Abstract
Globoid cell leukodystrophy (GLD; Krabbe disease), is a rare heritable metabolic disorder in humans, dogs, mutant twitcher mice, and rhesus monkeys that is caused by a deficiency in the lysosomal enzyme galactocerebrosidase (GALC). GALC deficiency results in the accumulation of psychosine, which is toxic to oligodendrocytes and Schwann cells of the central and peripheral nervous systems. Clinical signs include hypotonia, mental regression, and death by 2 years of age in most human patients. Here we describe a domestic longhaired kitten with rapidly progressive neurologic disease and brain and spinal cord lesions characteristic of GLD. Pathologic hallmarks of the disease reflect the loss of oligodendrocytes and include myelin loss, gliosis, and the perivascular accumulation of large mononuclear cells with fine cytoplasmic vacuoles (globoid cells) in the peripheral and central nervous systems. Globoid cells were CD68 and ferritin positive, confirming their monocytic origin, and cytoplasmic contents were nonmetachromatic and periodic acid-Schiff positive.
Keywords
Globoid cell leukodystrophy (GLD; Krabbe disease) is an autosomal recessive neurologic disease of dogs, mutant twitcher mice, rhesus monkeys, and humans that results from a deficiency in galactocerebrosidase (GALC) enzyme activity.6 In dogs, GLD has been described most commonly in Cairn Terriers and West Highland White Terriers, with clinical signs arising between 1 and 3 months of age, typically beginning with limb weakness and tremors and progressing to muscular atrophy and neurologic degeneration. Dogs are typically euthanized by 9 months of age because of the severity of the disease.7,9 Humans with the infantile form of Krabbe disease usually present 3–6 months after birth with irritability, spasticity, and mental regression that progresses to a severe decerebrate condition and death before 2 years of age.2,10. To date, only a single report has been made of feline GLD, which occurred in two genetically related shorthaired kittens.3 Here we report a naturally occurring case of GLD in a domestic longhaired cat.
A 12-week-old intact female domestic longhaired kitten presented to the Veterinary Teaching Hospital at Colorado State University with a 4-week history of body tremors that progressed to tetraparesis, rear limb extensor rigidity, and a firm distended urinary bladder that was difficult to express (upper motor neuron bladder). By 15 weeks of age, the cat had hind limb paralysis and at 21 weeks of age, she developed acute respiratory distress and died. The early posterior dysmetria and incoordination in this kitten were similar to the single previous report of GLD in the cat3 and the clinical course described in the dog,7 and resembled signs of cerebellar hypoplasia.
Significant gross lesions consisted of kyphosis of thoracic and lumbar vertebrae T10–L1 and hindlimb muscle atrophy. The vertebral bodies of T12 and T13 were slightly more narrow ventrally than dorsally, resulting in midly deformed wedge-shaped vertebrae. Histopathologically, marked alterations were found in all brain and spinal cord sections examined. These included cerebral cortex, striatum, thalamus, hippocampus, cerebellum, cerebellar peduncle, medulla oblongata, and spinal cord. All white matter areas had diminished stain intensity with Luxol fast blue (LFB) staining and white matter vacuolation occurred in areas of severe involvement. Vessels of the white matter had abundant accumulations of large, pale mononuclear cells with fine cytoplasmic vacuoles (globoid cells) located largely in the perivascular space but in vascular walls as well (Fig. 1). Globoid cells were 20–30 μm and had large, eccentric oval nuclei with coarsely stippled chromatin. White matter parenchyma had increased numbers of glial cells, including hypertrophic astrocytes. Gray matter areas were unaffected. No evidence of cerebellar hypoplasia was found in this kitten.
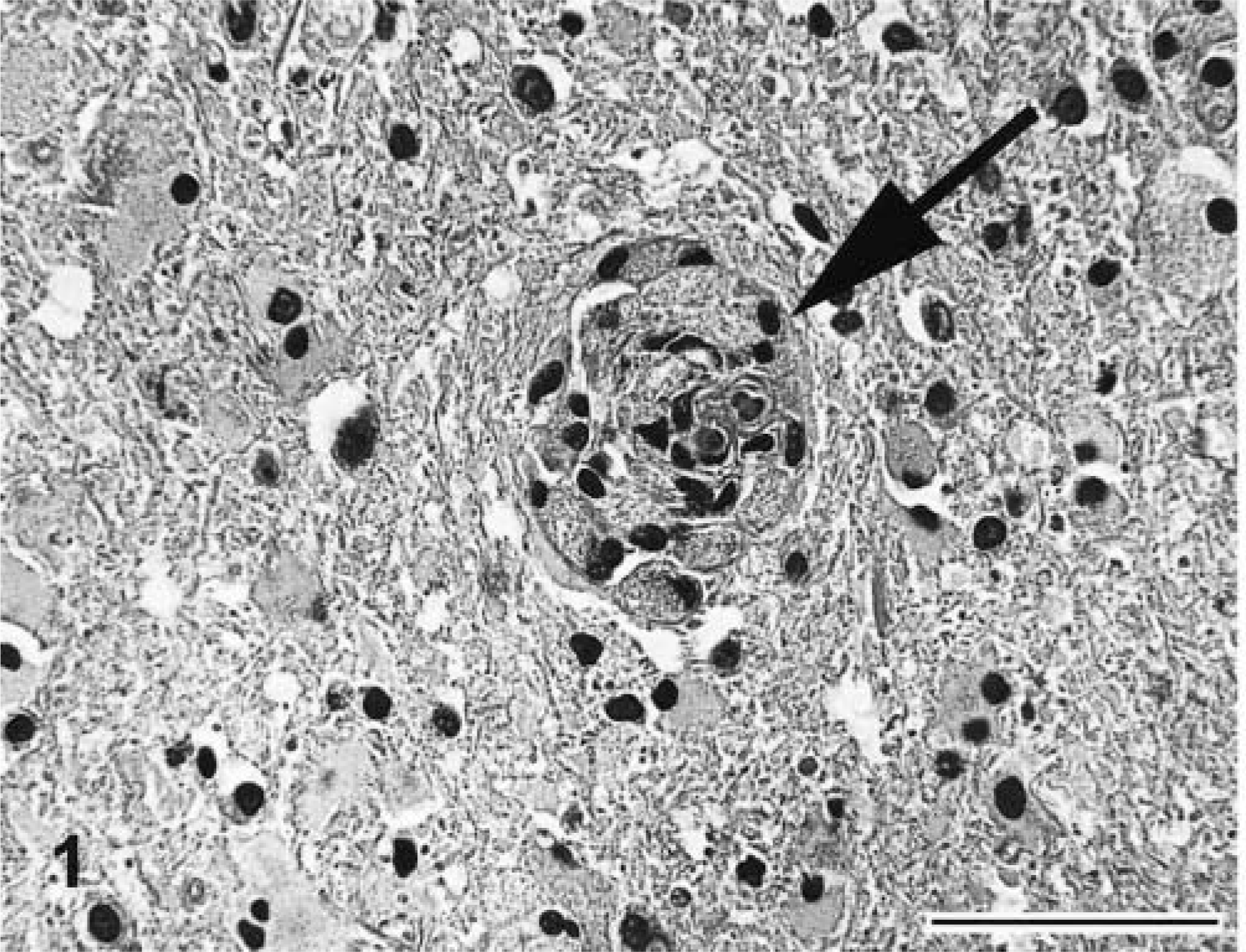
Brain at subcerebellar white matter; 21-week-old kitten. Large vacuolated mononuclear cells (globoid cells) accumulate in vascular walls and perivascular spaces (arrow) and are characteristic of GLD. HE. Bar = 100 μm.
Regional neuroanatomic differences were noted that included extensive vacuolation at the junction between the gray and white matter of the cerebral cortex; perivascular accumulations of eosinophilic, homogenous material indicative of vasogenic edema in the thalamus; especially prominent vascular accumulations of globoid cells in the white matter of cerebellar peduncles (Fig. 2); marked rarification of white matter parenchyma; and a small number of spheroids in the ventral funiculus of the spinal cord. Lung histopathology revealed diffuse mild congestion and edema. The cause of the respiratory distress is unknown.
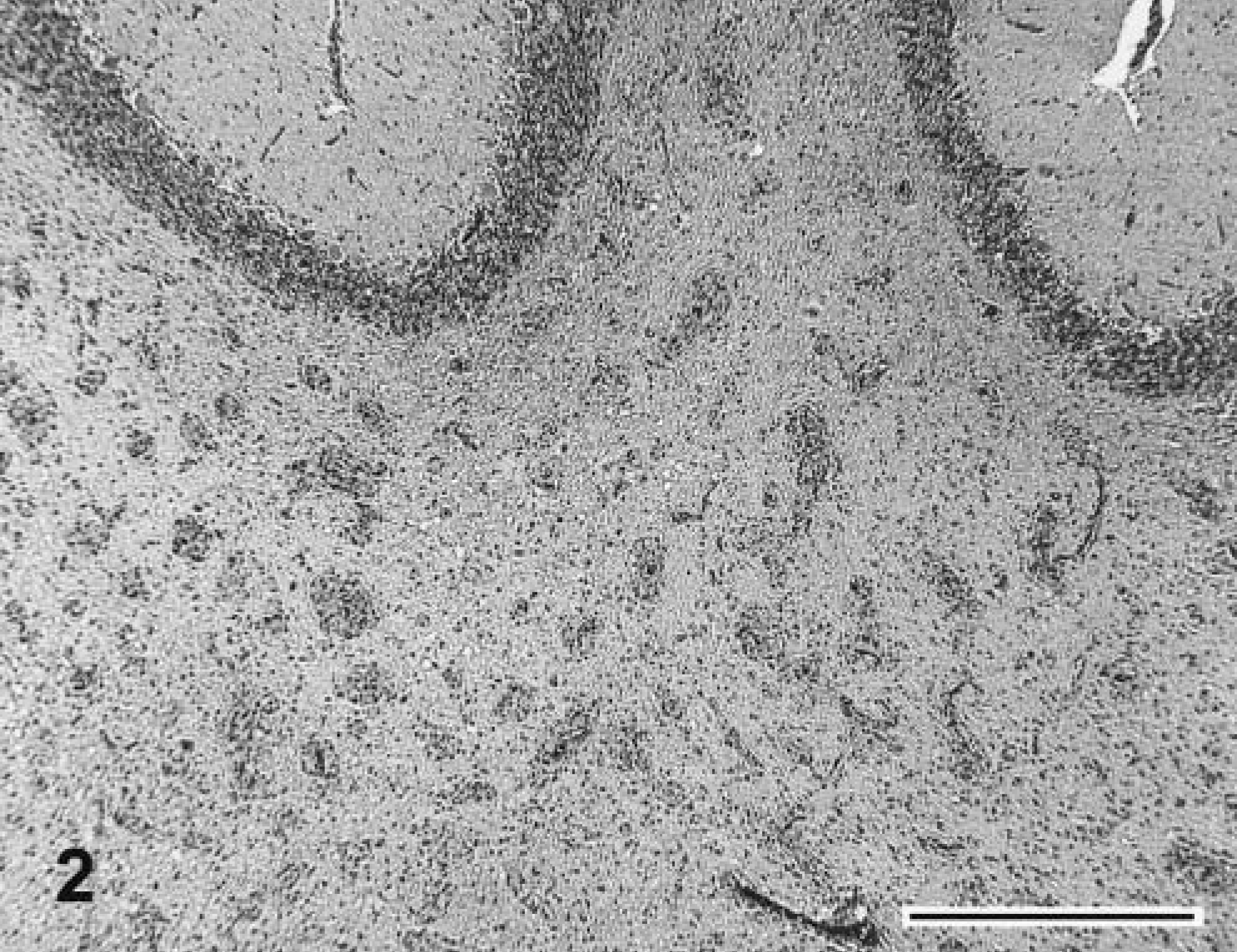
Brain at subcerebellar white matter; 21-week-old kitten. Prominent vascular accumulations of globoid cells and gliosis are evident. HE. Bar = 1 mm.
In the spinal cord, LFB staining was characterized by extensive, diffuse pallor of white matter indicative of myelin loss; however, spinal nerve roots had the staining characteristics of normal myelin. Myelin loss as assessed by LFB pallor was also significant in the subcortical white matter of the cerebral and cerebellar cortex, the pons, and the fimbria of the hippocampus. The thalamus was relatively spared of extensive myelin loss. Globoid cell cytoplasmic contents were periodic acid–Schiff stain positive and nonmetachromatic. Bodian staining demonstrated the presence of nerve fibers in all white matter areas, including those of marked pallor. By immunohistochemistry, globoid cells were CD68 and ferritin positive, suggesting an origin of bone marrow–derived macrophages. This finding is consistent with previous findings of GLD in humans.2 Controls used included the substitution of concentration-matched irrelevant mouse and rabbit antibodies.
GLD is an autosomal recessive disorder resulting in a deficiency of GALC enzyme activity (confirmed by enzyme analysis6). Unfortunately, enzyme analysis on this kitten was not possible because all tissues were formalin fixed. GALC is actolipids involved in myelin production.9 Therefore, in the absence of functional GALC, galactosphingolipids, including psychosine, accumulate. Psychosine is highly toxic to oligodendrocytes8 and myelin production and maintenance cease with degeneration of these cells.4 In GALC-deficient dogs, psychosine has been shown to accumulate during active myelination and is elevated by 4 months of age, resulting in toxicity and death of oligodendrocytes and a scarcity of myelin.7,9 Neuropathologic lesions of affected humans and other animals are typified by extensive demyelination, globoid cell accumulation in the white matter, and severe gliosis.2,8
In this kitten, the appearance of the globoid cells may raise the question as to the possible differential diagnosis of idiopathic granulomatous inflammation. However, the homogenous population of large globoid cells accumulating perivascularly along with the LFB stain demonstrating myelin loss characterize this disease as a myelin disorder. Moreover, the accumulation of globoid cells seen in this kitten is characteristic of GLD previously described in other species.9 In contrast to the previous report of feline GLD involving domestic shorthaired kittens, the present case involves a longhaired kitten. Unfortunately, this kitten was acquired from a pet store and we were unable to characterize the familial history. The accumulation of globoid cells in GLD differs significantly from other lysosomal storage diseases where storage of hydrophilic substrates in lysosomes occurs in many cell types.9
Recently, Im et al.1 showed the molecular target of psychosine to be a G protein–coupled receptor known as T cell death–associated gene 8. Moreover, the in vitro binding of psychosine to cells expressing this receptor leads to the formation of globoid multinuclear cells.1 Studies of humans with GLD treated early with bone marrow transplants have shown slowing of disease progression, providing hope for possible therapeutic intervention strategies.5 In summary, GLD does occur in the cat and the progression and neuropathology of the disease mirror those of the naturally occurring disease described in dogs, rhesus monkeys, mutant twitcher mice, and humans.
Footnotes
Acknowledgements
We thank Dr. Susan Kraft for reviewing the radiographs on this kitten.
