Abstract
Arthropathy in dogs induced by ofloxacin, a quinolone antimicrobial agent, was pathophysiologically investigated. In the in vivo studies, ofloxacin was administered orally once or twice at 20 mg/kg/day to male juvenile (3-month-old, n = 3) or adult (36-month-old, n = 2) dogs, and the humeral and femoral heads were examined pathologically. Unlike adult dogs, fluid-filled vesicles were macroscopically observed on the articular surfaces of one juvenile dog 24 hours after a single treatment with ofloxacin. These lesions were seen in all juvenile dogs by twice dosing. Microscopically, fissures or cavity formations in the middle zone of the articular cartilage were noted only in juvenile dogs. Furthermore, the cartilage matrix from the abnormal area to the articular surface showed a decreased safranin-O staining intensity, suggesting proteoglycan depletion. Ultrastructurally, chondrocytes in the middle zone of juvenile dogs displayed dilatation of the cisternae in the rough endoplasmic reticulum as an initial hallmark. In the in vitro studies, chondrocytes isolated from the articular cartilage of naive juvenile dogs were exposed to ofloxacin at 6.3–100 μg/ml for 24 hours. Although no changes were noted in the deoxyribonucleic acid synthesis, protein synthesis, or proteoglycan release at concentrations of up to 100 μg/ml, the proteoglycan synthesis was evidently decreased in a dose-dependent manner from 12.5 μg/ml. The results obtained suggest that the inhibitory action of ofloxacin on proteoglycan syntheses in the chondrocytes may largely contribute to the early morphologic features in the articular cartilage of the juvenile dog.
Quinolone antimicrobial agents (quinolones) have been widely used in clinical fields because of their bactericidal activity, broad spectra, and good pharmacokinetics. However, quinolones possess the potential to induce arthropathy in juvenile animals but not so in adult animals. Among the experimental animals such as mice, 17 rats, 14,15 guinea pigs, 1 rabbits, 16 dogs, 3,5,8,13 and non-human primates, 21 the juvenile dog is thought to be most susceptible to quinolone-induced arthropathy. 9,22
Broadly, quinolone arthropathy is characterized by fluid-filled vesicle formations in the articular surfaces, 9 but there have been only a few reports dealing with its underlying mechanism. In the report of Kato and Onodera, 14,15 the deoxyribonucleic acid (DNA) synthesis of the articular cartilage was first recognized to be inhibited by quinolones. Considering the inhibitory effect of quinolones on topoisomerase II, 7,12 they speculated that a trigger for the development of lesions was a depression in DNA syntheses of chondrocytes. Meanwhile, Bendele et al. 1 pointed out the extracellular matrix as a site for the initiation of the lesions, although no changes in degrading enzymes such as collagenase and neutral protease were detected. More recently, the inhibition in the synthesis of extracellular matrix components 4,11,16 and dysfunction in the mitochondria of chondrocytes 10 have been reported to be key processes in the occurrence of arthropathy. In addition, the importance of chelation of magnesium ions with quinolones resulting in a dysfunction of the integrin receptors on the chondrocytes has been suggested. 19,20
The present study was carried out to elucidate pathophysiologically the early target of quinolone arthropathy in dogs by use of both in vivo and in vitro systems. Namely, the early morphologic features in the articular cartilage of juvenile dogs receiving oral ofloxacin, a quinolone derivative, were examined. Afterward, the syntheses of DNA, protein, and proteoglycan, or release of proteoglycan in cultured canine articular chondrocytes exposed to ofloxacin, were investigated.
Materials and Methods
Animals
Male juvenile (3-month-old) Beagle dogs weighing 6.8 ± 1.1 kg and adult (36-month-old) Beagle dogs weighing 10.9 ± 0.9 kg were purchased from BMR Co. (Gifu, Japan) and Toyota Tsusho Co. (Tokyo, Japan), respectively. After 14-day quarantine, each dog was established to be healthy from the results of physical examinations. They were housed individually in a wire-mesh cage (65-cm width × 85-cm depth × 75-cm height). Dogs were allowed free running for 1 hour every day. However, we did not give toys as a source of environmental enrichment because it was a short-term experiment. The animal room was maintained at a temperature of 23 ± 2 C and a relative humidity of 60 ± 20% with a 12 hours light-dark cycle, and the dogs were allowed free access to a commercial laboratory diet (DM-2: Funabashi Farm, Chiba, Japan) and tap water. All dogs were treated humanely, and the study protocol was in accordance with the institutional guidelines of Daiichi Pharmaceutical Co., Ltd. (Tokyo, Japan) for use of laboratory animals.
In vivo studies
Ofloxacin (Fig. 1; Daiichi Pharmaceutical Co., Ltd.) contained in a 1/16 ounce gelatin capsule (Toropac Inc., NJ, USA) was administered orally once or twice to juvenile (n = 3) and adult dogs (n = 2) at 20 mg/kg/day (Table 1). The dosage levels used in the present investigation were selected on the basis of our previous published reports. 23–25 The dogs receiving oral administration of the gelatin capsule alone for 2 days served as the control (three juvenile and two adult dogs). They were euthanatized by exsanguination 24 hours after the first or second dosing under anesthesia with concurrent intravenous administration of xylazine (2 mg/kg, Bayer, Tokyo, Japan) and ketamine (40 mg/kg, Sankyo Co. Ltd., Tokyo, Japan). All dogs were fasted for at least 16 hours before anesthesia.
Distribution of dogs and number of tissue blocks and sections in the experiment.
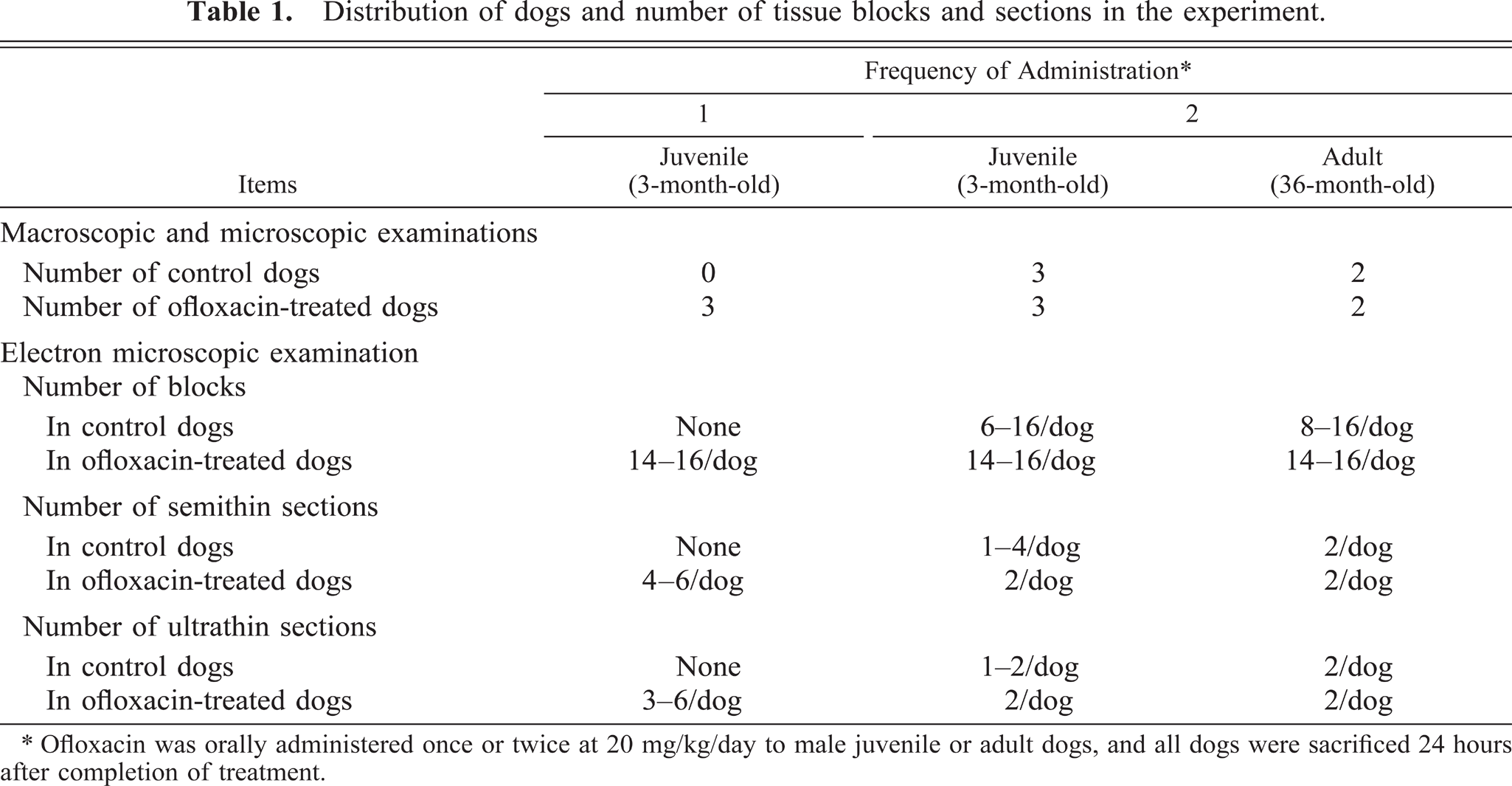
∗ Ofloxacin was orally administered once or twice at 20 mg/kg/day to male juvenile or adult dogs, and all dogs were sacrificed 24 hours after completion of treatment.
Chemical structure of ofloxacin.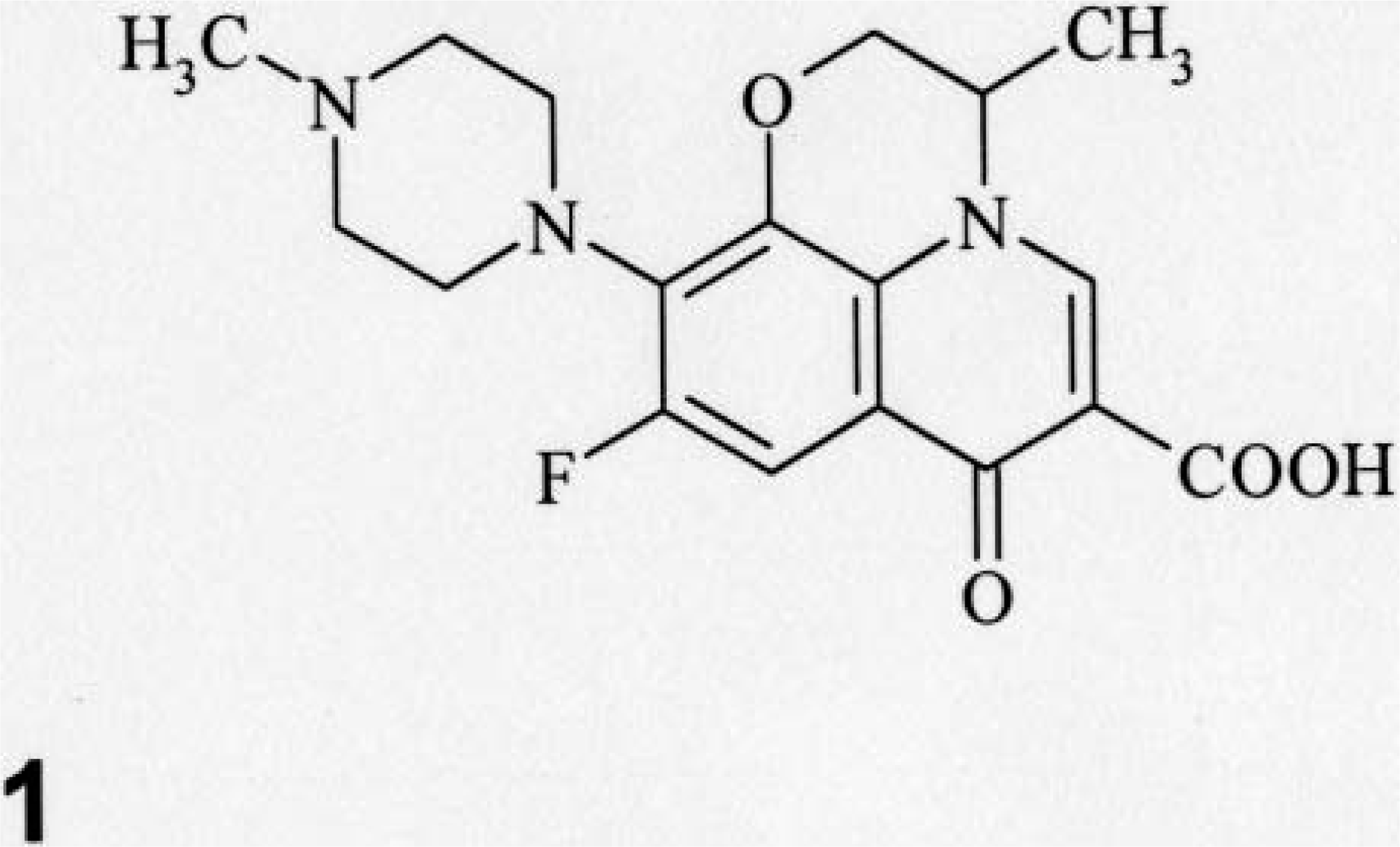
Macroscopic examination focused on the articular cartilages in the dorsal area of the humeral and femoral heads and around the insertion of the teres ligament because these regional areas have been known to be susceptible to the lesions in juvenile dogs (Fig. 2A, B)). 23–25 These cartilage tissues were cut off with a scalpel after completion of visual assessment. In juvenile dogs, the left-side specimens of the articular cartilage were fixed in 10% buffered-formalin solution without decalcification. In contrast, the articular tissues of adult dogs were fixed and decalcified in 10% ethylenediaminetetraacetic acid solution for 5 days. According to our previous report, 25 no artifacts were induced by these decalcification procedures. Subsequently, they were trimmed obliquely into 4-mm-thick slabs, embedded in paraffin wax, cut in a step section (4-µm thickness) at intervals of 20-µm thickness, and stained with hematoxylin and eosin or safranin-O. For evaluation of the extracellular matrix, right-side specimens were fixed overnight in 0.13% glutaraldehyde (pH 7.4) containing 0.1% ruthenium red for staining proteoglycans. Afterward, the specimens were rinsed three times by 0.1 M cacodylate buffer (pH 7.4), and they were further fixed for 5 hours in 2% osmium tetroxide (pH 7.4) containing 0.05% ruthenium red. The fixed specimens were dehydrated and then embedded in the epoxy resin (Table 1). Semithin sections (0.8-µm thickness) were prepared, stained with toluidine blue, and examined with a light microscope to choose the target region. Namely, the area showing the border of the vesicles was selected for ultrathin sections. After trimming, ultrathin sections (70-nm thickness) were stained with uranyl acetate and lead citrate and examined by transmission electron microscopy (LEM 2000, Topcon Ltd., Inc., Tokyo, Japan).
Schematic diagram of the left proximal humerus (Fig. 2A, caudal view) and femur (Fig. 2B, medial view). The gray areas within the broken lines represent the sites collected as articular specimens for microscopic and ultra-structural examinations. Insertion of the teres ligament (L).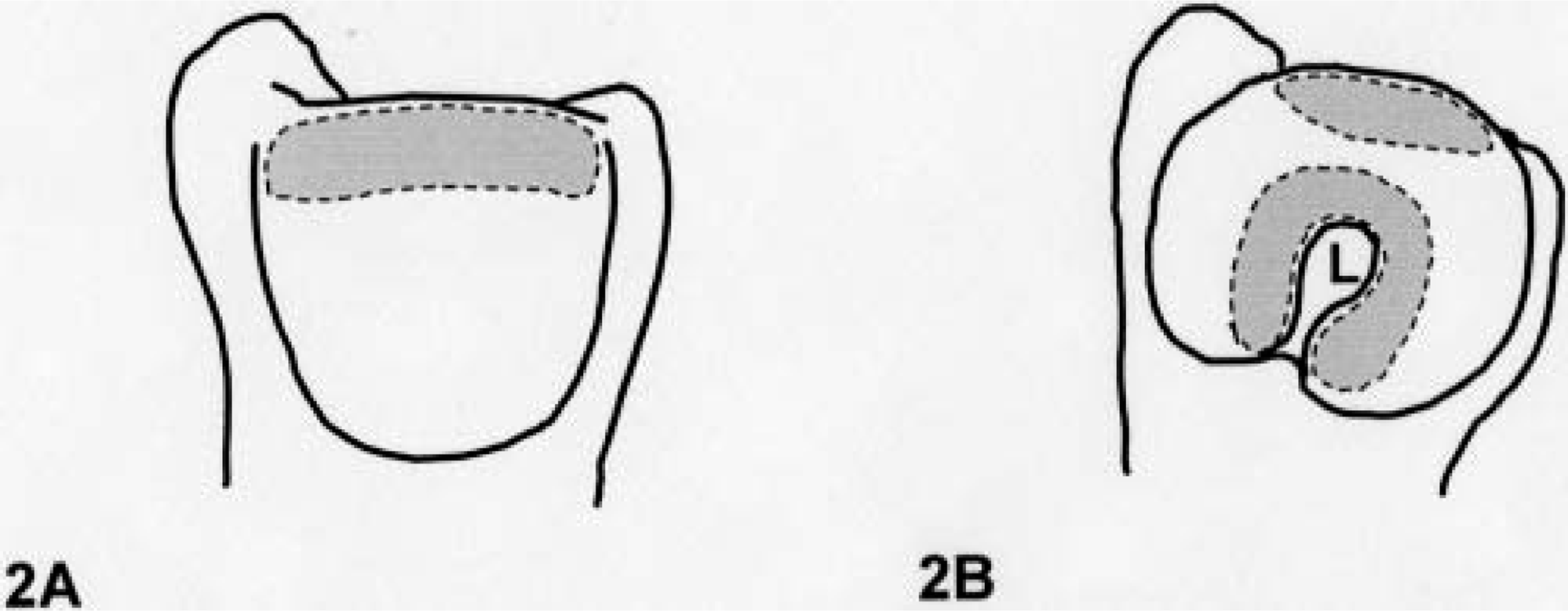
In vitro studies
Naive juvenile dogs (n = 2) were euthanatized under anesthesia as was the case with the aforementioned in vivo study. The articular cartilage was excised, and then chondrocytes were isolated and cultured by the method of Kato et al. 16 with a few modifications. In brief, cartilage tissues were collected from the proximal humerus and distal femur and cut into small pieces. They were treated at 37 C by sequential digestions in Dulbecco modified Eagles medium (DMEM, Sigma-Aldrich, Tokyo, Japan) containing 0.25% trypsin (Invitrogen, Tokyo, Japan) for 30 minutes, DMEM-0.1% hyaluronidase (Type I-S, Sigma-Aldrich) for 30 minutes, and DMEM-0.2% collagenase (Wako Pure Chemical Industries, Ltd., Osaka, Japan) for 3 hours. Isolated chondrocytes were suspended in DMEM with 10% fetal bovine serum (HyClone, Logan, UT), penicillin (Invitrogen) at 100 U/ml and streptomycin (Invitrogen) at 100 µg/ml, and seeded at 2.5 × 104 cells/cm2 in 24-well plastic plates (Asahi Technoglass Corporation, Chiba, Japan). Chondrocytes were then cultured in a humidified incubator with 5% CO2 at 37 C for 5 days until confluence. Chondrocytes expressed type-II procollagen messenger ribonucleic acid, confirmed by a reverse transcription-polymerase chain reaction, which suggested that they were phenotypically equal to the intact articular chondrocytes under these culture conditions (data not shown).
The chondrocytes were exposed to ofloxacin at 6.3–100 µg/ml for 24 hours. The drug concentration and exposure time were chosen on the basis of the results of our previous report. 23 The conditioned medium without ofloxacin was used as the concurrent control. To examine the syntheses of DNA, protein, and proteoglycan, 5 µCi/ml/well each of 3H-thymidine (PerkinElmer Life Science, Kanagawa, Japan), 3H-leucine (PerkinElmer Life Science) and 35SO4 (Perkin-Elmer Life Science) were added in sequence to the medium immediately after commencement of the incubation. After termination of the exposure, the cells were washed twice with phosphate-buffered saline (pH 7.2, Invitrogen), and dissolved in 2% sodium dodecyl sulfate (1 ml, Kishida Chemical Co., Ltd., Osaka, Japan). The cell lysate (1 ml) was transferred into a scintillation vial, and then the Aquasol II scintillation fluid (9 ml, PerkinElmer Life Science) was added to it. The radioactivity of the solution was measured with a liquid scintillation counter (LSC-600, Aloka Co. Ltd., Tokyo, Japan). To further assess proteoglycan releases, the chondrocytes were treated with 5 µCi/ml/well of 35SO4 for 24 hours before the exposure of ofloxacin. At the end of the incubation for 24 hours, the cultured medium (1 ml) was collected, and its radioactivity was counted. The cytotoxicity due to ofloxacin was evaluated by a trypan blue exclusion test. Each assay was performed in triplicate. Four wells per concentration were used to examine the syntheses of DNA, protein, and proteoglycan and releases of proteoglycan and three wells per concentration for evaluation of cytotoxicity.
Statistical analysis
Quantitative data from in vitro studies were expressed as the mean ± SD, and the differences between control and treatment groups were analyzed by Dunnett t-test (EXSAS, Version 5.00, Arm Corporation, Osaka, Japan). A P value of less than 0.05 was considered to be statistically significant.
Results
In vivo studies
Unifocal or multifocal fluid-filled vesicles were macroscopically observed in the articular surfaces in one juvenile dog 24 hours after the first dosing (Table 2). The lesion developed on the dorsal area of the humeral and femoral heads or around the insertion of the teres ligament. Twenty-four hours after the second dosing, the vesicle formation was seen in all juvenile dogs and erosion of vesicles was also noted in two dogs. Therefore, the incidence of articular lesions was apparently increased with twice dosing. Microscopically, fissures or cavity formations in the middle zone of the articular cartilage were observed only in juvenile dogs receiving ofloxacin (Figs. 3A, 4, 5). Degenerated or necrotic chondrocytes with the edematous matrix were noted around the lesions, and unmasked collagen fibers were seen in the edematous area (Fig. 6). Furthermore, the cartilage matrix from the abnormal area to the articular surface showed a decreased safranin-O staining intensity. Ultrastructurally, chondrocytes in the middle zone of juvenile dogs receiving ofloxacin displayed dilatation of the cisternae in the rough endoplasmic reticulum as an initial hallmark, in conjunction with decreased proteoglycan granules and edema in the territorial matrix (Figs. 7, 8). In more advanced cases, pyknotic nuclei, cytoplasmic vacuoles, and cell debris within the territorial matrix were also noted (Fig. 9). Necrotic chondrocytes were detached from the matrix with severe depletion of proteoglycan granules. In the interterritorial matrix, proteoglycan granules were also decreased, and collagen fibers were disarranged or had disappeared. No clear differences in ultrastructural lesions were observed between single and twice dosings. Meanwhile, the structure of the surface or deep zone of the cartilages away from the lesions, and the cell integrity of chondrocytes were maintained.
Macroscopic incidence of the articular lesions in male juvenile and adult dogs treated orally with ofloxacin.

∗ All dogs were sacrificed 24 hours after completion of treatment.
† Number of dogs affected/number of dogs used.
Microscopic appearance of the articular cartilage in a control juvenile dog (Fig. 3A) and adult dogs (Fig. 3B). Note the tidemark (arrowheads) between the deep and calcified zones in a adult dog. Safranin-O. Bar = 40 µm.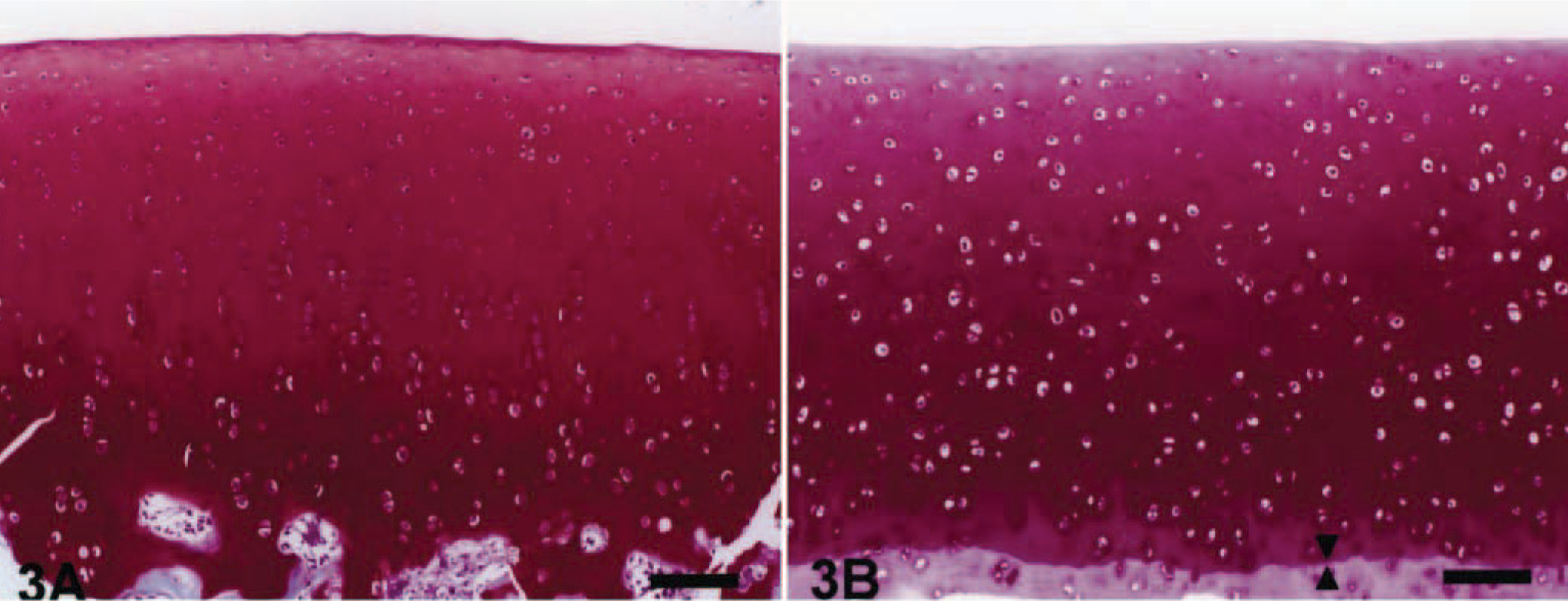
Microscopic appearance of the articular cartilage in a juvenile dog treated orally with ofloxacin at 20 mg/kg/day for 2 days. The dog was sacrificed 24 hours after the second dosing. Note fissure in the middle zone of the articular cartilage. Safranin-O. Bar = 40 µm.
Microscopic appearance of the articular cartilage in a juvenile dog treated orally with ofloxacin at 20 mg/kg/day for 2 days. The dog was sacrificed 24 hours after the second dosing. Note cavity in the middle zone of the articular cartilage. Safranin-O. Bar = 40 µm.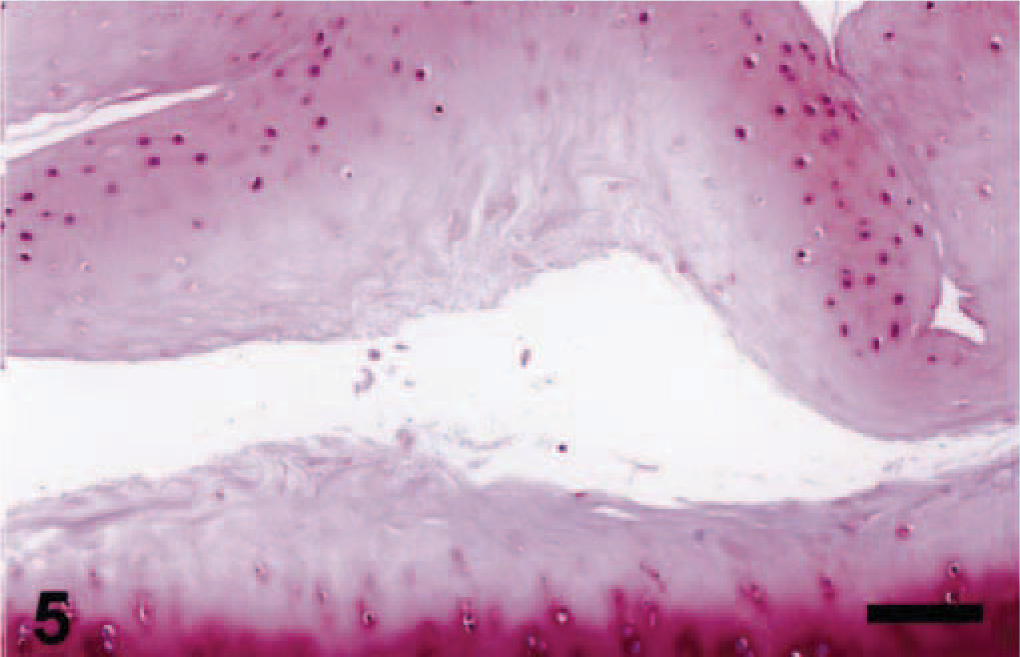
Microscopic appearance of the articular cartilage in a juvenile dog treated orally with ofloxacin at 20 mg/kg/day for 2 days. The dog was sacrificed 24 hours after the second dosing. Note cytoplasmic vacuolation (arrows), pyknotic nucleus (arrowheads) and edematous matrix (asterisk). Toluidine blue. Bar = 5 µm.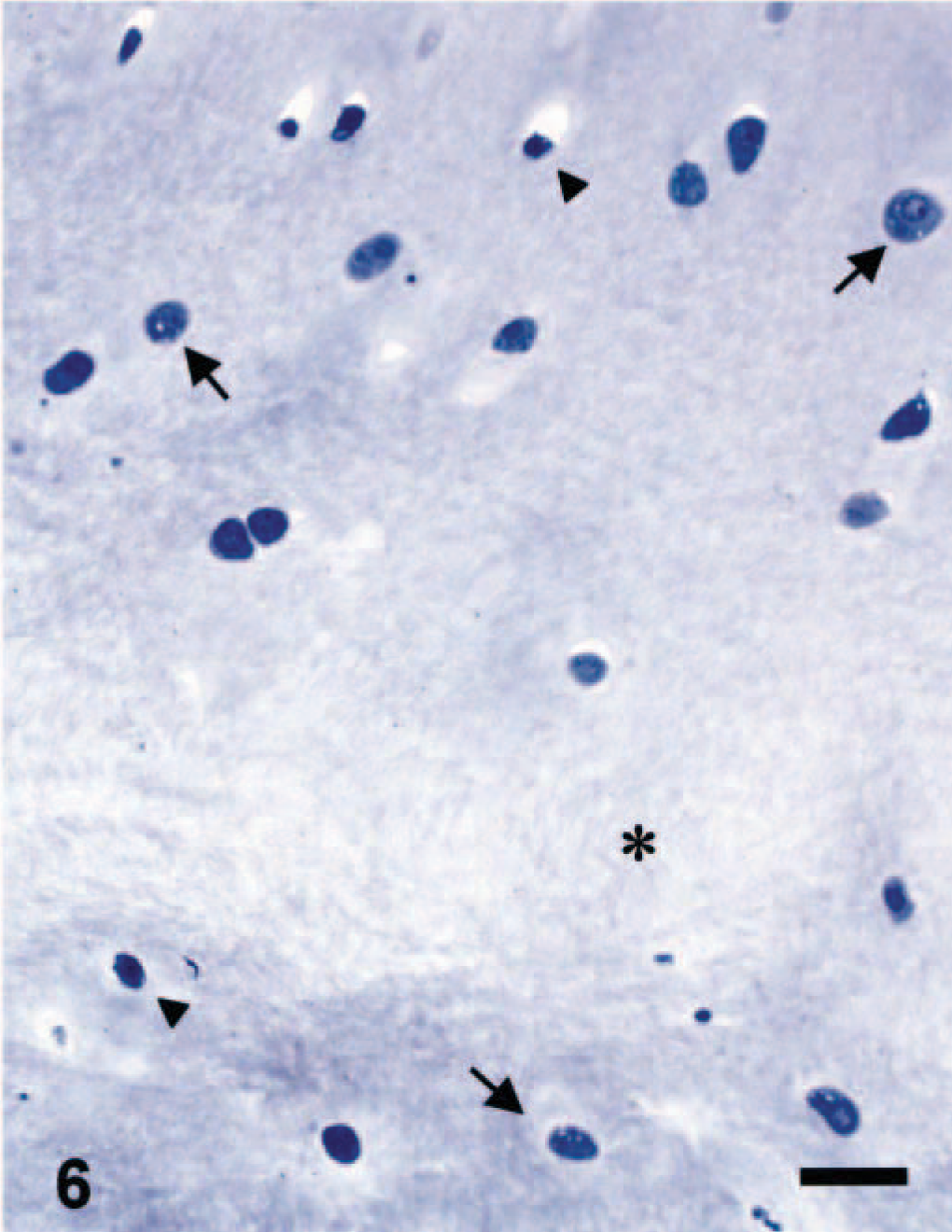
Ultrastructural appearance of the chondrocyte in the middle zone of the articular cartilage in a control juvenile dog. Note the well-developed rough endoplasmic reticulum and closely packed proteoglycan granules within the territorial matrix (asterisk). Ruthenium red fixation. Bar = 0.1 µm.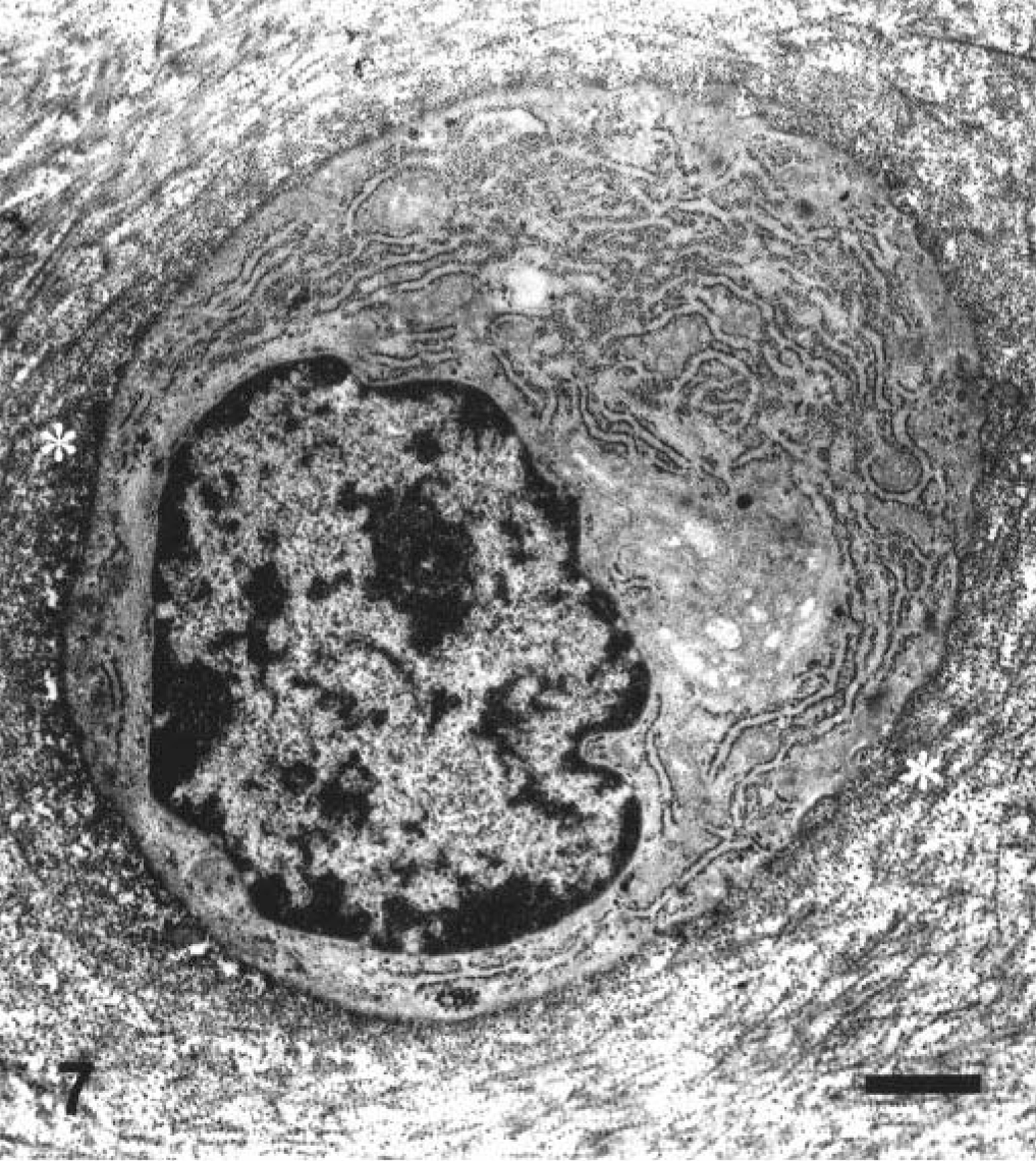
Ultrastructural appearance of the chondrocyte in the middle zone of the articular cartilage in a juvenile dog given a single oral treatment with ofloxacin at 20 mg/kg/day. The dog was sacrificed 24 hours after the first dosing. Note dilatation of the cisternae in the rough endoplasmic reticulum. Electron-dense proteoglycan granules decreased within the territorial matrix (asterisk). Ruthenium red fixation. Bar = 0.1 µm.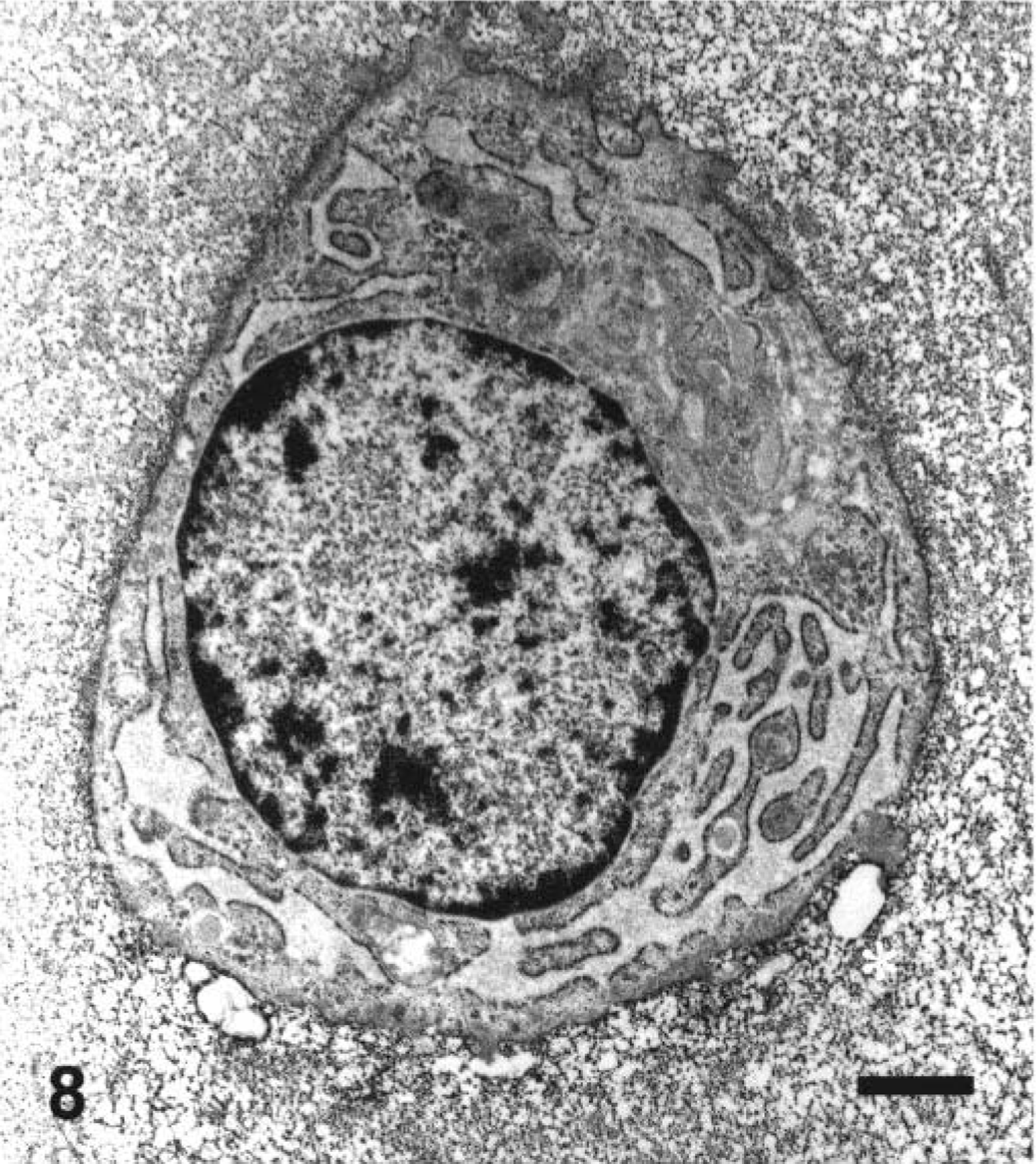
Ultrastructural appearance of the chondrocyte in the middle zone of the articular cartilage in a juvenile dog treated orally with ofloxacin at 20 mg/kg/day for 2 days. The dog was sacrificed 24 hours after the second dosing. Note pyknotic nucleus and severe depletion of electron-dense proteoglycan granules within the territorial matrix (asterisk). Ruthenium red fixation. Bar = 0.1 µm.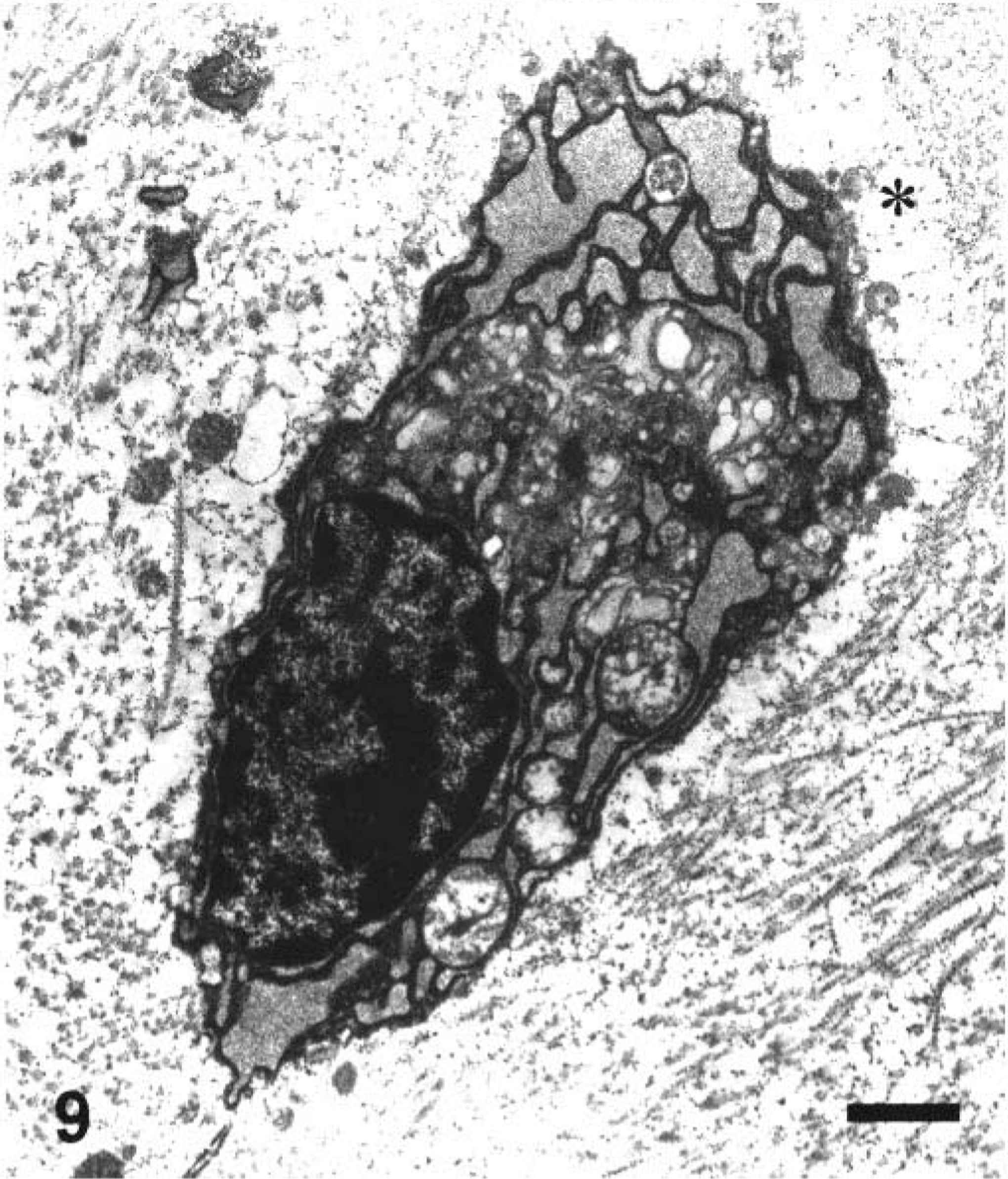
The articular cartilage of control adult dogs was microscopically thinner than that of control juvenile dogs, and was separated from the subchondral bone by the calcified zone. The tidemark was distinctly noted between the deep and calcified zones (Fig. 3A, B)). Moreover, the chondrocytes in the middle zone of the articular cartilage of control adult dogs ultrastructurally showed no obvious nucleoli or rough endoplasmic reticulum unlike control juvenile dogs (Figs. 7, 10). In adult dogs, ofloxacin did not affect the articular cartilages in any dose-regimen, and their morphologic appearance was kept intact as in control dogs.
Ultrastructural appearance of the chondrocyte in the middle zone of the articular cartilage in a control adult dog. Note clear differences between territorial matrix (arrowheads) and interterritorial matrix (asterisks). Ruthenium red fixation. Bar = 0.1 µm.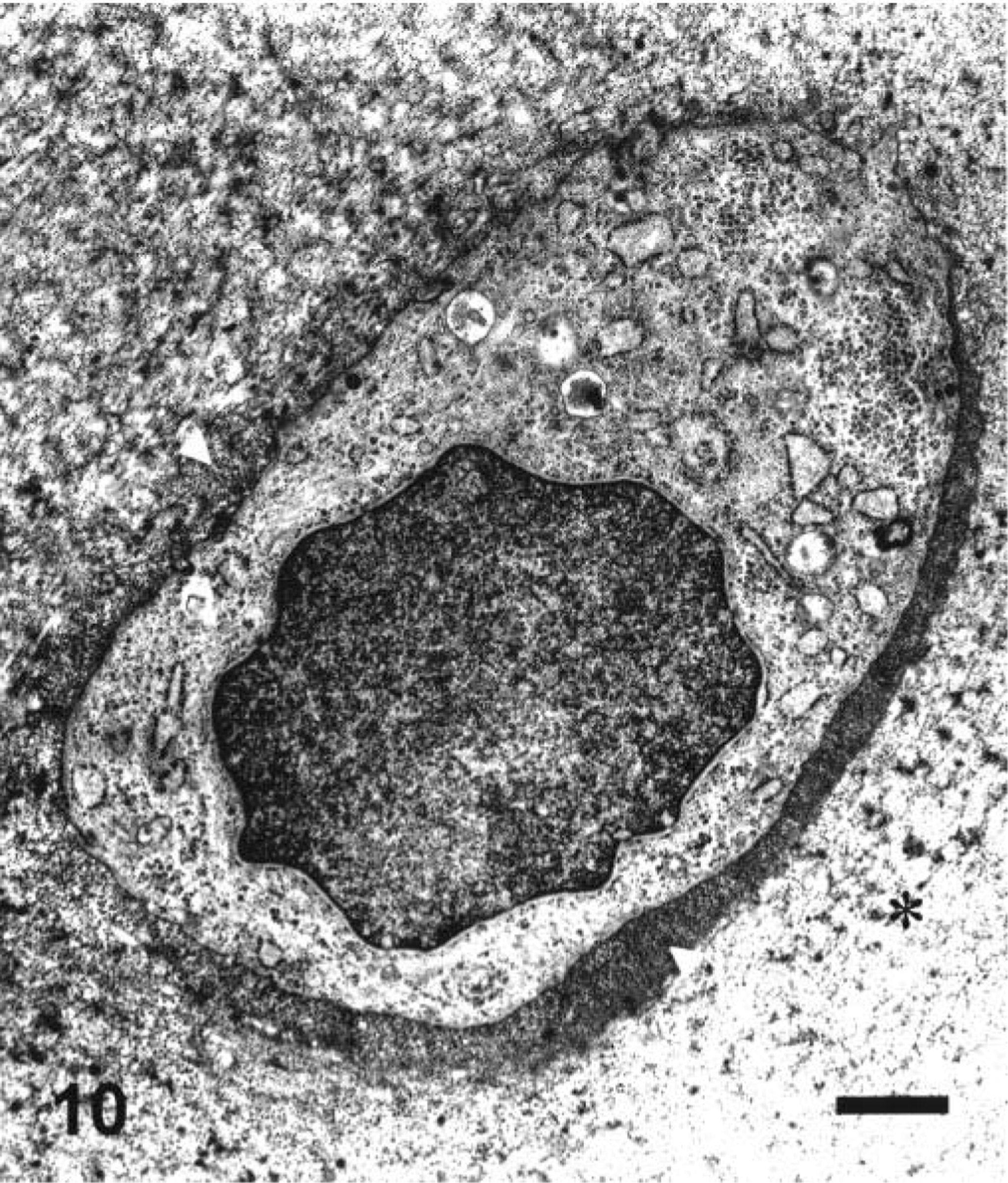
In vitro studies
Ofloxacin induced syntheses of neither DNA nor protein at concentrations of up to 100 µg/ml (Figs. 11, 12). In contrast, ofloxacin inhibited the synthesis of proteoglycan in a dose-dependent manner from 12.5 µg/ml without affecting the proteoglycan release at concentrations used (Figs. 13, 14). No cytotoxicity was seen in cultured cells used throughout all experiments under conditions of the present study (data not shown).
The effect of ofloxacin on DNA synthesis in the cultured canine articular chondrocytes. The chondrocytes were exposed to ofloxacin for 24 hours. Values represent means ± SD of four wells.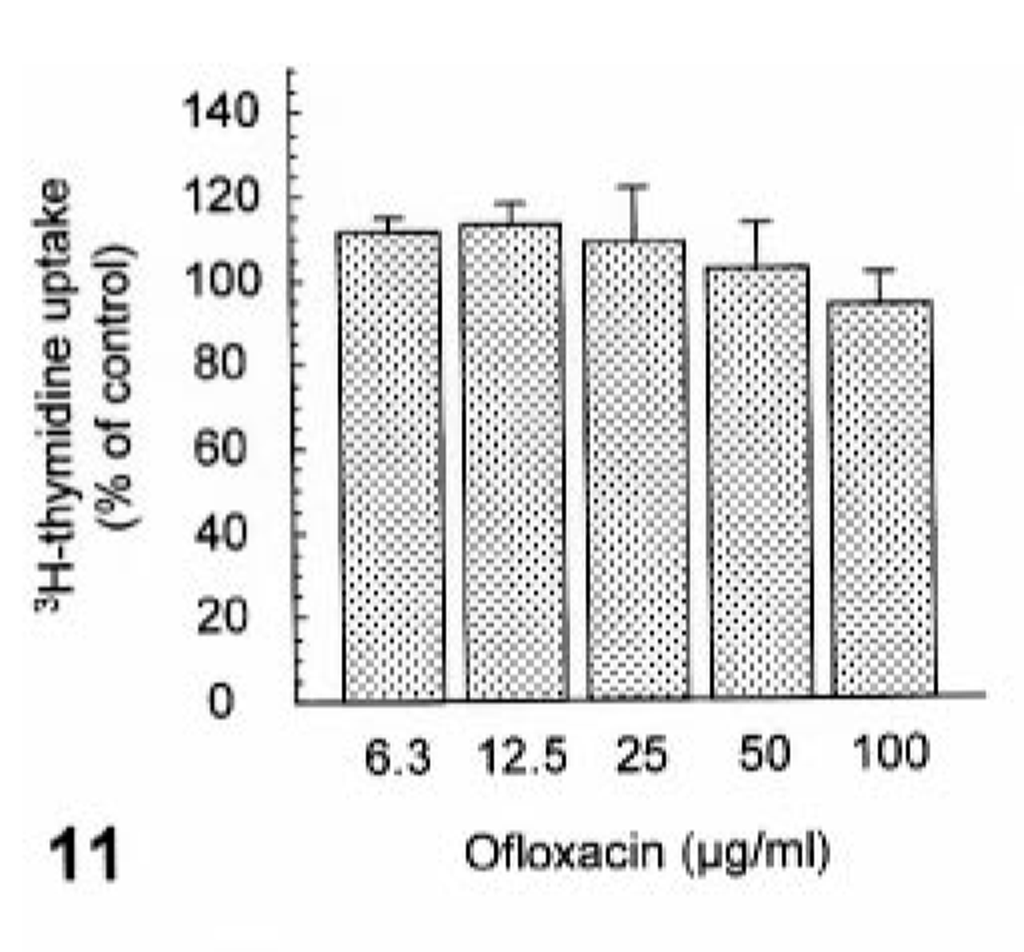
The effect of ofloxacin on protein synthesis in the cultured canine articular chondrocytes. The chondrocytes were exposed to ofloxacin for 24 hours. Values represent means ± SD of four wells.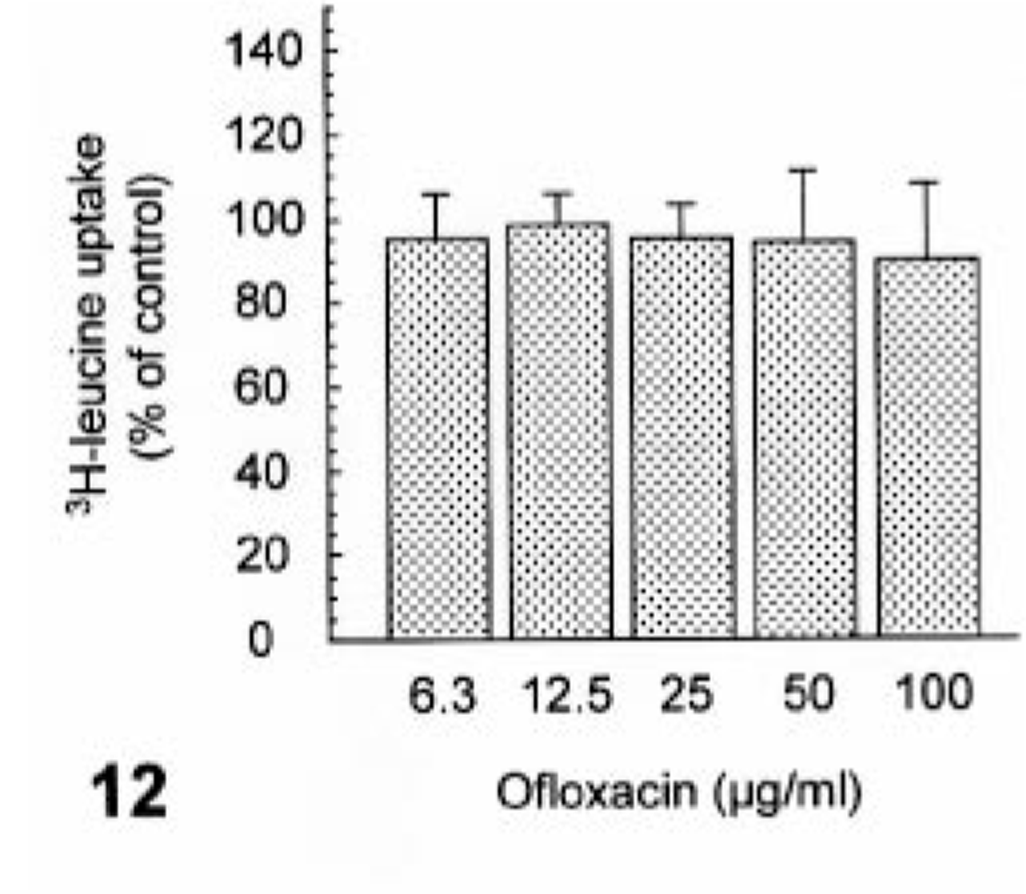
The effect of ofloxacin on proteoglycan synthesis in the cultured canine articular chondrocytes. The chondrocytes were exposed to ofloxacin for 24 hours. Values represent means ± SD of four wells. ##P < 0.01 = significant different from the vehicle control (Dunnett t-test).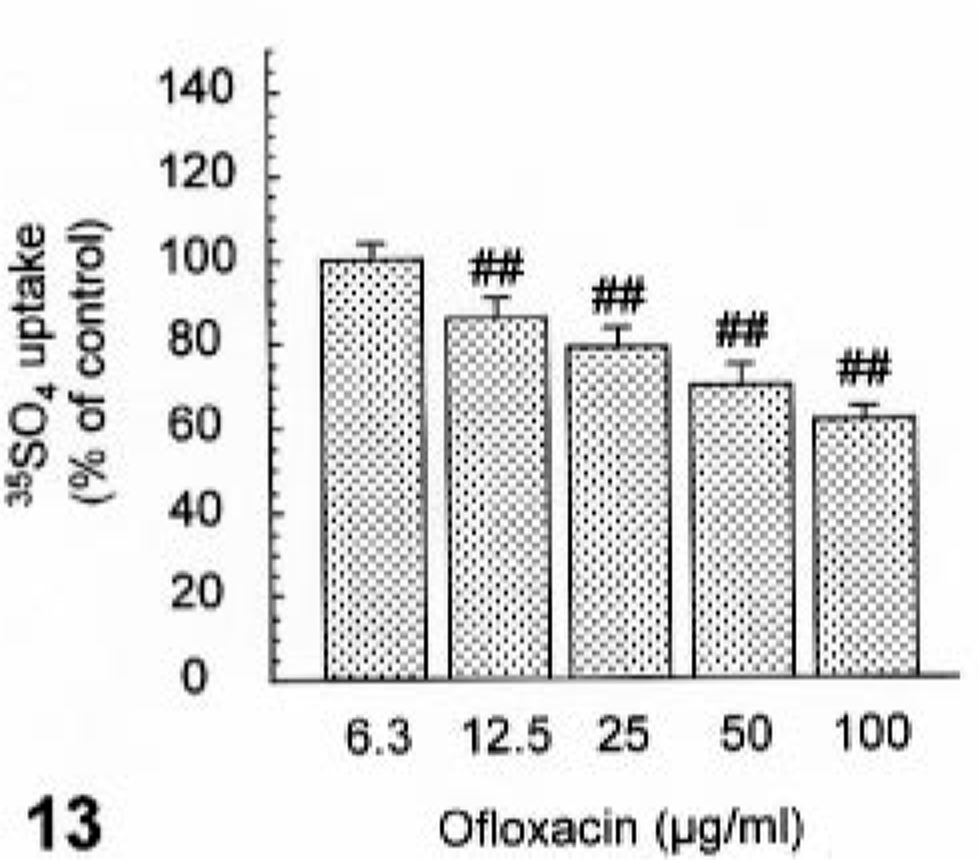
The effect of ofloxacin on proteoglycan release in the cultured canine articular chondrocytes. The chondrocytes were exposed to ofloxacin for 24 hours. Values represent means ± SD of four wells.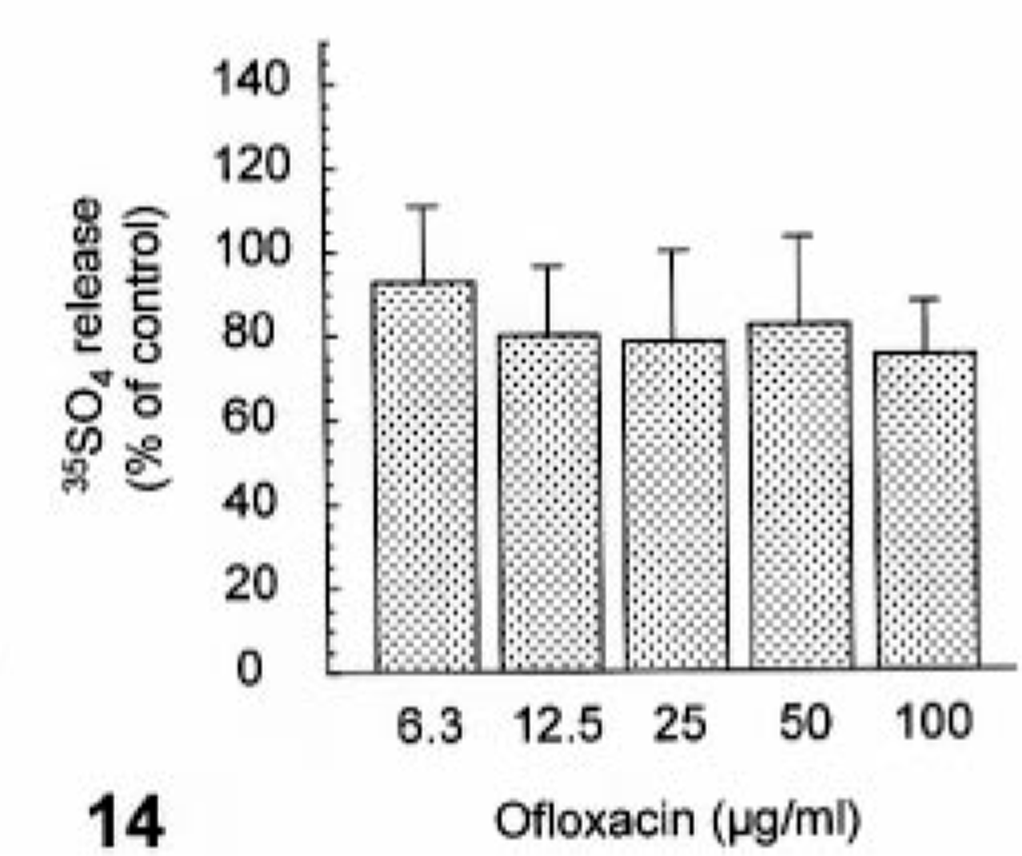
Discussion
The macroscopic and microscopic features of ofloxacin arthropathy in juvenile dogs seen in our work were essentially comparable with those of known quinolones. 3,8,13,22 The cartilage matrix from the abnormal area to the articular surface showed a decreased safranin-O staining intensity, suggesting proteoglycan depletion. As a novel ultrastructural finding, the earliest feature of the chondrocytes in the middle zone was thought to be dilatation of the cisternae in the rough endoplasmic reticulum, in conjunction with depletion in the proteoglycan granules and edema in the territorial matrix. The disorders such as disarrangement and disappearance of collagen fibers in the interterritorial matrix were likely to represent a later phase of the changes in the territorial matrix. Burkhardt et al. 5 have demonstrated that the initial change of the articular chondrocytes in juvenile dogs given oral difloxacin, a quinolone derivative, is swelling of mitochondria, leading to necrosis in the chondrocytes, and secondarily to disruption in the extracellular matrix. However, in our study, no changes in mitochondria were noted as an early event in the lesion development. Thus, subsequently observed necrosis of chondrocytes was presumably considered to be a result of perturbation in the cell-matrix interactions. Furthermore, dilatation of the cisternae in the rough endoplasmic reticulum was inferred to suggest a disturbance in the synthesis or release of proteoglycans in chondrocytes.
As with our ultrastructural findings, Kato and Onodera 15 have reported that juvenile rats receiving a single oral administration of ofloxacin reveal dilatation of the rough endoplasmic reticulum of the articular cartilage 5 hours after treatment. They have also observed nuclear condensation of these chondrocytes. However, in our study, no nuclear alteration was noted ultrastructurally in any chondrocytes showing the lesion of the rough endoplasmic reticulum in the early phase. Therefore, the direct effect of ofloxacin on nucleus functions such as DNA syntheses may not be essential for the lesion development.
The predilection site of ofloxacin-induced arthropathy was the dorsal area of the humeral and femoral heads or around the insertion of the teres ligament. These regions were consistent with the susceptible sites of juvenile dogs receiving oral difloxacin, a quinolone derivative, reported by Burkhardt et al., 3 suggesting the importance of weight bearing in the pathogenesis of the lesions. The articular cartilage is composed of large sulfated proteoglycan aggregates embedded in fibrillar networks of type-II collagen. 9 The polyanionic and hydrophobic natures of proteoglycans provide the tissue resiliency and rapidly reversible deformation against compressive forces, and the collagen fibers make the scaffolding that gives tensile strength to the articular cartilage. 9 Therefore, the fragile cartilages observed may be also indicative of the cavity formation as a consequence of the mechanical force. Furthermore, the increased incidence of vesicle formations was definitized by twice dosing, and erosion was thought to be a lesion secondary to the vesicle formation.
The decreased synthesis of proteoglycan has been reported to emerge during the maturation processes of the articular cartilage in humans. 2,6 Namely, the rate of proteoglycan synthesis in the articular cartilage of humans aged 12–17 years was approximately 2–3 times higher than that of those aged 18–71 years. As characteristics of quinolone arthropathy, an age-specific susceptibility existed between juvenile versus adult dogs, which suggested that the juvenile dogs were much more sensitive than adult dogs. This difference in aging may be explained by the presence of the tide-mark, which was distinctly noted between the deep and calcified zones only in adult dogs, demonstrating the mature articular cartilage tissue. The mature cartilages have been shown to be resistant to the onset of quinolone arthropathic lesions even at a dose that induced arthropathy in juvenile dogs. 25 In addition, different from juvenile dogs, nucleoli and rough endoplasmic reticulum of chondrocytes in adult dogs were ultrastructurally unremarkable. These morphologic aspects, at least in part, suggested that the chondrocytes of juvenile dogs were physiologically active, and these active immature cells were thought to be more vulnerable to ofloxacin than less active mature cells in adult dogs.
We have reported previously that the threshold of ofloxacin concentrations in the articular cartilage required to induce arthropathy is approximately 10 µg/g tissue in juvenile dogs after an 8-day repeated oral treatment. 23 In the present in vitro study, ofloxacin inhibited the proteoglycan synthesis in a dose-dependent manner from a concentration of 12.5 µg/ml that was nearly equal to the arthropathic drug concentration. Nevertheless, ofloxacin did not affect the syntheses of DNA and protein or the release of proteoglycan. These results implied that ofloxacin specifically inhibited proteoglycan synthesis in chondrocytes. In support of our data, Kato et al. 16 have reported that levofloxacin, an optically active isomer of ofloxacin, initially inhibits the proteoglycan synthesis and subsequently DNA synthesis at actual arthropathic concentrations in cultured rabbit chondrocytes. Moreover, in the previous in vitro studies, difloxacin has been also shown to inhibit proteoglycan synthesis in organ cultures of canine articular cartilage and has elicited distention of rough endoplasmic reticulums in affected chondrocytes. 4
From the viewpoint of the chemical structure of ofloxacin, this compound possesses a positively charged piperazinyl moiety at the 7 position. Meanwhile, proteoglycans consist of a core protein and negatively charged polysaccharide chains (glycosaminoglycans). 9 The core protein has been reported to be synthesized in the endoplasmic reticulum, followed by attachment of the saccharides in the Golgi apparatus. 18 Taken together, these data suggest the possibility that the cationic moiety at the 7 position of ofloxacin may interact with negatively charged glycosaminoglycans and thereby reduce the biosynthesis of proteoglycans. On this basis, the accumulation of the incomplete formations of proteoglycans may lead to dilatation of the cisternae within the endoplasmic reticulum of chondrocytes. However, a Golgi lesion was not apparent in the present study. Further studies would need to confirm the effect of ofloxacin on biosynthetic pathways of proteoglycans.
In conclusion, the inhibitory action of ofloxacin on proteoglycan synthesis in the chondrocytes would largely contribute to the early morphologic features seen in the articular cartilage of the juvenile dog, and consequently, the depletion of proteoglycans in the articular cartilage matrix may play a critical role in the cavity formation, preceded by dilatation of the cisternae in the rough endoplasmic reticulum.
Footnotes
Acknowledgements
We thank Dr. Michiyuki Kato, Dr. Tetsuyo Kajimura, and Dr. Kiyonori Kai for their critical review and helpful advice and also Ms. Keiko Okado, Mr. Takanori Enkawa, Ms. Satoko Tago, and Mr. Takashi Hirayama for their excellent technical support.
