Abstract
A 2-year-old, castrated, male European Shorthair cat was presented with inappetence, vomiting, and fever. Ascites and an irregularly nodular liver were observed on laparotomy; in view of the poor prognosis, the animal was euthanatized. Histopathologic examination of the liver showed a chronic hepatitis with cirrhosis associated with massive accumulation of copper in hepatocytes and macrophages, particularly in the fibrotic areas between the regenerative nodules. Electron microscopy and energy-dispersive X-ray analysis revealed lysosomal accumulation of copper in the hepatocytes. Analytical determination showed very high copper concentrations in the liver. No evidence for increased dietary or environmental copper could be found. This and the pattern of hepatic copper storage, the associated inflammation and fibrosis, strongly suggest a primary copper storage disorder.
Hepatic copper accumulation resulting in necrosis and inflammation of the liver is well known in dogs. It occurs as a familial, inherited disorder in the Bedlington Terrier, 8 West Highland White Terrier, Skye Terrier, 3 and the Dalmatian. 9 In these animals, copper accumulates in the hepatocytes, starting in the centrolobular regions, and with progressive accumulation results in hepatocellular necrosis, inflammation with copper-laden macrophages, and, finally, in chronic hepatitis and cirrhosis. Dogs of other breeds, such as Doberman Pinschers, 6 Labrador Retrievers, and American and English Cocker Spaniels, have been reported to have elevated copper concentrations in association with chronic hepatitis, but it remains to be determined whether this copper accumulation is primary or secondary to chronic inflammation, fibrosis, and cholestasis. Healthy dogs with normal livers may have copper levels up to 500 µg/g dry weight (dw). Hepatic copper levels in breeds with primary copper storage disease vary between the individual animals and between breeds. Bedlington Terriers, West Highland White Terriers, and Dalmatians with copper-associated liver damage reportedly have liver copper concentrations greater than 2,000 µg/g dw. In the other breeds, copper levels vary but usually range from 800 to 2,200 µg/g dw (World Small Animal Veterinary Association [WSAVA]: Liver Diseases and Pathology Standardization Research Group, T. S. G. A. M. van den Ingh, personal communication). The underlying genetic defect has been unraveled only in the Bedlington Terrier and consists of a deletion in the MURR1 gene causing absence of the MURR1 protein. 5 Its function is not yet known, but this protein is associated with a vesicular intracytoplasmic compartment in hepatocytes and many other cells. 5
Extensive hepatic copper accumulation has been reported only once in a Siamese cat. 2 The animal showed marked centrolobular copper accumulation associated with hepato-cellular degeneration, hepatic necrosis, and inflammation. In this case, the liver copper value was 4,074 µg/g dw, whereas normal hepatic copper levels are 148–180 µg/g dw. 2 Other authors have observed slight, probably secondary copper accumulation in the periportal hepatocytes of cat livers with chronic nonsuppurative cholangiohepatitis. 1
The purpose of this report is to describe severe hepatic copper accumulation in a cat, associated with chronic hepatitis and cirrhosis.
A castrated, male, European Shorthair was presented at 2 years of age with inappetence, vomiting, and fever. The animal had been retrieved from an animal shelter at the age of 6 months. The history of the animal had been uneventful, except that it had fallen into a pond near the owner's house a year before, followed by an episode of fever, which had resolved on antibiotic therapy. At that point of time, the animal was tested negative for feline leukemia virus and feline immunovirus. The owners reported that at least after the accident, but probably already before, the animal was sleeping a lot and did not play much.
During explorative laparotomy, the animal showed marked ascites consisting of a clear watery fluid and an irregular, nodular liver. Because of the suspected poor prognosis, the animal was euthanatized. A wedge biopsy of 1 cm3 size was taken and fixated in 10% neutral-buffered formalin for histopathologic examination. Macroscopically, the fixated liver biopsy was brown-yellowish and had an irregular surface with multiple, pale, often coalescing small nodules up to 3–4 mm in diameter. On histopathologic examination, the liver had an abnormal architecture characterized by multiple parenchymal hyperplastic nodules of varying size separated by broad bands of fibrous tissue (Fig. 1). These fibrous septa showed marked biliary proliferation, individual and small groups of heavily gray-brown–pigmented hepatocytes, gray-brown–pigmented macrophages, and a moderate inflammatory infiltrate of plasma cells, lymphocytes, and some small groups of neutrophils (Fig. 2). Heavily pigmented hepatocytes regularly showed individual cell necrosis characterized by cytoplasmic condensation and nuclear pycnosis (Fig. 3). The parenchymal nodules had an abnormal structure and consisted of pale, glycogen-rich hepatocytes with solitary cell necrosis as well as a slight mixed inflammation. The rubeanic acid stain for copper revealed very high quantities of copper in the fibrous septa within macrophages (Fig. 4). A slight to moderate amount of copper was present in the parenchymal nodules and was mainly observed in hepatocytes in the still recognizable centrolobular areas (Fig. 5). Quantitative copper analysis was performed on a piece of formalin-fixed liver and revealed a copper concentration of 4,170 µg/g dw. As a control, hepatic copper levels in fresh liver tissue of three young (2-year-old) and two old (14- and 18-year-old) cats with normal livers ranged within 26–174 µg/g dw (absolute values 86, 96, 174, 26, 120 µg/g dw, respectively).
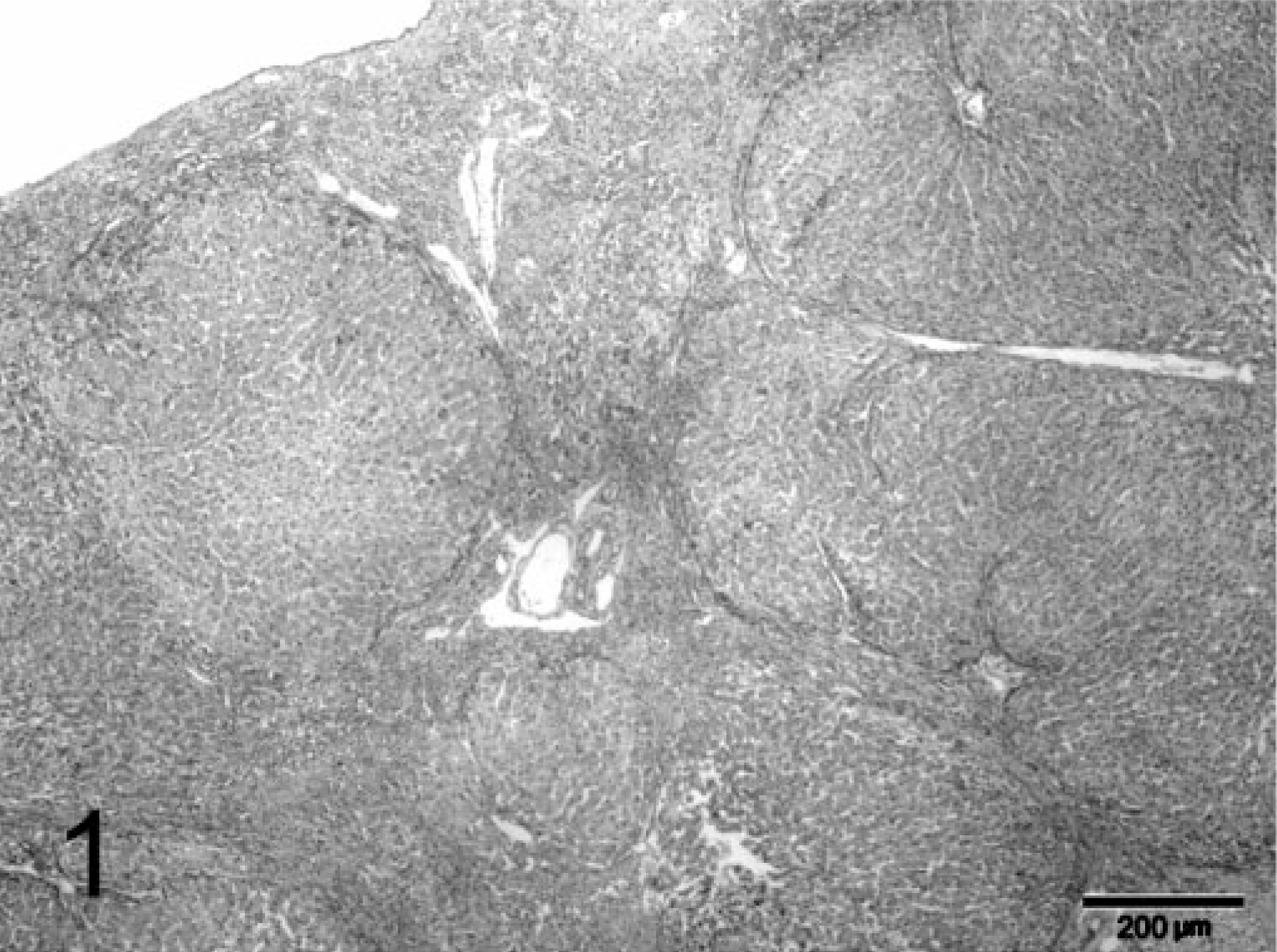
Liver; feline. Cirrhotic liver with multiple parenchymal hyperplastic nodules separated by fibrotic bands. HE stain. Bar = 200 µm.
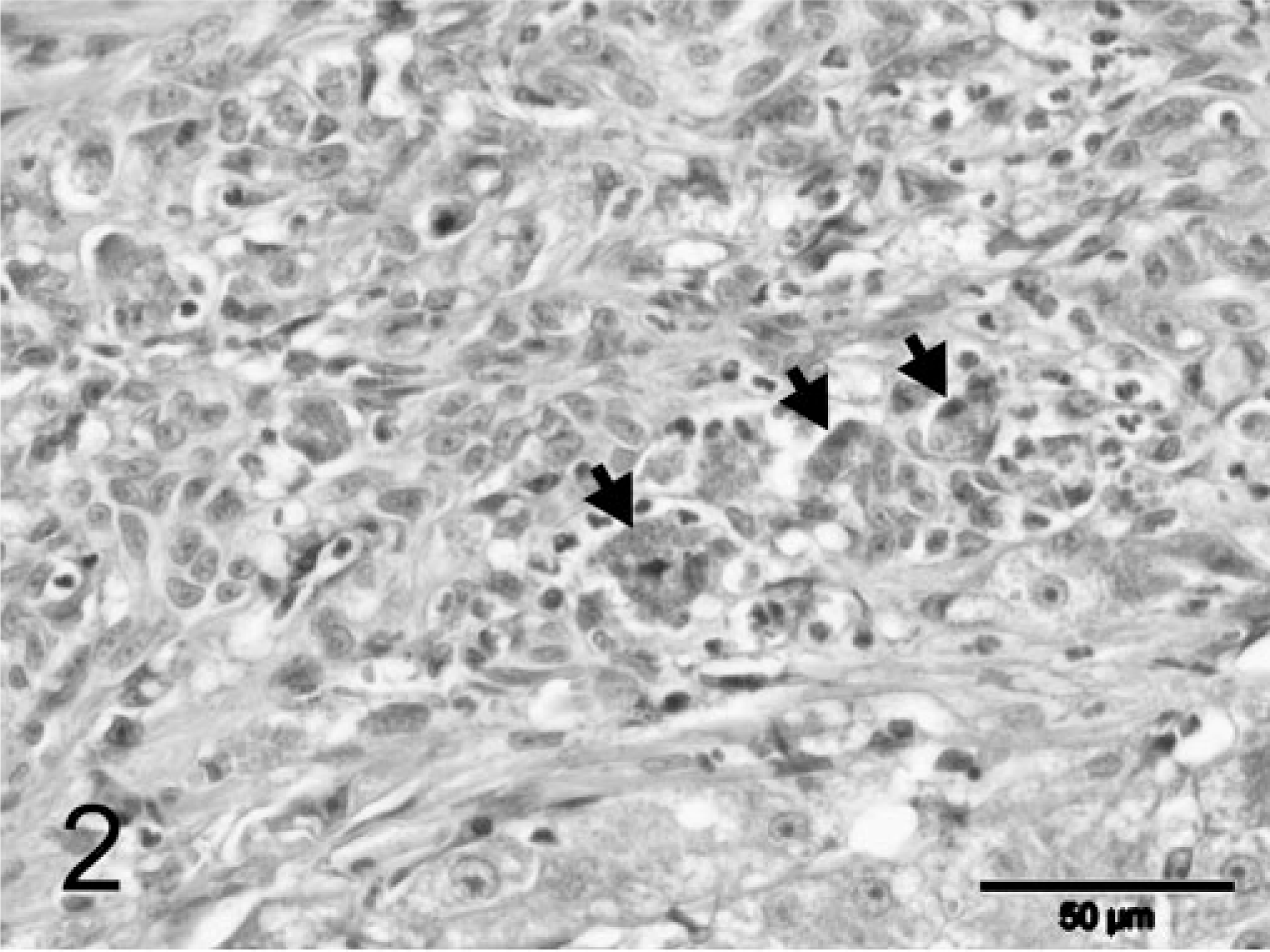
Liver; feline. Fibrous septa with marked biliary proliferation, abundant gray-brown–pigmented macrophages (arrows), and a slight inflammatory infiltrate. HE stain. Bar = 50 µm.
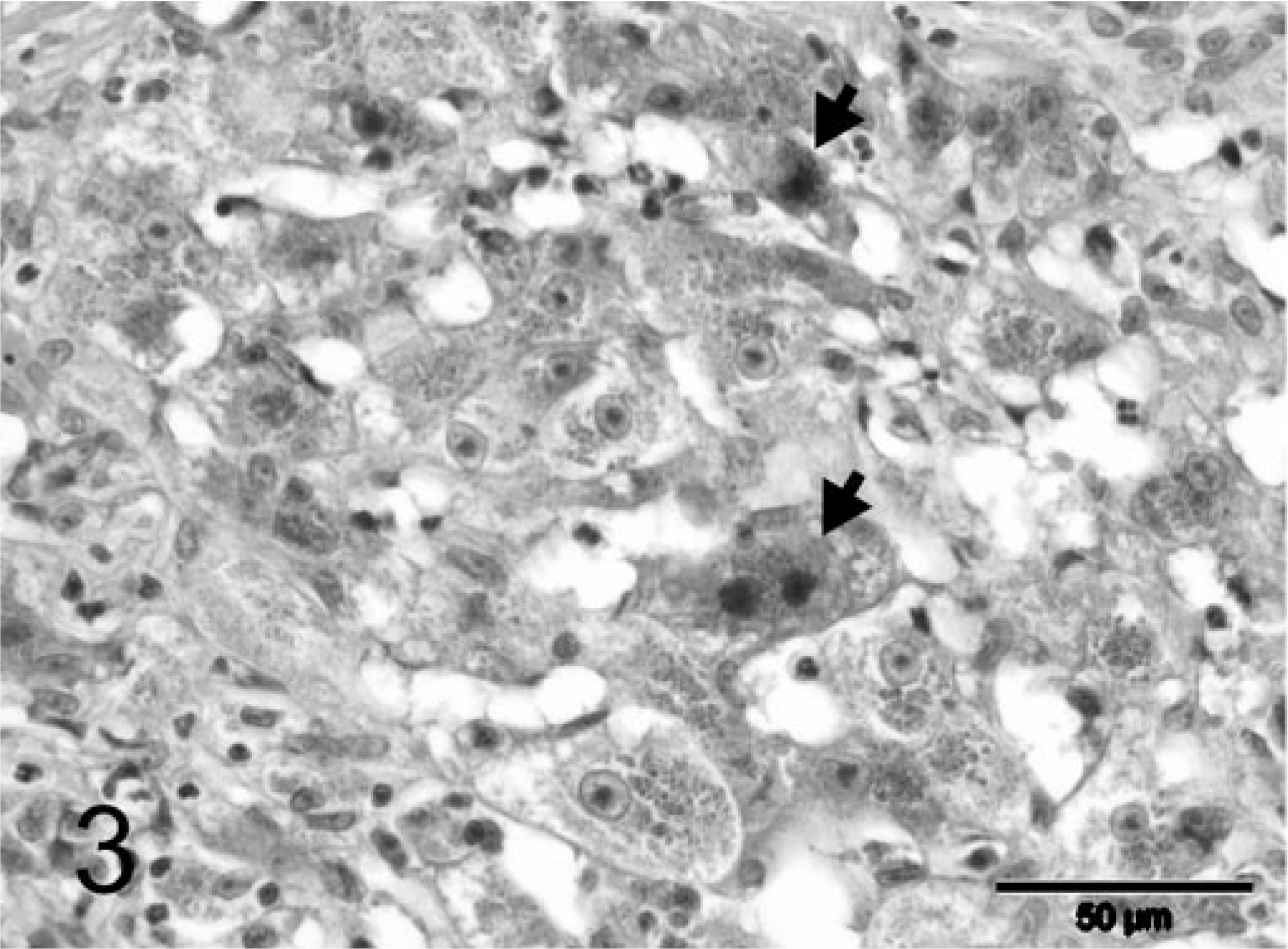
Liver; feline. Heavily pigmented hepatocytes, sometimes with condensation of the cytoplasm, and nuclear pycnosis (arrows). HE stain. Bar = 50 µm.
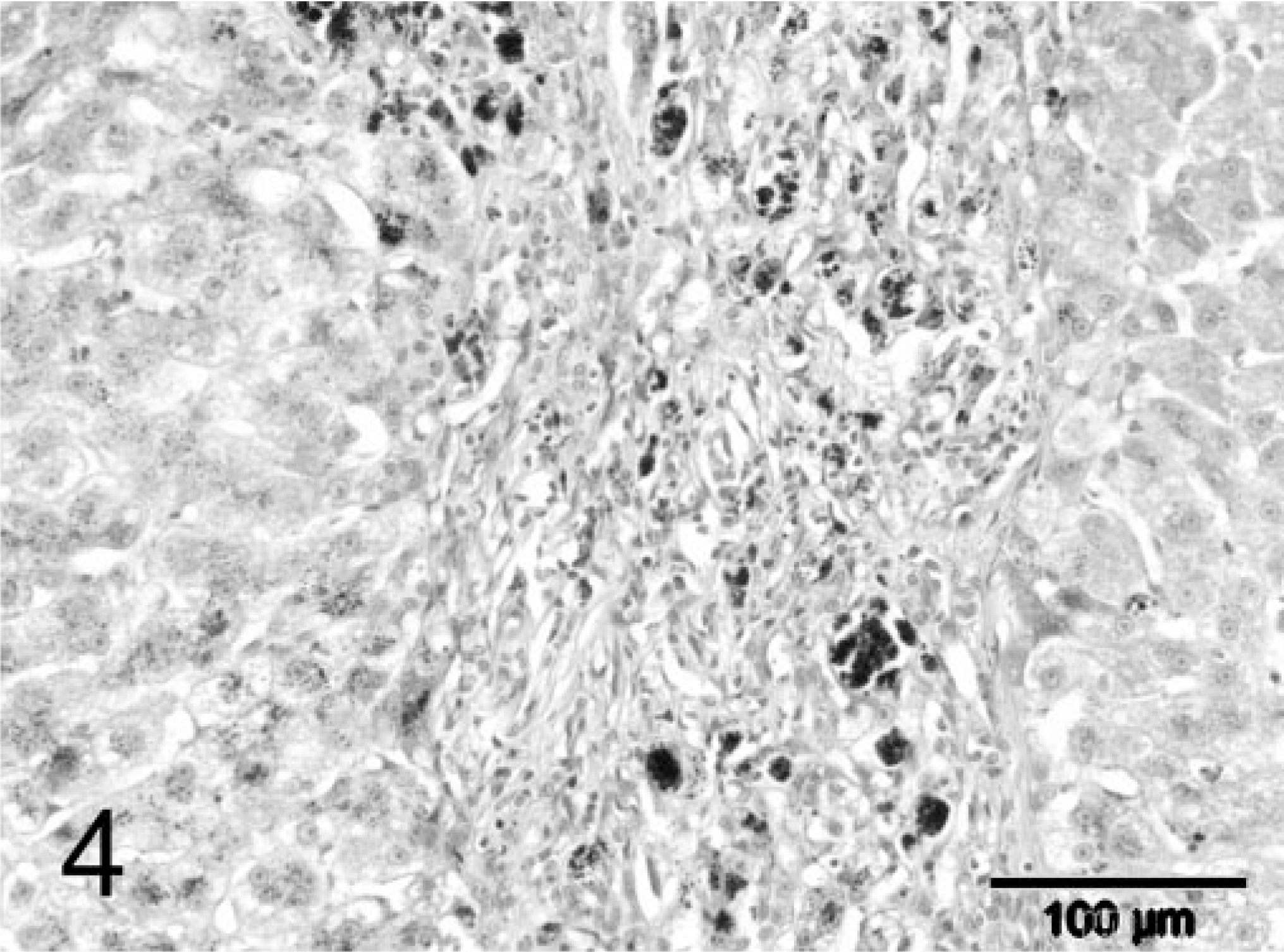
Liver; feline. Fibrous septum with large amounts of copper pigment in macrophages. Rubeanic acid stain. Bar = 100 µm.
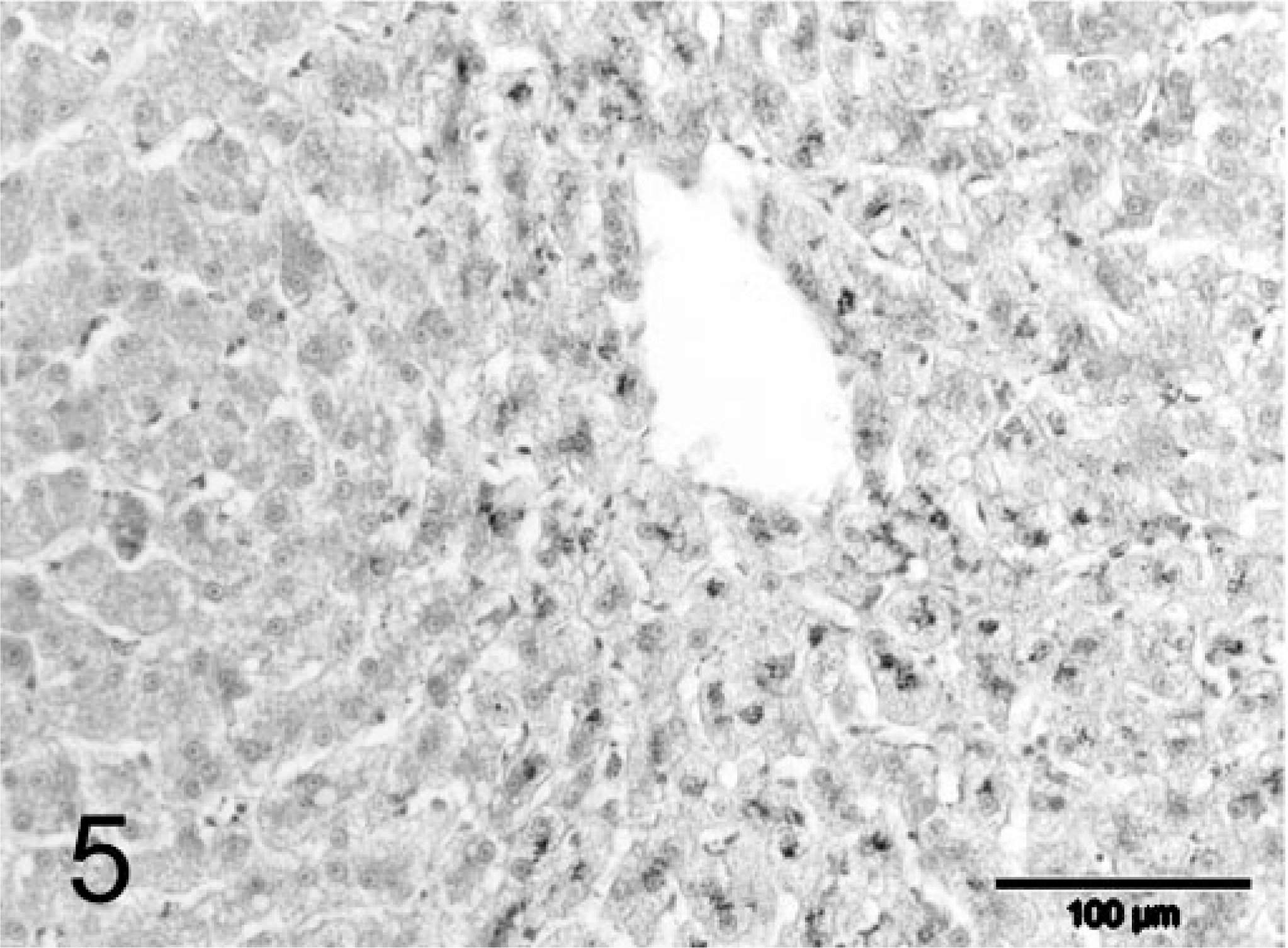
Liver; feline. Large parenchymal regenerative nodule with a moderate amount of copper in centrolobular hepatocytes. Rubeanic acid stain. Bar = 100 µm.
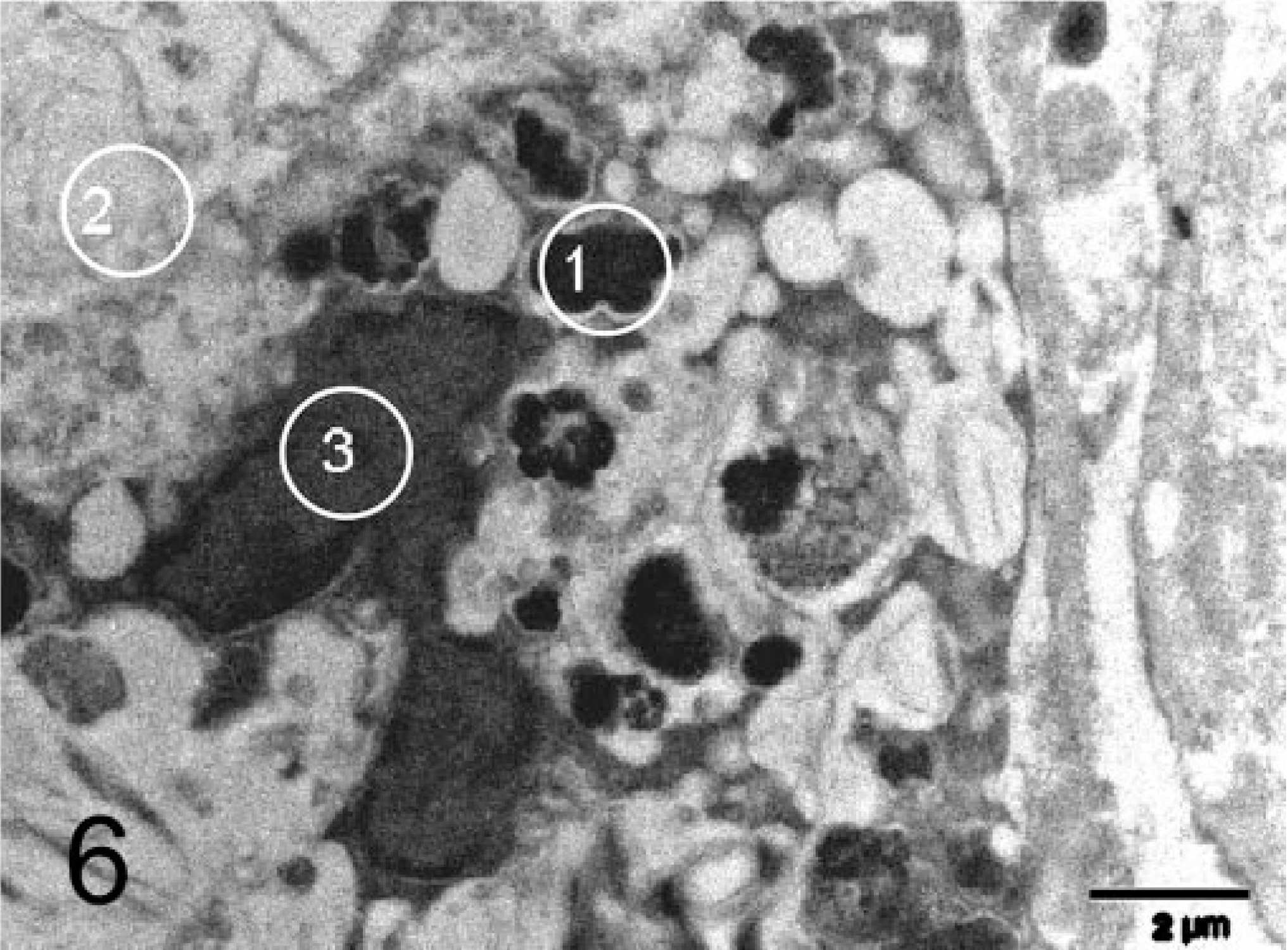
Liver; feline. Macrophage with multiple electron-dense aggregates. Electron microscopic energy-dispersive X-ray analysis was performed on (1) electron-dense intracytoplasmic aggregate, (2) nucleus, (3) normal cytoplasm. EM. Bar = 2 µm.
Liver tissue was transferred to Karnovsky's solution and routinely processed for electron microscopy. Large, electron-dense aggregates up to 1.5 µm, probably lysosomes, were seen in hepatocytes and macrophages in the areas of fibrosis (Fig. 6). Energy-dispersive X-ray analysis of these aggregates proved them to consist mainly of copper and sulfur. The sulfur peaks were higher than the copper peaks, suggesting a ratio of copper to sulfur atoms of about 1 : 4 to 1 : 7. Semiquantitative analysis of all identified peaks revealed significantly higher levels of both copper and sulfur in these aggregates (copper 0.499–0.875%, sulfur 3.65–4.33%) as compared with other cytoplasmic densities in the same cells (copper 0%, sulfur 0.039%) (Fig. 6) and in the cytoplasm of expected copper-free cells such as fibroblasts (copper 0%, sulfur 0.086%).
Hepatopathy associated with excessive hepatocellular copper has been described only once in a 2-year-old cat, 2 but the gross and microscopical findings were very different from those presented in this study. The liver of that cat showed a dark yellow aspect on gross examination and hepatocellular degeneration and necrosis on histopathologic examination. These findings suggest an acute disease course associated with increased hepatic copper levels. In our cat, the cirrhotic liver is clearly associated with chronic liver disease, consistent with the clinical history of sleepiness and apathy for a period of at least 1 year. The presence of massive copper accumulation in hepatocytes in the fibrous bands and the associated hepatocellular necrosis, large numbers of copper-laden macrophages, biliary proliferation and inflammation suggest a primary copper storage disease in our cat. Also, the presence of copper in the centrolobular areas of the parenchymal hyperplastic nodules supports an ongoing, probably primary, copper storage disease.
Reported normal hepatic copper levels in cats are 148–180 µ/g dw according to Haynes and Wade 2 and 100–150 µg/g dw according to Thornburg. 7 We found comparable ranges in our five control cats. The hepatic copper concentration of our cat (4,170 µg/g dry weight) was therefore more than 20-fold increased. The presence of regenerative parenchymal nodules containing only small amounts of copper indicates even higher copper concentrations in the fibrotic bands.
Electron microscopy showed electron-dense aggregates, probably lysosomes, in hepatocytes and macrophages. They are comparable with those described in Bedlington Terriers with inherited copper toxicosis. 4 The combination of copper and sulfur in the electron-dense intracytoplasmic aggregates might be explained by the presence of large amounts of sulfur-containing proteins such as metallothioneins, which bind the intracellular free copper to protect the cell from its toxic effects.
Footnotes
Acknowledgements
We thank Ton Ultee (electron microscopy) and Dr. Hans Meeldijk (electron microscopic energy-dispersive X-ray analysis) for their technical support.
