Abstract
Alopecia areata (AA) can be induced in C3H/HeJ mice by grafting full-thickness AA-affected skin. An 8- to 12-week delay between surgery and overt hair loss onset provides an opportunity to examine disease pathogenesis. Normal haired C3H/HeJ mice were sham-grafted or grafted with AA-affected skin. Mice were euthanatized 2, 4, 6, 8, 10, and 12 weeks after surgery along with chronic AA-affected mice as a positive control. Until 6 weeks after grafting, inflammation was only evident around anagen-stage hair follicles in host skin adjacent to but not distant from the AA-affected graft. From 8 weeks on, AA-grafted but not sham-grafted mice exhibited a diffuse dermal inflammation at distant sites that progressively focused on anagen-stage hair follicles at 10 and 12 weeks. Perifollicular inflammation was primarily composed of CD4+ and CD8+ cells associated with follicular epithelium intercellular adhesion molecule -1 expression. Only CD8+ cells penetrated intrafollicularly by 12 weeks after surgery, although both CD4+ and CD8+ intrafollicular cells were observed in chronic AA-affected mice. Under electron microscopy, intrafollicular lymphocyte and macrophage infiltration associated with hair follicle dystrophy was prominent 10 weeks after surgery, primarily within the differentiating outer and inner root sheaths. This study shows that focal follicular inflammation develops some time in advance of overt hair loss and focuses on the differentiating root sheaths in C3H/HeJ mice. The severity of inflammation and the degree of hair follicle dystrophy induced by the infiltrate appear to reach a threshold level before overt hair loss occurs.
Alopecia areata (AA) is a disease in which dystrophic hair follicles are the focus of a peri- and intrafollicular inflammatory infiltrate. Histopathologic examination shows a significant infiltrate of primarily CD4+ and CD8+ cells in and around AA-affected anagen hair follicles. 7,8,11,18,19,23 In active disease, there is increased expression of major histocompatibility complex class I and II antigens along with increased numbers of dendritic cells in AA lesions. 11,24 This and other circumstantial and indirect evidence suggest that AA is an autoimmune disease with as yet unidentified target antigens within the hair follicle. 16
Experiments with humans and rodent models have demonstrated that inflammatory cells are important in maintaining hair follicles in a dystrophic disease state and that suppressing or modifying the infiltrate using drugs or monoclonal antibodies enables hair to regrow. 6,7,10,13–15 However, research has so far been limited to examining the effects of inflammation after the onset of overt hair loss. Nothing is known concerning the disease development process between the disease activation event and onset of alopecia. Until recently, it was not possible to anticipate disease development in specific, spontaneously affected rodents and humans until hair loss was actually observed.
A simple method to transfer AA from spontaneously affected C3H/HeJ mice to unaffected individuals has been identified in previous studies. 12 Grafting full-thickness AA-affected C3H/HeJ mouse skin to unaffected mice induces development of AA, which is grossly visible approximately 8–12 weeks after the surgical procedure. The delay between the disease trigger event (skin grafting) and onset of alopecia suggests that key disease initiation events occur before hair loss can be clinically observed. Because the stages of disease development can be anticipated by timing the interval between surgical procedure and experimental analysis, this technique can be used as a model to define the dynamics of AA after the disease activation event, in advance of overt hair loss, and during the initial stages of clinical hair loss. Here, we report the development of inflammatory cell infiltrate and hair follicle dystrophy using histology, immunohistology, and electron microscopy in a time-course analysis.
Materials and Methods
All C3H/HeJ mice were obtained from stocks at the Jackson Laboratory's (Bar Harbor, ME) specific pathogen–free production facility. Mice received autoclaved food pellets (NIH 31 diet code 5K52, PMI, Richmond, IN) and acidified water (pH 2.8–3.0) ad libitum. Normal haired mice were grafted with AA-affected skin using procedures described previously. 12 For comparative controls, age-matched C3H/HeJ mice were sham grafted. Each mouse was housed in a separate cage for the duration of the study. Mice were euthanatized 2, 4, 6, 8, 10, and 12 weeks after surgery. For each time point, three mice grafted with AA-affected skin and two sham-grafted control mice were euthanatized. Seven mice with chronic skin graft–induced AA persisting for more than 6 months were used as positive controls.
Each mouse was euthanatized by CO2 asphyxiation and necropsied. Tissue was taken across the junction between the skin graft and adjacent host skin and at distant sites from ventral and dorsal skin. Biopsies were not taken during the course of the study on account of the hair wave induction effect of surgery and concern about complications from scarring. 22 Excised tissue from each mouse was divided for routine histology, immunofluorescence, and electron microscopy. Biopsies were fixed in Fekete's acid-alcohol-formalin solution, processed routinely, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. Adjacent tissue was snap-frozen in liquid nitrogen, embedded in OCT compound (Sakura Finetek USA Inc., Torrance, CA), and stored at −70 C for immunofluorescence. 20,22 Small 1- to 2-mm3 samples of host skin, proximal to the graft site, were fixed in 2.5% phosphate-buffered glutaraldehyde, pH 7.2; postfixed in 1% osmium tetroxide; and processed routinely for transmission electron microscopy. 1
Indirect immunofluorescence was performed on 5-µm frozen skin sections placed on aminoalkysilane (Sigma, St. Louis, MO)–coated slides. Rehydrated tissue was incubated with one of the following primary antibodies: rat anti-mouse granulocyte (Gr)–1 (Ly-6G, clone RB6-8C5, Pharmingen, Uppsala, Sweden), hamster anti-mouse CD3e (clone 145-2C11, Pharmingen), rat anti-mouse CD4 (clone GK1.5, Pharmingen), rat anti-mouse CD8a (clone 53-6.7, Pharmingen), and hamster anti-mouse intercellular adhesion molecule (ICAM)–1 (CD54, clone 3E2, Pharmingen). Slides were washed, incubated with a relevant anti-rat or anti-hamster fluorescein isothiocyanate–conjugated secondary antibody (Pharmingen) for 60 minutes at room temperature, washed, mounted in glycerol (Sigma), and examined with a fluorescence microscope. Controls included normal anagen C3H/HeJ pelage hair follicle cryosections and replacement of the primary antibody with normal rat serum.
Results
Histology
Histologic progression studies revealed that 2 weeks after surgery, there was limited scarring associated with both the sham grafts of control mice and AA-affected skin grafts, enabling easy identification of the skin graft borders with normal host skin. By 4 weeks, in all mice, the skin graft procedure had induced a narrow but consistent area of anagen-stage hair follicles in the host skin adjacent to the surgical site, a phenomenon acknowledged in other studies on wound healing. 2 Beyond this border of anagen-stage hair follicles in the host skin, the follicles were predominantly in telogen stage (Fig. 1A, B).
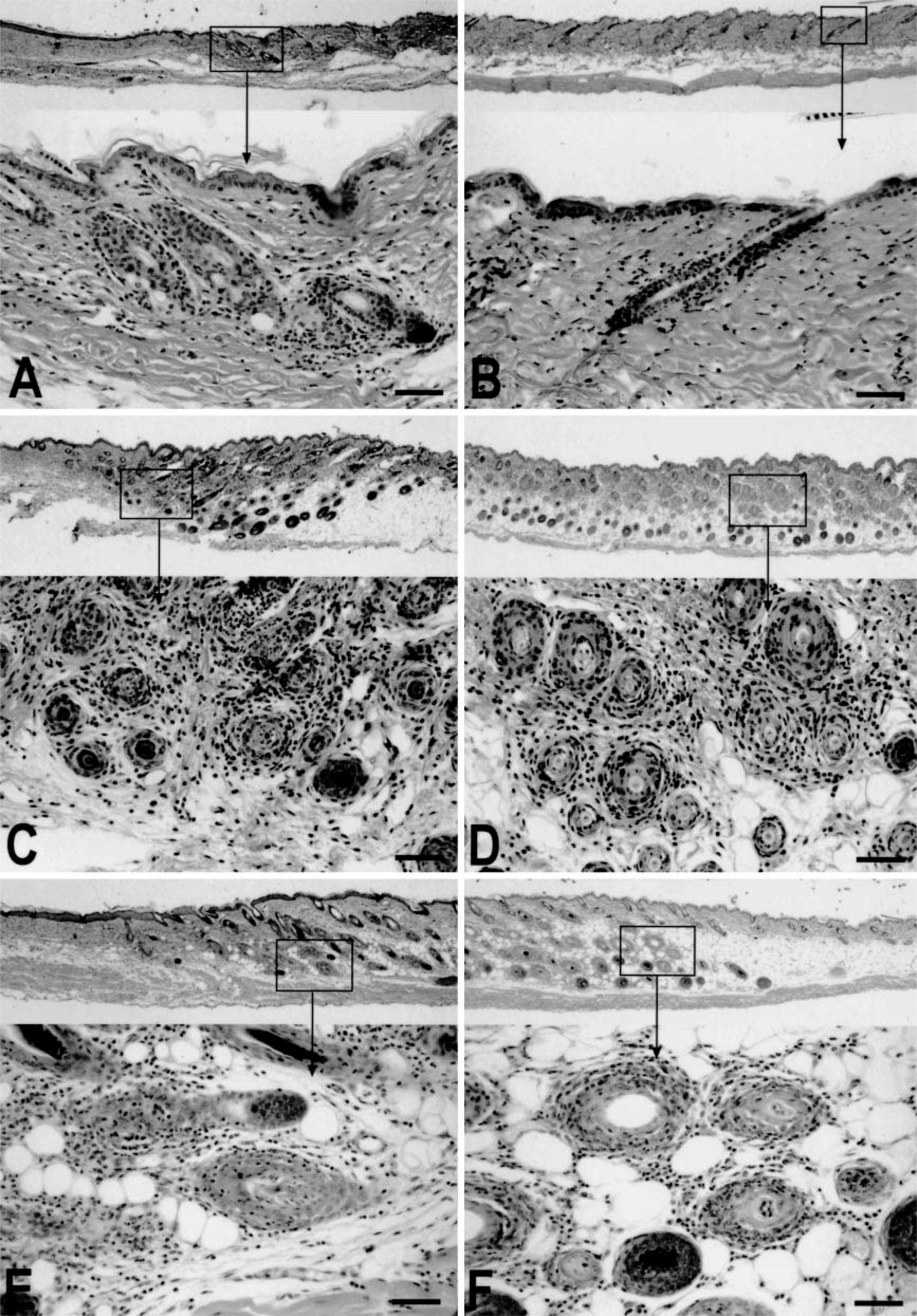
At 2 and 4 weeks after surgery, sham-grafted mice had little to no inflammation in or around the anagen hair follicles in the graft itself, the normal host skin adjacent to the graft, or at distant sites. Where inflammation was observed, the inflammatory cells were located at the graft–host skin junction associated with scar tissue and did not demonstrate any apparent organized, focal inflammation of hair follicles. By 6 or more weeks after surgery, sham-grafted control mice exhibited no apparent dermal inflammation or anagen-stage hair follicle disorganization by histology, immunohistology, or electron microscopy.
In contrast, all mice grafted with skin from AA-affected mice gradually developed an extensive, primarily lymphocytic, peri- and intrafollicular inflammation of anagen-stage hair follicles. At 2, 4, and 6 weeks after surgery, inflammation was limited to AA-affected hair follicles within the grafted skin and around anagen-stage hair follicles in host skin immediately adjacent to the graft (Fig. 1A, B). The observed localized infiltrate was typical of inflammation observed in chronic spontaneous C3H/HeJ mouse AA.
The first histologic changes beyond the very limited areas at the immediate border of the graft and host skin were not evident until 8 weeks after surgery (Fig. 1C, D) and became progressively generalized in the successive 4 weeks. Whereas inflammation was tightly focused on anagen-stage hair follicles in chronic AA–affected control mice, the initial inflammatory infiltrate 8 weeks after grafting presented with a comparatively diffuse pattern in the dermis. At 10 and 12 weeks after surgery, dermal inflammatory infiltrates became progressively focused on anagen-stage hair follicles (Fig. 1E, F). Late anagen–and early catagen–stage hair follicles from the AA-affected skin graft recipients had a prominent inflammatory component and marked follicular dystrophy in the region of the matrix above the bulb associated with disintegration of hair fiber.
In three mice, isolated hair follicles had prominent granulocytic inflammation (suppurative folliculitis). One mouse had prominent follicular abscesses immediately adjacent to the surgical site, suggesting that this was a focal infection or complication of the surgery itself. The other two mice had a mixture of neutrophils and lymphocytes (confirmed by immunohistology), with a predominance of the former around wound-edge anagen-stage hair follicles. These also were interpreted to be minor surgical complications identified microscopically but not clinically. These did not appear to alter the induction of generalized AA at sites distant from the graft.
Overt hair loss was first observed limited to the ventral skin of mice necropsied at 10 weeks, although intense focal inflammation could be observed around anagen-stage hair follicles located on both the ventral and dorsal skin in these mice. Mice at 12 weeks after grafting had extensive ventral alopecia and limited patchy dorsal hair loss associated with focal inflammation of dystrophic anagen-stage hair follicles.
Immunohistology
Immunohistology confirmed the general observations made above. Grafts from AA mice to normal recipients contained predominantly CD4+ and CD8+ cells within the graft on day 0. Isolated CD3+, CD4+, and CD8+ cells were observed around anagen-stage hair follicles adjacent to the graft by 4 weeks and at distant sites by 6 weeks. However, only from 8 weeks onward were intense CD3+, CD4+, and CD8+ cell infiltrates observed in perifolliclular locations distant from the graft site (Fig. 2A, B). CD8+ T cell infiltration in the dermis was more diffuse and less focused on hair follicles in the graft-recipient mice at this time point than observed in chronic spontaneous AA–affected mice. By 10 weeks after surgery, although both CD4+ and CD8+ cells were found in perifollicular positions, only CD8+ cells had penetrated to intrafollicular locations (Fig. 2C). Similar features were observed at 12 weeks after surgery. However, both CD8+ cells and isolated CD4+ cells were observed intrafollicularly in severely disrupted hair follicles in chronic AA–affected control mice.
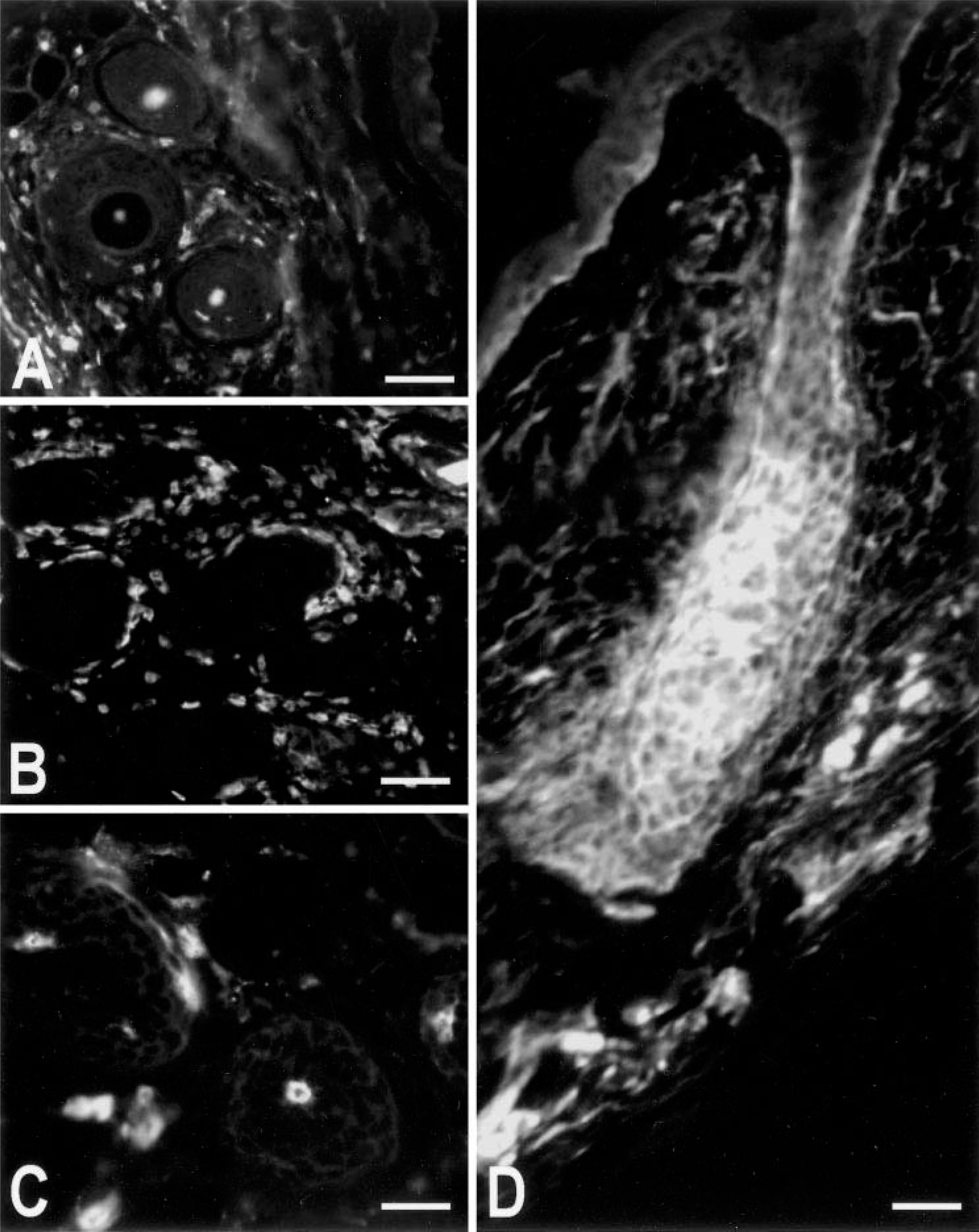
ICAM-1 expression was observed on hair follicle keratinocytes from 4 weeks onward. Fluorescence was primarily limited to differentiating keratinocyte cells of the outer and inner root sheaths (Fig. 2D). ICAM-1 expression intensity varied between adjacent anagen-stage hair follicles, and qualitative observation of serial sections stained with CD4+ and CD8+ cells suggested that high ICAM-1 expression was associated with intense perifollicular CD4+ and CD8+ cell inflammation. ICAM-1 expression could also sometimes be observed in dermal tissue immediately adjacent to the lower differentiating regions of individual hair follicles. Isolated Gr1+ granulocytes were evident at all stages after surgery, with no significant variation in distribution or number except in the three mice with wound complications described above. In these mice, isolated hair follicles adjacent to the graft site had focal perifollicular Gr1+ cell infiltration.
Electron microscopy
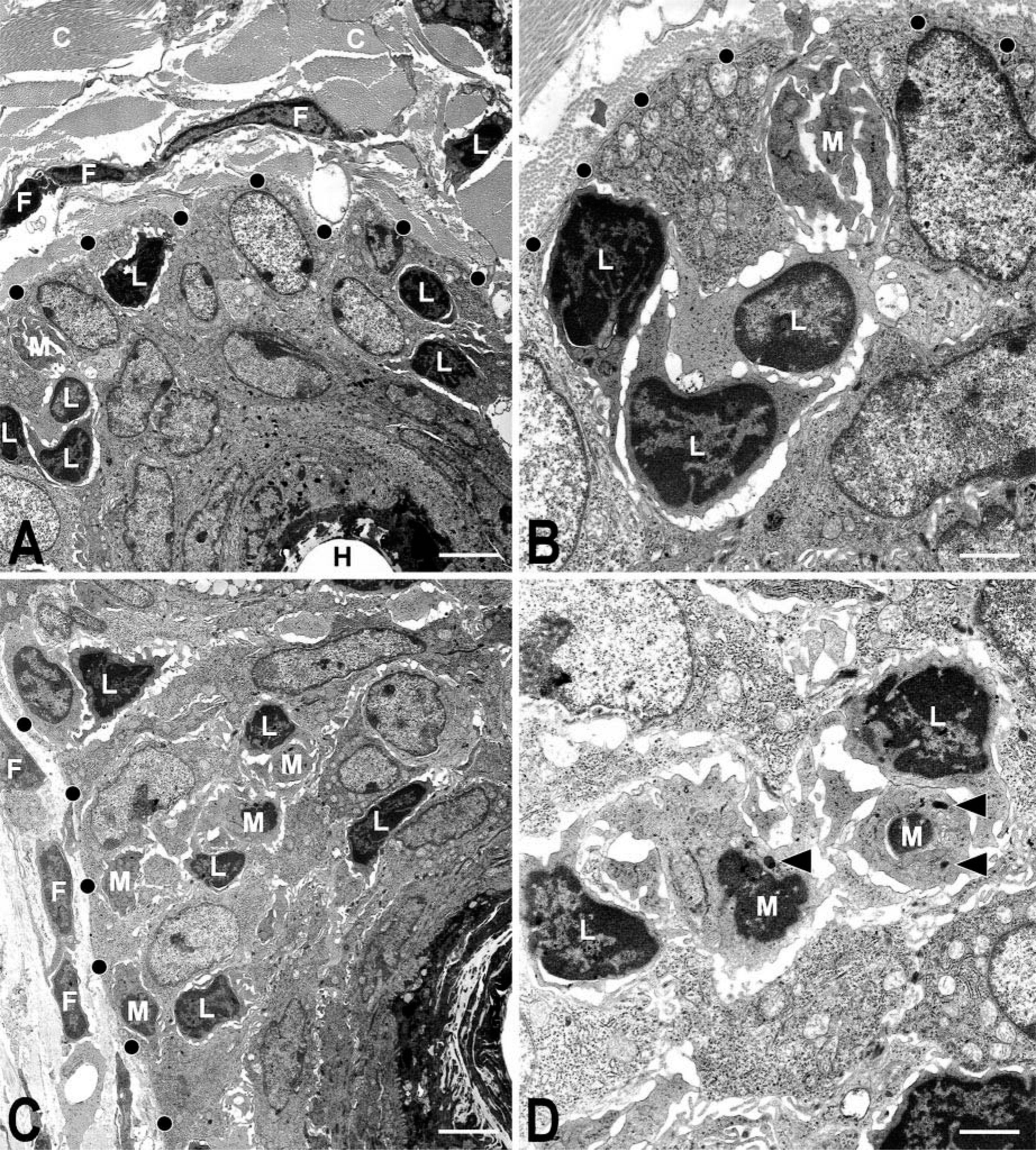
Transmission electron microscopy of skin samples taken from the immediate vicinity of the graft site revealed isolated lymphocytes in a perifollicular location by 6 weeks of grafting. However, disorganization of anagen-stage hair follicle root sheaths was first evident by 8 weeks of grafting, and extensive hair follicle dystrophy was apparent by 10 weeks of grafting. At this time, lymphocyte and macrophage infiltration was prominent in peri- and intrafollicular locations primarily located within the differentiating outer and inner root sheaths (Fig. 3A–D). Limited perifollicular polymorphonuclear cell inflammation was observed at 8 weeks after surgery, but only isolated polymorphonuclear cells penetrated to intrafollicular positions in severely disrupted hair follicles at 12 weeks after surgery. Mast cells also were observed but were present with a similar frequency on the skin of both AA-affected skin–grafted mice and sham-grafted control mice.
Discussion
The ability to induce AA-like hair loss in C3H/HeJ mice using a simple full-thickness skin grafting technique enables a wide range of projects to be undertaken to understand the pathogenesis of AA before overt hair loss. The objective of this study was to investigate changes in hair follicle morphology and associated inflammatory infiltrate between the disease activation event and onset of overt hair loss and to compare these results with the chronic spontaneous AA form found in this strain.
The results circumstantially suggest that two pathogenic actions may take place subsequent to skin grafting normal haired mice with AA-affected skin. Initially, focal inflammation was observed around host hair follicles adjacent to the graft. It is possible that the inflammatory cells observed migrated directly from the graft. In previous studies, a ring of hair loss in host skin around the graft was observed in some individual mice before the onset of hair loss distal to the graft. 12 More generalized inflammation distant from the graft occurred from 8 weeks onward. Given the extent of affected regions and the intensity of inflammation, it is likely that the cells involved were primarily derived from the host immune system by lymphocyte activation.
It is probable that only a small minority of the inflammatory cells around hair follicles in AA are specific for localized antigen(s) and have a direct destructive action. This heterogeneity in CD4+/CD8+ cell populations has been recognized for some time in relation to other autoimmune diseases. 19,20 The majority of the inflammatory cells may be irrelevant to AA pathogenesis other than having indirect effects via cytokine production. This mechanism may be reflected in reports of hair regrowth promotion in AA-affected individuals by modifying rather than removing the perifollicular infiltrate composition and associated cytokine production using contact sensitizers. 6,9 Only cells penetrating the intrafollicular positions are likely to exert a direct destructive influence on hair follicle epithelium. Previous immunohistologic studies on AA-affected rodents revealed a predominance of CD8+ cells in intrafollicular locations within the hair follicle structure. 6,25 Similarly, in this study, intrafollicular inflammatory lymphocytes were primarily CD8+ cells. Immunohistologic studies on humans are less conclusive, but both CD4+ and CD8+ cells were observed in intrafollicular locations. 8,18,19,23
The apparent primary morphologic targets of attack for intrafollicular migrating inflammatory cells were the differentiating keratinocytes of the outer and inner root sheaths. Isolated inflammatory cells also were observed within the differentiating hair cortex. Inflammatory cells were not observed to focus on other proposed candidate such as the dermal papilla or melanocytes. 17 Transmission electron microscopy confirmed these observations on intrafollicular inflammatory cells. Activated macrophage-like and lymphocyte-like cells were identified along with isolated polymorphonuclear cells in the root sheaths associated with root sheath disruption.
Polymorphonucleocytes most likely are bystanders in the development of AA, as demonstrated in previous studies where AA-affected skin was grafted to C3Smn.CB17-Prkdc scid /J (previous designation: C3HSmn.C-Prkdc scid /J) mice. In graft recipients, in the absence of lymphocytes, a pronounced perifollicular polymorphonuclear cell infiltrate was identified, but no hair loss developed. When C3Smn.CB17-Prkdc scid /J mice were grafted with AA-affected skin and injected with lymph node–derived cells from immunocompetent normal haired C3H/HeJ mice, hair loss was induced with a mixed lymphocytic and polymorphonuclear cell infiltrate. 12
Development of AA in the C3H/HeJ AA skin graft model involves immune system activation before onset of overt hair loss. The time between disease activation by skin grafting and alopecia development may be up to 12 weeks. During this time, key events such as initial antigen presentation and lymphocyte cell proliferation are likely to occur. This has been confirmed using flow cytometry and sequential gene array analysis of transcripts and blockade of lymphocyte costimulatory pathways with monoclonal antibodies. 3,26 The severity of inflammation and the degree of hair follicle dystrophy induced by the infiltrate appears to reach a threshold level before overt hair loss occurs. The same may be true for other rodent models and for humans.
Although numerous secondary morphologic targets may come under attack through epitope spreading in chronic diseases, 4 the physical location of the initial inflammatory cell infiltrate suggests that the primary antigenic targets for inflammatory cells are located within the differentiating inner and outer root sheaths in C3H/HeJ mice. These structures may be worth researching for identifying antigenic targets involved in primary disease activation. This inducible model of AA should enable significant progress in defining the initial disease activation events that occur in AA.
Footnotes
Acknowledgements
We gratefully acknowledge the assistance of P. Jewett for histologic services. This work was supported by the Dermatology Foundation/Glaxo Dermatology (K. J. McElwee), Ernst Schering Research Foundation (K. J. McElwee), National Alopecia Areata Foundation (K. J. McElwee, J. P. Sundberg), National Institute of Arthritis and Musculoskeletal and Skin Diseases (AR43801, J. P. Sundberg), National Center for Research Resources (RR173, J. P. Sundberg), National Cancer Institute (CA34196, J. P. Sundberg), and funds from the Department of Veterans affairs (L. E. King, Jr).
