Abstract
Fatal adverse reactions to vaccination are uncommon and poorly documented. To our knowledge, this is the first case report of fatal adverse reaction to an inadvertent intravenous vaccination in three calves vaccinated against respiratory (Somnustar Ph) and clostridial (Tasvax 8) diseases. All three calves had severe acute interstitial pneumonia with multifocal pulmonary hemorrhages that resulted in fatal respiratory failure. Qualitatively, the pulmonary lesions in these calves were similar to those in septicemic/endotoxemic calves; however, the severity and extensity of pulmonary hemorrhages were of a higher degree than those usually observed in clinical septicemia/endotoxemia. In addition, approximately 30% of the arterioles and small arteries were surrounded by hemorrhages, which occasionally extended around adjacent bronchioles. A unilateral perijugular hematoma with recent transmural perforation of jugular vein found in all three calves was believed to have been caused by the injection needle during vaccination, and the fatal pulmonary changes were believed to have been secondary to the intravenous injection of vaccine.
Vaccination plays a very important role in the management of animal and public health. Accordingly, each year, billions of animals are vaccinated and thus exposed to various antigens and adjuvants present in vaccines to stimulate their immune response and induce resistance to various diseases. The vaccinated animals may have undesired local or systemic adverse reactions associated with vaccination. Local reactions to vaccines are relatively well documented and are usually observed by clinicians. Most common local reactions to vaccines are transient erythema, edema, and swellings. 2 Less common but well-documented and potentially detrimental local adverse reactions are alopecia in dogs, 14 vaccine-associated sarcoma in cats, 6 muscle necrosis and spinal cord inflammation in lambs, 9 peritonitis in salmonids vaccinated against furunculosis, 11 and granulomas in commercial birds. 4 Systemic adverse reactions to various vaccines are not as common, and clinical cases of these reactions are relatively poorly documented in professional literature. 5 To our knowledge, this is the first case report of clinical fatal adverse reaction to an inadvertent intravenous vaccination of calves against bacterial respiratory (Somnus-tar Ph) and clostridial diseases (Tasvax 8).
A group of 75 Red Angus calves, all roughly aged 2 months, were restrained in a calftilt-chute and vaccinated by an inexperienced person with two different bacterial vaccines (i.e., Somnustar Ph [Novartis Animal Health, Mississauga, Ontario] and Tasvax 8 [Schering-Plough, Kenilworth, NJ]). Somnustar Ph consisted of the outer membrane components of Haemophilus somnus and Pasteurella haemolytica and of the attenuated leukotoxin of P. haemolytica emulsified in an adjuvant. Tasvax 8 contained adjuvant with toxoids of various Clostridium spp.: C. chauvoei, C. novyi, C. perfringens types B, C, and D, C. septicum, C. haemolyticum, and C. tetani. Vaccines were intended to be injected into the subcutaneous tissue in the ventral portion of the neck. Approximately 2 hours after the vaccination, 19 of the 75 calves showed signs ranging from mild depression to severe dyspnea with red, foamy nasal and oral discharge. When the veterinarian arrived on the farm, three of the more severely affected calves died. All other vaccinated calves were treated with dexamethasone (Dexamethasone 5, Vetoquinol, Lavaltrie, Quebec) and epinephrine (Epichlor, Rafter 8, Calgary, Alberta), and there were no additional mortalities in the herd associated with this vaccination.
The three calves (Nos. 1–3) that died were submitted for postmortem examination to Prairie Diagnostic Services, Western College of Veterinary Medicine, University of Saskatchewan. On gross examination, all three calves were in good body condition, and each calf had 1) severe diffuse pulmonary edema, congestion, and multifocal-to-coalescing hemorrhages in the dorsal pulmonary parenchyma, which failed to collapse when the thoracic cavity was opened (Fig. 1); 2) a unilateral 3- to 4-cm-long hematoma surrounding the jugular vein on the right side of the neck approximately 15–20 cm ventrally from the ipsilateral mandibular ramus (the location of the peri-jugular hematoma corresponded to the vaccination site according to history); 3) an approximately 0.5-mm-diameter defect in the tunica intima of the jugular vein corresponding to the location of the peri-jugular hematoma (Fig. 2); and 4) subendocardial hemorrhages.
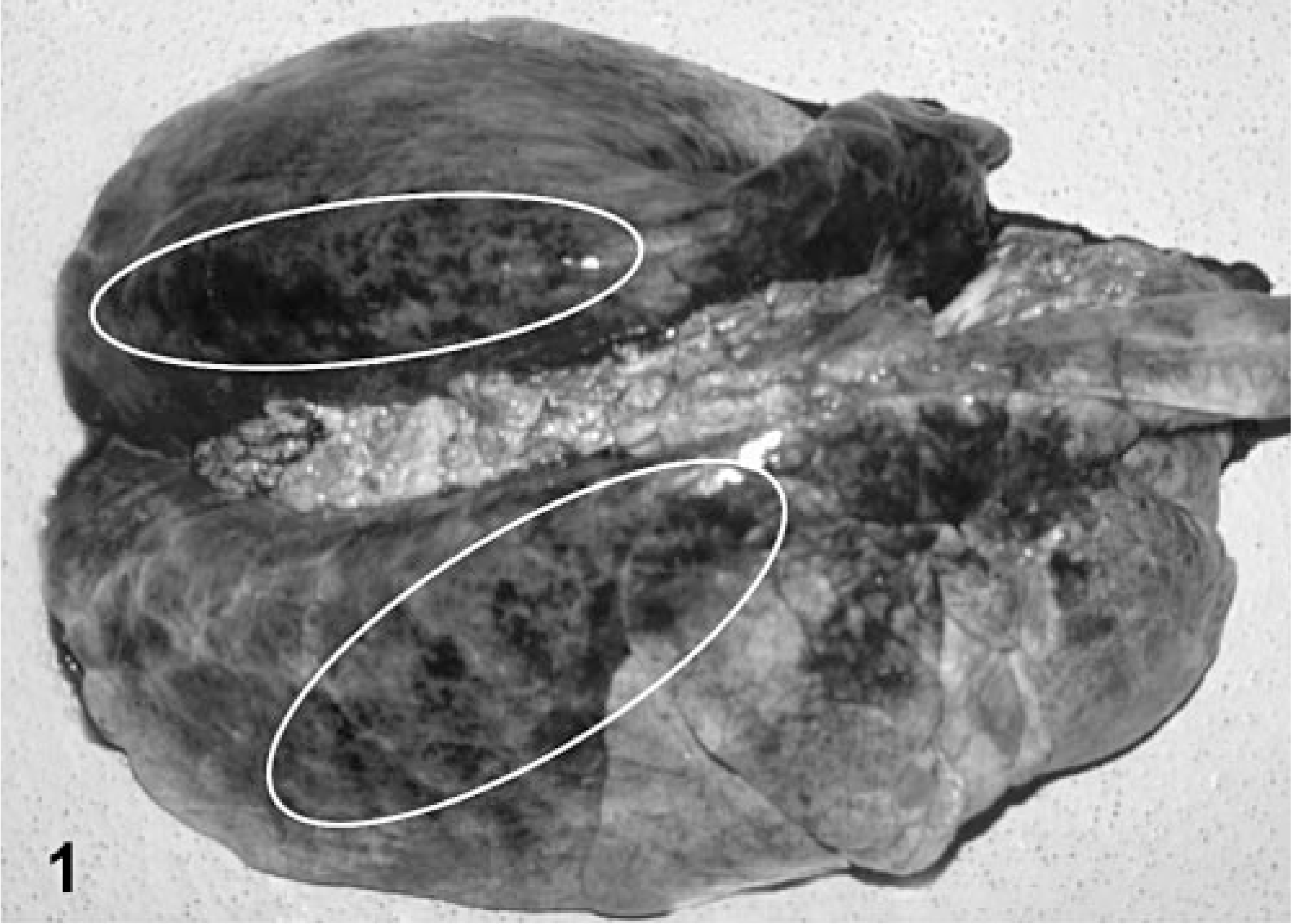
Lungs, calf No. 1. The lungs failed to collapse and were affected by diffuse pulmonary edema, congestion, and multifocal-to-coalescing hemorrhages (circled with white line).
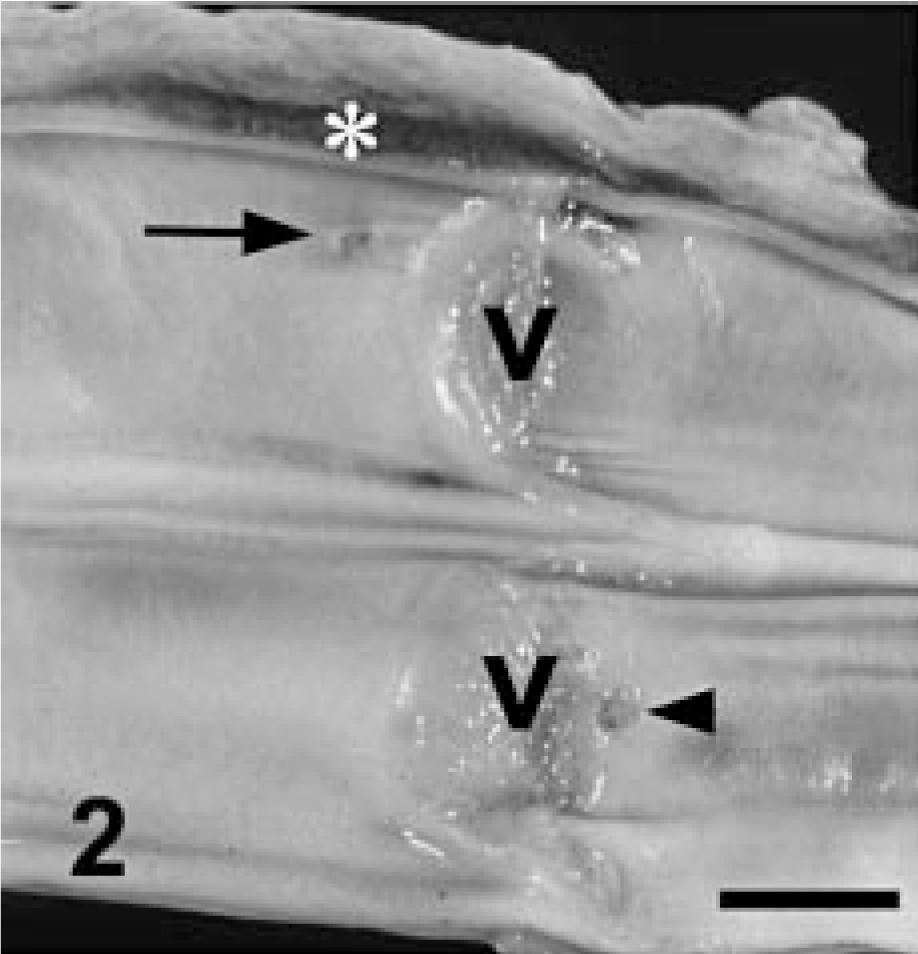
Jugular vein, calf No. 1. The opened partially fixed jugular vein with exposed venous valves (v) is affected by a peri-jugular hemorrhage (∗), a transmural perforation of jugular vein (arrow), and a focal erosion of tunic intima on the opposite side of the perforation (arrowhead). Bar = 5 mm.
Histopathologic changes were similar in all three calves (Nos. 1–3). The lungs were affected by acute interstitial pneumonia with multifocal-to-coalescing severe intraalveolar and interstitial hemorrhages. The terminal bronchioles and alveoli were filled diffusely with proteinaceous edema fluid containing a moderate number of macrophages and a multifocally variable number of neutrophils. The interalveolar septa were thickened by interstitial edema, diffuse activation of pulmonary macrophages and endothelial cells, and congested/hyperemic capillaries containing a multifocally increased number of neutrophils. Occasional capillaries contained fibrillar proteinaceous material consistent with polymerized fibrin. Interlobular septa and pleural interstitium were affected by diffuse edema and multifocal hemorrhages. Approximately 30% of the arterioles and small arteries were surrounded by hemorrhages (Fig. 3). Serial histologic sections of the affected jugular vein from each calf (Nos. 1–3) showed that the 0.5-mm defect in the tunica intima, observed during gross examination, was an approximately 0.2-mm transmural perforation of the jugular vein. The adjacent tunica adventitia was affected by a severe hemorrhage corresponding to a peri-jugular hematoma observed during the postmortem examination (Fig. 4). Hepatic Kupffer cells and glomerular mesangial cells were activated, and multiple parenchymal organs were congested. The periacinal hepatocytes and multifocal renal tubular epithelial cells were affected by cytoplasmic hypereosinophilia, nuclear pyknosis, and occasional cellular disintegration consistent with ischemic degeneration and necrosis.
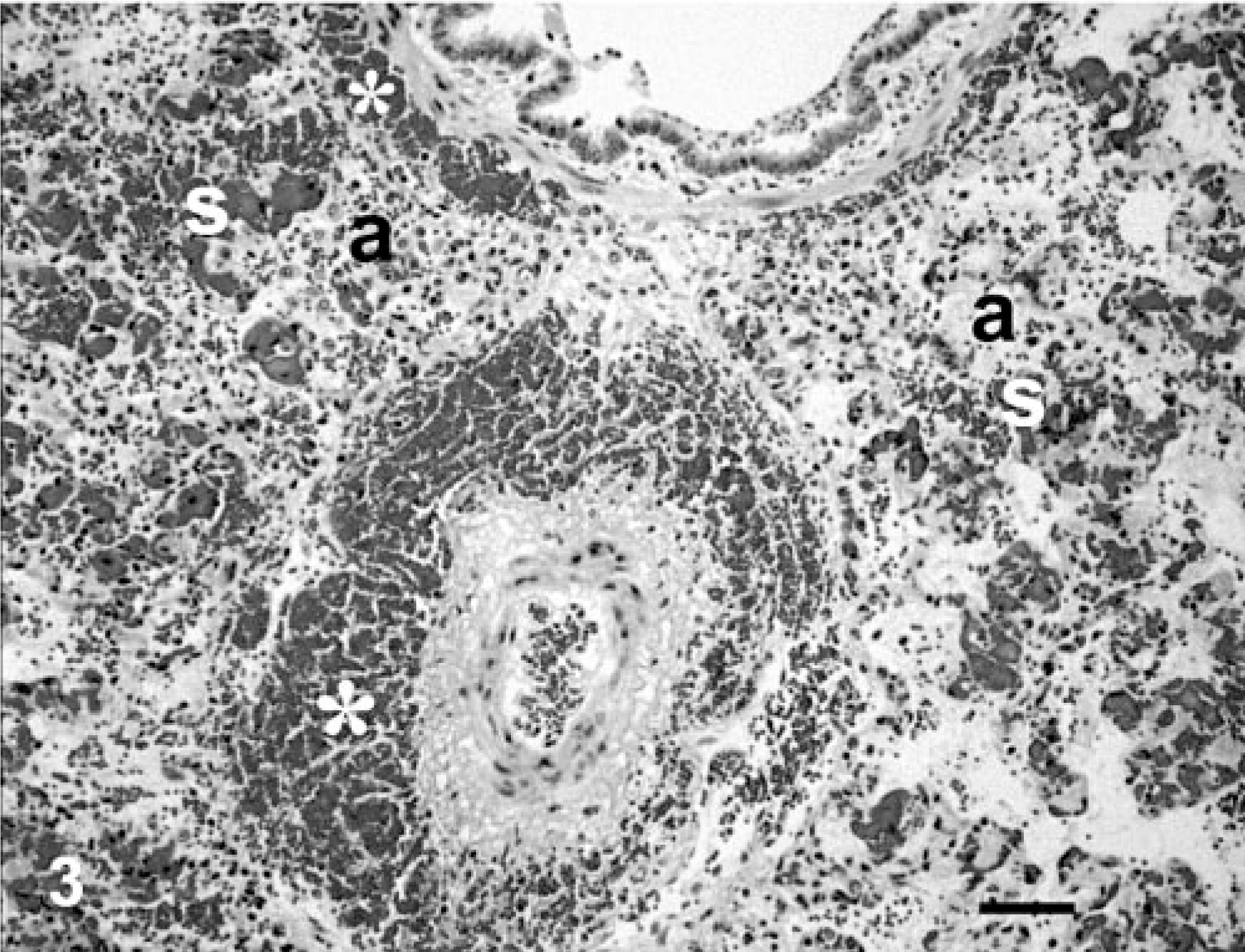
Lung, calf No. 1. Histologic appearance of the lungs affected by acute interstitial pneumonia characterized by thickened, congested, and collapsed alveolar septa (s), intraalveolar proteinaceous edema with macrophages and erythrocytes (a), and periarteriolar and peribronchiolar hemorrhages (∗). HE. Bar = 25 μm.
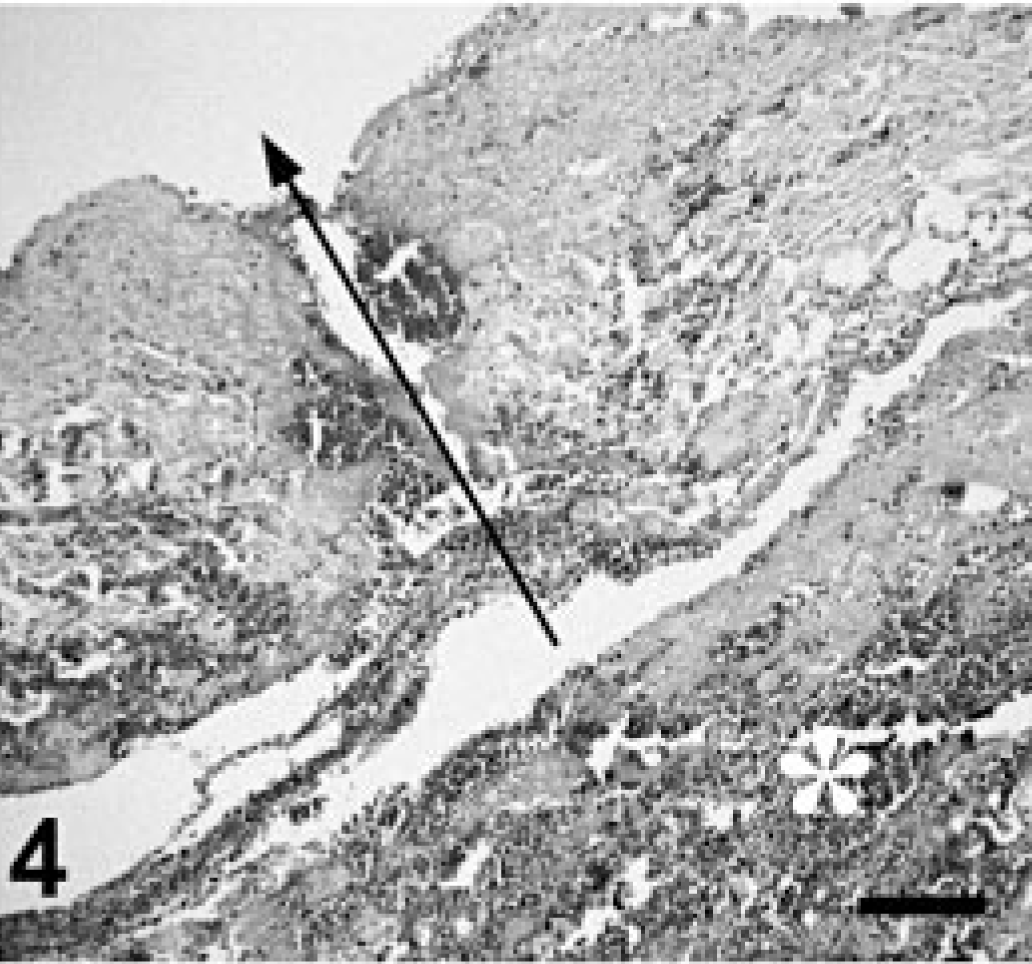
Jugular vein, calf No. 1. Histologic appearance of the transmural perforation (arrow) of the jugular vein (sectioned at the site indicated by the arrow in Fig. 2) and adjacent peri-jugular hemorrhage (∗). HE. Bar = 100 μm.
Samples from the lung, liver, kidney, and spleen of each calf were submitted for bacteriology. No bacteria were isolated from any of the tissues from calf No. 1, but from calf Nos. 2 and 3, a scant growth of contaminants (i.e., nonhemolytic Escherichia coli, alpha Streptococcus spp., or Lactobacillus spp.) was detected in some tissues.
The pulmonary changes observed in all three calves were consistent with severe pulmonary changes induced by septicemia or endotoxemia in spontaneous and experimental cases. Slocombe et al. 12 reported that the intravenous injection of lipopolysaccharide (LPS) isolated from P. haemolytica caused pulmonary edema with intraalveolar macrophages and multifocal intraalveolar hemorrhages, thickened and hypercellular interalveolar septa, and multifocal alveolar collapse in calves. All of these pulmonary changes, but with greater severity, were present in the lungs of the three calves of our study. In addition, approximately 30% of the pulmonary arterioles and small arteries were surrounded by hemorrhages in all three calves.
The hepatic and renal changes observed in all three calves were considered secondary to septicemia and ischemia. An activation of the Kupffer and glomerular mesangial cells is commonly observed in animals that die due to septicemia and endotoxemia. Degeneration and necrosis of the periacinal hepatocytes and renal tubular epithelium were thought to have been caused by the terminal ischemia associated with fatal respiratory failure due to severe diffuse pulmonary changes.
The macroscopic and microscopic changes observed in the right jugular vein of each calf were attributed to a recent needle perforation. Given that none of calves was given any injection, except for vaccination, it stands to reason that the transmural perforations in jugular veins were caused incidentally during the vaccination procedure. Even though we do not have unequivocal evidence that any amount of the vaccine was injected intravenously, considering the severity of the pulmonary changes and mortality 2 hours after the vaccination, we believe that the fatal pulmonary changes were caused by vaccine components injected intravenously in these three calves.
The specific list of vaccine components and their concentrations is proprietary information and was not available to the authors. However, on the basis of product specifications, Tasvax 8 contains toxoids of various clostridial pathogens (listed above), and Somnustar Ph consists of the outer membrane components of H. somnus and P. haemolytica and of the attenuated leukotoxin of P. haemolytica. Both P. haemolytica and H. somnus are gram-negative bacteria that have abundant LPS, also called endotoxin, on their outer membrane. LPS is composed of a polysaccharide of variable chain length (O-polysaccharide and core polysaccharide) and a covalently bound lipid component, termed lipid A. The lipid A portion of LPS is responsible for the biologic effects of endotoxin during endotoxemia. 10
Septicemia or endotoxemia is initiated by macrophages activated by LPS. The activated macrophages secrete multiple inflammatory mediators (e.g., tumor necrosis factor alpha, IL-1, IL-6, IL-8, platelet-activating factor, eicosanoids, chemokines) that have profound local and systemic effects. Some of these mediators activate endothelial cells with a consequent increased capillary permeability and expression of adhesion molecules, which facilitates the extravasation of activated neutrophils. This proinflammatory cascade, if uncontrolled, can escalate and result in septicemic or endotoxemic shock, characterized by fever or hypothermia, peripheral vascular dilation, endothelial damage and leakage, disseminated intravascular coagulation, tachycardia, tachypnoea, hypovolemia, and death (reviewed by Van Amersfoort et al. 13 ).
The response that an individual has to endotoxin is influenced by the dose of endotoxin received, the route of administration, and the age, genetics, and immune status of the animal. 3, 12 We suspect that, in our study, the most important factors responsible for the different reactions of calves to vaccination were the route of administration and the volume of vaccine that was injected intravenously. Accordingly, the three calves that died most likely received a higher dose of vaccine intravenously than did those that had the above-described clinical signs but survived. The calves that did not show any clinical signs associated with vaccination had most likely been injected subcutaneously with the entire amount of vaccine.
Even though the pulmonary lesions observed in these calves were similar to those present in septicemic or endotoxemic calves, LPS present in Somnustar Ph vaccine may not have been the only pathogenic factor that caused this lesions, because other components of gram-negative and gram-positive bacteria (e.g., superantigens) are able to initiate and perpetuate the fatal cytokine cascade, resulting in morphologic lesions identical to lesions of septicemia or endotoxemia. It is not clear if, or to what extent, toxoids from Tasvax 8 contributed to the lesions in these calves. By definition, toxoids are immunogenic but not pathogenic substances; however, the intravenous injection of toxoids and adjuvant of Tasvax 8 may have contributed to the pathologic changes observed in these three calves.
Ellis et al. 5 reported that 1 of 21 calves (30 days or younger) died 8–10 hours after intramuscular vaccination with a viral and bacterial combination vaccine (Virabos 4 + HS, Vetrepharm, London, Ontario) that was not recommended for use in calves younger than 3 months. The vaccine contained inactivated bovine viral diarrhea virus, parainfluenza-3 virus, bovine respiratory syncytial virus, an H. somnus bactrin, and an adjuvant. Pulmonary lesions in the calf that died were considered a cytokine-mediated injury induced by endotoxins or other bacterial components with potential synergistic enhancement by adjuvants. All pathologic pulmonary changes described in their case report were similar to the ones found in the calves of our study, except for periarteriolar/arterial hemorrhages, which were not reported in their calf. In addition, the onset of clinical signs and mortality was reported 6–8 hours later than in our calves. These differences may be related to the different administration routes of vaccines; namely, the bacterial components and adjuvants injected intravenously in the calves of our study had more rapid and probably more severe systemic and pulmonary effects than the effects induced by intramuscular injection.
Diagnosis of anaphylactic reaction to vaccine might be implied by the fact that the calves that survived were treated with epinephrine and steroids. Systemic anaphylaxis is a life-threatening type 1 hypersensitivity caused by a sudden release of potent vasoactive and proinflammatory mediators from mast cells and basophils. Anaphylaxis is initiated within minutes of exposure to a particular allergen to which an animal is presensitized. Ladiges et al. 7 sensitized calves with an intravenously injected antigen and then challenged them with the same antigen after 2 weeks to induce an experimental anaphylactic reaction. Severely affected calves collapsed within minutes after challenge and either recovered or died within 1 hour. Postmortem examination showed severely high proteinaceous pulmonary edema and emphysema accompanied by a variable number of intraalveolar macrophages. 7 The clinical incidence of systemic anaphylactic reaction in humans and animals is poorly documented, but in general, it is thought to be less than 1 per 10,000 animals 1, 8 . The severe pulmonary hemorrhages observed in all of the dead calves of our study were not described in experimental anaphylaxis. 7 In addition, it would be extremely unlikely that 19 of 75 calves would have an anaphylactic reaction unless they had all been previously sensitized to the vaccine, which was not the case. Anaphylaxis would also be expected to occur very rapidly following administration, whereas the calves described in this case report did not react for about 2 hours postadministration. Accordingly, we did not consider systemic anaphylaxis as a cause of death in the calves of our study, and the effects of treatment on the survival of the other vaccinated calves are arguable in this study.
The severe acute interstitial pneumonia with hemorrhages, recent jugular vein perforation with peri-jugular hemorrhage, and 4% mortality that occurred after vaccination were the basis for our conclusion that endotoxemia initiated by intravenous vaccination was the cause of death in the three calves that we examined postmortem. It cannot, however, be proven beyond a doubt that the vaccine was given intravenously without actually observing the vaccination procedure or demonstrating the adjuvant within pulmonary tissues, neither of which was done in these animals.
To improve the margin of safety and decrease undesired reactions, pharmaceutical companies provide specific guidelines for the administration of each product, including vaccines. However, even when vaccines are administered correctly, there is still the potential for an adverse reaction in a small proportion of the population. The number of adverse reactions in vaccinated animals can be greatly increased if the vaccine is not administered according to the manufacturer's instructions. Therefore, it is essential to educated producers to administer appropriately vaccines and to conform to the product specifications.
Footnotes
Acknowledgements
We thank the Department of Biochemistry and the Department of Experimental Pathology and Oncology, University of Florence, Italy, for their facilities and instruments used for image acquisition and processing. This manuscript has been prepared in the Uniform Requirements format.
