Abstract
The role of recruited neutrophils in Mannheimia haemolytica infection is controversial. We hypothesized that the neutrophilia induced by recombinant bovine granulocyte colony-stimulating factor (GCSF) would lead to rapid bacterial clearance and less severe lesions after infection with M. haemolytica. Two experiments (A and B) were conducted in which four calves per experiment were treated daily with 5 μg/kg GCSF and four calves per experiment were treated with saline. All 16 calves were challenged with 5 × 109 colony-forming units (cfu)/ml (experiment A) or 4.5 × 108 cfu/ml (experiment B) of M. haemolytica bacteria, into the right bronchus by bronchoscope-placed catheter. The mean maximal blood neutrophil counts in non-GCSF-treated and GCSF-treated calves before bacterial challenge were 5.6 ± 0.7 × 109/liter and 25.4 ± 2.7 × 109/liter, respectively. Two untreated calves became neutropenic and were euthanatized 2 days after infection because of severe respiratory distress. GCSF-treated calves had a 37% reduction in lung lesions compared with nontreated calves, and this difference was significant (P = 0.04) when the effect of previous antibody titre to leukotoxin was considered. The effect of GCSF treatment on the severity of clinical signs seemed to be influenced by the antibody titre to M. haemolytica leukotoxin, although this effect could not be conclusively addressed. In conclusion, GCSF induced neutrophilia and partially protected calves against experimental infection with M. haemolytica. These results imply that increased numbers of neutrophils may, under some circumstances, protect against severe pneumonia caused by M. haemolytica.
Keywords
Introduction
Shipping fever pneumonia is the leading cause of death and economic loss for the beef industry. Mannheimia haemolytica, the most common cause of shipping fever pneumonia, colonizes the nasopharynx of many normal cattle. Although calves are innately resistant to pulmonary infection with M. haemolytica, these bacteria infect the lower respiratory tract and cause pneumonia during times of stress, adverse climatic conditions, and respiratory viral infection. 1,20,21
Neutrophils are rapidly recruited to the lung after bacterial challenge or after administration of inflammatory cytokines. 15 Neutrophils defend the lung against bacterial infection by phagocytosing bacteria and killing them using oxygen radicals, antimicrobial peptides, and proteolytic enzymes. 27 However, secretion of these same proteases and oxygen radicals may cause pulmonary injury, including necrosis of epithelial cells and degradation of matrix proteins, and the physical presence of neutrophils in alveoli and bronchioles precludes ventilation of the alveoli. Three studies have examined the effect of prior depletion of neutrophils on the response to pulmonary challenge with M. haemolytica, with somewhat variable results. In general, neutrophil depletion reduced the development of gross pulmonary lesions, neutrophil exudation into alveoli, and necrosis of alveolar septa. Neutrophil depletion had little effect on the development of fever, alveolar edema and hemorrhage, and thrombosis, which may represent direct effects of lipopolysaccharide (LPS) or LPS-induced cytokines. 3,23,24 Treatment with a selectin inhibitor, which impairs neutrophil infiltration into alveoli and bronchioles, reduced oxidative injury to lung tissue in neonatal calves infected with M. haemolytica (albeit not significantly). 18,19 These findings imply that in calves challenged with high numbers of M. haemolytica, the neutrophil responses are harmful and cannot effectively eliminate the bacterial infection.
Nevertheless, the role played by recruited neutrophils at the time of acute bacterial infection is complex and probably depends on the magnitude of bacterial challenge, the responsiveness of the pulmonary innate defenses, and the concentrations of pathogen-specific antibody in serum and lung fluids. On this basis, we hypothesized that the balance between the beneficial and harmful effects of neutrophil responses to M. haemolytica infection depends on the challenge dose of bacteria and the level of serum antibody to leukotoxin. We evaluated this possibility by using granulocyte colony–stimulating factor (GCSF) to induce blood neutrophilia in calves before challenge with M. haemolytica. GCSF enhances neutrophil production by stimulating the proliferation of bone marrow neutrophil precursor cells and also stimulates neutrophil release from the bone marrow. 7 Therapeutic uses of GCSF in human medicine include treatment of neutropenia and prevention of postoperative infections. The effectiveness of GCSF therapy in nonneutropenic human patients with sepsis and bacterial pneumonia remains controversial. 8,16,17,22,26, Bovine GCSF increases the expression of CD11a, CD11b, CD11c, and CD18 and primes neutrophils for a greater response to M. haemolytica. 9,15 However, bovine GCSF had no effect on oxidative burst of neutrophils, as measured by reduction of nitro blue tetrazolium. 7 This study evaluates the impact of GCSF treatment on bovine neutrophil function in vivo and the effect of GCSF-induced neutrophilia on the response to challenge with M. haemolytica.
Materials and Methods
Reagents
All chemicals and reagents were obtained from Canadian Life Technologies (Burlington, ON, Canada), with the following exceptions: N-[2-Hydroxyethyl] piperasine-N′-[2-ethanesulfonic acid] (HEPES), fluorescein isothiocyanate (FITC), zymosan A, cytochalasin B, phorbol-13-myristate-12-acetate (PMA), and 3,3′,5,5′-tetramethylbenzidine (Sigma, St. Louis, MO); 2,7-dichlorodihydrofluorecein diacetate (H2DCFDA) and calcein AM (Molecular Probes, Eugene, OR); and hydrogen peroxide and sodium citrate dihydrate (Fisher, Fairlawn, NJ). Recombinant bovine GCSF was a gift from Amgen Inc. (Thousand Oaks, CA).
M. haemolytica was kindly supplied by Dr. P. Shewen, University of Guelph. Bacteria were cultured on blood agar and subcultured in brain-heart infusion broth (BHIB) on a 37 C shaker. When the absorbance at 525 nm reached 0.4–0.5, the bacteria were washed once in cold phosphate-buffered saline (PBS), maintained at 4 C, and the concentration was estimated by measuring the absorbance at 525 nm. This bacterial suspension was used as the challenge inoculum, as outlined below. The bacterial concentration was confirmed the next day by culturing diluted aliquots of the inoculum on blood agar and counting the resulting colonies.
Neutrophil function assays
Bovine blood neutrophils were isolated as described previously. 5 The oxidative burst function of the neutrophils was measured by a flow cytometric method. 15 In brief, neutrophils were reconstituted to 2 × 107 cells/ml with Roswell Park Memorial Institute (RPMI) medium containing 20 mM HEPES and 5% fetal bovine serum (RPMI-H-FBS). The neutrophils were incubated with 10 mM H2DCFDA at 37 C for 30 minutes. Neutrophils were incubated with medium or with PMA (200 ng/ml in RPMI-H-FBS) at 37 C for 30 minutes. The reaction was stopped by placing the mixture in an ice bath, and the samples were analyzed within 20 minutes by flow cytometry. Fluorescence was measured on 10,000 cells within the gated neutrophil population by calculation of the difference between the mean fluorescence of the test and chilled samples.
Phagocytosis was measured by the uptake of FITC-labeled Staphylococcus aureus. FITC-labeled S. aureus was prepared by overnight incubation of S. aureus in BHIB at 37 C. Bacteria were heat killed in a 65 C water bath for 90 minutes, washed twice in PBS, resuspended in 1.0 M bicarbonate buffer (pH 9.5) containing 0.04% FITC, and incubated for 2 hours at 37 C in the dark. Bacteria were washed twice in PBS, resuspended in PBS, and stored in the dark at −20 C. Before the assay, the bacteria were washed twice with RPMI-H-FBS and resuspended to 108 bacteria/ml. Neutrophils were prepared to the concentration of 107 cells/ml with RPMI-H-FBS. A total of 5 × 107 bacteria/ml and 5 × 106 neutrophils/ml were incubated in 5-ml round-bottom tubes in a 37 C shaking water bath for 30 minutes. Phagocytosis was stopped by adding an equal volume of cold PBS containing 4% trypan blue and 2.5 mM ethylenediamine-tetraacetic acid, and the samples were held on wet ice. Negative control samples were prepared by combining cells and bacteria at 4 C. Samples were analyzed using flow cytometry as described above.
Neutrophil chemotaxis toward zymosan-activated serum (ZAS) was measured. Washed zymosan was suspended in normal bovine serum at a concentration of 7.1 mg/ml, incubated at 37 C for 40 minutes, and centrifuged to remove the zymosan, and the ZAS was stored at −80 C. Chemotaxis was measured using 30-µl disposable 96-well chemotaxis chambers with polyvinylpyrrolidone-free membranes containing 5 µm diameter pores (ChemoTx, NeuroProbe, Gaithersburg, MD). Neutrophils were prepared to a concentration of 107 cells/ml in Hanks' balanced salt solution containing 20-mM HEPES (HBSS-H) and labeled with 5-µg/ml calcein AM at 37 C for 30 minutes. After washing twice in HBSS-H, neutrophils were resuspended to 107 cells/ml. Lower chambers were loaded with 29 µl of 10% ZAS in HBSS-H or medium alone, the membrane was applied, and 29-µl neutrophils was added to the membrane surface. Wells containing 30 µl of neutrophils in the lower chambers served as the positive control to estimate the total fluorescence of the sample. All samples were prepared in triplicate. Plates were incubated at 37 C for 60 minutes, and with the filter still in place, the upper surface of the membrane was gently washed to remove nonmigrated neutrophils. The plate was centrifuged at 1,800 rpm for 10 minutes to force membrane-adherent neutrophils into the lower wells. The membrane was then removed and fluorescence in each well was measured on a Fluoromark fluorescence microplate reader using excitation and emission wavelengths of 485 and 538 nm, respectively. Chemotaxis was calculated as the difference in migration of neutrophils toward ZAS and toward HBSS, expressed as a percentage of the total fluorescence.
Experimental design
Sixteen 4- to 6-month-old Holstein bull calves, weighing 200–314 kg (experiment A) and 131–191 kg (experiment B), were acclimatized for 1 week before the experiments. Serum antibody titers to M. haemolytica leukotoxin were measured before the experiments. 10 Calves in experiment A had relatively high leukotoxin titres (7–10) and were challenged with 12.5 × 1010 cfu of M. haemolytica bacteria, whereas calves in experiment B had lower leukotoxin titres (2–5) and were challenged with 1.1 × 1010 cfu of bacteria. Calves were grouped on the basis of antibody titres and then randomly assigned within titre blocks to GCSF treatment groups. In experiment A, calves in the high- and low-titre groups had leukotoxin titres of 9–10 and 7–8.5, respectively. In experiment B, calves in the high- and low-titre groups had leukotoxin titres of 5–8 and 2–2.5, respectively. Calves in the GCSF treatment groups were administered 5 µg/kg GCSF once daily by subcutaneous injection. Calves in the non–GCSF-treated groups were administered the same volume of saline. All calves were challenged at day 8 (experiment A) or day 7 (experiment B) of treatment, with 25-ml cold PBS containing 12.5 × 1010 cfu (experiment A) or 1.1 × 1010 cfu (experiment B) of M. haemolytica bacteria, into the right bronchus by bronchoscope-placed catheter.
Clinical signs were measured once daily before challenge and three times daily after challenge by a veterinarian who had no knowledge of the treatment status (S. A. Youssef). Objective clinical parameters, including temperature, anorexia, strength, respiratory rate, rhinitis, tracheitis, and respiratory distress, were measured using predefined criteria from zero to five. A total objective clinical score was defined by the average of strength, respiratory rate, and respiratory distress. In addition, the overall clinical disease severity was subjectively estimated. Calves that were euthanatized were assigned no score for subsequent time points. All surviving calves were euthanatized at 5 days after bacterial challenge.
Assessment of gross and histologic lesions
Gross and histologic evaluation of the lung lesions was conducted without knowledge of the animal identification or treatment status. At necropsy, the percent lung involvement was estimated visually and by palpation, and the weights of the right and left lung were measured. Six sections were harvested from standardized areas of the right lung, and one section was taken from the left cranial lung lobe. Sections were fixed in 10% buffered formalin, processed routinely, and stained with hematoxylin and eosin.
Histologic lung lesions were scored as follows: 0—no lesions, 1—mild, 2—moderate, and 3—severe. Histologic parameters included the severity of alveolar and airway filling with fibrin and neutrophils, interlobular and pleural fibrin deposition, and foci of parenchymal necrosis. In mild lesions, alveoli and airways contained low amounts of inflammatory cells, fibrin, and edematous fluid but necrotic areas were absent. Moderate lesions contained extensive fibrin deposition, interlobular edema, and necrotic areas affecting < 50% of the section. Severe lesions contained extensive fibrin deposition and areas of necrosis affecting > 50% of the pulmonary tissue. The mean histologic lesion score for each calf was calculated by averaging the lesion scores of the seven sections of each lung.
Bacteriologic analysis
Lung tissue samples from predetermined sites (three from the right lung and one from the left lung) were collected at necropsy. Using a sterile tissue homogenizer, 1.0 g of tissue from each site was homogenized in 9 ml PBS. Tenfold dilutions were prepared, and 50 µl of each dilution was plated on trypticase soy agar and incubated at 37 C for 24 hours. Colonies were counted and the results expressed as the bacterial count per gram of lung tissue. A swab from one kidney of each calf was cultured routinely on blood agar.
Statistical analysis
The hematology data were analyzed by repeated measures two-way analysis of variance (ANOVA). The clinical signs scores and the necropsy lesions were analyzed by ANOVA and considered the parameters of experiment number, GCSF treatment, and titre group. Within-experiment data were evaluated by two-way ANOVA and Bonferroni multiple comparisons test. The necropsy lung weight : body weight (LW : BW) ratios were analyzed by one-way ANOVA with Bonferroni multiple comparisons test. All values are listed as mean ± SEM.
Results
Hematology
Treatment with GCSF induced a significant (P < 0.0001), time-dependent increase in the blood neutrophil count (Fig. 1). Significantly increased numbers of mature and band neutrophils were detected in the blood within 5 and 6 days of treatment in experiments B and A, respectively. The mean maximal blood neutrophil counts in non–GCSF-treated and GCSF-treated calves before bacterial challenge were 5.6 ± 0.7 × 109/liter and 25.4 ± 2.7 × 109/liter, respectively. The mean maximal band neutrophil counts in non–GCSF-treated and GCSF-treated calves before bacterial challenge were 8.4 ± 1.3 × 109/liter and 0.4 ± 0.2 × 109/liter, respectively (Fig. 1).
Changes in blood mature and band neutrophil numbers in calves treated with GCSF and challenged with M. haemolytica. Compared with untreated calves (white bars), GCSF-treated calves (black bars) developed increased numbers of mature neutrophils (A,C) and band neutrophils (B,D) in both experiment A (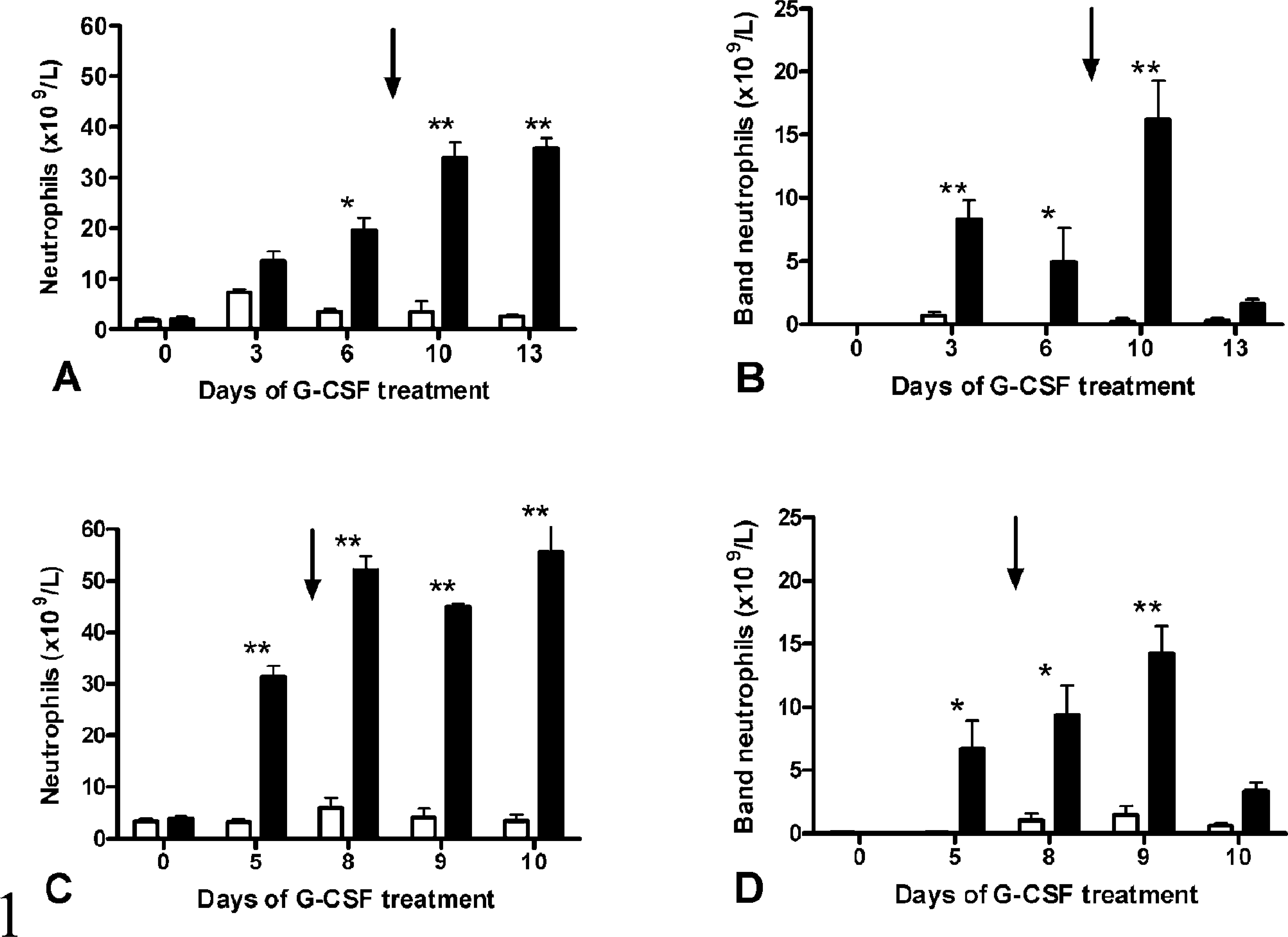
In experiment A (in which calves received a higher challenge dose of M. haemolytica), all non–GCSF-treated calves developed a reduction in blood neutrophil numbers after bacterial challenge (Fig. 1A)). Band neutrophils were present in low number in these calves before bacterial challenge (0.22 ± 0.14 × 109/liter, mean ± SEM) but increased after challenge (0.40 ± 0.23 × 109/liter) (Fig. 1B)). In contrast, in the GCSF-treated calves, mature neutrophil numbers remained stable or continued to rise after bacterial challenge. The average blood neutrophil counts in non–GCSF-treated and GCSF-treated calves, in experiment A at 5 days after bacterial challenge, were 2.5 ± 0.5 × 109/liter and 35.9 ± 2.5 × 109/liter, respectively (Fig. 1A)). Two non–GCSF-treated calves became severely neutropenic (0.12 × 109/liter and 0.16 × 109/liter) 2 days after challenge.
In experiment B (in which calves received a lower challenge dose of M. haemolytica), mature neutrophil numbers continued to rise after bacterial challenge in the GCSF-treated calves, and the numbers of band neutrophils reached a zenith at 9 days of GCSF treatment (3 days after bacterial challenge) and declined by day 10 (Fig. 1C)). The non–GCSF-treated calves also developed neutrophilia with band cells after bacterial challenge but this was of a lower magnitude compared with the GCSF-treated calves (Fig. 1D)).
Clinical signs
After challenge with M. haemolytica, all calves exhibited depression and a variable degree of tachypnea, anorexia, and recumbency. Coughing and nasal discharge were uncommonly observed. Two non–GCSF-treated calves in experiment A developed neutropenia, labored breathing, and profound depression and were euthanatized 2 days after bacterial challenge (Fig. 2).
Clinical signs in untreated and GCSF-treated calves after intrabronchial challenge with M. haemolytica in experiments A (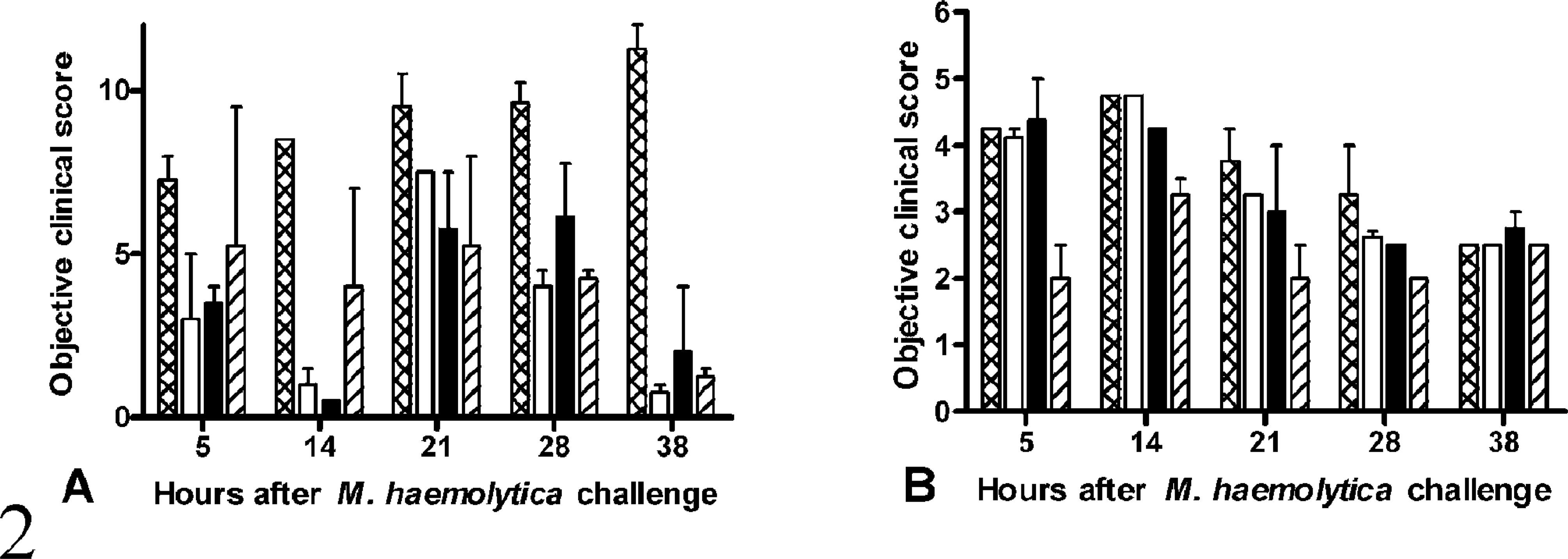
Uniform differences in clinical signs between the GCSF-treated and untreated calves were not obvious during the experiment. However, ANOVA of the clinical scores revealed significant effects of GCSF treatment groups (P = 0.001), antibody titre to M. haemolytica leukotoxin before the experiment (P < 0.001), and differences between experiments A and B (P < 0.001), as well as significant interactive effects among these parameters. In the calf groups with intermediate antibody titres to M. haemolytica leukotoxin (i.e., the low-titre group in experiment A and the high-titre group in experiment B, with titres of 5–8.5), GCSF-treated calves had lower clinical scores than non–GCSF-treated calves. This effect was significant for experiment A (P = 0.004, objective clinical score, assessed by repeated measures two-way ANOVA) but was not significant for experiment B (P = 0.067). In contrast, GCSF treatment did not affect the clinical scores in calves with either very high titres (experiment A, high-titre group, which had titres of 9–10; see Fig. 2A) or very low titres (experiment B, low-titre group, which had titres of 2–2.5; see Fig. 2B).
Neutrophil function assays
Neutrophil oxidative burst measurements varied significantly (P < 0.01) from day to day, but treated calves did not have consistently high or low responses compared with untreated animals. There were no consistent differences in neutrophil phagocytosis or chemotaxis between the non–GCSF-treated and GCSF-treated calves.
Gross lesions
The pulmonary lesions in all calves were qualitatively similar but differed in severity and extent. Acute fibrinous, hemorrhagic, and necrotizing pneumonia was the most characteristic lesion (Fig. 3). Affected lung tissue was consolidated and congested and had prominent interlobular septa due to fibrin deposition and edema. When cut transversely, irregularly shaped areas of necrosis were often present. These areas were gray, round to oval, 0.1–2 cm in diameter, dry in consistency, and occasionally outlined by a thin, red inflammatory rim. Frequently, acute fibrinous adhesions between the right lung lobe and parietal pleura were present. These lesions were mainly present in the right lung and to a limited extent in the left lung.
Typical gross lesions after intrabronchial challenge with M. haemolytica included consolidation, reddening, and fibrinous pneumonia, affecting mainly the right lung lobe.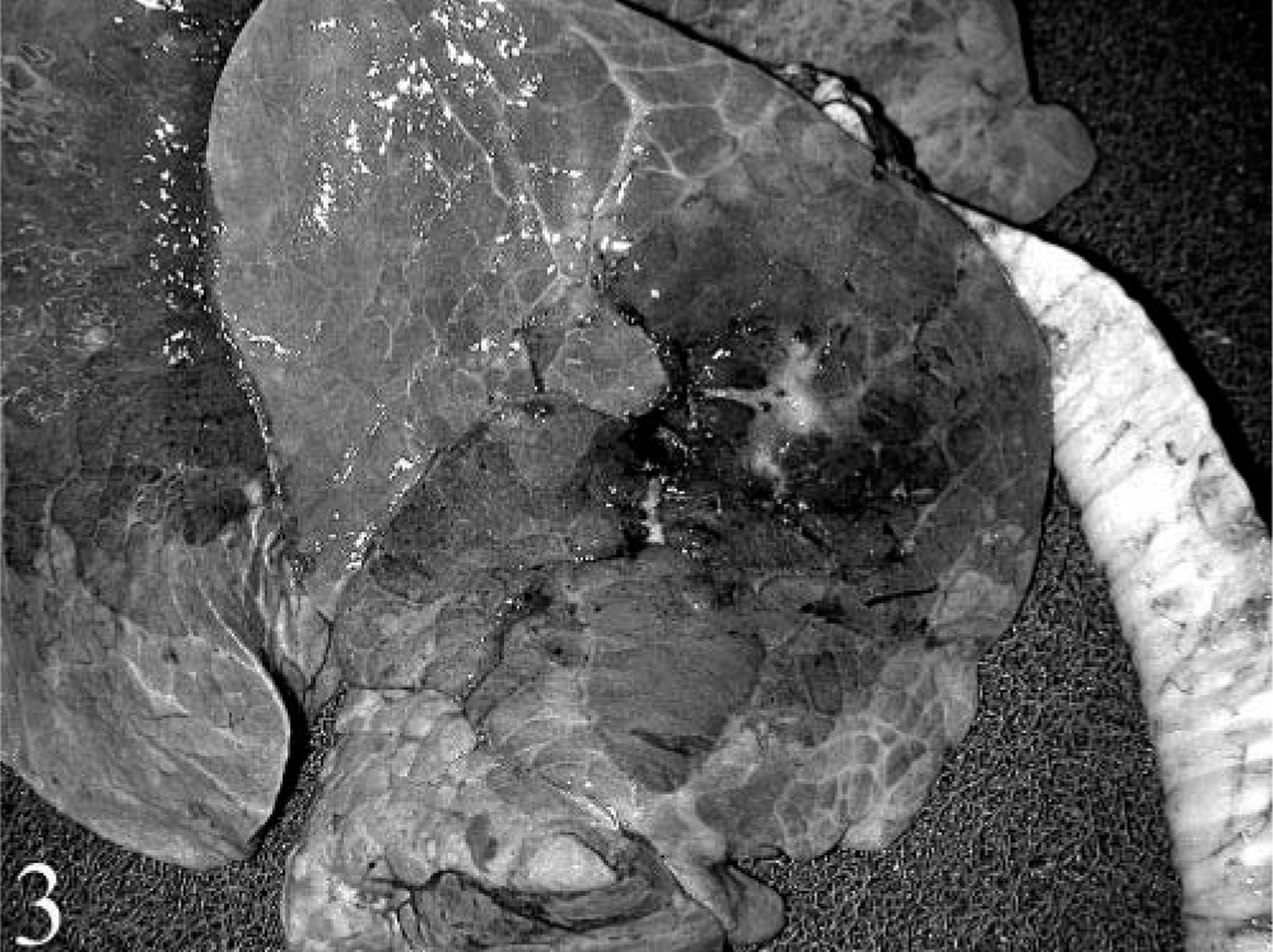
GCSF-treated calves had a 37% reduction in gross lung lesions compared with nontreated calves (Fig. 4). This difference was significant (P < 0.05) when the effect of previous antibody titer to leukotoxin was considered. Overall, LW : BW ratios were similar in GCSF-treated and nontreated calves (Fig. 5). In the low-titre group in experiment A, GCSF-treated calves had lower LW : BW ratios than non–GCSF-treated calves (P = 0.02), but significant differences were not present in the other groups.
Percent of lung affected by consolidation after intrabronchial challenge with M. haemolytica in experiments A (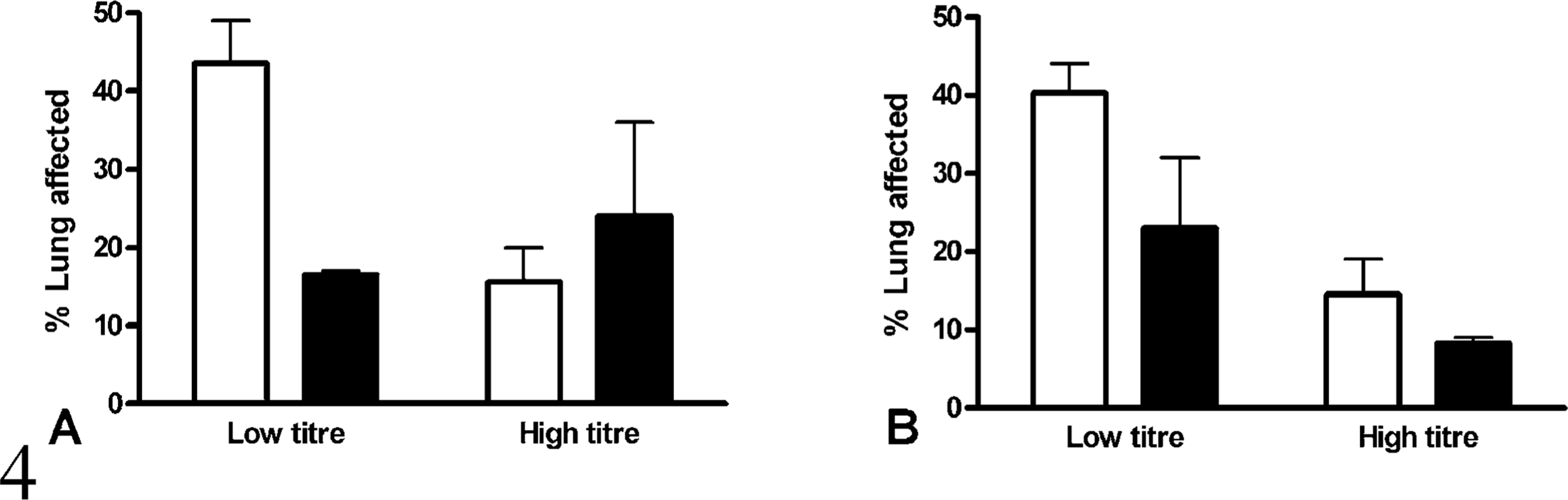
Lung weight : body weight (LW : BW) ratios were used as a measure of pulmonary edema and cellular infiltration after intrabronchial challenge with M. haemolytica. The graphs represent experiments A (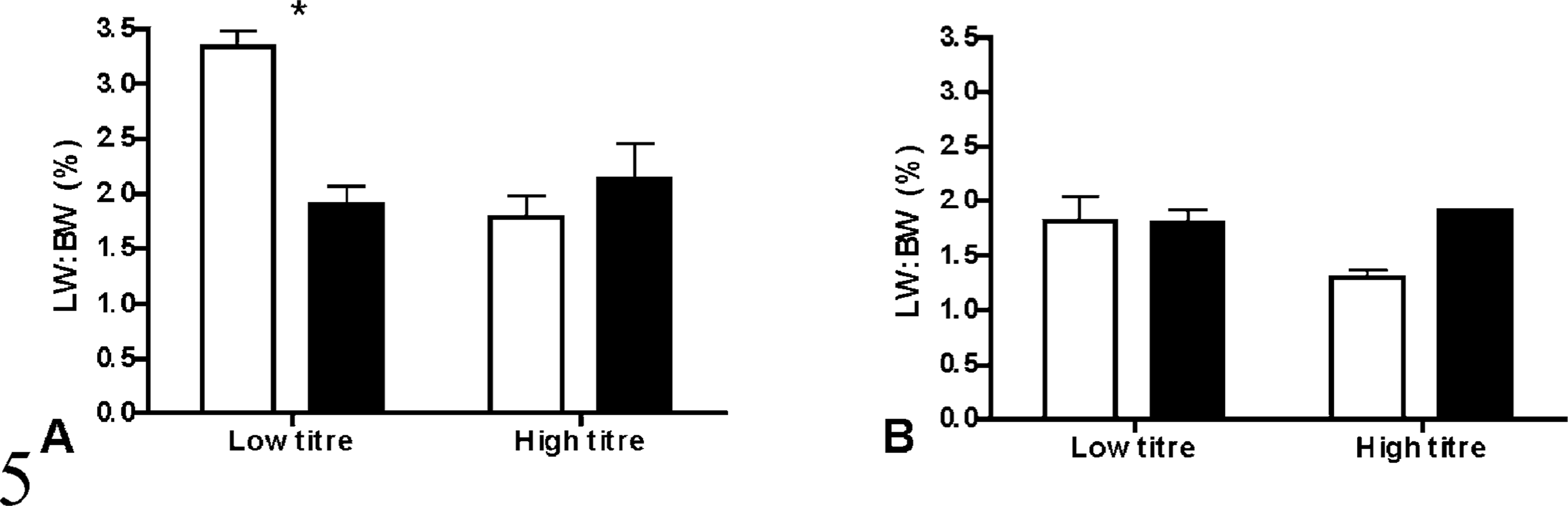
The bronchial lymph nodes of all calves were enlarged and edematous. In euthanatized calves from the untreated group, generalized hemorrhage was present in the bronchial lymph node. Acute multifocal petechial hemorrhages were present in the subcutaneous tissue of the head and neck of the two calves of the untreated groups that were euthanatized 2 days after bacterial challenge.
Histologic lesions
Generally, the most severe lesions of acute fibrinous or fibrinosuppurative and necrotizing pneumonia were present in the right cranial lung lobe of the GCSF treated and untreated calves. Lesions were subtle or absent in sections taken from the left lung. There was extensive filling of the alveoli and airways with fibrin, necrotic neutrophils, and few macrophages. Multifocal, coalescing, irregularly shaped areas of coagulation necrosis were present and were lined by a thin zone of intense neutrophil infiltration (Fig. 6). The visceral pleura and the interlobular septa were extensively filled with fibrin and their lymphatic vessels had numerous fibrin thrombi. Significant differences in histologic scores between GCSF-treated and untreated calves were not detected.
Alveoli are filled with fibrin, neutrophils, and proteinaceous fluid and focal areas of coagulation necrosis demarcated by a band of necrotic neutrophils (arrowheads). HE Bar = 200 µm.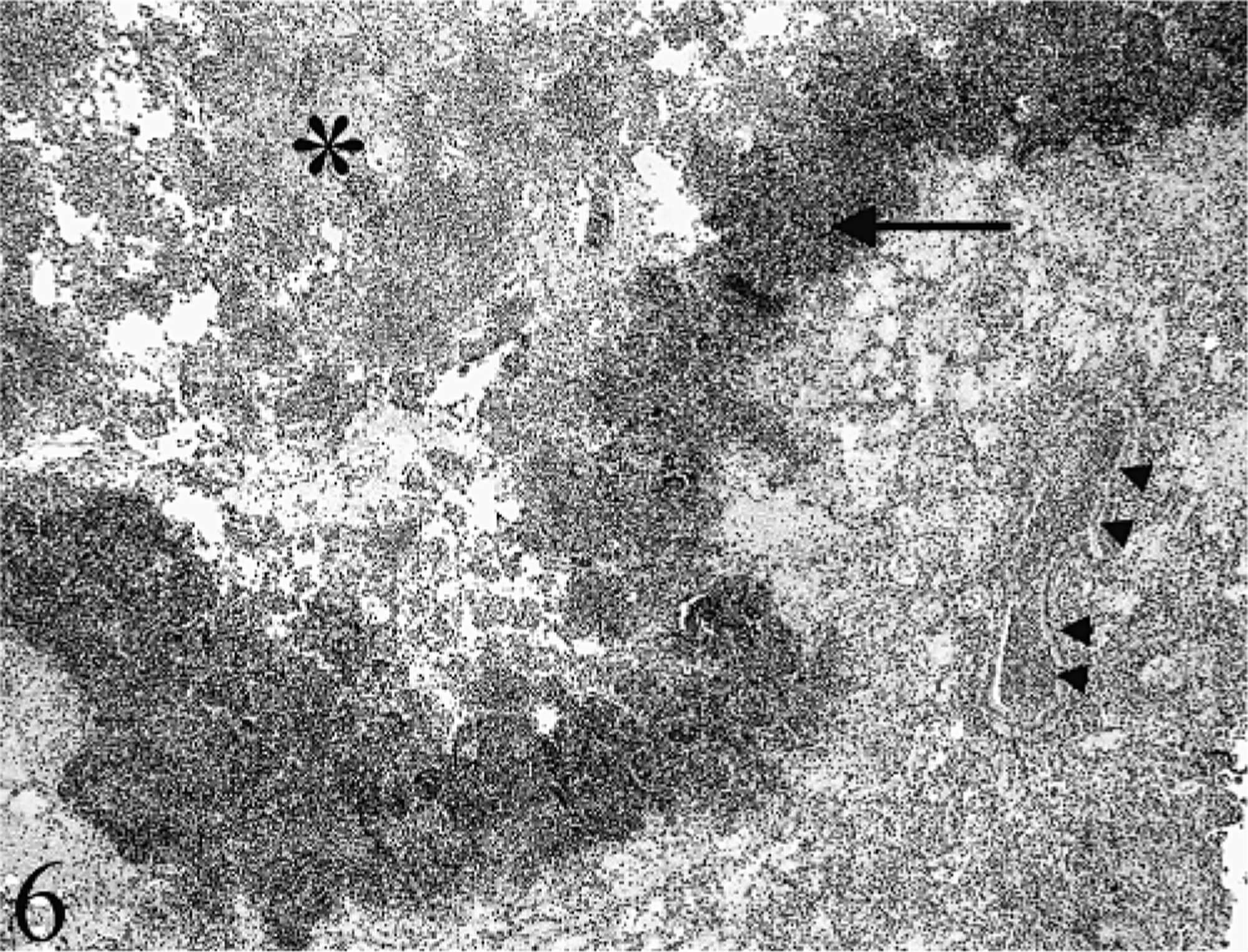
Bacterial culture
Pure cultures of M. haemolytica were recovered from the lung of all calves. In experiment A, GCSF treatment was associated with a nonsignificant decrease in the average M. haemolytica concentration in the lung. The M. haemolytica concentrations were 10.5 ± 1.3 × 107 cfu/g and 8.4 ± 1.2 × 107 cfu/g of lung tissue in the untreated and GCSF-treated calves, respectively. M. haemolytica bacteria were isolated from the kidney of two of four untreated calves (three colonies per plate) and three of four GCSF-treated calves (one colony per plate).
Discussion
The aims of this study were to evaluate the effect of GCSF on bovine blood neutrophil numbers and function and to investigate the effect of GCSF-induced neutrophilia on the response to pulmonary challenge with M. haemolytica. Calves that were treated with GCSF developed neutrophilia but did not have detectable changes in neutrophil function. After challenge with M. haemolytica, GCSF treatment protected against the development of neutropenia and reduced the size of the pneumonic lung lesion, although the effect on the severity of clinical signs was complex.
GCSF induced a neutrophilia that was detectable within 5 days of treatment, similar to previous studies. 6,7 Neutrophil functions, including chemotaxis, oxidative burst, and phagocytosis, were not significantly different between the GCSF treated and nontreated groups. These results are in general agreement with a previous study, where recombinant human GCSF did not affect a panel of function tests, using neutrophils from clinically normal and postparturient cows with retained placenta, metritis, or mastitis. 4 In contrast, other studies found that neutrophils from prepartum cows treated with GCSF had enhanced phagocytosis of S. aureus and neutrophil-mediated cytotoxicity and decreased chemotactic function, iodination, and bactericidal activity, but no significant difference in the oxidative burst. 11 In this study, two nontreated calves became severely neutropenic and were euthanatized 2 days after challenge. None of the GCSF-treated calves experienced neutropenia. Thus, one of the beneficial effects of GCSF therapy in calves with pneumonia may be to augment marrow production of neutrophils during an acute inflammatory response. GCSF also prevents development of severe neutropenia in a cecal puncture model of sepsis in rats. 13,14 Whether this ability to sustain neutrophil recruitment to the lung would, by itself, affect clearance of bacteria from the lung and overall disease severity is uncertain.
Treatment of calves with GCSF before challenge with M. haemolytica significantly reduced the gross lung lesions when the effect of previous antibody titre to leukotoxin was considered. The effect of GCSF treatment on other parameters of disease severity was more complex. In calves with low or high serum antibody titres to M. haemolytica leukotoxin, GCSF treatment did not confer a reduction in the severity of clinical signs. In contrast, in the calves with intermediate titres, clinical signs were lower in GCSF-treated compared with non–GCSF-treated calves. Although the results were significant at P < 0.05, caution is warranted in the interpretation of these interactive effects because each combination of GCSF treatment, antibody titre, and experiment number was represented by only two animals per group. Nevertheless, the data suggest that GCSF treatment may partially protect calves with intermediate antibody titres against M. haemolytica challenge, whereas animals with low and high antibody titres are susceptible and resistant, respectively, to M. haemolytica regardless of GCSF treatment.
As an explanation for this phenomenon, we speculate that increased numbers of neutrophils may not confer additional resistance in calves that already have high protective antibody titres, and calves with little or no antibody to M. haemolytica may be susceptible to challenge with high numbers of bacteria regardless of their neutrophil status. However, calves with intermediate antibody titres may benefit from augmented neutrophil responses, particularly if these cells are effective in phagocytosing and killing bacteria that have been opsonized by antibodies to M. haemolytica surface proteins. As an alternative explanation, neutrophil responses may be ineffective and potentially harmful in calves with low antibody titres to leukotoxin because the leukotoxin induces lysis of neutrophils as they infiltrate the lung. In this situation, neutrophils may no longer be able to effectively kill bacteria, yet may continue to cause injury to lung tissue. In contrast, neutralization of leukotoxin in calves with intermediate or high titres may augment neutrophil survival in the presence of M. haemolytica infection, allowing these cells to ingest and kill bacteria. Thus, the contribution of neutrophils to protection of the lung against M. haemolytica infection must be considered in the context of other interacting mechanisms of pulmonary defense.
Because our in vitro studies did not show any significant effect of GCSF on neutrophil function, we infer that the reduction of the pneumonic lung lesions in GCSF-treated calves was an effect of the neutrophilia induced by GCSF. This result is in agreement with previous results where the GCSF-induced neutrophilia in rat models of sepsis resulted in rapid bacterial clearance and a milder disease. 12,13 We cannot completely rule out the possibility that GCSF has effects on innate immunity through other mechanisms. However, the published literature suggests that the effect of GCSF on monocyte/macrophage function is minor compared with that of other colony-stimulating factors. 2,25 Similarly, although GCSF may promote release from the marrow of immature neutrophils that have a reduced capacity to infiltrate the lung tissue, the finding of a similar degree of neutrophil infiltration in the GCSF-treated versus untreated calves argues against this possibility.
The roles played by neutrophils in the pathogenesis of bovine pneumonic pasteurellosis are diverse and complicated. Pneumonia caused by M. haemolytica has been a paradigm of neutrophil-induced tissue injury in which the characteristic lesions of pulmonary necrosis are thought to represent tissue injury induced by inflammatory mediators arising from the excessive inflammatory response. These concepts are based largely on three previous studies of experimental M. haemolytica challenge in neutrophil-depleted calves, which generally developed less severe lung lesions than their neutrophil-replete cohorts. 3,23,24 In contrast, this study demonstrates that the neutrophilia induced by GCSF treatment does not result in more severe disease after M. haemolytica challenge and in fact lessens some measures of disease severity. There are several differences among these studies that may explain some of the observed differences. First, the studies by Slocombe and by Weiss used calves that were less than 3 weeks of age, whereas this study and that of Breider used 4- to 6-month-old and 130–230 kg calves, respectively. Because age-related changes in neutrophil function have been demonstrated in calves and other species, these studies may be more relevant to the pathogenesis of pneumonia in feedlot calves. Second, the challenge doses of M. haemolytica used by Breider were comparable to those in this study but 40- to 100-fold higher than in the other studies. Third, calves in the previous studies were euthanatized much earlier—at 6 hours—after bacterial challenge, whereas our study was extended to allow us to monitor the development and resolution of clinical signs. Finally, the antibody titres to M. haemolytica were quite variable in this study but were either more uniform or not determined in the previous studies. Although this titre variation probably led to variability in the severity of disease, including these titres in the analysis did permit us to investigate the idea that antibody titre may partially determine whether GCSF treatment influences the severity of the disease.
In conclusion, GCSF treatment induced neutrophilia and protected calves from the severe neutropenia that is noticed in some challenged but non–GCSF-treated calves. The neutrophilia induced by GCSF treatment reduced the severity of the pneumonia that developed after M. haemolytica challenge, and this protective effect of GCSF treatment was most noticeable in calves with intermediate titres to M. haemolytica leukotoxin. The protection conferred by GCSF treatment was only partial and probably would not justify use of this cytokine for the prevention of shipping fever pneumonia, at least under the conditions used in this study. Nevertheless, the results challenge the belief that neutrophil responses are always harmful in pneumonic pasteurellosis and suggest that the contribution of neutrophils to bacterial clearance and to tissue injury are complex and probably depend on the age of the calf, the antibody titre, the challenge dose of bacteria, and the stage of the disease. Finally, this study suggests that strategies for augmenting neutrophil responses may be useful in the prophylaxis of bacterial pneumonia in cattle.
Footnotes
Acknowledgements
We thank Betty Anne McBey, Doug Hodgins, and Patricia Shewen for measuring antibody titres and for assistance in developing the infection protocol. This study was funded by the Natural Sciences and Engineering Research Council of Canada and by the Ontario Ministry of Agriculture and Food Beef Research Program.
