Abstract
The status of the erbB-2 (human epidermal growth factor receptor 2/neu) proto-oncogene in canine osteosarcoma (OSA) has not been reported previously. In this study we used real-time reverse transcriptase polymerase chain reaction to evaluate erbB-2 expression in seven canine OSA cell lines and 10 canine OSA tissue samples. We determined erbB-2 to be significantly overexpressed in 86% (six of seven) of the cell lines and 40% (4 of 10) of the OSA tissues samples. Given the importance of erbB-2 in human breast cancer, the finding of erbB-2 overexpression in canine OSA may be important in further understanding the pathogenesis and possible therapies of OSA.
Osteosarcoma (OSA) is the most frequently occurring highly malignant bone tumor in canine as well as human patients. 3,5,8 The use of canine OSA as a model of human OSA is compelling because they bear the following striking resemblances: males are more commonly affected in both species, metaphyseal sites in appendicular bones are most often affected, the etiology is unknown, and less than 10% of patients have documented metastasis at presentation. Over 90% of canine OSAs show high-grade histology with the metastatic rate more than 80% with amputation alone, primarily of the lung. 2,12,14 Research into the molecular mechanisms underlying canine OSA would provide the necessary information toward better treatment of both canine and human patients.
erbB-2 is a proto-oncogene that encodes human epidermal growth factor receptor 2 (HER-2). HER-2 is a 185-kd transmembrane glycoprotein of the tyrosine kinase family of receptors. 4 To date, no known ligand has been found for HER-2; however, activation of HER-2 induces a cascade of mechanisms resulting in cell transformation and growth. 6 HER-2 has been extensively evaluated in both human and canine breast cancer. 1 In both spaces HER-2 overexpression is correlated to poor prognosis. In addition, a correlation between HER-2/erbB-2 overexpression and poor prognosis and decreased survival in human OSA patients has been proposed in several recent studies. 5,9 In light of these data, HER-2/erbB-2 expression may be a useful prognostic indicator in canine OSA.
We measured erbB-2 expression levels in mouse fibroblast cells, cell lines derived from canine osteogenic sarcomas, and canine OSA samples, with the ultimate purpose of evaluating the potential utility of canines as models for human OSA as well as measuring erbB-2 gene expression as a diagnostic indicator in canine OSA. To accomplish this goal, NIH/3T3 and B104-1-1 (NIH/3T3 cells engineered to overexpress erbB-2), 7 both mouse fibroblast cell lines, and seven osteogenic canine cell lines were used. OSA cell lines were grown in modified minimum essential medium and 10% fetal bovine serum (FBS). NIH/3T3 and B104-1-1 cells were grown in Dulbecco modified Eagle medium (GIBCO, Grand Island, NY) and 10% FBS. All cells were maintained at 37 C in 5% CO2. OSA and control bone samples from the same individual were also obtained from 10 different canine patients presenting to the Colorado State University Veterinary Teaching Hospital. Histopathology was performed on all samples to confirm the presence or absence of OSA. In addition, samples were flash frozen in liquid nitrogen and stored at −80 C until RNA isolation. Cell lines were detached from flasks with 0.25% trypsin and disrupted using QIAshredder (Qiagen, Valencia, CA). Total RNA was then isolated from the samples using RNeasy Mini Kit (Qiagen). RNA from OSA tissue and normal bone was isolated by homogenizing the tissue under liquid nitrogen, disrupting the homogenate in a QIAshredder (Qiagen), and collecting RNA using the RNeasy Mini Kit (Qiagen). One microgram of total RNA was DNase treated and reverse transcribed using Invitrogen reagents (Invitrogen, Grand Island, NY). For each sample two polymerase chain reactions (PCRs) were performed using glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and erbB-2 primers. The sequences of primers used were GAPDH forward 5′-GGC AAA TTC CAC GGC ACA GTC AAG GC-3′, reverse 5′-CAG AGG GGC CGT CCA CGG TCT TCT GGG TGG-3′; erbB-2 forward 5′-ACC AGG CCC TGC TCC ATA GT-3′, reverse 5′-GTA CTC GGC ATT CCT CCA CG-3′. PCR reactions totaled 50 µl and comprised 6 µl of complementary DNA, 1× PCR buffer (Sigma, St. Louis, MO), 2 mM MgCl2, 0.2 mM deoxynucleoside triphosphate, 0.12 mM each of forward and reverse primers, 1 unit of Platinum Taq (Invitrogen), and PCR-grade water to volume. Thermocycler parameters were 15 seconds at 95 C, 30 seconds at 55 C, and 1 minute at 72 C for 30 cycles. Each reaction included an initial denaturation at 95 C for 5 minutes and a final extension at 72 C for 2 minutes.
We performed real-time reverse transcriptase (RT)–PCR on all samples using a Bio-Rad iCycler (Bio-Rad, Hercules, CA). For this assay, two PCR reactions per sample were performed using the parameters described above with the following addition: 1 µl Sybr Green (1:1,000 of 10,000× stock; Molecular Probes, Eugene, OR), 1 µl fluorocein (10 nM; Bio-Rad). After PCR amplification a melt curve was performed to confirm that nonspecific products were not being amplified.
Each real-time reaction was analyzed by selecting the amplification cycle where the PCR product of interest first crosses the threshold of baseline fluorescence. This threshold cycle (CT) was further defined as the fractional cycle number at which the fluorescence reaches 10 times the standard deviation of baseline. The relative expression of erbB-2 in each sample was calculated as described in the footnotes to Tables 1–3.
Real-time RT-PCR measuring expression of erbB-2 in NIH/3T3 and B104-1-1 (NIH/3T3 cells engineered to overexpress erbB-2) cell lines.

∗ All samples were run in triplicate.
† ΔCT was calculated as (gene of interest CT) – (house keeping gene CT).
‡ Relative expression was calculated as 2−((tumorΔCT) − (normal ΔCT)).
Using GADPH as a control for the level of gene expression, we compared the levels of erbB-2 expression in the B104-1-1 and NIH/3T3. Real-time RT-PCR of the B104-1-1 cells showed that, as expected, they expressed erbB-2 33.8 times higher than NIH/3T3 cells (Table 1). These data are consistent with other data from our laboratory, which show real-time technology to be a more highly sensitive methodology to study gene transcription. erbB-2 expression analysis of the seven osteogenic canine cell lines revealed that six of the seven cell lines (86%) significantly overexpressed erbB-2 (Table 2), further confirming erbB-2 overexpression in a high percentage of canine OSAs. Comparison of canine OSA with unaffected bone from the same patient was also done to minimize the effects of interbreed variation in erbB-2 expression patterns. When these tissues were evaluated with real-time RT-PCR, 4 of the 10 samples (40%) analyzed significantly overexpressed erbB-2 (Table 3).
Expression of erbB-2 in canine OSA cell lines compared with the average of normal bone samples.
∗ All samples were run in triplicate.
† ΔCT was calculated as (gene of interest CT) – (house keeping gene CT).
‡ Relative expression was calculated as 2-((tumor ΔCT) - (normal ΔCT)).
§ Average of 10 normal bone samples was used as a comparison. ΔCT = 13.062 ± 0.849.
‖ erbB-2 is positive if relative expression is 2 or more.
Real-time RT-PCR evaluating the expression of erbB-2 in canine OSA compared with normal bone, within the same animal, and survival time.
∗ All samples were done in triplicate.
† ΔCT was calculated as (gene of interest CT) – (house keeping gene CT).
‡ Relative expression was calculated as 2-((tumor ΔCT) - (normal ΔCT)).
§ erbB-2 is positive if relative expression is 2 or more.
‖ Data point was removed from survival data because of surgical complications.
Immunohistochemistry (IHC) was used to determine whether HER-2 expression was correlated to real-time PCR erbB-2 data in NIH/3T3, B104-1-1, and the seven osteogenic canine cell lines; in addition, 2 of the 10 clinical cases were also stained for HER-2. Cells were plated onto permanox chamber slides (Nalge Nunc, Naperville, IL) and incubated at 37 C in 5% CO2 for 48 hours. Cells were then washed with phosphate-buffered saline (PBS), and slides were fixed for 10 minutes in 1% paraformaldhyde and allowed to air dry before staining. Fixed-cell slides were rinsed with PBS and permeabilized with 0.1% Triton-X 100 and 0.4% goat serum (Vector Laboratories, Burlingame, CA) for 20 minutes at 37 C. Cell slides were then incubated with HER-2 (Neu c18) primary antibody (Santa Cruz Biotechnology, Santa Cruz, CA) for 15 minutes at 37 C. After the PBS wash, cell slides were incubated with a biotinylated secondary antibody (Vector Laboratories) for 30 minutes at 37 C. The cell slides were then washed in PBS and incubated with avidin biotinylated horseradish peroxidase complex (Vector Laboratories) for 30 minutes at 37 C. After the PBS wash cell slides were immunodetected with 3-amino-9-ethylcarbazole (Biomedia, Foster City, CA) for 10 minutes at 37 C. Finally, the cell slides were counterstained with Gills III hematoxylin and coverslipped. Tissue sections (4 µm) were mounted on plus slides and deparaffinized using two Xylene washes and rehydrated in graded ethanol. Sections were peroxidase blocked using 3% hydrogen peroxide for 5 minutes. Antigen retrieval on rehydrated sections was done in 1× Citra Antigen Retrieval (Bio Genex, San Ramon, CA). Sections were washed in PBS and blocked with 0.4% goat serum. After the blocking step, IHC proceeded as above.
Cells and sections stained for HER-2 were evaluated under 40× objective and scored as HER-2 positive or HER-2 negative compared with the mouse fibroblast cell lines. Consistent with our analysis of erbB-2 gene transcription, the OSA cell lines showed differential HER-2 expression (Figs. 1, 2, 3–4). This differential expression of HER-2 closely followed the expression levels for the erbB-2 real-time data presented here. In addition, IHC HER-2 expression in the sections from clinical samples paralleled real-time erbB-2 expression from these same samples (Figs. 5, 6, 7–8) but to a lesser extent than was observed in the cell lines. This result was expected because IHC is inherently a less-sensitive technique than quantitative real-time PCR. Additionally, within different areas of the same tumor there are different percentages of tumor anaplasia, collagen, necrosis, and inflammation, all of which could alter the IHC staining pattern. These results support the usefulness of real-time PCR as a rapid, exquisitely sensitive, and effective way to determine genetic expression patterns within these tumors.
NIH/3T3 mouse fibroblast cell line without intracytoplasmic IHC reactivity for HER-2. The chromogen is AEC, and Gills III hematoxylin is the counterstain. Bar = 20 µm.
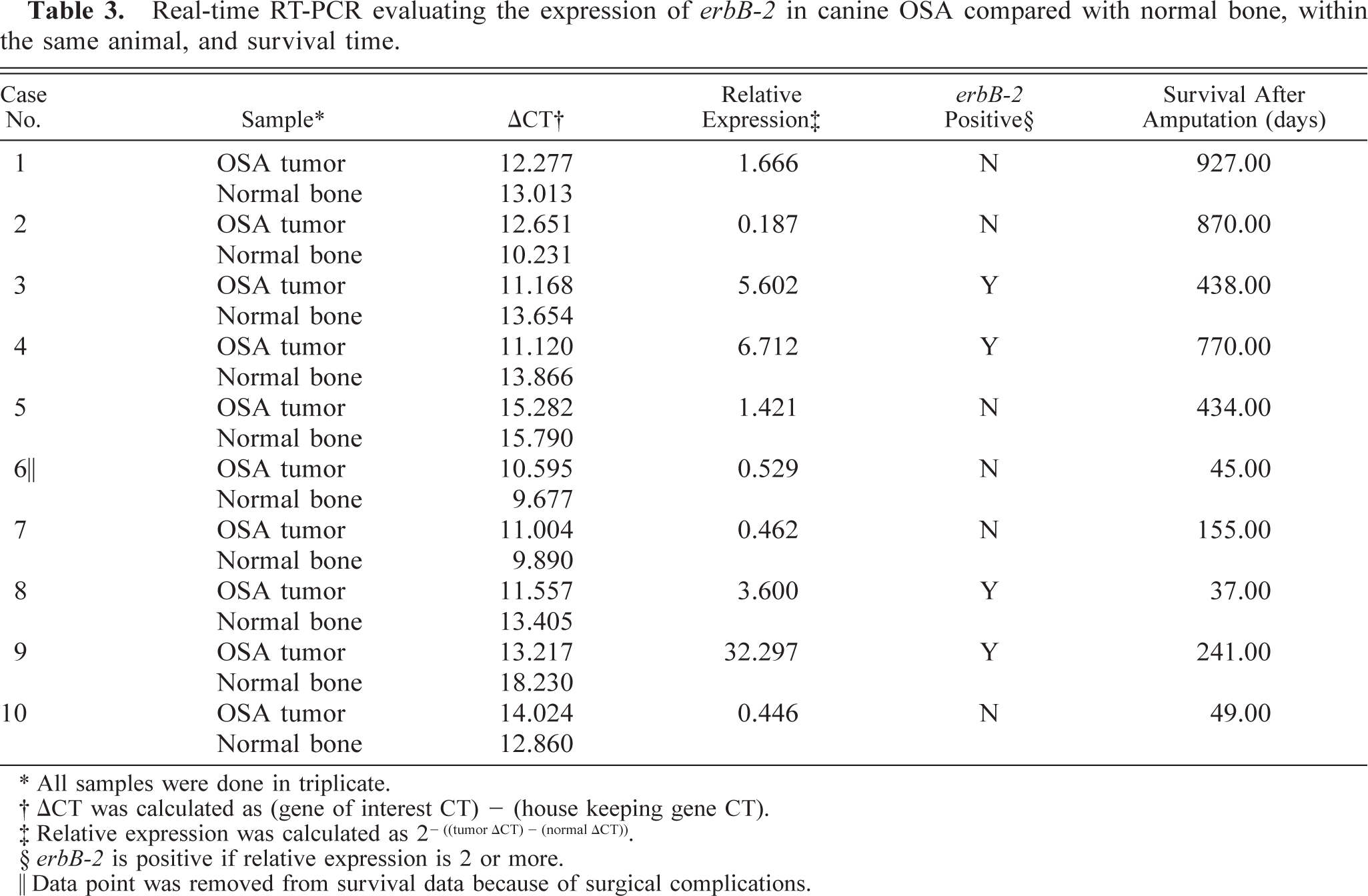
B104-1-1 mouse fibroblast cell line (NIH/3T3 engineered to overexpress erbB-2). Cells exhibit diffuse intracytoplasmic IHC reactivity for HER-2 (arrows). The chromogen is AEC, and Gills III hematoxylin is the counterstain. Bar = 20 µm.
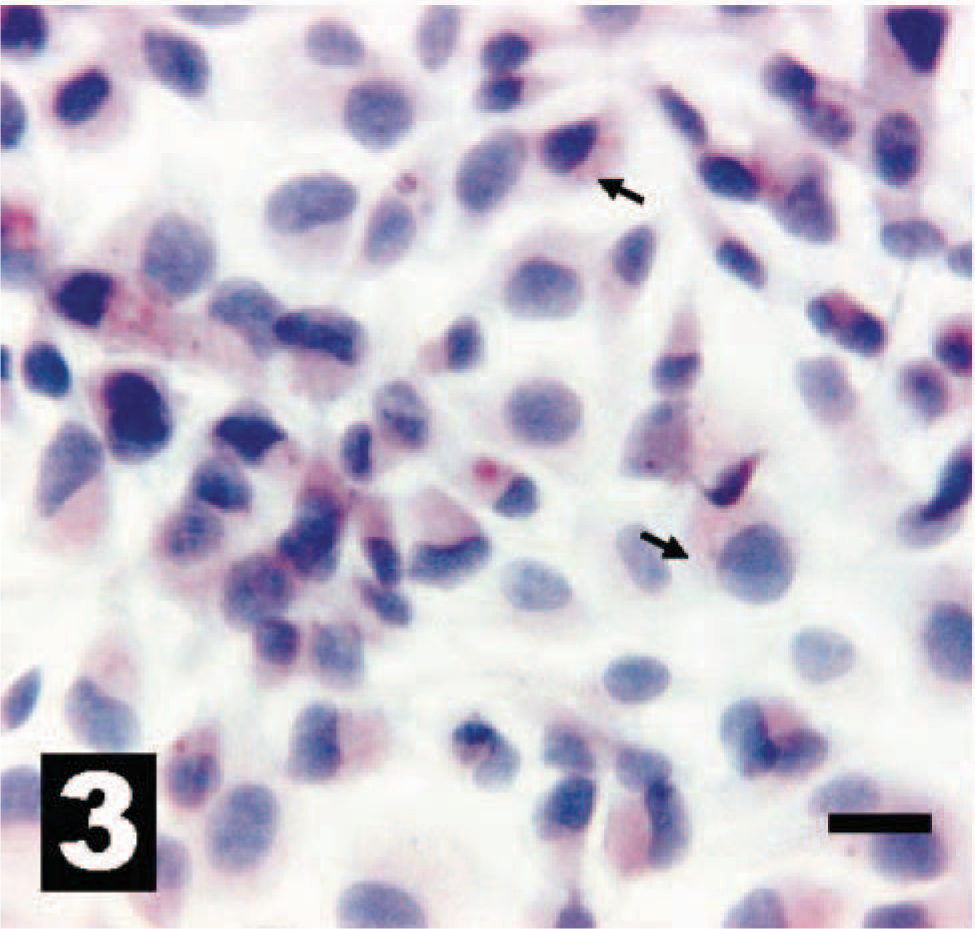
Canine osteosarcoma cell line OSA-CL2 weakly overexpresses erbB-2 by real-time PCR analysis (Table 2) and exhibits light to moderate intracytoplasmic IHC reactivity for HER-2 (arrows). The chromogen is AEC, and Gills III hematoxylin is the counterstain. Bar = 20 µm.
Canine osteosarcoma cell line OSA-CL5 overexpresses erbB-2 by real-time PCR analysis and exhibits diffuse intracytoplasmic IHC reactivity for HER-2 (arrow). The chromogen is AEC, and Gills III hematoxylin is the counterstain. Bar = 20 µm.
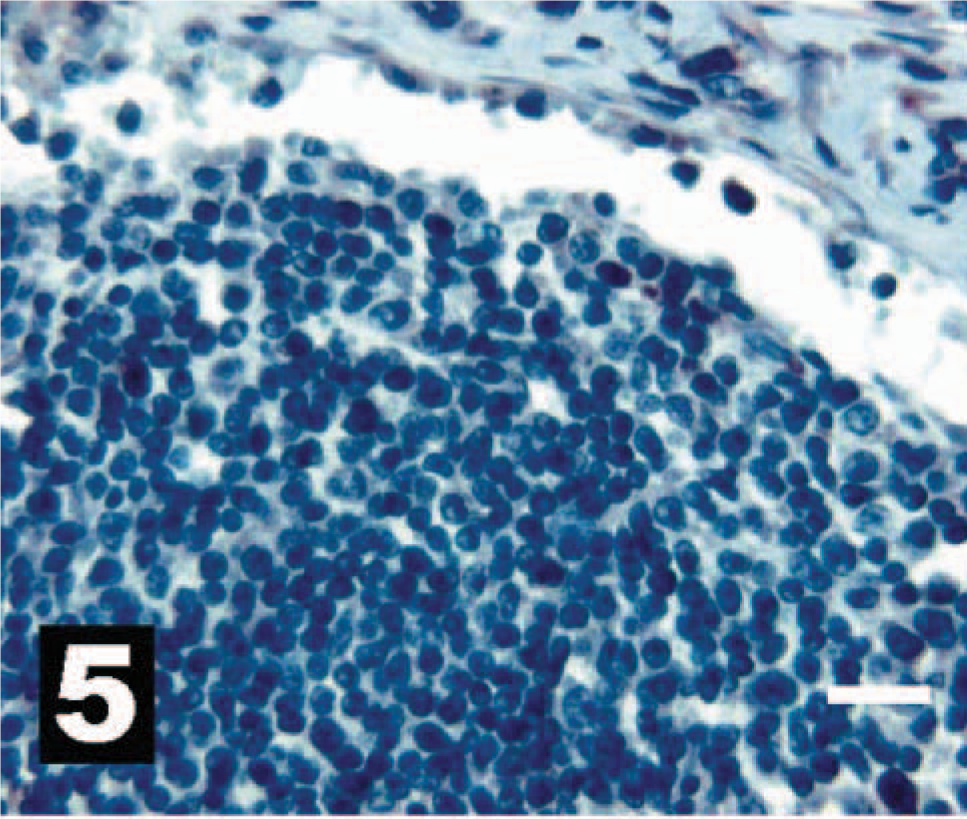
Canine lymph node, as a negative IHC control, with no intracytoplasmic reactivity for HER-2. The chromogen is AEC, and Gills III hematoxylin is the counterstain. Bar = 20 µm.
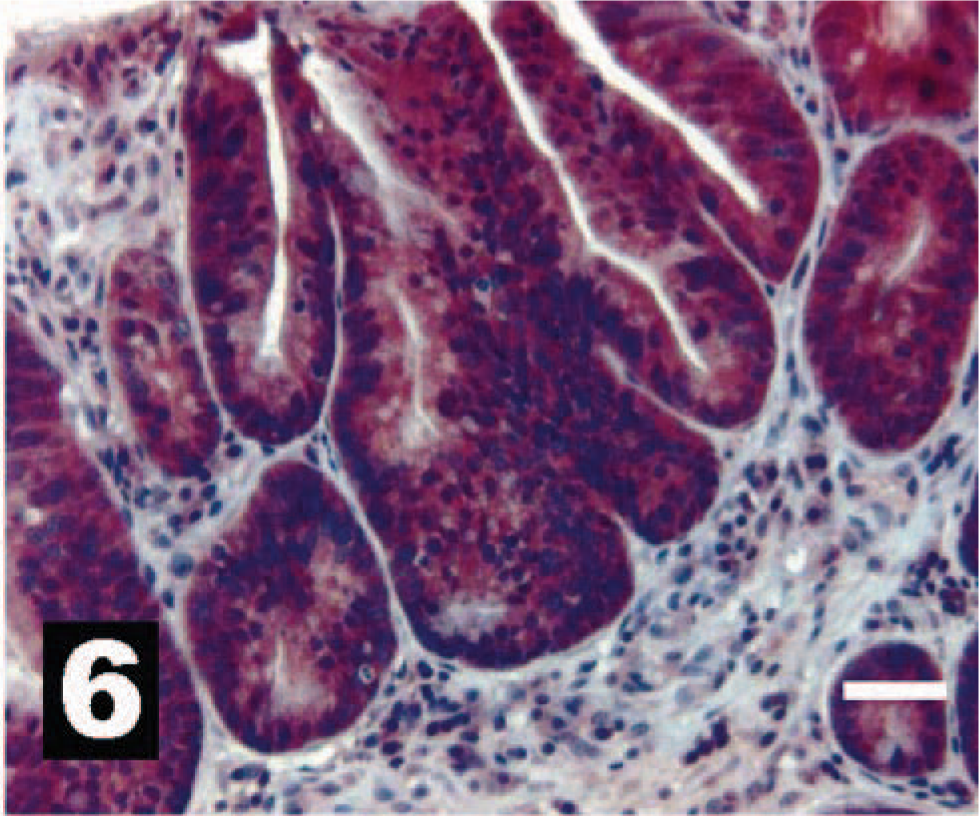
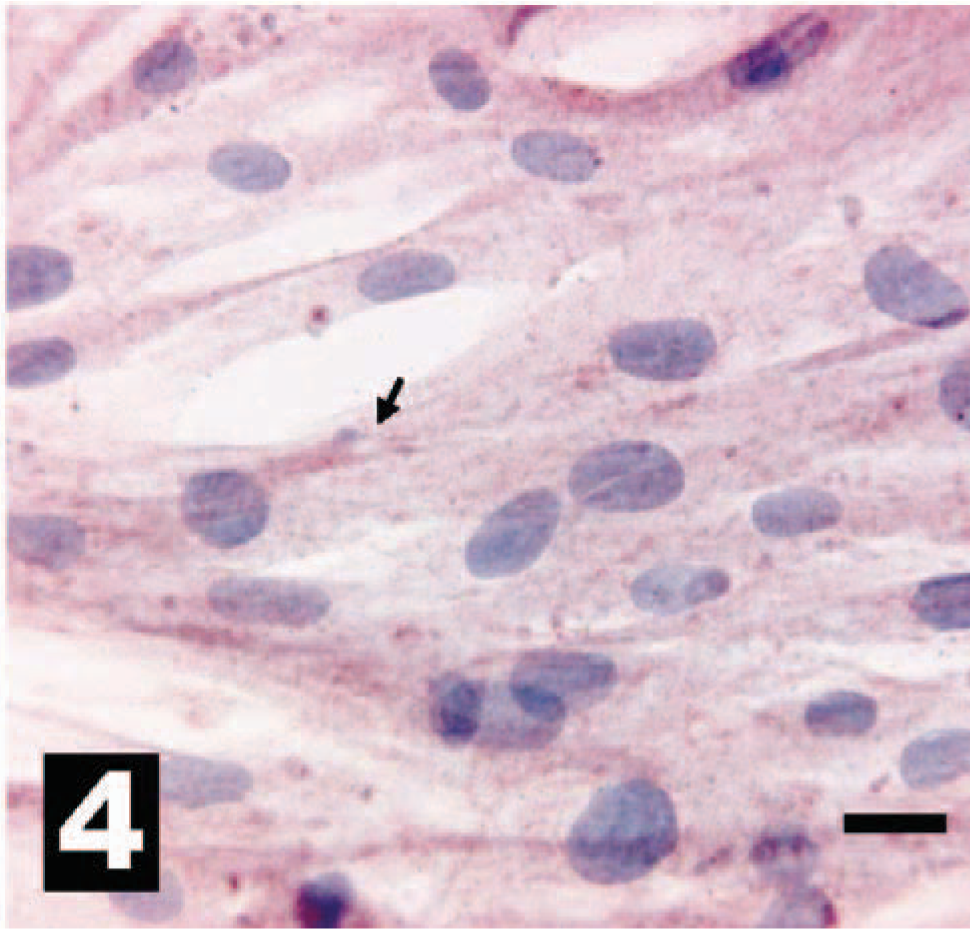
Canine intestinal epithelium, as a positive IHC control, with intense intracytoplasmic reactivity for HER-2. The chromogen is AEC, and Gills III hematoxylin is the counterstain. Bar = 20 µm.
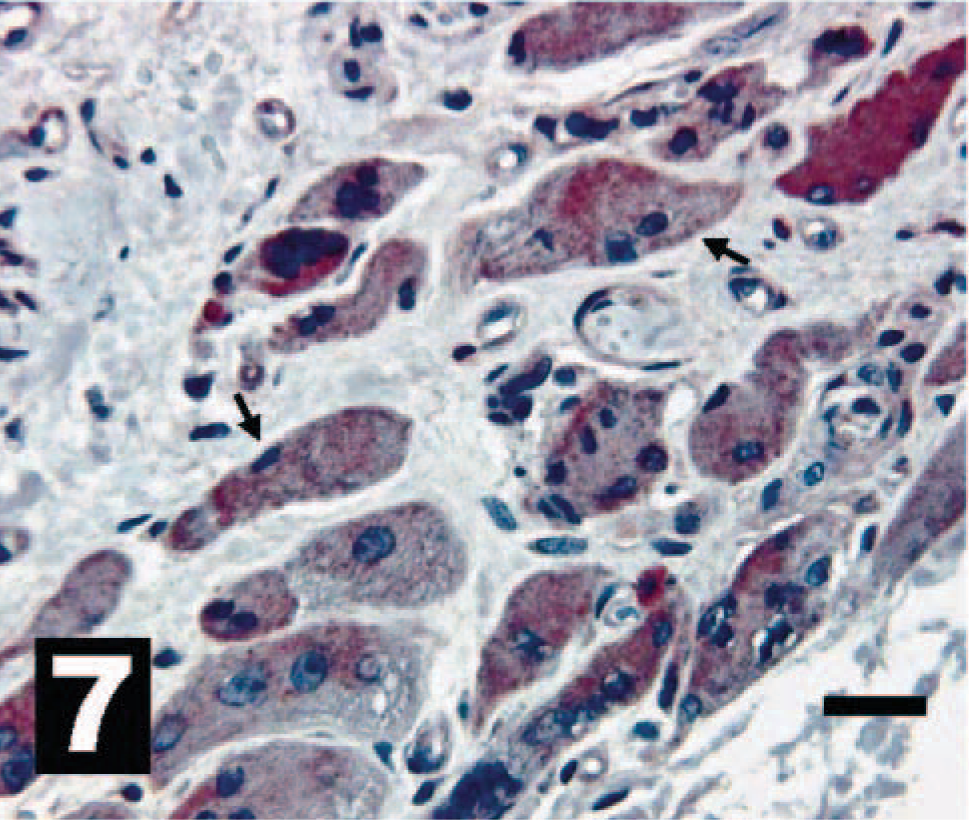
Canine osteosarcoma exhibiting moderate to intense intracytoplasmic IHC reactivity for HER-2 in osteoclastic-like cells (arrows). The chromogen is AEC, and Gills III hematoxylin is the counter stain. Bar = 20 µm.
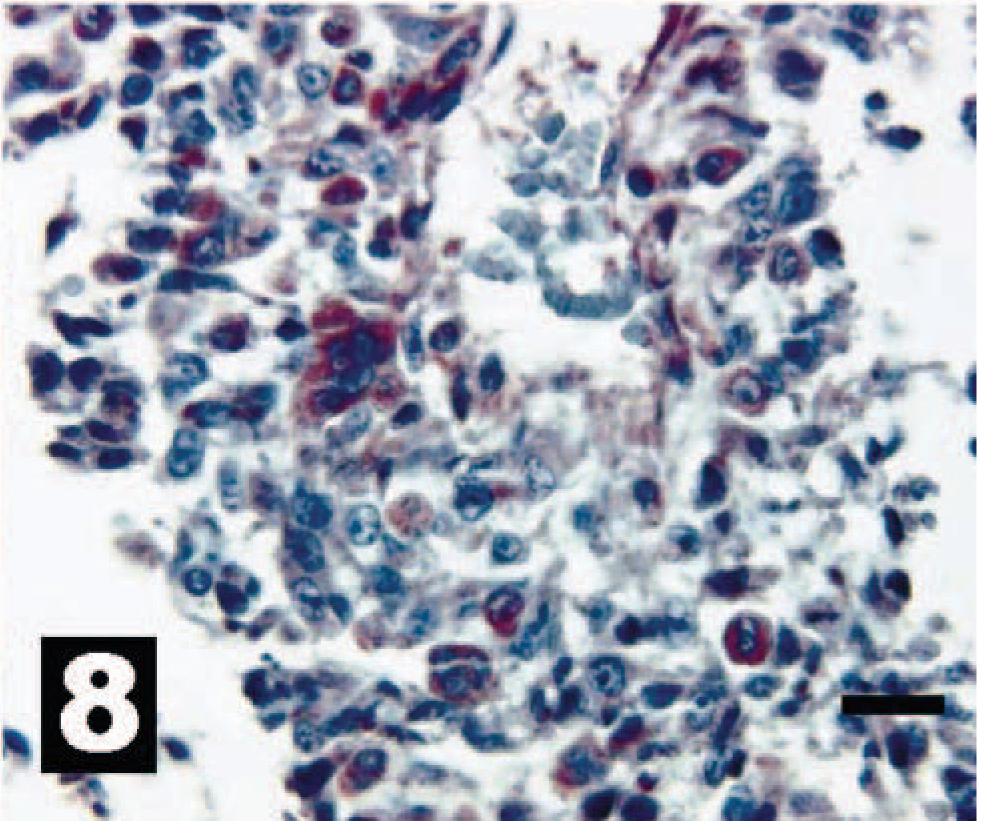
Canine osterosarcoma exhibiting varied intracytoplasmic IHC reactivity for HER-2. The chromogen is AEC, and Gills III hematoxylin is the counter stain. Bar = 20 µm.
Of all the cancers occurring in dogs, about 10% of the caseload seen at Colorado State University Veterinary Teaching hospital (oncology unit) is OSA. Moreover, OSA is extremely difficult to treat and represents one of the most common causes of death among small-animal cancer patients. 3,12 Unfortunately, traditional treatment strategies such as surgery, radiotherapy, and chemotherapy have been unsuccessful in controlling these tumors. 2,12 The high prevalence of metastases and the recurrence after surgery have prompted a search for methods of identifying patients that would benefit from early and aggressive, yet highly specific, treatments for OSA. A firm and thorough understanding of the cellular events underlying this disease process would allow the potential development of effective and specific treatment regimens for both human and canine patients affected with OSA.
Overexpression of erbB-2 is associated with OSA as well as with other neoplastic transformation in a variety of human tumors including breast 11 and ovarian. 10 Moreover, overexpression of erbB-2 is strongly correlated with early pulmonary metastasis and poor survival rates for human patients with these cancers. 5,9,13 The data presented here support the hypothesis that overexpression of erbB-2 in OSA may be a useful prognostic marker for canine patients. Moreover, our IHC analysis of HER-2 shows that gene expression patterns for erbB-2 are consistent with the subsequent expression of functional HER-2, thus further implicating canine HER-2/erbB-2 as a possible therapeutic target. To further substantiate the prognostic value of erbB-2 overexpression, longevity after diagnosis was calculated for the 10 clinical cases described in this study. Analysis of these data shows that overexpression of erbB-2 decreases longevity by 115.5 days (Table 4). However, given the limited number of dogs in our study, this period of time was not statistically significant, yet the trend exists.
Comparison of erbB-2–positive dog survival with erbB-2–negative dog survival after amputation. Dogs negative for erbB-2 survived 115 days longer.
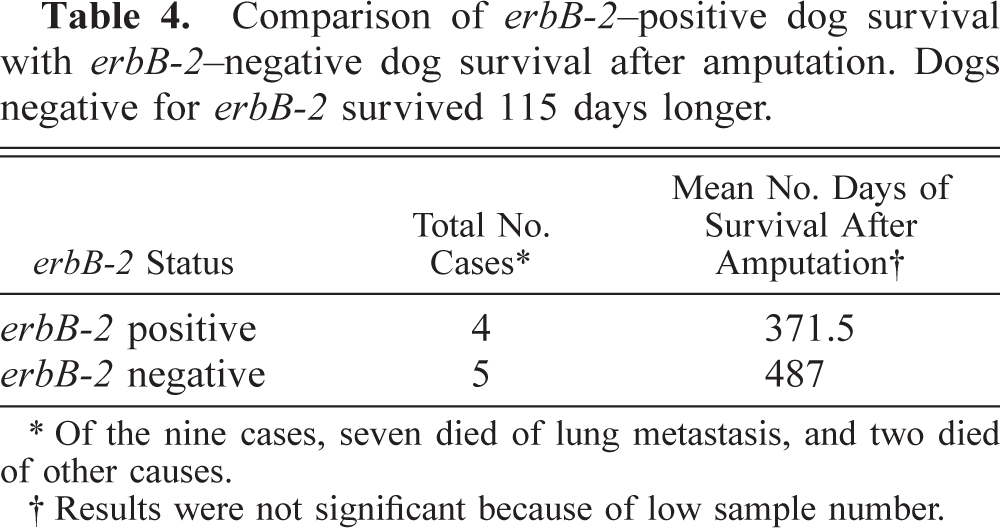
∗ Of the nine cases, seven died of lung metastasis, and two died of other causes.
† Results were not significant because of low sample number.
Our initial results demonstrate that overexpression of HER-2/erbB-2 is detected in a significant percentage of canine OSA, thereby lending credence to the fact that HER-2/erbB-2 may be an important prognostic indicator and therapeutic target for the initial diagnosis and treatment of canine OSA. In conclusion, our results closely follow other studies in which HER-2 overexpression in human OSA patients was an indicator of poor prognosis. 5,9
erbB-2 has been extensively studied for its involvement in a host of different human tumors. In most cases overexpression of the erbB-2 gene correlates well with a poor prognosis; thus, therapeutic approaches have been used to functionally inactivate it through the use of a monoclonal anti–erbB-2 antibody (Herceptin™). On the basis of our current data it is plausible to hypothesize that a similar approach could be used in the dog, thus resulting in improved treatments for canines afflicted with OSA.
Footnotes
Acknowledgements
The canine cell lines derived from OSAs were a generous gift from Dr. Greg MacEwen, University of Wisconsin, Madison, Wisconsin. We would also like to thank Dr. Lisa Parshley, who helped isolate some of the messenger RNA samples and Dr. Ruth Billings for her RT-PCR data analysis and careful review of this manuscript.
