of C. difficile. Inoculation of young hares resulted in inflammation and necrosis, principally of the duodenum, cecum, and colon. 32 Additionally, severity of the disease was greater for co-infections with C. perfringens or C. tertium, or both, even though these species failed to cause disease in monoassociated hares. To the authors' knowledge, the susceptibility of adult hares to CDAD has not been described.
Horses are susceptible as adults or foals, and may develop CDAD in the first few days of life. 70, 92, 157 In contrast, spontaneous disease in pigs is apparently an exclusive disease of neonates, with a mean age of 5 days (range, 1–14 days). 156 There is a single case report of pseudomembranous typhlocolitis in 8-week-old pigs, but Salmonella typhimurium was isolated in addition to C. difficile. 69 Our unpublished data and anecdotal evidence confirm this as a neonatal disease of pigs.
Dietary factors reportedly may also contribute to development of the disease. An atherogenic diet, high in saturated fat and cholesterol, when fed to Syrian hamsters, purportedly caused mortality attributable to C. difficile. 12 High amounts of dietary casein and corn starch greatly increased the likelihood that C. difficile–associated typhlitis would develop in hamsters, after treatment with vancomycin. 100 Diet is known to impact the balance of bacterial species in the indigenous flora, and such changes could alter the host's colonization resistance to C. difficile. 38, 49
Sensitivity and Host Susceptibility
Hamsters are exquisitely sensitive to C. difficile infection and, if pretreated with clindamycin, can develop fatal disease when inoculated with as few as one colony-forming unit of C. difficile. 84 TcdA administered intragastrically (IG) in doses as low as 0.08 mg/kg of body weight causes diarrhea and lesions in hamsters; 0.16 mg/kg is uniformly fatal. 90 Mice rarely develop spontaneous disease, but they are moderately susceptible to the toxins and experimentally induced infection. Intragastric inoculation of mice with 2 mg of TcdA/kg causes diarrhea and lesions, but not death. 90 Rats seem to be the most resistant laboratory animals to the effects of C. difficile toxins; the same dosage (2 mg/kg administered IG) that causes disease in mice and is fatal for hamsters has no effect on rats. 90 Despite their decreased sensitivity to toxin, rats are often used as experimental models, especially in situations where a surgical model (e.g., inoculation of intestinal loops) is needed, and mice or hamsters are simply too small.
Clostridium difficile–associated disease develops in laboratory rabbits, although C. spiriforme is probably a more important cause of spontaneous disease. 110, 113 Peracute death, without clinical signs of disease, is a common feature of CDAD in rabbits. 24, 113 Rabbits are an adequate experimental model, but like rats, are most often used in experiments requiring surgery, particularly the ligation of intestinal loops.
Clostridium difficile seems to be an uncommon pathogen of domestic carnivores, with limited case reports. Chronic diarrhea in dogs was attributed to C. difficile infection, which persistently relapsed after cessation of metranidazole therapy. 8 Clostridium difficile–associated disease in two cats was diagnosed on the basis of detection of toxins, responsiveness of clinical signs of disease to metranidazole therapy, and, for one of the cats, exclusion of other potential etiologic agents. 162
Clostridium difficile–associated disease in nonhuman primates is rarely reported, but Old World and New World monkeys are known to be susceptible to naturally acquiring disease and to the effects of purified toxins. Postantibiotic diarrhea in juvenile and adult cotton-top tamarins (Saguinus oedipus) was reportedly attributed to C. difficile. 128 The two individuals with the most severe clinical signs of disease had mild lesions, consisting of only mucosal congestion and crypt dilatation. Individuals with severe lesions had mucosal to transmural necrosis extending from the cecum to the anus. Long-tailed macaques (Macaca fascicularis) and pig-tailed macaques (M. nemestrina) developed diarrhea that was correlated with C. difficile toxins in fecal extracts. 150 Rhesus monkeys (M. mulatta) are susceptible to intravenous or intraperitoneal inoculation of TcdA and TcdB, but to our knowledge, spontaneous disease has never been reported. 2
There are apparently no published reports of C. difficile causing disease in apes, but it has been diagnosed in a single orangutan (Pongo pygmaeus) (J. S. Brazier, personal communication). Case reports of CDAD in primates describe a clinical syndrome and lesions similar to that described in other species. It may be that the relative infrequency of cases is due to underreporting or a lack of predisposing factors, rather than marked resistance to infection or to the effects of C. difficile toxins.
Humans seem to have moderate susceptibility to C. difficile. Clostridium difficile–associated disease of humans is generally limited to the combination of advanced age, administration of antibiotics, and hospitalization, whereas most other species do not have such clearly defined risk factors. 65 Also, most hospital patients infected with C. difficile are asymptomatic, suggesting that they are not unduly susceptible to development of CDAD. 67, 98 Possible contributing factors to the large number of human CDAD cases include an aging population, widespread administration of broad-spectrum antibiotics, and a large number of susceptible, debilitated individuals in hospitals, where spore densities are highest. 66, 80
Though it is generally a pathogen of mammals, C. difficile has been isolated from asymptomatic birds and some poikilotherms. There is one case report of CDAD in a group of 9-day-old ostriches with severe hemorrhagic typhlitis and colitis. 42 Clostridium difficile is also reportedly an infrequent cause of hepatitis in ostrich chicks. 136
Clinical Syndromes and Lesions
The distribution and severity of lesions varies among susceptible species infected with C. difficile. However, the lesions associated with naturally acquired CDAD generally have an aboral distribution, principally affecting the colon or cecum, or both, and variably, the ileum. Clinical signs of disease can vary greatly within a species, and this has been most consistently documented in humans. Most of the people infected with C. difficile remain asymptomatic, but a portion will develop diarrhea with or without evidence of colitis. Usually diarrhea develops in the absence of grossly visible colitis or systemic signs of disease. 98 In most instances, colitis is not associated with pseudomembrane formation, but may be characterized by diffuse or patchy erythema. Clinically, such patients describe watery diarrhea, abdominal pain, nausea, anorexia, and malaise. 80 Some also have low-grade fever and polymorphonuclear leukocytosis. A more severe colitis is characterized by pseudomembrane formation, usually confined to the rectosigmoid region. 80 It occasionally extends to more proximal parts of the colon, rarely involving the entire colon. The clinical syndrome associated with pseudomembranous colitis is similar to that already described, but symptoms are often more severe. A minority of patients, approximately 3%, will develop severe colitis that may progress to more serious, life-threatening complications such as chronic ileus, megacolon, and intestinal perforation. 130 Lesions in species other than humans generally maintain the aboral distribution, but are usually more extensive.
The distribution of lesions in hamsters and guinea pigs is predominantly cecal, with occasional extension into the ileum or colon. 4, 87, 88, 123 The cecum is usually distended by fluid and has hemorrhages that vary in severity from multiple petechiae to diffuse hemorrhage of the cecal wall (Fig. 4). 4, 12 Affected hamsters die after a brief bout of diarrhea or without any obvious clinical signs of disease. 4, 12, 132 There are also a few reports of chronic CDAD in hamsters that was characterized by chronic typhlitis with mucosal hyperplasia. 27, 50, 131 Cholangiohepatitis, and amyloidosis of liver, kidneys, and intestinal wall, especially the cecum, were also described. Healthy, age-matched controls did not develop amyloidosis and, in those cases with chronic CDAD, it was attributed to chronic inflammation. 27 Though spontaneous amyloidosis is a common age-dependent lesion in hamsters, it is not usually associated with the gastrointestinal tract. 48
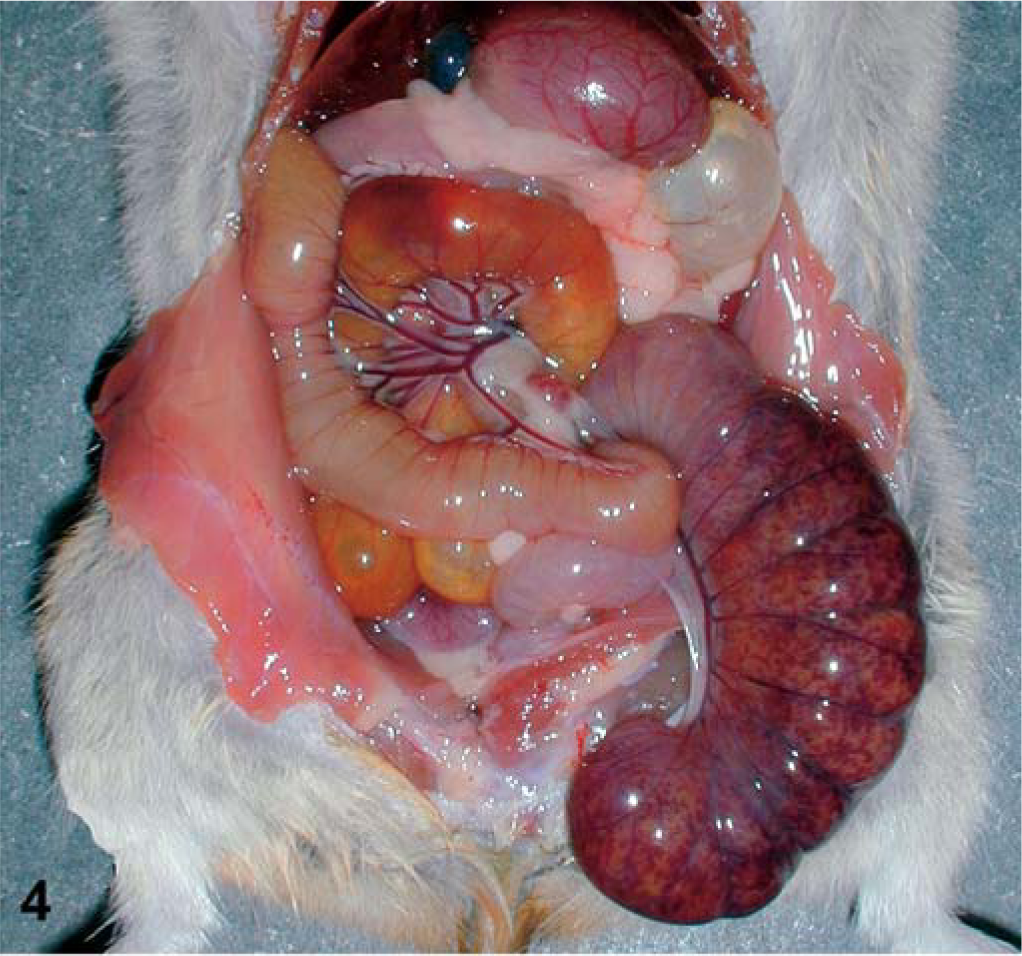
Abdominal viscera, adult hamster with CDAD. The cecum is hyperemic, has multiple ecchymotic hemorrhages, and is distended by fluid. The small intestine and colon are slightly distended by fluid, but lack the mucosal necrosis and inflammation microscopically evident in the cecum.
Inoculation of C. difficile into axenic mice induces ulcerative typhlitis. 105 However, lesions did not develop after inoculation of mice that had a full constituency of normal flora, even after treatment with cefoxitin. 105
Mucosal lesions of pigs are limited to the cecum and colon. They are typically mild, but vary from grossly inapparent, multifocal necrosis of surface epithelial cells to transmural necrosis. 156 Pigs with spontaneous disease typically have small microscopic ulcers with effusion of fibrin and neutrophils into the lumen, so-called “volcano ulcers” (Fig. 5). Edema is a common feature and manifests as hydrothorax, ascites, or mesocolonic edema, or all 3 (Fig. 6). 156 Diarrhea is variably present, and some pigs with mild disease are apparently obstipated. Other clinical signs of disease include dyspnea, mild abdominal distention, and scrotal edema. 138, 156
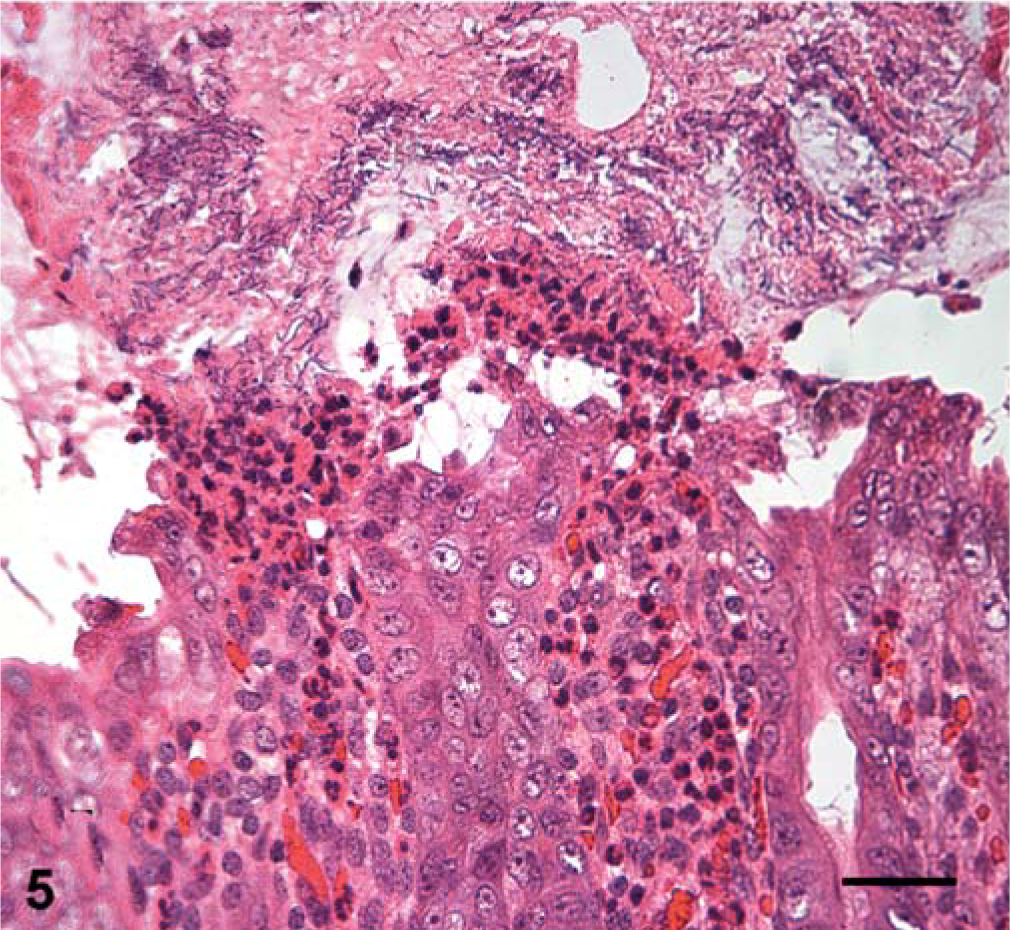
Colon; neonatal pig with CDAD. Small ulcers are associated with effusion and massive migration of neutrophils. Many rod-shaped bacteria, consistent with C. difficile, adhere to the protein-rich exudate. Though they are not pathognomonic, such lesions are associated with C. difficile infections in many species. In more severe cases, the ulcers can coalesce and necrosis may affect deeper tissues. HE. Bar = 25 µm.
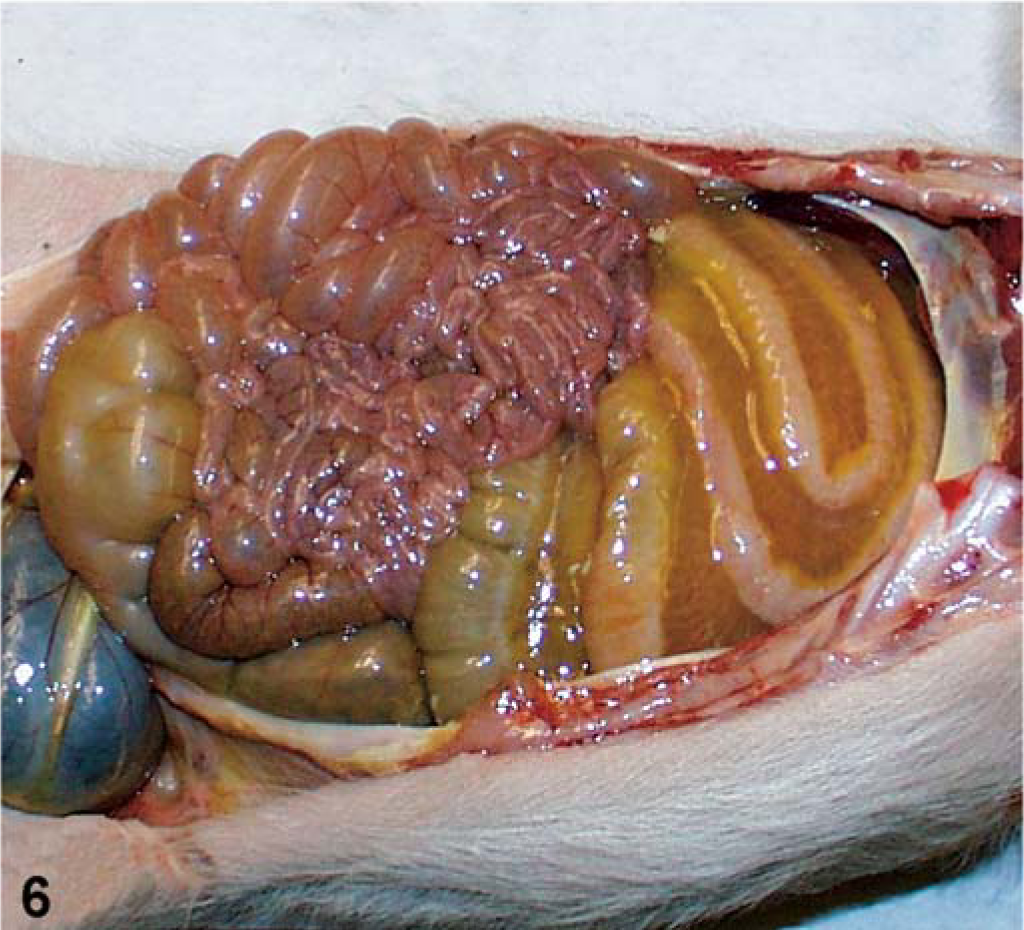
Abdominal viscera; neonatal pig with CDAD. The mesentery of the spiral colon is greatly distended by edema. Such mesocolonic edema can be caused by other pathogens, but is common in neonatal pigs with spontaneous disease and in controlled experimentally induced infections with C. difficile.
As with many other species, lesions in adult horses are isolated to the cecum and ascending colon. 54, 114 Affected segments are often edematous with superficial ulcers or erosions, but in more severe cases, hemorrhagic necrotizing typhlocolitis has been reported. 54, 114 Microscopic lesions do not differ remarkably from those associated with CDAD in other species, and include edema, variable vascular necrosis, and multifocal mucosal necrosis, erosions, and ulcers that vary with severity of the disease. 114
In contrast to adult horses and most other susceptible species, foals consistently develop severe lesions in the small intestine. The small intestine and colon of diseased foals may be hemorrhagic and have patchy mucosal erosions or ulcers, with exudation of fibrin and adherent ingesta. 70, 93 Severe necrosis of villus epithelium, with adherent gram-positive, rod-shaped bacteria, is observed microscopically. Crypt epithelium may be intact. 70 Foals with colitis have been reported, but such cases apparently are rare. 93 Foal disease can begin immediately after birth and is associated with high mortality, even with intensive medical therapy. 148 Like foals, rabbits are most likely to develop lesions in the small intestine. Severe lesions are seen in the ileum, whereas those in the cecum are milder and more variable. 113
A different distribution or density of toxin receptors, or both, in the gastrointestinal tract may explain the species-specific distribution of C. difficile–associated lesions, but may also relate to the regions of the gastrointestinal tract in which C. difficile is able to proliferate to substantial numbers. Higher numbers of C. difficile probably produce greater quantities of toxin, increasing the potential for local lesion development. For instance, though C. difficile infection culminates in predominantly cecal lesions in hamsters and mice, 4, 12, 105 direct application of culture filtrate to either species indicates that stomach and small intestine are also susceptible to toxin effects. 90 Experimental inoculation of clindamycin-treated hamsters is associated with proliferation of C. difficile in the cecum but not in the small intestine. 167 Therefore, the more limited distribution of lesions in naturally acquired disease may reflect the distribution of C. difficile in the gastrointestinal tract.
Other Clostridial Enteric Diseases
As a group, members of the genus Clostridium induce a multitude of widely differing disease syndromes, including a number of enteric diseases important to various mammals and birds. Most other clostridial enteropathies have lesions that are not limited to the distal portion of the gastrointestinal tract, thus differentiating them from CDAD. There are rare exceptions to this difference, and they will be discussed individually. Excellent reviews of clostridial enteropathies are available that cover the subject in much greater detail than can be presented here. 14, 59, 139, 140
Clostridium perfringens is probably the most important clostridial cause of intestinal disease. 140 The species is classified into five types, A–E, on the basis of production of the four major toxins, α, β, ∊, and ι. 19, 97, 129 A diversity of syndromes is attributed to C. perfringens and includes enterotoxemia and enteritis. 39, 140, 152 Specific clinical signs and lesions of disease vary with the type of C. perfringens involved and the species affected. 140 Unlike CDAD, lesions attributable to C. perfringens tend to be found in the proximal segments of the gastrointestinal tract, affecting the small intestine and even the stomach or rumen. Even when the colon is involved, as reported in goats and pigs, lesions extend more proximad than do those in cases of CDAD. 29, 101, 107 Foals, however, can apparently develop enteritis with extensive necrosis in the small intestine attributable to C. difficile and C. perfringens. 21, 109 Therefore, both agents should be included in the differential diagnosis of necrotizing enteritis of young foals.
Clostridium spiroforme causes iota enterotoxemia of rabbits. 15, 22, 23, 58 Like CDAD, this condition is associated with disruption of the gastrointestinal flora, either by antibiotic therapy, or at weaning. 15, 22, 78 Lesions associated with C. spiroforme have been most often identified in the cecum, with lesions extending to the proximal portion of the colon or distal portion of the ileum. 110 Spontaneous CDAD in rabbits is principally associated with lesions in the small intestine, especially the ileum. 113 Both diseases cause mucosal necrosis, and the microscopic lesions may be similar, with the possible exception of tissue distribution.
Malignant edema, a common wound-associated condition of horses, sheep, and cattle, is the disease most commonly attributed to C. septicum. 59, 140 As an agent of enteric disease, it is the cause of braxy, also known as bradsot, an acute infection of lambs and older sheep. 14, 45 The disease has also been reported in calves. 36 It occurs principally in Great Britain, Ireland, Norway, Iceland, and the Faroe Islands, although it has been infrequently reported from Europe, Australia, and the United States. 36, 140 The ingestion of cold or frozen feed is associated with penetration of the pathogen through the mucosa of the abomasum, causing mural edema, hemorrhage, and variably, mucosal necrosis. 36 There is probably little confusion of the lesions associated with C. septicum and C. difficile due to the extreme proximal distribution of the lesions in braxy and the occurrence of the disease in a limited number of species.
Diagnosis of CDAD
Clostridium difficile–associated disease should be suspected in any case of necrotizing enteritis, particularly if it affects the distal portion of the gastrointestinal tract. However, lesions may be located more craniad in some species (e.g., foals and rabbits), and cranial lesions should not be the basis for ruling out CDAD in those species, or in species for which CDAD is poorly documented. The diagnosis of CDAD is contingent on isolation of the pathogen from fecal samples or detection of C. difficile toxins, or both. 33 A history of antibiotic use and the susceptibility of the species and age class of the animals examined may help increase the suspicion of CDAD. Some individuals are asymptomatic carriers of C. difficile; therefore, other necrotizing enteritides with similar lesion distribution should be ruled out. The antemortem diagnosis of CDAD is often difficult due to the necessity to rule out a large number of other potential causes of diarrhea that may be discounted in the postmortem examination on the basis of the distribution and microscopic appearance of the lesions.
Clostridium difficile is isolated on selective media that are commercially available or made fresh according to published techniques. 37, 47, 165 The contents of the large intestine or fecal samples should be submitted in an anaerobic transport medium unless they can be delivered to the bacteriology laboratory within 24 hours. 160 If samples cannot be delivered to the laboratory within 24 hours they should be sent refrigerated to be received as soon as possible.
Toxin is detected by ELISA or by assessing the cytopathic effect of fecal filtrate on cell lines. The ELISA is rapid, sensitive, and specific, and is commercially available. 116, 135 The cytotoxin assay is specific and more sensitive, but requires cell culture facilities and takes considerably more time than the ELISA. 116, 144 Toxin production by isolates is determined by use of polymerase chain reaction assays, using primers specific for TcdA and TcdB 53, 145 Toxin is also identified in culture filtrate by ELISA or cell culture assays. 89 It is important to determine the potential of isolates to produce toxin, as some isolates lack one or both toxin genes or essential elements of the genes. 11, 95
Clostridium difficile–associated disease is poorly documented in various species, and this may be due to its underdiagnosis. The disease has only recently been recognized with appreciable frequency in horses and pigs. Increased awareness of the disease and its lesions will likely result in the diagnosis of CDAD in other species not known to be susceptible. Further research may help alleviate problems associated with CDAD by revealing species-specific risk factors and new prophylactic measures. Additional information about the pathogenesis of the toxins and the mechanisms of host colonization is particularly important. Such information could improve animal welfare and livestock revenues as well as generate better understanding of the disease in humans.
