Abstract
A nephrogenic rest was found in the kidney of an 11-week-old male Crl:CD (SD)IGS BR rat. Histologically, the rest was within the renal cortical interstitium and consisted of glomeruloid structures (primitive glomeruli) and inconsistently distinct basophilic tubules lined by a single layer of cuboidal to columnar cells and variably bordered by blastemal cells. Nephrogenic rests have not been reported previously in this rat strain.
The term nephrogenic rest has been proposed for abnormally persistent foci of embryonal cells that are capable of developing into nephroblastomas.8 In other reports, the generic term nephrogenic rest has been used for a focus of abnormally persistent nephrogenic cells, retaining cells that can be induced to form a Wilms' tumor.1 Lesions characteristic of nephrogenic rests have been reported in children, the Sprague-Dawley rat,6 and the Mastiff dog.11 When present in diffuse or multifocal distribution, nephroblastomatosis more specifically defines the morphologic change observed. In humans, nephrogenic rests are classified into perilobar and intralobar types. Both types are subdivided into categories that reflect their development and morphology. Incipient rests are microscopic lesions without indications of proliferation, maturation, or involution that occur in newborns or infants. Dormant rests are those lesions present in an older patient. Involuting (sclerosing) rests contain well-formed tubular structures with a single layer of low cuboidal basophilic epithelium with few or no mitotic figures surrounded by a dense collagenous stroma. Involuting rests progress to obsolescent rests, which are composed completely of collagenous stroma and are difficult to identify without the presence of other rests. Hyperplastic rests are diffuse or focal proliferative overgrowths and tend to preserve the original shape of the rest.1,8
An 11-week-old male Crl:CD (SD)IGS BR rat weighing 419 g and housed and maintained for 38 days in accordance with established guidelines10 was euthanatized at the end of a 28-day oral toxicity study. The animal had no prior history of illness, clinical signs of disease, or external or gross tissue abnormalities at the time of necropsy. Standard protocol tissues were collected and fixed in 10% neutral buffered formalin with the exception of the eyes and testes/epididymides, which were fixed in Davidson's solution4 and Bouin's solution,3 respectively. Tissues were processed, embedded in paraffin, sectioned at 4–6 μm, and stained with hematoxylin and eosin (HE). During routine histopathologic evaluation, the nephrogenic rest was observed in the midcortex of the right kidney.
Microscopically, the rest was within the cortical interstitium and was composed of inconsistently distinct basophilic tubules lined by a single layer of cuboidal to columnar cells variably surrounded by blastemal cells with formation of glomeruloid structures (primitive/abortive glomeruli) (Figs. 1, 2). There was no apparent compression or evidence of invasion of adjacent cortical parenchyma (Fig. 1). Multiple sections of paraffin-embedded tissue did not demonstrate any additional morphologic change, anaplasia, or evidence of nephroblastoma; the rest diminished in size in subsequent sections.
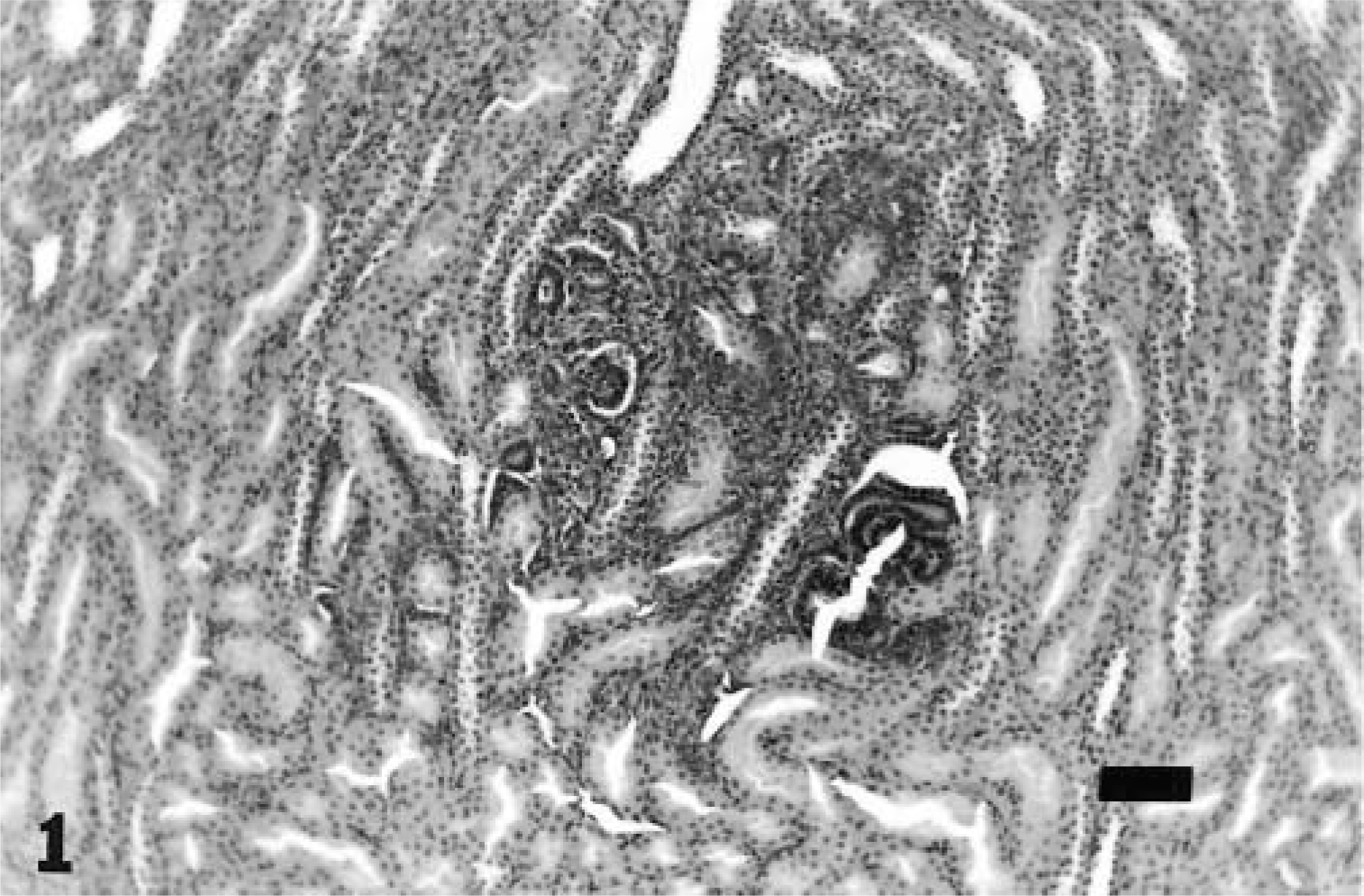
Kidney; 11-week-old rat. Nephrogenic rest within the renal cortex with no compression or evidence of invasion of adjacent cortical parenchyma. HE. Bar = 75 μm.
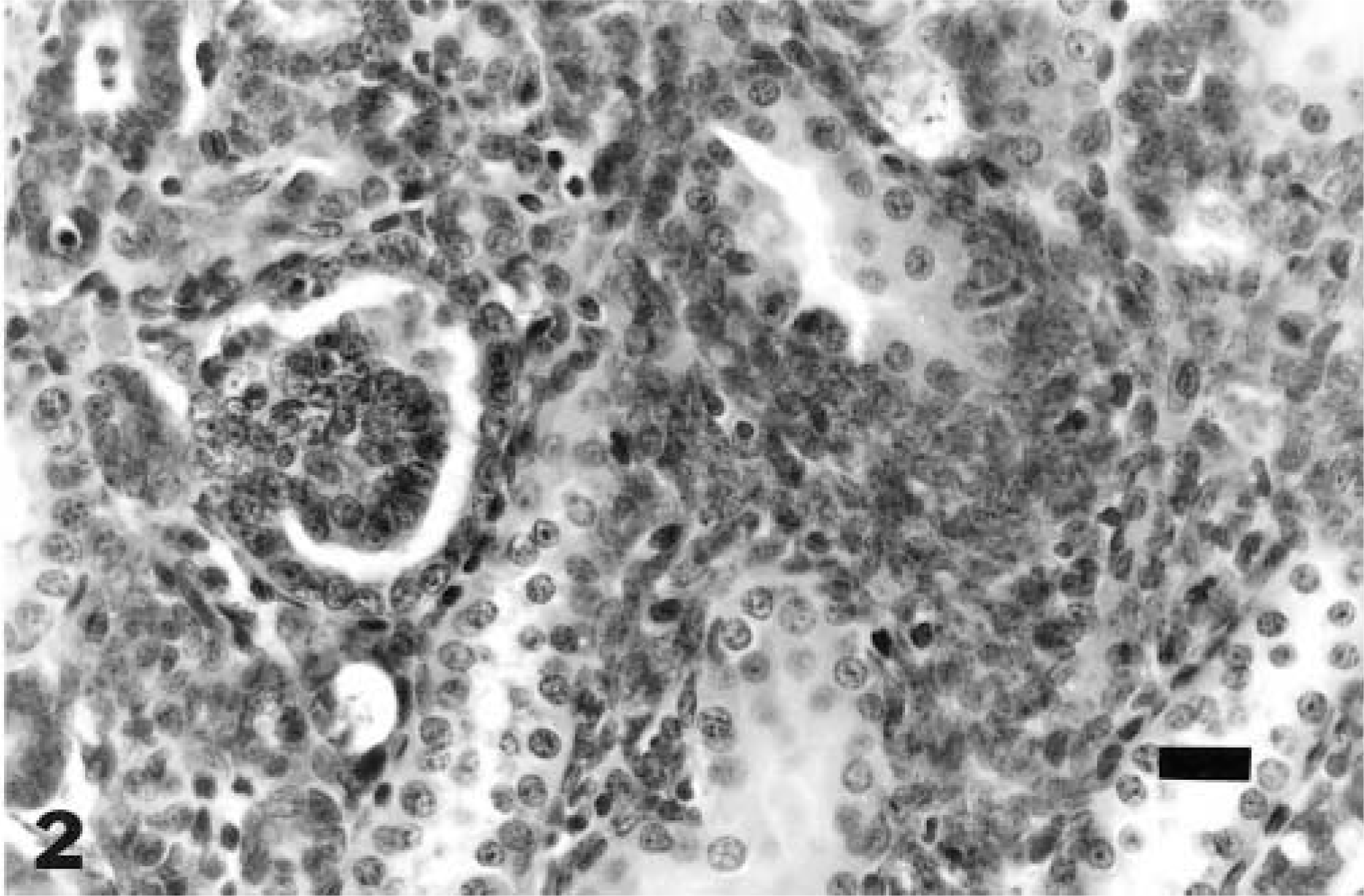
Kidney; 11-week-old rat. Basophilic tubules are lined by a single layer of cuboidal to columnar cells and bordered by blastemal cells and with glomeruloid structures (abortive/primitive glomeruli). HE. Bar = 20 μm.
In this rat, the nephrogenic rest was an incidental finding and was not observed in any other animals in the study. Based on previously defined terminology, this lesion would be considered an incipient rest because it was without indications of proliferation, maturation, or involution. The lack of expanding nodules with compressed peripheral zones and lack of connection with a larger solid tumor (as indicated by deeper sectioning) are consistent with characteristics of nephrogenic rests as reported in other species.11 Nephrogenic rests have the potential to progress toward hyperplasia, may regress and become obsolescent, or may progress to nephroblastoma.8 This proliferative potential and the neoplastic sequelae of nephrogenic rests are important considerations when laboratory animals are used as test systems for risk assessment of the effects of pharmaceuticals and chemicals in humans. If proliferative precursor lesions are found in laboratory animals, the proliferative potential may exist for humans exposed to similar compounds. Nephrogenic rests may arise during an initial, possibly inherited genomic failure of the ureturic bud to initiate normal nephrogenesis and may undergo neoplastic transformation with subsequent loss of certain cancer suppressor genes.11 Nephrogenic rests and nephroblastomatosis, precursors of nephroblastoma, are found in 25–40% of humans with similar lesions and are observed during approximately 1% of routine infant postmortem examinations.8 Nephroblastomas are true embryonal tumors arising from primitive nephrogenic blastema and in foci of renal dysplasia. Their presence in cartilage, collagen, bone, skeletal muscle, and adipose tissues normally not associated with the kidney indicates a pluripotential mesenchymal origin before the tissue becomes metanephrogenic. In some tumors, the mesenchymal components may be more predominant than the epithelial component, particularly in ruminants.5
Synonyms for nephroblastoma include embryonal nephroma, renal adenosarcoma, embryonal carcinoma, Wolffian duct tumor, and Wilms' tumor. These lesions occur primarily in younger animals2 and are usually unilateral.5 Nephroblastoma in rats generally have a cellular pattern characterized by blastemic, epithelial, and stromal elements. Blastemic cells form nests, cords, and alveolar and papillary structures. Within blastemic proliferations formed by cuboidal cells, organoid epithelial differentiation resembles glomeruli and tubules. Focal blastemic proliferations may represent early nephroblastomas.7 Nephroblastoma is the most common primary renal tumor of pigs and chickens. Incidence of 3.5 per million and 43.5 per million have been reported in the United Kingdom and United States, respectively. Nephroblastomas are reported less often in calves and dogs and are uncommon in other species. Various carcinogens, e.g., dimethylnitrosamine, may induce nephroblastoma and other tumors. Metastases to lung and liver occur in >50% of canine cases but are rare in pigs and calves. Differentiation to tubular and glomerular components indicates a good prognosis; anaplasia and sarcomatous stroma are associated more with metastases and thus have a poorer prognosis.5 Wilm's tumor 1 (WT1) gene activity on human chromosome 11p13 indicates its suppressive function in the development of Wilms' tumor, aniridia, genitourinary defects, and mental retardation (WAGR) syndrome. WT1 is expressed at late stages during urogenital system development in mammals. In Japanese eels, cloning of the WT1 counterpart (EWT1) demonstrated that the EWT1 protein may function in eel kidney development and may be a participant in the genesis of nephroblastoma, as in mammals.9
Nephrogenic rests raise questions of what molecular events cause rest formation and which factors controlling hyperplastic overgrowth, involution, and tumor induction within rests impact Wilms' tumor pathogenesis. Gene studies of nephrogenic rests and related material (most Wilms' tumors lack a deletion involving 11p1312) may prove beneficial in determining renal cellular differentiation. More than one type of molecular event in differentiation may be involved. Research on the morphologic differences between nephrogenic rests and renal neoplasia and on the relationship between diffuse overgrowth of rests and neoplastic changes will be important to clinicians, pathologists, and tumor biologists.1
Footnotes
Acknowledgements
We thank L. J. Watson and L. J. Meyers for histology and R. Phillips for her literature search.
