Abstract
We showed earlier that Tritrichomonas foetus-specific bovine immunoglobulin (Ig)G1 and IgA antibodies in uterine and vaginal secretions are correlated with clearance of this sexually transmitted infection. Eosinophils have been noted in previous studies of bovine trichomoniasis but the role of mast cells and IgE responses have not been reported. The hypothesis that IgE and mast cell degranulation play a role in clearance was tested in 25 virgin heifers inseminated experimentally and infected intravaginally with T. foetus strain D1 at estrus and cultured weekly. Groups were euthanatized at 3, 6, 9, or 12 weeks, when tissues were fixed and secretions were collected for culture and antibody analysis. Immunohistochemistry using a monoclonal antibody to a soluble lipophosphoglycan (LPG)-containing surface antigen (TF1.17) demonstrated antigen uptake by uterine epithelial cells. Lymphoid nodules were detected below antigen-positive epithelium. Little IgG2 antibody was detected but IgG1, IgA, IgM, and IgE T. foetus-specific antibodies increased in uterine secretions at weeks 6 and 9 after infection. This was inversely proportional to subepithelial mast cells numbers and most animals cleared the infection by the sampling time after the lowest mast cell count. Furthermore, soluble antigen was found in uterine epithelium above inductive sites (lymphoid nodules). Cross-linking of IgE on mast cells by antigen and perhaps LPG triggering appears to have resulted in degranulation. Released cytokines may account for production of predominantly Th2 (IgG1 and IgE) and IgA antibody responses, which are related to clearance of the infection.
Trichomoniasis is a human and bovine sexually transmitted infection, which results in adverse outcome of pregnancy. The bovine disease is caused by Tritrichomonas foetus, an extracellular parasite that colonizes the uterine, vaginal, and preputial epithelium of female and male cattle, respectively. It is most often found deep in uterine glands or preputial crypts, probably because it requires anaerobic conditions. The prevalence is high in beef-producing areas of the world or whenever natural breeding is used. For example, in a recent random survey in California, 16% of herds were infected. 4 Control has been primarily by culling infected animals because of the long-term nature of the infection. Chemotherapeutic agents such as the imidazoles are not allowed because of concerns about toxic residues in meat. Because the disease is of considerable economic importance, there has been much interest in immunoprophylaxis and immunotherapy. This has resulted in a commercially available vaccine 20, 21 and much information on protective immune responses. A major surface antigen, detected by monoclonal antibodies TF1.17 and TF1.15, 16 was found to contain a lipophosphoglycan (LPG) protein complex and to be released from the surface as soluble glycosylated antigen (SGA). 35 A related LPG-containing surface antigen has been described by Shaia et al. 32 We have shown that systemic or local (intranasal or intravaginal boosts after systemic priming) immunization with an immunoaffinity-purified protective surface antigen (TF1.17 antigen) induces a slight immunoglobulin (Ig)M–specific antibody response but primarily IgA and IgG1 responses in genital secretions and IgG1 antibodies in serum. 5, 9, 10 Almost no IgG2 antibodies specific for TF1.17 antigen were detected. In cattle, IgG1 responses are believed to be stimulated by Th2 cytokines and IgG2 by Th1 cytokines, 14, 15 so there seems to be strong bias for Th2 responses to this antigen. Furthermore, in infected but nonvaccinated controls, there is a strong local uterine and vaginal IgA and IgG1 response to TF1.17 antigen but little systemic response. 2, 9 This suggests that induction takes place locally. However, the induction of local immune responses in the female genital tract of any species is poorly understood. In our studies of virgin noninfected heifers, lymphoid follicles were not seen in the uterine mucosa but, in infected heifers, accumulations of immunocytes, lymphoid nodules, and follicles (some with germinal centers) were detected under the epithelium and adjacent to infected glands. 2, 9, 10 We concluded that uterine inductive sites develop after antigenic stimulation. Several questions arise. First, how is the formation of inductive sites initiated? Second, why is there a Th2 bias? Last, how does the IgG1 antibody cross into secretions because, unlike IgA, it does not have secretory piece to mediate transport across epithelial cells? Mast cells link the innate and acquired immune responses, 38 so they may be an important key to these puzzles. We addressed this hypothesis by studying the relationships between uterine mast cells, IgE- and IgG1-specific antibodies in uterine secretions of heifers experimentally infected for 3, 6, 9, or 12 weeks.
Materials and Methods
Infection
T. foetus strain D1 was obtained from a cow with trichomonad-induced pyometra and has been used in several experiments to infect heifers and to study immune responses. 5, 9, 10, 36 The organism was grown in modified Diamond TYM medium 11 with 10% calf serum and numbers of organisms were estimated by counting in a hemacytometer.
Virgin beef heifers were confirmed to be T. foetus negative by culturing vaginal secretions three times at weekly intervals as above. Then, 25 heifers were synchronized for estrus with 500 μg of synthetic prostaglandin F2α (Estrumate;™Bayer; Shawnee Mission, KS) intramuscularly at days −16 and −3. At day −9, 86 μg of synthetic gonadotrophin release hormone (Fertagyl™; Intervel, Millsboro, DE) was given. The first estrous behavior was observed at day −1. At day 0, all heifers were inseminated into the body of the uterus, using frozenthawed semen from a single ejaculate. Insemination was immediately followed by instillation of 106 T. foetus strain D1 into the cranial vagina (according to our standard procedures for infection). 5 Six heifers were euthanatized at weeks 3, 6, and 9 with seven heifers at week 12 after insemination/infection. Because the animals were in estrus at day 0 and the estrus cycle is approximately 21 days in cattle, nonconceiving animals in each group should have been at or near estrus at each time of euthanasia. This was critical because cyclic changes in mast cell numbers are known to occur. 23, 43 Assignment to groups was randomized at day 0. Two noninfected/noninseminated heifers and two inseminated heifers infected with Neospora caninum 3 served as controls. The latter animals were not pregnant at the time of euthanasia but were included to control for the possibility that the mast cell changes may be a nonspecific response to any protozoal infection of the female genital tract. Both sets of controls were synchronized and euthanatized at estrus. Weekly samples of cervicovaginal mucus were collected for culture of T. foetus. Cultures were read daily for 7 days. Just before euthanasia, a cotton tampon was placed in the cranial vagina so that vaginal fluids would not flow back into the uterus when the carcass was hung. Immediately after euthanasia, the entire reproductive tract was removed, placed in ice, and carried back to the laboratory (∼10 minutes). Secretions were aspirated from the vagina and flushed from the uterus with 5 ml of phosphate-buffered saline. An aliquot of each was cultured and the remainder frozen for antibody analysis (see below).
Pregnancy determination
Early pregnancy was monitored by enzyme immunoassay for plasma progesterone in samples collected twice weekly, up to day 24. Heifers whose progesterone patterns showed a sustained rise and remained elevated above 1.2 ng per ml at days 20–24 were presumed to be pregnant as of day 21. 6 Ultrasound examination at day 28 and transrectal palpation at day 35 were used to confirm pregnancy status. At euthanasia, after flushing of the uterine lumen, the extirpated uterus was opened along the entire length of both horns to search for a conceptus.
Histopathology
At necropsy, the genital tract was immersed in 10% buffered formalin for 48 hours before cutting in blocks for paraffin embedding and sectioning. Adjacent sections of inter-caruncular areas were stained with hematoxylin and eosin for general morphology or Giemsa stain for detection of metachromatic granules in mast cells. Although formalin fixation is not used for mast cell detection in rodents or humans, it was found to be as good as Carnoy fixative for mucosal or connective tissue mast cells of cattle. 46 Also, Chen et al. 8 observed more mast cells in the bovine lung fixed in neutral buffered formalin than in adjacent tissue fixed in isotonic formalin acetic acid as recommended by Enerback 13, 14 for rat mucosal mast cells. Mast cells were quantitated without knowledge of group assignment. Ten high-power fields (400×) for uterine surface epithelium (stratum compactum) and 10 for the glandular area (stratum spongiosum) were selected randomly. For each location, the mean of the total number of mast cells in 10 fields for each animal was reported. Inflammatory cell infiltrates were characterized according to severity. A mild infiltrate consisted of diffusely scattered inflammatory cells in the stratum compactum only. Moderate infiltration referred to a diffuse distribution of inflammatory cells in the stratum compactum and focal accumulations in the stratum spongiosum. Severe inflammation was defined as diffuse infiltration in the stratum compactum and multifocal to coalescing inflammatory infiltration in the stratum spongiosum. Infiltration of eosinophils was graded as 0, 1+, 2+, or 3+. In selected tissues, the eosinophil infiltration was further confirmed by Luna's method for staining eosinophil granules. 24
Immunohistochemical studies of formalin-fixed, paraffin-embedded tissues were done with TF1.15 mAb, which recognizes the same LPG-protein complex as TF1.17 mAb. Procedures for reaction with the mAb followed by peroxidase-labeled anti-mouse Ig were as reported previously. 29 Vaginal and uterine sections were chosen at random from three animals at each time point.
Antibody analysis
Uterine fluids were assayed for isotype-specific antibodies to TF1.17 antigen as described previously. 2 In brief, half-well microtiter plates (Costar, Cambridge, MA) were coated with immunoaffinity-purified TF1.17 antigen 5 at 3 μg of protein/ml in carbonate buffer, 50 μl per well. Alternatively, wells were coated with whole T. foetus cells at 105 cells per well as reported previously. 5 After blocking with 3% gelatin (Difco Laboratories, Detroit, MI) and washing, uterine flushes were diluted 1 : 20 for IgG1, IgG2, IgM, or IgA or 1 : 5 for IgE enzyme-linked immunosorbent assays (ELISAs). Samples were added in duplicate and incubated 90 minutes at 37° C. Wells were again washed, incubated with secondary murine monoclonal antibodies to bovine isotypes (see below) for 90 minutes at 37° C and washed. The reaction was detected with horseradish peroxidase–conjugated mouse anti-IgG, -IgM, and -IgA (Zymed Labs Inc., San Francisco, CA) for 30 minutes at 37° C, developed for 15 minutes in 3, 3′, 5, 5′ tetramethyl-Benzedrine™, plus hydrogen peroxide substrate (Kirkegaard and Perry Laboratories Inc., Gaithersberg, MD) and was stopped with 0.3 M phosphoric acid. Absorbances were read at A450 and A650. The average background readings (from wells with all reagents except primary antibody) were subtracted from all values.
Murine monoclonal antibodies were used for detection of bovine IgG1 (DAS 16), IgG2 (DAS 2), IgM (DAS 11), and IgA (DAS 7), at 1 : 8,000, 1 : 2,000, 1 : 4,000, and 1 : 8,000 respectively. All were gifts from Al Guidry (Beltsville, MD). Results with IgG2 are not presented in this study because the readings were essentially the same as the background (with antigen, but no primary antibody), as in our previous studies. 2, 9, 10, 17 The IgE levels in uterine secretions were detected with a murine monoclonal antibody to bovine IgE as reported previously. 30, 40 Because the previous study showed that assays to detect IgE in bovine serum were more sensitive after ammonium sulfate precipitation of other isotypes, we tested this possibility. Preliminary studies showed that 27.5–28% ammonium sulfate was optimal. Therefore, aliquots of all uterine secretions were precipitated with ammonium sulfate at a final concentration of 27.5% for 1 hour at room temperature, vortexing every 15 minutes. Precipitates were removed by centrifugation. IgE values for these aliquot supernatants were compared with nonprecipitated aliquots by ELISA against both whole T. foetus antigens and TF1.17 immunoaffinity-purified antigen. Overall, IgE antibody values dropped more after precipitation than those of other Ig classes. Therefore, values on nonprecipitated uterine secretions are reported.
Statistical analysis
Mast cell numbers in the uterine mucosa at weeks 3, 6, 9, and 12 were compared by analysis of variance with polynomial contrasts.
Results
Heifers gradually cleared the infection from both the uterus and the vagina, in that there were fewer culturally positive animals at 12 weeks than at 3, 6, or 9 weeks (Table 1). The lower percentage of heifers with positive uterine cultures, as compared with vaginal cultures, at week 3 is consistent with the concept that trichomonads take time to colonize the uterus after being deposited in the vagina. Pregnancy determinations by plasma progesterone concentrations at week 3 suggested that as many as nine heifers conceived, but concepti were found in only five heifers at euthanasia, including two from the group euthanatized at week 3, and one each in the 6, 9, and 12 week euthanasia groups. Ultrasound examination and transrectal palpation confirmed the three pregnancies that remained after euthanasia of the 3-week group. No heifers lost a known pregnancy between ultrasound and euthanasia. There were no lesions noted in the fetal membranes examined from the three concepti large enough to process. Uterine fluids appeared grossly normal in the pregnancies.
Tritrichomonas foetus culture results at necropsy of 25 T. foetus–infected heifers.

∗ Weeks after infection at euthanasia and necropsy.
Histopathologic examination of the vagina showed diffuse variably intense suppurative and lymphoplasmacytic mucosal and submucosal inflammation, with moderate exocytosis and occasionally dense eosinophilic infiltrates. Some cows had prominent lymphocytic aggregates in the vaginal submucosa. Lesions in the uterus consisted of variable degrees of suppurative and eosinophilic endometritis with a diffuse lymphoplasmacytic infiltrate, mast cells, and mild eosinophilic infiltration of the myometrium. Lymphoid aggregates were evident in the periglandular areas of the inter-caruclular endometrium after week 6. There was relatively little inflammation in the endometrium at 3 weeks after infection, but at 6 and 9 weeks after infection, the majority of heifers had moderate to severe inflammation. At 12 weeks, most animals had mild to moderate endometrial inflammation. Mast cell numbers were quantitated more precisely (Fig. 1) because of their role in linking innate and specific immune responses. Separate values are given for superficial mast cells, in the stratum compactum just under the surface epithelium, and those surrounding the glands, in the stratum spongiosum (Fig. 2a, b). Mast cell populations were similar in noninfected controls, N. caninium–infected controls, and in animals at 3 weeks after infection with T. foetus. At 6 weeks after T. foetus infection, numbers of mast cells fell in both superficial and glandular areas with lowest numbers at week 9 and increasing values at week 12 (Fig. 1). Loss of detectability is presumably because of degranulation and thus lack of staining of metachromatic granules. Statistical analysis showed that the mast cell numbers at weeks 3, 6, 9, and 12 were curvilinear. This was stronger for the superficial mast cells (cubic, P = 0.045) than for glandular mast cells (quadratic, P = 0.004). It should be noted that the lowest percentage of animals with positive uterine cultures (Table 1) was at the next sampling time after lowest numbers of granulated mast cells were detected in the uterine mucosa (Fig. 1). Unlike mast cells stained for metachromatic granules, there was very little eosinophil infiltration at 3 weeks but moderate to severe infiltration was detected by 6 weeks (Table 2). The peak number of eosinophils was detected mainly under the uterine surface epithelium and adjacent to glands in the stratum compactum (Figs. 3, 4). Luna-stained sections confirmed the presence of eosinophils in 6-week sections and differentiated them clearly from neutrophils that were also present in variable numbers (Fig. 5). Although both mast cells and eosinophils numbers are known to vary with the stage of the estrus cycle, 23, 27 this is not thought to be important in our studies because heifers were synchronized to be in estrus at time 0. Sampling periods thereafter were at 3-week intervals, so nonconceiving heifers should have been at, or close to, estrus at each sampling period.
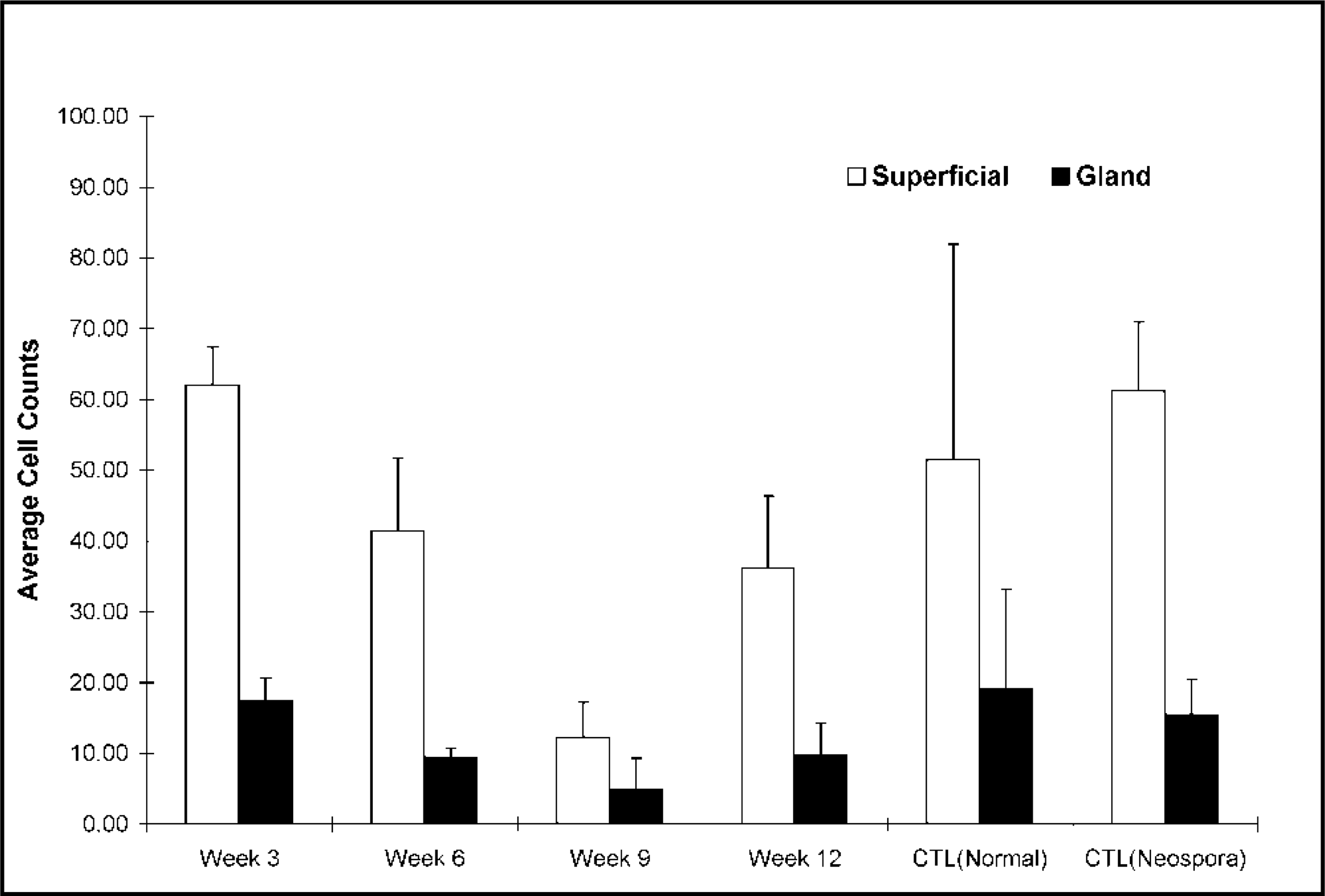
Mast cell counts under the uterine superficial and glandular epithelium in heifers euthanatized 3, 6, 9, or 12 weeks after infection with T. foetus. Mast cells were detected with Giemsa stain for metachromatic granules.
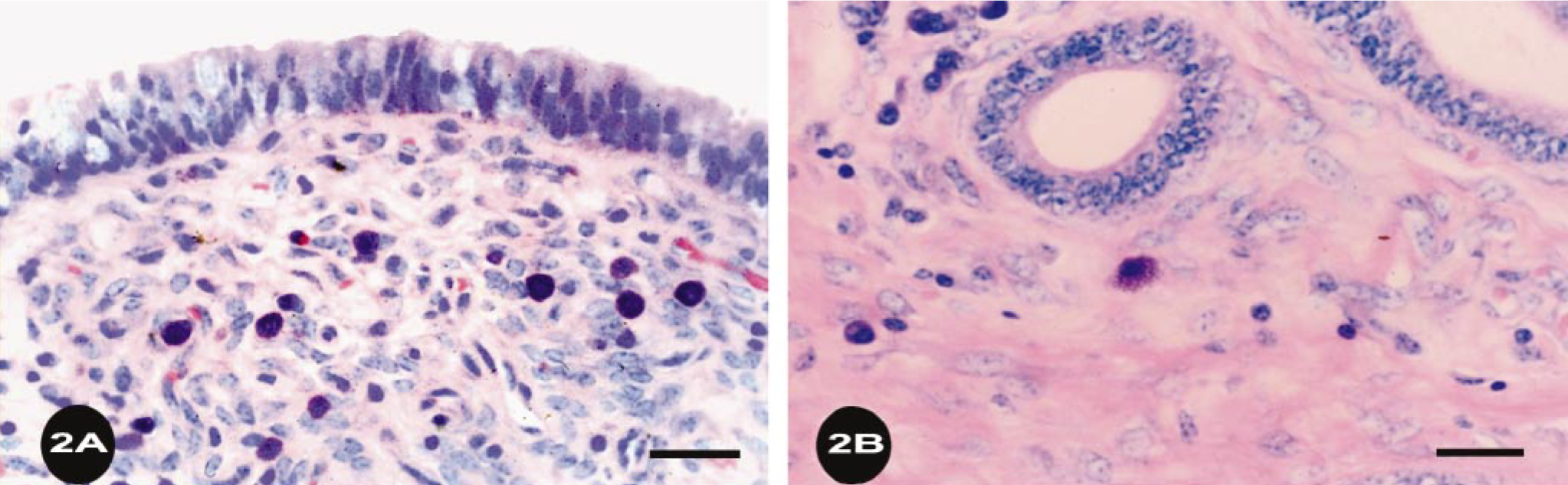
Endometrium; bovine, heifer No. 95, experimentally infected with T. foetus for 6 weeks. Note mast cells with metachromatic granules under the (
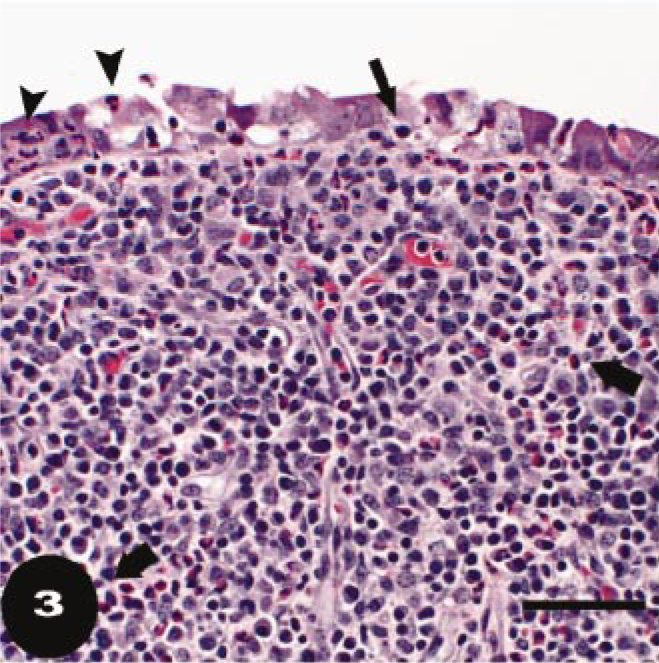
Endometrium; bovine, heifer No. 95, infected with T. foetus for 6 weeks. Inflammatory infiltrates, including numerous eosinophils (thick arrows), lymphocytes, and plasma cells, expand the stratum compactum and extend into the deeper endometrium. Eosinophils are abundant immediately beneath the mucosa and adjacent to glands. Neutrophils (arrowheads) and lymphocytes (thin arrow) are detected within the mucosal epithelium. HE. Bar = 500 μm.
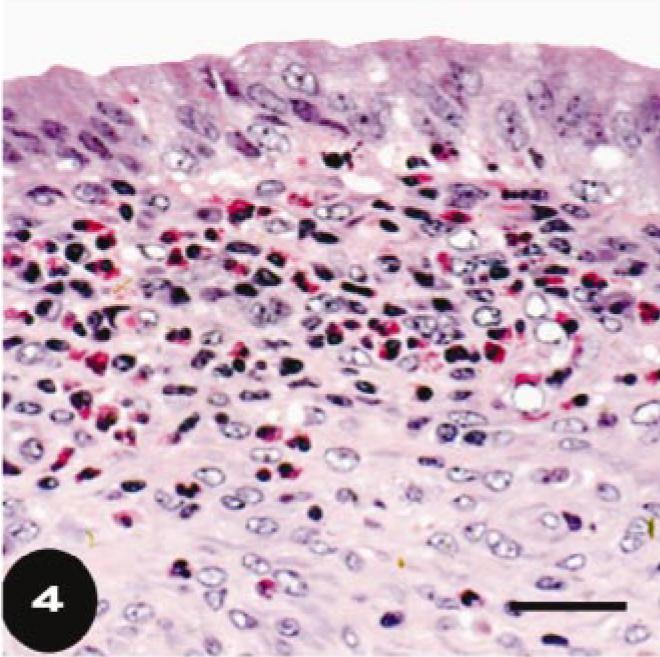
Endometrium; bovine, heifer No. 95, infected with T. foetus for 6 weeks. In many areas, focal concentrations of eosinophils under the surface epithelium dominate the inflammatory infiltrate. HE. Bar = 150 μm.
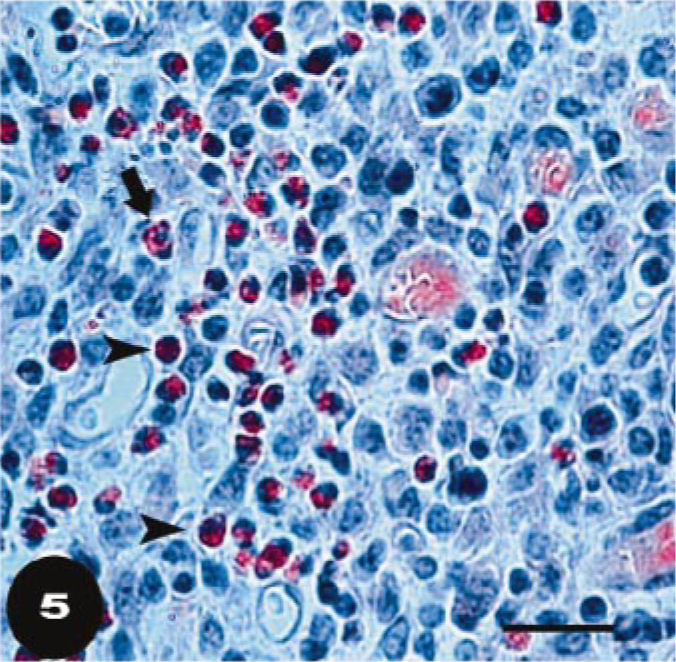
Endometrium; bovine, heifer No. 95, infected with T. foetus for 6 weeks. Many eosinophils (arrowheads) are present in the stratum compactum. Note the relatively pale pink staining cytoplasm and multilobulated nucleus that distinguishes neutrophils (arrow) within the mucosa of an endometrial gland from neighboring eosinophils (arrow head). Luna's stain. Bar = 100 μm.
Semiquantitation of eosinophils in uterine infiltrates.

∗ Weeks after infection at euthanasia and necropsy.
Immunohistopathologic studies demonstrated that the uterine epithelium (Fig. 6a, b) contained the TF1.17 LPG/peptide antigen because cells stained for the antigen from the apical to the basal membrane. Staining was diffuse or finely punctuate, not indicative of cross sections of whole T. foetus. Thus, staining appeared to be because of uptake of SGA. Mononuclear cells accumulated below this antigen-positive epithelium (Fig. 6). In the uterus, some glands contained trichomonads and others did not, as is usual. Similarly, antigen was detected in only some glands, suggesting that only epithelial cells in infected glands pinocytosed shed antigen (Fig. 6a). A focal accumulation of lymphocyte-like mononuclear cells was detected around those glands positive for antigen. Some cells with typical macrophage or dendritic cell morphology stained heavily for TF1.17 antigen (Fig. 6b). These putative antigen-presenting cells were found below the basement membrane of the surface and the glandular epithelium, which contained the antigen.
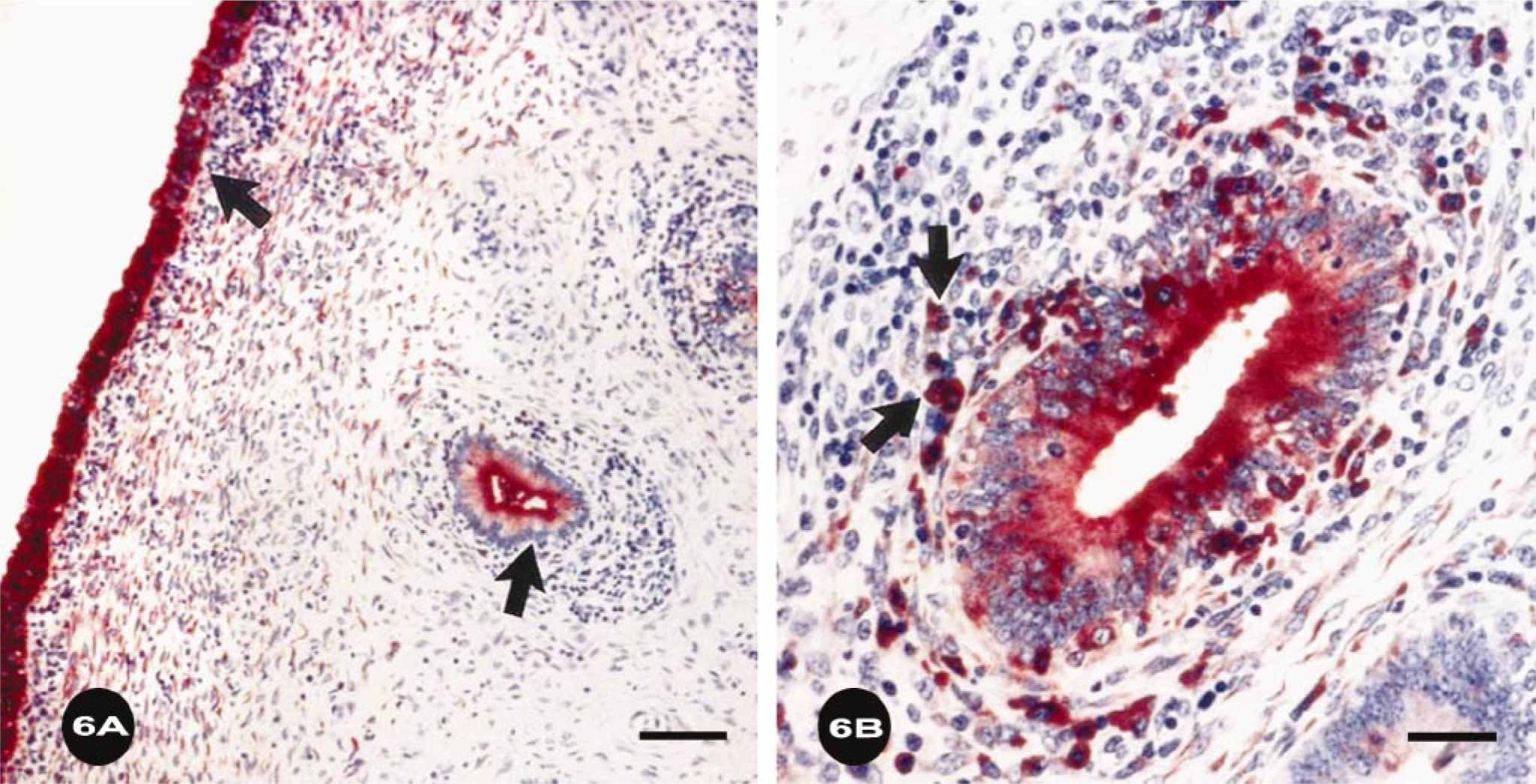
Endometrium; bovine, heifer No. 86, infected with T. foetus for 12 weeks. T. foetus soluble LPG antigen (SGA) was detected immunohistochemically with TF1.15 mAb. (
Specific antibody analyses were quite revealing. IgG2 data is not shown because there was essentially no IgG2 response at any time point, as in our previous studies. 5, 9 With the other four Ig classes, there was essentially no antibody to either whole cell or the surface antigen (TF1.17) at 3 weeks after inoculation, again suggesting that the uterine infection was just getting started 3 weeks after vaginal inoculation (Fig. 7). Also, it is clear that, for all Ig classes of antibody, the response to whole cells is greater than that to the single purified antigen (TF1.17). This is to be expected because a polyclonal response to infection should recognize more than one antigen of the protozoan. In comparing graphs between isotypes, it should be noted that the absorbance scales are different. Also, the sensitivity and dilution of the monoclonal second antibodies differ for each isotype. Therefore, absorbances should not be compared from one isotype to another but kinetic patterns of responses may be compared. Thus, for all isotypes, responses peak at 6 to 9 weeks and decrease by 12 weeks after infection (Fig. 7). Importantly, the peak antibody response corresponds to the time of fewest detectable mast cells (Fig. 1) and precedes clearance of infection in most animals.
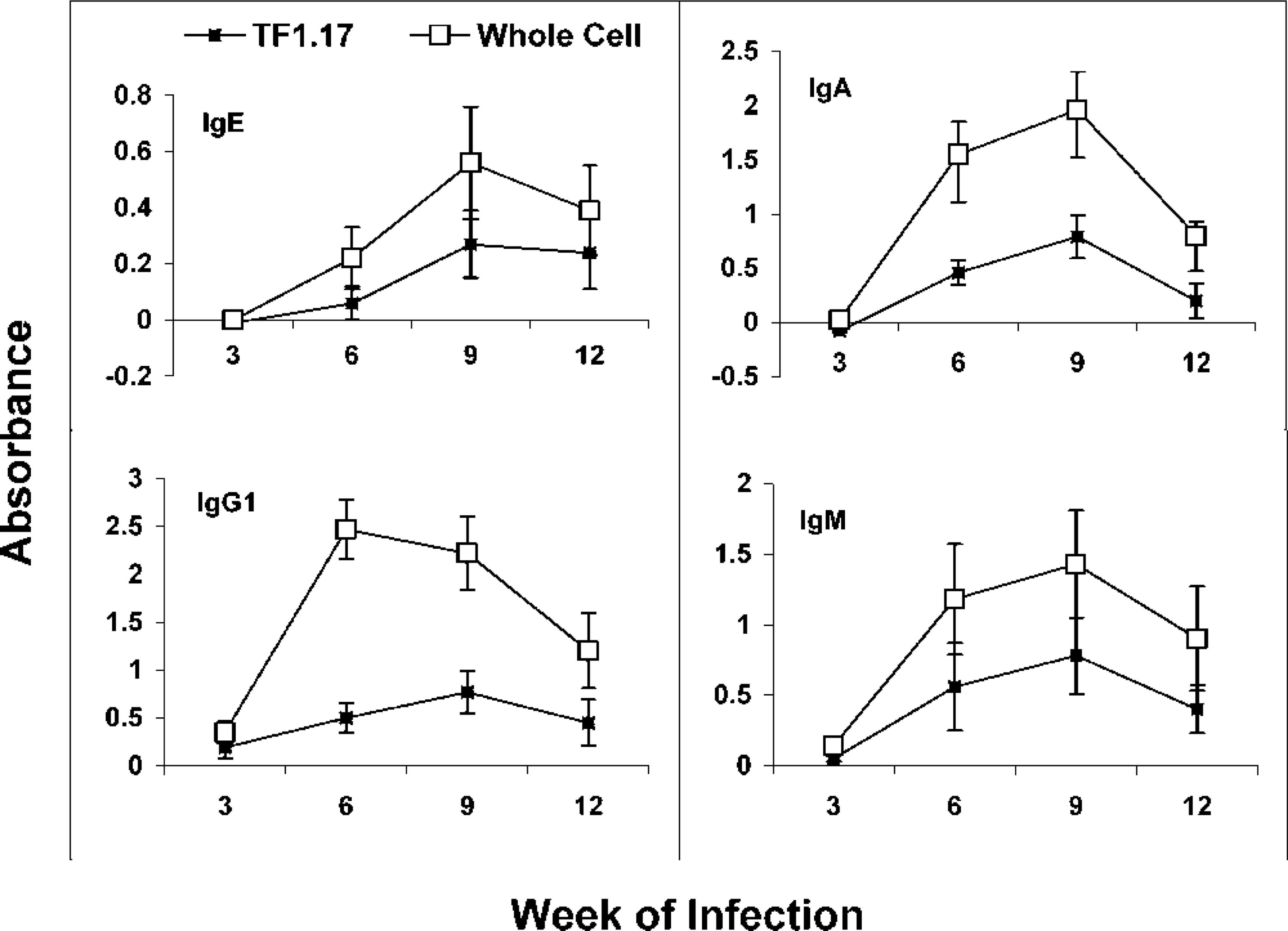
Isotype-specific ELISA of antibodies in uterine secretions of heifers infected 3, 6, 9, or 12 weeks. TF1.17 antigen (TF1.17) or whole T. foetus cells (whole cell) antigen was on the solid phase.
Discussion
The kinetics of isotype-specific antibody responses, mast cell degranulation and clearance of infection demonstrate that defense of the uterus is related to increasing antibody levels and decreasing detectable subepithelial mast cells. Both pregnant and nonpregnant animals maintained the infection. The apparent attrition of pregnancy in this study was considerably more than the estimated embryonic death after maternal recognition of pregnancy in cattle of 15–20%, in absence of infectious agents. 33, 37 Perhaps the higher rate in this study was because of trichomoniasis. The relationships between IgA/IgG1 antibodies specific for T. foetus 5, 36 or TF1.17 antigen 2, 9, 10 and clearance of infection have been demonstrated previously. Although eosinophils have been noted previously in the subepithelial inflammatory infiltrates in uterine trichomoniasis, 10 the association with uterine IgE responses has not been reported in trichomoniasis, to our knowledge. Similarly, mast cell and IgE antibodies specific for T. foetus or TF 1.17 antigen have not been reported before. Also, the mechanism of IgG1 transport to the uterine secretions should be addressed. We propose that IgE cross-linking by soluble T. foetus antigen stimulated release of mediators by mast cells in the uterine mucosa. This may alter the integrity of the uterine epithelium as has been suggested for the gastrointestinal tract, 22 thus facilitating IgG1 transport to secretions and clearance of infection.
Mast cells from different mucosal surfaces have unique characteristics. 18 Human uterine mast cells respond strongly to anti-IgE in vitro 26, 41 and it has been suggested that they may contribute to embryo implantation. The mouse uterine mast cell has been shown to evoke uterine contractility in vitro 31 which could contribute to reproductive failure if massive degranulation (as in this study of bovine trichomoniasis) occurs early in gestation. Equine uterine mast cells appear to be of two subtypes, tryptase positive/chymase negative or both tryptase and chymase negative. 43 Little is known about bovine uterine mast cells but it may be assumed that they secrete some of the usual mast cell mediators, such as histamine, leukotrienes, prostaglandins, heparin, and proteinases as well as proinflammatory cytokines and Th2 related cytokines. Many of these mediators may be involved in the secretory immune response. For example, tryptase is known to activate matrix metalloproteinases, which degrade matrix to alter mucosal epithelial integrity. 22 This plus the effect of mast cell mediators on vascular permeability and mucosal secretion 1 may account for the increases in IgG1 antibody in uterine secretions as mast cells degranulate at 6 and 9 weeks of infection. Proinflammatory cytokines may also contribute to damage of the epithelium with IgG leakage. Certainly, in our studies, inflammatory cell infiltration was noted to increase at 6 and 9 weeks of infection and to slightly decrease at 12 weeks when detectable granulated mast cell numbers again increased.
The propensity of mast cells to produce Th2 type and inflammatory mediators may be responsible for the moderate to severe eosinophil infiltration in some infected animals by 6 weeks after infection. Others have shown that interleukin (IL)-4 and tumor necrosis factor α cause 10- to 20-fold increases in release of eotaxin. 28 Caron et al. 7 showed that histamine release polarizes dendritic cells to prime for Th2 responses. The production of Th2 cytokines may also account, in part at least, for the IgE and IgG1 antibody responses with very little IgG2 response to TF1.17 antigen. We noted the skewed IgG1 over IgG2 response in T. foetus–infected cattle in the past 2, 9, 10 but had no explanation. Because bovine IgG1 is characteristic of a bovine Th2-type response, 14, 15 it is likely that mast cell release of IL-4 plays a role.
Mast cell mediators may be released because of antigen/IgE cross-linking of IgE receptors or by non–receptor-mediated mechanisms. 1, 39 Soluble lipopolysaccharide (LPS) from gram-negative bacteria is well known as a non–receptor related mast cell activator. 39 The T. foetus surface glycolipid, LPG, is somewhat similar to LPS and is a major surface antigen, with about 106 molecules per cell. 34 This is a component of the TF1.17 antigen and the complex is released from the cell surface in soluble form as SGA. 35 The presence of SGA in uterine epithelial cells and some cells below the basement membrane is similar to that detected in preputial epithelium of the bull. 29 The fact that epithelial cells in the female rat genital tract 44, 45 and macrophage/dendritic cells can present antigen to T cells probably accounts for stimulation of the local immune response in lymphoid nodules under the surface and glandular uterine epithelium in infected animals. 2, 9 Furthermore, if LPG has a similar effect to LPS in stimulating mast cells, it would explain some of the early leakage of IgG1 into uterine secretions. Because LPS priming also results in increases in eosinophils, 25 LPG may be involved in the uterine eosinophil accumulations reported in this study. Last, enzymatic degradation products of fibrinogen and fibronectin are known to activate mast cells. 1 Earlier, we showed that T. foetus expresses extracellular cysteine proteinase 19 (TFECP) in vivo and that TFECP cleaves both bovine fibrinogen and fibronectin. 42 These cleavage fragments may very well contribute to mediator release and passive transport of IgG1. Later in infection, the IgE response would play a role in mast cell degranulation and mediator release. This is consistent with the lowest number of detectable mast cells at week 9 when IgE levels of antibody to T. foetus were highest. IgE antibodies may also play a unique role in induction of endometrial immune responses. In mice with induced airway allergy, IgE immune complexes in respiratory secretions induced higher levels of IL-4 and eosinophils than controls. 47 It was proposed that antigen-specific IgE captured airborne antigens to form immune complexes, which potentiate immune responses. We have shown that T. foetus releases soluble LPG/protein antigen (TF1.17 antigen or SGA) in vitro. 35 The immunohistochemical studies reported here indicate that this antigen is present in epithelial cells and large mononuclear cells in vivo. Also, we showed that IgE antibodies specific for TF1.17 antigen are present in uterine secretions during infection. Similar to the mouse allergy model, 47 a Th2-type response resulted, perhaps because of IgE immune complexes.
As far as we know, this is the first report of uterine IgE responses to pathogens in cattle and perhaps other species. In this study, IgE antibody responses to the dominant surface and secreted antigen of T. foetus were associated with mast cell degranulation and clearance of infection.
Footnotes
Acknowledgements
We thank Karen Arnold, John Eddow, and Karen van Hoosear for technical assistance. We also thank Dr. Robert R. Corbeil, Emeritus Professor of Math and Computer Science, University of San Diego, for statistical analysis and Dr. Al Guidry, Beltsville MA, for the gift of monoclonal antibodies. This work was funded, in part, by USDA NRI grant numbers 97-35204-4771, 98-35204-6401 and 2001-35204-10803.
1Patologia Veterinaria, INTA, Balcarce, Argentina.
