Abstract
Twenty pigs were inoculated with a virulent isolate (Quillota strain) of classical swine fever (CSF) virus to determine the chronological development of lesions in bone marrow. Histopathologic, ultrastructural and immunohistochemical (detection of viral antigen gp55, myeloid-histiocyte antigen, CD3 antigen, and FVIII-rag), and morphometric techniques were employed. Viral antigen was detected from 2 days postinfection (dpi) in stromal and haematopoitic cells, and severe atrophy related to apoptosis of haematopoitic cells was observed. Megakaryocytes (MKs) did not show significant changes in number, but there were important qualitative changes including 1) increased numbers of cloud-nuclei MKs, microMKs, apoptotic MKs, and atypical nucleated MKs and 2) decreased number of typical nucleated MKs. Morphometric study of these cells showed a decrease in cytoplasmic area. MK infection was detected from 2 dpi, but in a small percentage of cells. Myeloid cells showed quantitative changes, with an increase in granulocyte numbers. Apoptosis of lymphocytes and viral infection of erythroblasts were also observed. The main changes in stroma were depletion of T lymphocytes in the middle phase of the experiment and macrophages. Viral infection was also observed in these cells. MK lesions suggest dysmegakaryocytopoiesis, which would aggravate the thrombocytopenia already present and could be responsible for it. Granulocyte changes would lead to the appearance of circulating immature forms, whereas lymphocyte apoptosis in bone marrow would contribute to lymphopenia.
Introduction
A number of viral diseases cause thrombocytopenia and marked alterations in the differential WBC count; in some diseases, rapid return to normal values is impossible because of the direct action of the virus on hematopoietic cells, an infection of stromal cells of the marrow, or a cytotoxic immune response. 20 This is clearly seen in the Flavivirus infections (Dengue) 20 and in the classical swine fever (CSF). 29 In their acute phase, these diseases are characterized by severe leukopenia, with marked lymphocyte and neutrophil depletion.
Although a number of studies in vitro have demonstrated the susceptibility of stromal cells to CSF virus, 23 high virus titers in bone marrow at early stages, 8 megakaryocyte (MK) degeneration, 5 , 6 , 15 and changes indicative of necrosis and apoptosis in hematopoietic cells, 15 , 26 no detailed morphological study has yet been performed to determine the involvement of bone marrow in leukopenia, thrombocytopenia, and altered differential WBC count. However, knowledge of the mechanisms responsible for these events has improved. Both lymphocyte and neutrophil depletion have been attributed to apoptosis unrelated to the direct action of the virus on lymphocytes 22 , 24 and neutrophils, 25 although both cells are susceptible to infection and the virus might directly induce this type of cell death. 4 The onset of thrombocytopenia appears to be linked to massive platelet aggregation and activation in various organs; 3 neither platelet infection 31 nor MK infection 12 seem to play a major role in these processes, and disseminated intravascular coagulation does not appear to be involved. 5 , 6 , 11 , 14 , 21
This article reports a structural, immunohistochemical, and ultrastructural study of bone marrow in pigs inoculated with a moderately virulent strain of CSF. The study was designed to determine the changes taking place in stromal and hematopoietic cells during the course of the disease and the involvement of these changes in its pathogenesis.
Materials and Methods
Animals, virus and experimental designs
Twenty-two Large White × Landrace pigs of both sexes were used for this study; body weight at the start of the study was roughly 30 kg. All animals were clinically healthy and serologically negative for CSF. Pigs were housed in isolation at the Centro de Investigación en Sanidad Animal in Valdeolmos, Madrid, Spain. Two pigs were used as controls and 20 received an intramuscular inoculation of 105 TCID50 of the virulent CSF virus isolate “Quillota.” Clinical symptoms and rectal temperature were monitored daily.
Preinoculation blood samples were taken from all pigs to obtain baseline values. Considerable care was taken in the collection of the blood samples to avoid hemolysis and tissue contamination. Blood samples were obtained from the anterior vena cava with plastic syringes and disposable 18-gauge needles. Blood was mixed with 1% ethylenediaminetetraacetic acid (9:1 ratio by volume) and diluted 1:200 with 1% ammonium oxalate in distilled water. A hemocytometer chamber was used to determine the platelet count. Blood samples were taken on the day of slaughter.
Processing of specimens for light and electron microscopy
Pigs inoculated were slaughtered in batches of four at 2, 4, 6, 8 and 10 days postinoculation (dpi). Before slaughter, animals were sedated with Combelen® (Pfizer Inc., Sandwich, NJ, USA), then anaesthetized and killed with T61® (American Hoechst Corp., Somerville, NJ, USA). Bone marrow tissue samples were fixed by immersion in 4% formaldehyde for light microscopy and 2.5% glutaraldehyde for electron microscopy. Samples were embedded in paraffin and Epon 812® (Fluka Chemie AG, Buchs, Switzerland) using routine techniques. Paraffin-embedded sections were stained with hematoxylin-eosin and by methanamine silver staining; Epon-812-embedded sections 1-μm thick were stained with toluidine blue (1% aqueous solution). Fifty-nanometer sections of bone marrow for ultrastructural examination were counterstained with uranyl acetate and lead citrate and viewed using a Philips CM-10 transmission electron microscope.
Immunohistochemistry
The avidin–biotin–peroxidase complex (ABC) technique was used to demonstrate CSF virus glycoprotein 55 (gp55) (Central Veterinary Laboratory, Weybridge, UK) and to identify MKs (FVIII-rag) (Dako®, Hamburg, Germany), myeloid-histiocyte cells (MAC387) (Dako®), and T lymphocytes (CD3) (Dako®). Phosphate-buffered saline and nonimmune serum were used in place of specific primary antibodies as negative controls. Samples from two uninoculated animals were used as controls.
Cell counts and statistical analysis
Cell counts were performed on 4-μm sections of bone marrow from all inoculated and control animals; cells were identified by immunostaining for gp55 viral antigen, FVIII-rag, MAC-387, and CD3. The number of cells per unit area was determined as follows: the number of 0.3 mm2 counting areas (chosen randomly in four diagonally arranged squares) necessary to obtain 200 cells was determined, and the distribution was expressed as cells/mm2 (MKs, myeloid cells, and T lymphocytes). Percentage types of MKs were obtained during the counts of these cells.
Data on immunopositive cell counts were analyzed by analysis of variance, followed by the Newman-Keuls statistic for multiple comparisons. Differences between counts for control and inoculated animals were considered to be significant at P < 0.05. Results are expressed as mean ± standard deviation (x ± SD) and mean (types of MKs).
Results
Clinical signs
The clinical signs observed in these animals were characteristic of CSF. From 2 dpi, the animals showed persistent pyrexia (40.5–41.5 C), varying degrees of anorexia, and constipation. Slight diarrhea, erythema of the skin, and an ocular discharge associated with conjunctivitis were present from 4 to 5 dpi. From 6 dpi onward, deterioration was rapid, with temperatures of >42 C and markedly decreased activity. From 8 dpi, the typical staggering gait associated with CSF and a cyanotic discoloration of the skin of the abdomen, snout, ears, and legs were also observed. Control animals remained healthy.
Changes in blood cells
Total white blood cell (WBC) number, absolute number of WBC types, and platelets counts were measured during CSF virus infection. A marked and statistically significant decrease in total WBC number was observed at 4 dpi onward, with lymphopenia, monocytopenia, and neutropenia, although there was a significant increase in the number of band neutrophils and metamyelocytes from 6 dpi (data not shown). The number of eosinophils remained unchanged. Pigs infected with CSF virus showed a decrease in platelets from 475 × 103/μl to 342 × 103/μl in the first 48 hours, coinciding with pyrexia and becoming more severe during the next few days.
Morphological findings
General aspects
As the disease progressed, there was evidence of generalized hypocellularity of the bone marrow, which became extremely severe from 6 dpi onward. At 2 dpi, scattered pyknotic cells were observed; these were more abundant at 4 and 6 dpi and were associated with abundant cell debris composed mainly of pyknotic nuclear debris, some undergoing phagocytosis by macrophage, and fibrin strands (Fig. 1). These areas, in which background stroma was loosely arranged, also contained microhemorrhages. Ultrastructurally, pyknosis was accompanied by other subcellular changes resembling apoptosis. There was uniform or peripheral condensation of chromatin, with loss of the nuclear membrane, fragmentation of nuclear chromatin, the presence of intracytoplasmic granular chromatin masses, and cytoplasmic condensation. The cytoplasmic membrane remained intact, although at later stages chromatin debris was engulfed by a membrane unit, and fragmentation of cytoplasm into apoptotic bodies occurred (Fig. 2). Apoptotic bodies were engulfed by adjacent macrophages. By 8 dpi, cell debris and fibrin strands had disappeared. An increase in the reticulin fiber network was observed at 8 and 10 dpi.
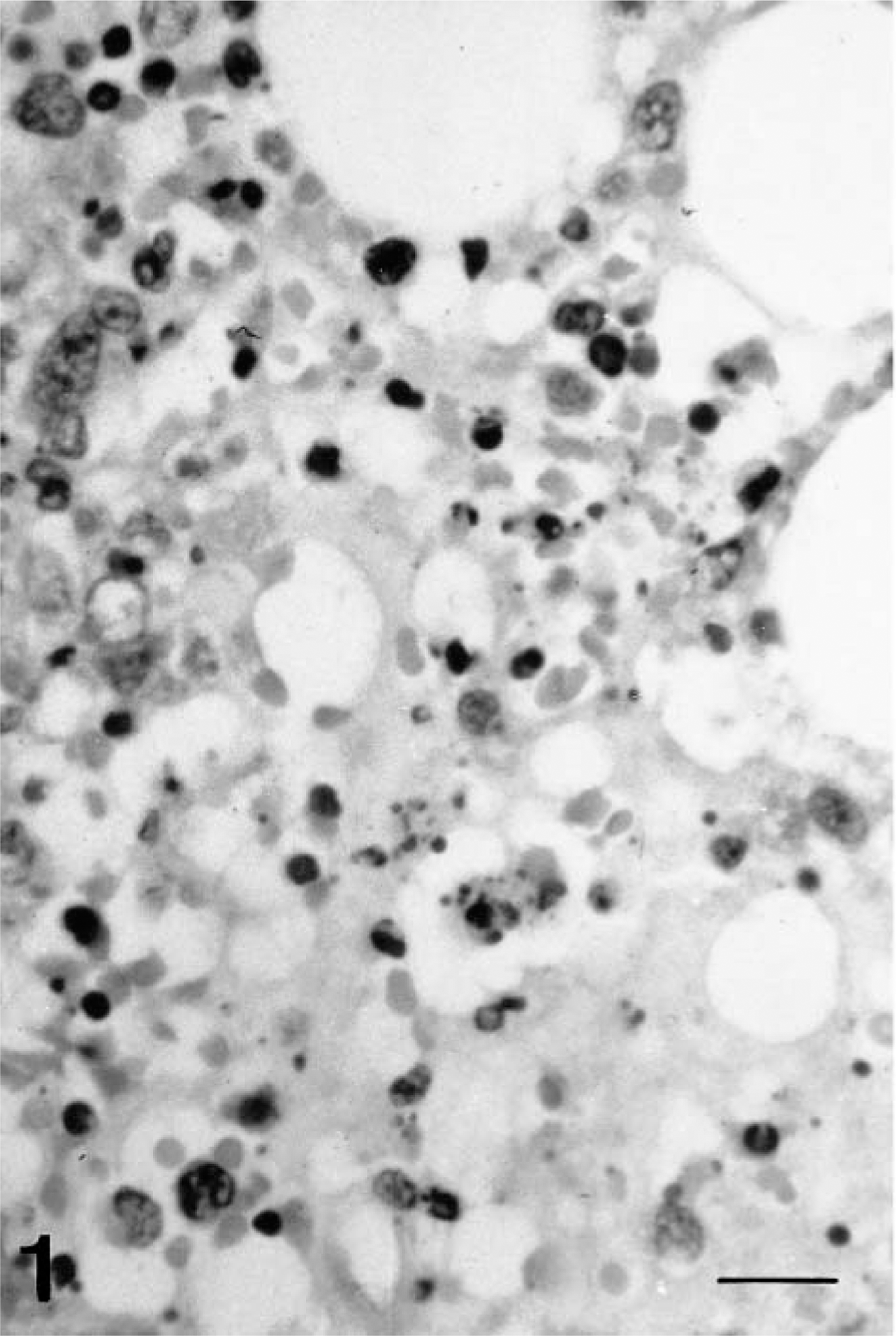
Bone marrow; pig 4 days after CSF virus infection. Focal hypocellularity with cells debris. HE. Bar = 65 μm.
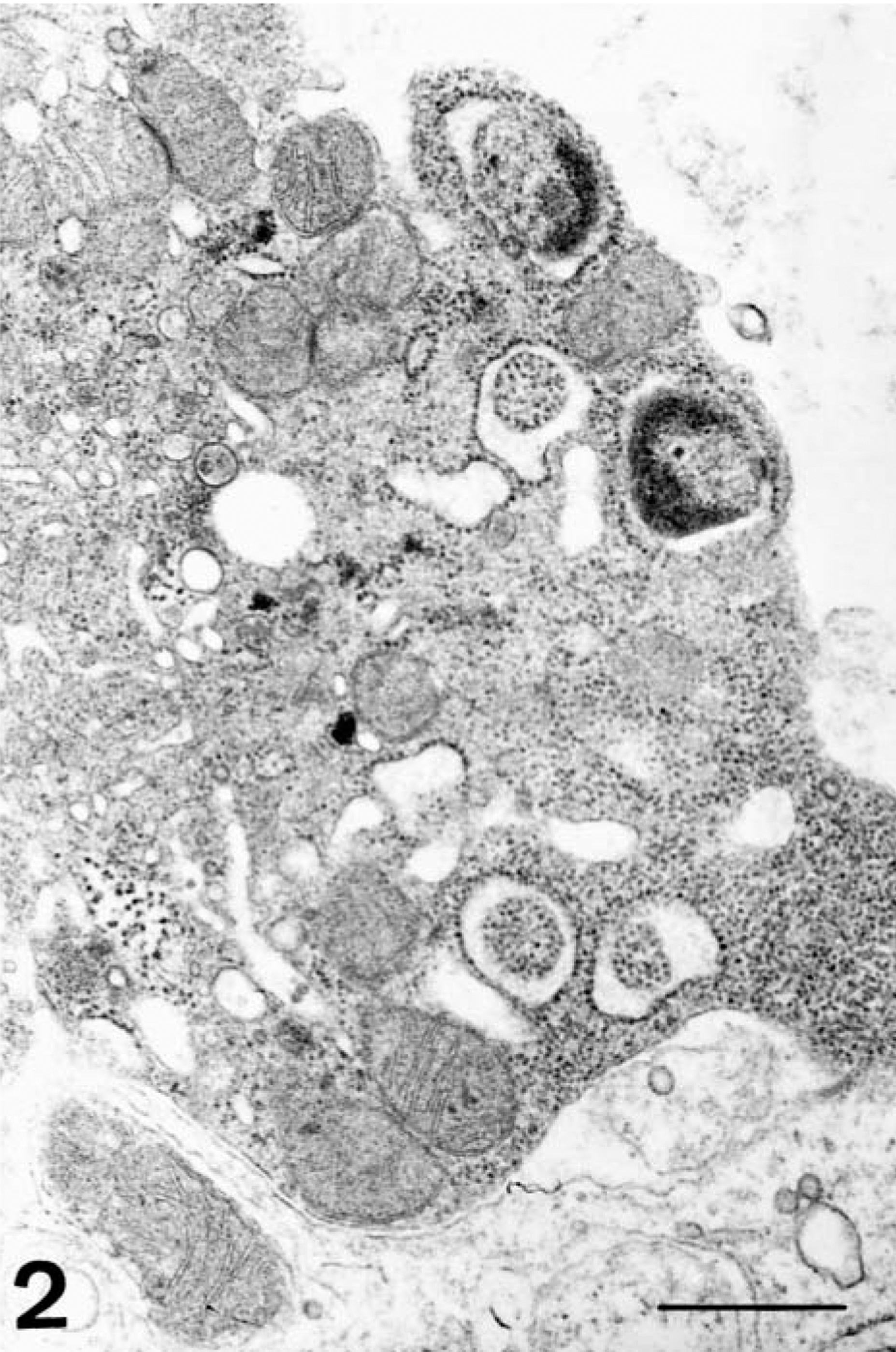
Electron micrograph. Bone marrow; pig 6 days after CSF virus infection. Apoptotic cells with fragmentation of nucleus and initial phase to make apoptotic bodies. Bar = 500 nm.
Immunocytochemical analysis for CSFV gp55 demonstrated infection of hematopoietic cells from 2 dpi; the percentage of infected cells rose from 10–15% at 4 dpi to 65–80% at 10 dpi. From 6 to 8 dpi, the rate fluctuated between 35% and 50% (Fig. 3). Scattered apoptotic cells were positive for CSF virus gp55 between 4 and 10 dpi.
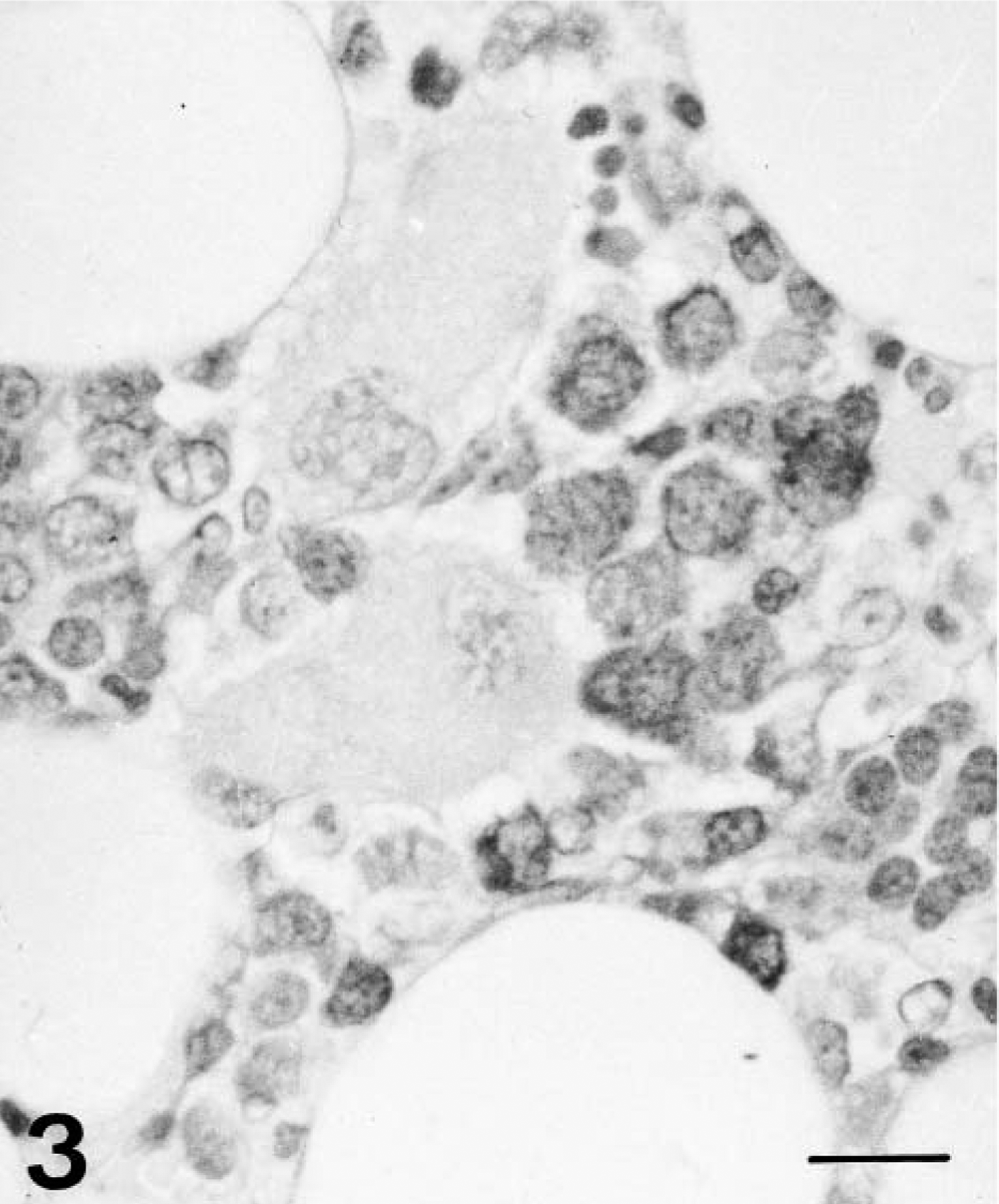
Bone marrow; pig 6 days after CSF virus infection. Immunohistochemical detection of viral antigen showing abundant immunopositive cells and two cloudlike MKs. ABC technique. Bar = 65 μm.
Hematopoietic cells
From 4 dpi, numerous MKs displayed elongated cytoplasm and a round, balloon-shaped nucleus located at one end of the cell (cloud-nuclei MKs) (Fig. 3); these were generally arranged in clusters to form MK “nests,” and differed from typically nucleated MKs. Upon ultrastructural examination, both types of MKs displayed rudimentary cytoplasmic granules and a well-developed membrane demarcation system (Fig. 4), although these structures were absent in a few MKs from 6 dpi. Globose cytoplasmic processes containing organelles, granules, or membranes were observed in these cells.
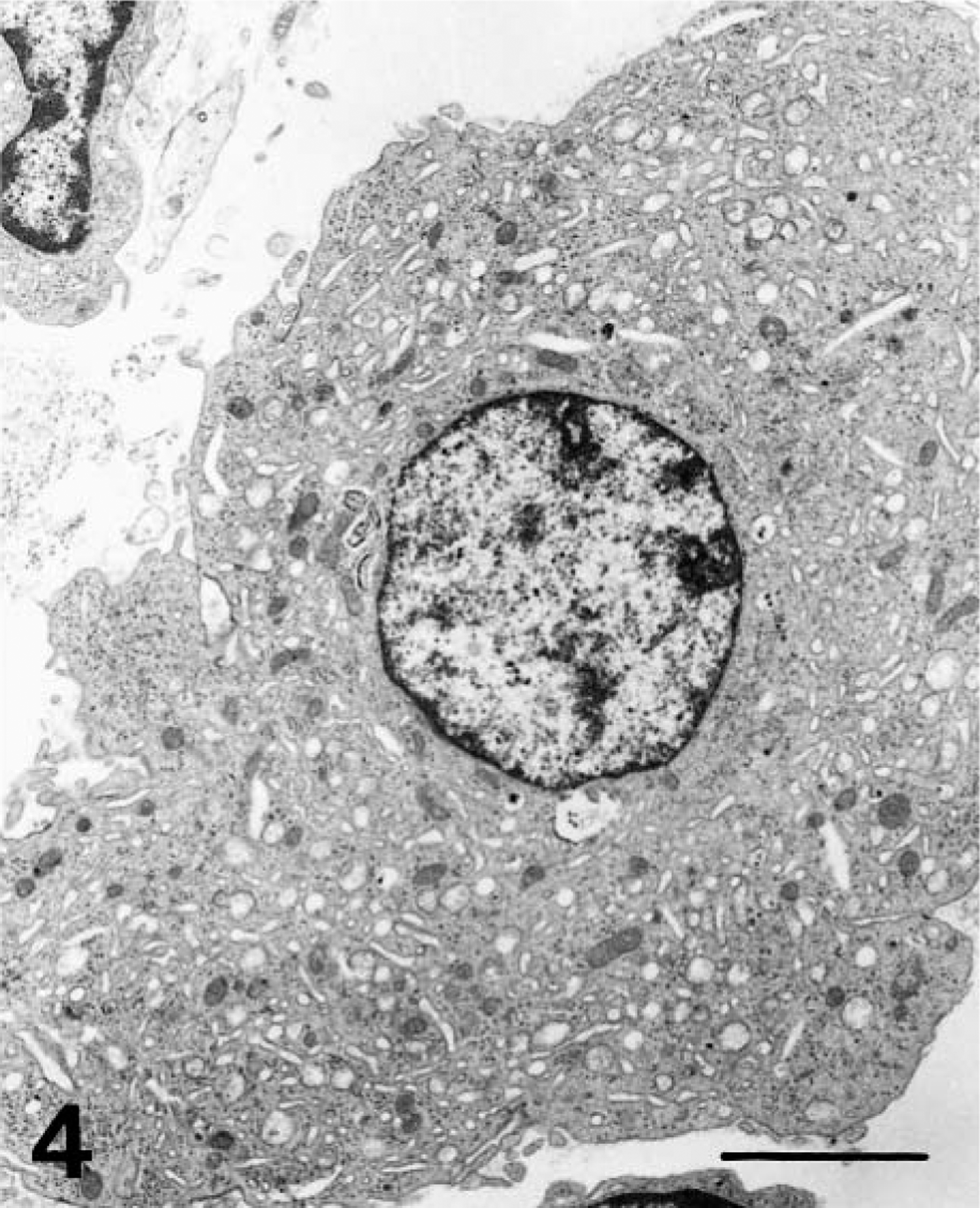
Electron micrograph. Bone marrow; pig 10 days after CSF virus infection. MK showing a developed membrane demarcation system and absence of cytoplasmic granules. Bar = 2.5 μm.
From 6 dpi, some apoptotic MKs were observed. These displayed a condensed, compact hyperchromatic nucleus; cytoplasm was either absent or formed a small perinuclear halo. MicroMKs, representing an immature MK phase, were small, with a rounded nucleus, and were only identifiable by FVIII-rag staining.
A further change observed in MKs was emperipolesis, seen as morphologically normal leukocytes, almost always neutrophils, embedded in MK cytoplasm. This was rarely observed in the first few days of the experiment but became increasingly common from 6 dpi onward. By 8 and 10 dpi, emperipolesis was frequent, and the number of embedded leukocytes had increased (Fig. 5).
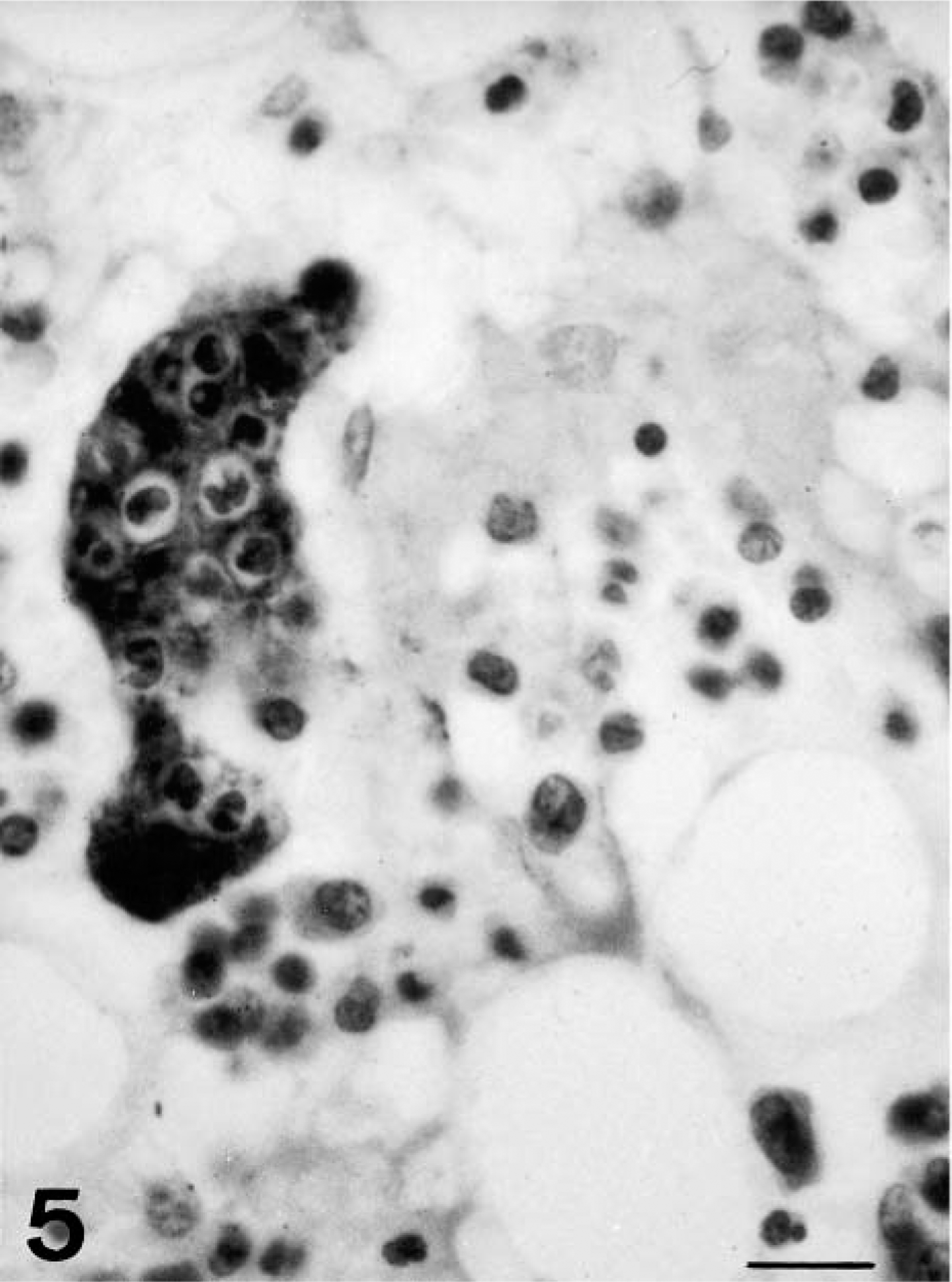
Bone marrow; pig 10 days after CSF virus infection. MKs showing emperipolesis of a great number of cells. ABC technique. Bar = 65 μm.
The ABC immunostaining technique used for detection of viral antigen gp55 revealed positive staining in 2.5–9% of all types of MKs from 2 dpi, including typically nucleated, cloud-nuclei, and apoptotic MKs. Infection of MKs was confirmed by ultrastructural examination.
Morphometric analysis of MKs (Table 1) showed a decline in nuclear and total area during the course of the experiment, in both typically nucleated and cloudlike MKs. Decreases were found to be statistically significant in animals slaughtered at 4 dpi but thenceforth ceased to be significant. The nuclear area-total area ratio increased significantly during the course of the experiment.
Morphometrical study of nuclear and total area of megakaryocytes of pigs inoculated with CSF virus.
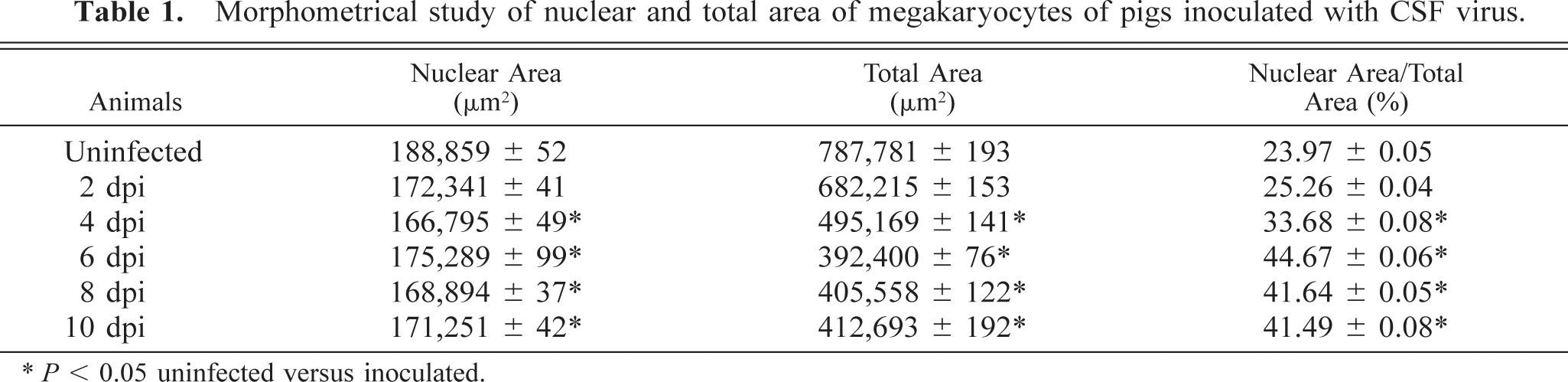
∗ P < 0.05 uninfected versus inoculated.
Immunocytochemical analysis for gp55 antigen detection showed infection of myeloid cells, especially monocytes, from 2 dpi. The infection rate rose from 10% at 2 dpi to 40–50% from 6 dpi onward. Electron microscopy revealed infection of promonocytes and monocytes, as well as promyelocytes, metamyelocytes, and neutrophilic myelocytes.
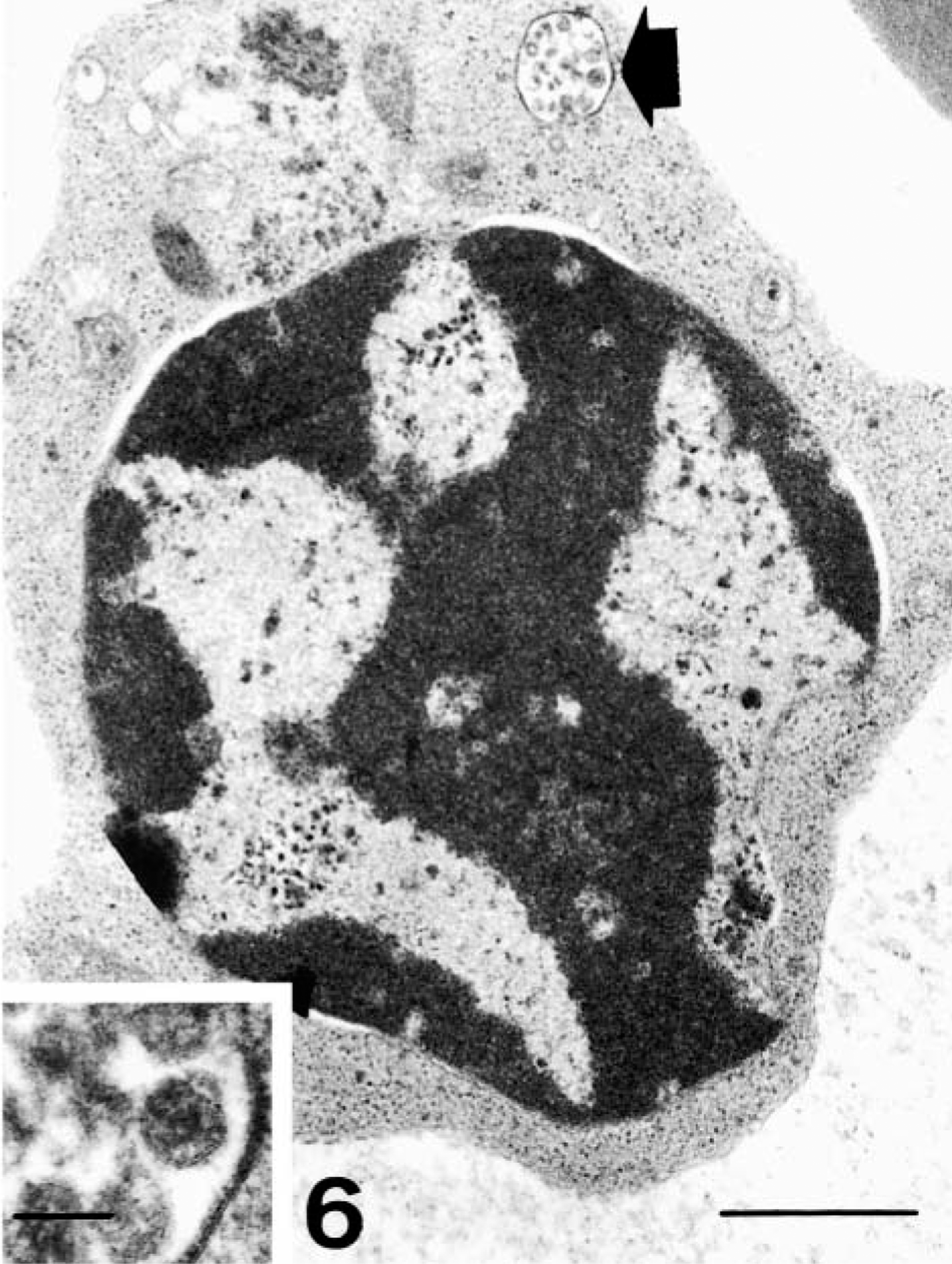
Electron micrograph. Bone marrow; pig 6 days after CSF virus infection. Erythroid cell with ultrastructural evidence of viral infection represented by a vesicle containing viruslike particles (arrow). Bar = 1 μm. Inset: Detail of this vesicle. Bar = 75 nm.
Cells in stroma
At 2 dpi, positive staining for viral antigen was observed in macrophages, and ultrastructural examination revealed viral structures and virions in the cytoplasm of these cells. Macrophages in areas of apoptosis displayed phagocytosis of cell debris and fibrin strands. One significant change observed in bone marrow macrophages was evidence of activation, in the form of increased cell size and proliferation of organelles, particularly Golgi complexes and rough endoplasmic reticulum cisternae, from 4 dpi.
Viral infection of reticular cells was confirmed by immunohistochemical and ultrastructural methods. Medullary sinus endothelial cells showed no significant structural or ultrastructural changes. However, a few capillary endothelial cells showed signs of virus infection from 4 dpi.
Immunocytochemical analysis using anti-CD3 antibody revealed significant T lymphocyte depletion from 4 dpi onward, with values dropping by the end of the experiment to half those in controls. Only a very small number of cells showed evidence of CSF virus infection.
Discussion
Analysis of bone marrow cell composition during the course of acute CSF revealed marked hypocellularity from 6 dpi onward, preceded by occasional foci of apoptosis affecting lymphocytes, neutrophils, and MKs. Apoptosis, though not associated with direct virus infection of the apoptotic cells themselves, was related to the presence of viral antigen in stromal and hematopoietic cells from 2 dpi, an earlier stage than that reported by other authors. 25 Lymphocyte apoptosis in bone marrow in CSF is similar to that found in other lymphoid organs and would account for the severe, early-onset lymphopenia characteristic of the disease. 10 , 22 , 24 , 27 Granulocyte proliferation would account not only for the presence of circulating immature forms but also for a significant increase in their relative proportion; however, subsequent apoptosis-induced granulocytopenia was not to be counteracted by regeneration. 25 Infection of stromal cells may play a major role in lymphocyte apoptosis and in depletion of the myeloid population due to indirect mechanisms after modification of the hematopoietic microenvironment regulating these cells. 7 , 17 , 33
Studies addressing the changes taking place in MKs in CSF commonly report both degeneration and depletion. 6 , 14 However, immunohistochemical examination of MKs stained for FVIII-rag showed that there is no significant change in MK numbers during the course of the disease. It has not been possible to establish any link between MK depletion and thrombocytopenia. The results obtained in this study suggest that degenerative processes are neither as marked nor as early in onset as those reported by other authors. 6 , 14 However, there is considerable morphological change indicative of a progressive functional alteration in MKs. Such changes include an increase in microMK numbers, the predominance of MKs with cloud-shaped nuclei, and a decrease in both cytoplasmic area and the ratio of nuclear area to total area. These changes are characteristic of the state of dysmegakaryocytopoiesis reported in myelodysplastic syndromes 19 and in HIV infection. 2 , 28 Proliferation of reticulin fibers is also a common finding in this type of pathology. 19 , 30 Impaired megakaryocytopoiesis in CSF may be related to various events taking place during the course of the disease. Chief among these are 1) the infection of stromal cells, mainly macrophages and reticular cells, both of which play a major role in regulating hematopoiesis in general and megakaryocytopoiesis in particular, through soluble chemical factors 1 , 13 , 32 , 33 and 2) depletion of T lymphocytes, which are also involved in modulating hematopoiesis. 13 , 17 These events may lead to impairment of the humoral balance by which megakaryocytopoiesis is regulated, giving rise to the MK seen. The fact that platelet-forming MKs are affected may be of some relevance with regard to the thrombocytopenia occurring at an earlier stage (2 dpi), which becomes more severe, without return to normal values of platelets.
Impairment of the humoral balance may also play an important role in the increased number of MKs undergoing apoptosis, 16 , 18 probably the degenerated MKs reported by Hoffmann et al. (1971) and Calderón et al. (1997), although they could be the result of an attempt by the bone marrow to counteract earlier thrombocytopenia, leading to exhaustion of platelet-forming MKs. 2 Given that macrophages retain their normal morphological and functional integrity and there is evident phagocyte activation, it is unlikely that the increase in apoptotic MKs is due to impaired phagocytosis by macrophages. 34 The low percentage of apoptotic MKs suggests that this phenomenon does not play an important role in the pathogenesis of thrombocytopenia in CSF, mechanism reported in subacute African swine fever. 9
If inhibition of thrombocytopoiesis were the cause of thrombocytopenia, one would expect an increase in the area of MK cytoplasm. Morphometric analysis of MKs in the present study suggested no such increase, indicating that this mechanism can be ruled out as a possible cause of thrombocytopenia in CSF. Moreover, the presence of viral antigen in circulating platelets 31 may be attributed to infection of MKs 12 together with effective thrombocytopoiesis.
Finally, evidence of erythroid cell infection suggests that this cell population may serve to spread the virus throughout the organism; erythroblasts and reticulocytes are frequently encountered in the general circulation in the final and late phases of the disease. 29
Footnotes
Acknowledgements
This work was supported financially by grants from DGESIC-MEC (PB98-1033) and Plan Andaluz de Investigación (AGR-0137). We appreciate the technical assistance of Central Servicie for Support for Research, University of Córdoba, Spain.
