Abstract
Lesional skin of cats with allergic dermatitis has a cellular infiltrate and a CD4/CD8 ratio comparable to that in humans with atopic dermatitis. CD4+ helper T cells and in particular cells belonging to the Th2 subset play an important role in disease pathogenesis in humans. We investigated the cytokine pattern of CD4+ T cells in situ, with special emphasis on the putative presence of cells producing interleukin 4 (IL4), in cats with allergic dermatitis. Immunohistochemical procedures were used to determine that CD4+ T cells in lesional and nonlesional skin of cats with allergic dermatitis can produce IL4, as occurs in humans. Lesional and nonlesional skin of cats with allergic dermatitis had significantly more IL4+ T cells (P = 0.001) than did skin of healthy control cats. Double staining indicated that all IL4+ cells were positive for pan-T or CD4 markers. Double labeling for mast cell chymase and IL4 stained primarily different cells. Western blotting demonstrated cross-reactivity between the antibody against human IL4 and a feline recombinant IL4. These results indicate that IL4 is primarily produced by CD4+ T cells and is also present in clinically uninvolved skin, indicating a role in the pathogenesis of allergic dermatitis in cats.
Keywords
Atopic dermatitis in humans is mediated by antigen-specific IgE, and an important role has been ascribed to CD4+ helper T cells (Th), in particular cells of the Th2 subset.20 Interleukin 4 (IL4) is produced primarily by Th2 cells in humans and has many biologic activities, including (with IL13) stimulation of the production of IgE.14,15 In lesional skin of human patients with atopic dermatitis, IL4-producing CD4+ allergen-specific T cells occur in high density.22 Mast cells are considered another potential source of IL4; however, studies on IL4 production by cutaneous mast cells have produced conflicting results.12,20
In cats, the diagnosis of atopic dermatitis is generally based on a history of pruritus, recurrent or chronic crusted papules defined as miliary dermatitis, results of intradermal skin tests, response to glucocorticoids, histopathology, and the exclusion of other pruritic skin diseases such as food hypersensitivity, dermatophytosis, flea bite hypersensitivity, and parasitic diseases.18,25 Histology of lesional skin of cats with allergic dermatitis (AD) reveals a perivascular to diffuse infiltrate of mast cells, eosinophils, lymphocytes, and macrophages,10 comparable to the infiltrate that occurs in humans with atopic dermatitis.19 Lesional skin of cats with AD shows a predominant increase of CD4+ T cells and a CD4/CD8 ratio of 3.9 ± 2.0 (± SD),16 similar to that found in humans.28 A putative feline IgE1,7–9 and the involvement of heat-stabile cytophilic immunoglobulins17 have been reported in cats with AD. These observations indicate a comparable disease pathogenesis of atopic dermatitis in cats and in humans. Because interleukins play an important role in disease pathogenesis, the next step in the characterization of AD in cats was aimed at the elucidation of the cytokine pattern of CD4+ T cells in situ.
Materials and Methods
Animals
Skin biopsies were obtained from five client-owned domestic shorthair cats with a history of recurrent miliary dermatitis and pruritus responsive to glucocorticoids. These cats were 3–10 years of age (○ = 6.5 years; one spayed female and four castrated males). Food hypersensitivity, dermatophytosis, and ectoparasites had been excluded by standard diagnostic procedures.18 Intensive measures to prevent flea infestation were carried out for all animals before skin testing. Except for the described skin problems, the animals were healthy. All cats were withdrawn from injectable glucocorticoids for at least 6 weeks and received no antihistamines or oral glucocorticoids for a minimum of 4 weeks prior to skin testing. Only cats with the histopathologic features of AD were included in this study.10 Five healthy domestic shorthair cats 2–5 years of age (○ = 4.0 years; two spayed females and three castrated males) were used as normal control animals. Histopathology of skin biopsies of these cats was compatible with that of normal skin.
Skin testing
All cats were examined using a commercially available intradermal skin test for dogs, which contains 22 standardized allergens, phosphate buffer as a negative control solution, and 0.01% histamine solution as a positive control (ARTU Biologicals, Lelystad, the Netherlands). The cats were tested while sedated with 10 mg/kg ketamine, 1 mg/kg xylazine, and 50 µg/kg atropine sulfate administered intramuscularly. Intradermal skin tests were performed after clipping the hair from nonlesional skin of the lateral thorax. The skin was monitored for 30 minutes after injection of the allergens, and reactions were interpreted at 20 minutes after injection with transillumination of the injection site. Reactions were considered positive when injection sites were larger than half of the sum of the diameters of injection sites of the two control solutions.
Sample collection
Subsequent to the skin test, 6-mm punch biopsies of lesional skin and nonlesional skin were obtained under sedation, immediately snap-frozen in liquid nitrogen, and stored at −80 C until used. Two biopsies of comparable lesions were used for hematoxylin and eosin (HE) staining to establish the histopathologic features of the AD. The skin biopsies from the control animals were also obtained under sedation and processed in the same way.
Immunohistochemistry
Single staining was performed with a mouse monoclonal antibody (mAb) to human IL4 (1-41-1, Novartis, Vienna, Austria). Biopsy sections were cut at 6 μm, air dried overnight, and fixed in acetone for 7 minutes. Nonspecific binding was blocked by preincubation for 25 minutes with phosphate-buffered saline (PBS) containing 10% horse serum and 10% cat serum. The mAb to human IL4 was applied for 1 hour in different dilutions. Horse anti-mouse biotin-conjugated antibody (Vector, Burlingame, CA) was used as a second step followed by alkaline phosphate-conjugated streptavidin (Dakopatts a/s, Glostrup, Denmark). Staining was developed using Naphthol AsBi (N-2250, Sigma, St. Louis, MO) as substrate and New Fuchsin as a chromogen, resulting in pink-red staining. The sections were counterstained with diluted hematoxylin. Control staining was performed with an isotype-matched antibody (mouse IgG1 standard 102-01, Southern Biotechnology Association, Birmingham, AL). Two mouse mAbs (MD1 and MD2) to human interferon (IFN-γ) and a polyclonal rabbit anti-human IFN-γ antibody (all three antibodies from P. H. van der Meide, TNO, Rijswijk, the Netherlands) tested in different dilutions on cryostat sections did not result in any positive staining.
For the double staining procedures, sections were air dried overnight and fixed in acetone. Nonspecific binding was blocked by preincubation for 25 minutes with PBS containing 10% horse serum and 10% cat serum. As first primary antibody, a dilution of the mouse mAb to human IL4 was applied for 1 hour. A horse anti-mouse biotin-conjugated antibody (Vector) was used as a second step, followed by blocking with rabbit anti-mouse serum (Dakopatts a/s). Murine mAbs to feline pan-T (1.5721) or feline CD4 (vpg 3427) were administered and left for 1 hour as the second primary antibody, followed by streptavidin–β-galactosidase (Crowley Down, Sussex, UK) and rabbit anti-mouse alkaline phosphate-conjugated serum (Dakopatts a/s). After staining the slides with X-gal (5-bromo-4-chloro-3-indolyl-β-
Positively stained cells in the superficial dermis were counted with a square graticule in four adjacent high-power fields (400×). Cells in hair follicle walls were not counted because of differences in hair follicle density and plane of cutting. The deep dermis was excluded because of variation in depth of dermis.
Cloning and sequencing of feline IL4
Peripheral blood lymphocytes from a specific-pathogen-free cat were stimulated in vitro with 6 µg/ml Concanavalin A and 100 U/ml IL2 for 48 hours. Cells were pelleted (800 × g) for 10 minutes and then resuspended in 1 ml Trisolv (Biotecx Laboratories, Houston, TX) for total RNA extraction according to the procedure recommended by the manufacturer. RNA was resuspended in 40–60 µl diethyl pyrocarbonate–treated water with 0.5 µl RNase inhibitor and 1 µl DNase. The extracted RNA was incubated for 1 hour at 37 C to remove DNA and then heated for 5 minutes at 95 C to inactivate DNase. RNA concentration was determined spectrophotometrically by measuring the A260 in a microcuvette (Genequant II, Pharmacia Biotech, Piscataway, NJ). Total RNA (1 µg) was reverse transcribed (42 C for 1 hour, 95 C for 10 minutes) using an RT-Kit (Promega Corp., Madison, WI) and an oligo-dT primer. The feline IL4 cDNA was amplified with a single-round polymerase chain reaction (PCR) performed in a Biotherm Bio-oven III (Biotherm Corp., Fairfax, VA).
The PCR amplification consisted of 35 cycles (30-second template denaturation at 94 C, 30-second primer binding at 55 C, and 45-second elongation at 72 C), with an initial 5-minute denaturation at 94 C and an extra 10-minute elongation at 72 C for completion. The primers used were derived from the published feline sequence (GenBank accession No. X87408) with an added BamHI or EcoR1 restriction enzyme site on the forward (5′-GCGGGATCCATGGGTCTCACCTACCAAC-3′) and reverse (5′-GCGGAATCCCAGCTTCAGTGCTTTGAG-3′) primer, respectively. The PCR product was ligated into a pCR3.1 plasmid and transformed into Escherichia coli strain One Shot (TOP10F′) by TA cloning (Invitrogen Corp., Carlsbad, CA). The feline IL4 sequence was cut out of the pCR3.1 at the BamHI and EcoR1 sites, ligated into pGEX2T (Pharmacia Biotech), and transformed into One Shot cells. Sequences inserted into pGEX2T are expressed as a fusion protein with glutathione-S-transferase (GST). The sequence and coding frame of the fIL4-pGEX2T was verified using the chain-termination reaction (Sequenase, U.S. Biochemical, Cleveland, OH) and standard techniques with 5′ and 3′ pGEX sequencing primers (Pharmacia Biotech).
Expression and western blotting of recombinant feline IL4
The fIL4-pGEX2T plasmid was transformed into E. coli strain BL-21 (Novagen, Madison, WI). Transformed bacteria were grown at 30 C until the A600 reached 0.6. Protein expression was then induced by adding isopropyl-β-
Bacterial lysates were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis and then transferred to nitrocellulose membranes. The membranes were probed with goat anti-GST polyclonal antibody (Pharmacia Biotech) or mouse anti-human IL4 mAb (1-41-1, Novartis). The membranes were then incubated with the appropriate anti-goat or anti-mouse antibody conjugated to horseradish peroxidase. The membranes were developed using Sigma FAST diaminobenzidine with metal enhancer in 0.1 M imidazole (pH 7).
Recombinant feline IL2, used as a positive control, was generated as described for IL4. The recombinant IL2 is biologically active and was used to generate mAbs against feline IL2 (G. Dean, unpublished data).
Statistical analysis
For the statistical analysis of the immunohistochemistry data, Student's t-test (P < 0.05) was used.
Results
Histopathology
In the biopsies of lesional skin of the five cats with AD, the epidermis was hyperplastic, with occasional exocytosis of lymphocytes. A mixed perivascular infiltrate with a variable number of mast cells, eosinophils, neutrophils, macrophages, and lymphocytes was present in the dermis. The epidermis of the nonlesional skin was either normal or showed minimal hyperplasia. The cellular infiltrate in the dermis of nonlesional skin contained only a few mast cells and lymphocytes and sometimes a few eosinophils. Neutrophils, however, were not present. In the control cats, the epidermis was thin, with minimal cellularity of the dermis.
Intradermal skin test
Three of five cats with AD had immediate skin test reactivity to one or more allergens. These cats had one to three positive reactions to the following allergens: grass pollen, weed pollen, Tyrophagus putrescentiae, Dermatophagoides farinae, and D. pteronyssinus. One cat had a positive reaction to flea allergen in addition to the positive reactions to the aeroallergens. Two cats had negative skin test results but showed positive reactions to the histamine control. The group of five cats with miliary dermatitis was further defined as having AD; this group included cats with atopic dermatitis and others with additional flea bite hypersensitivity. Positive reactions to the allergens did not occur in the control cats.
Immunohistochemistry
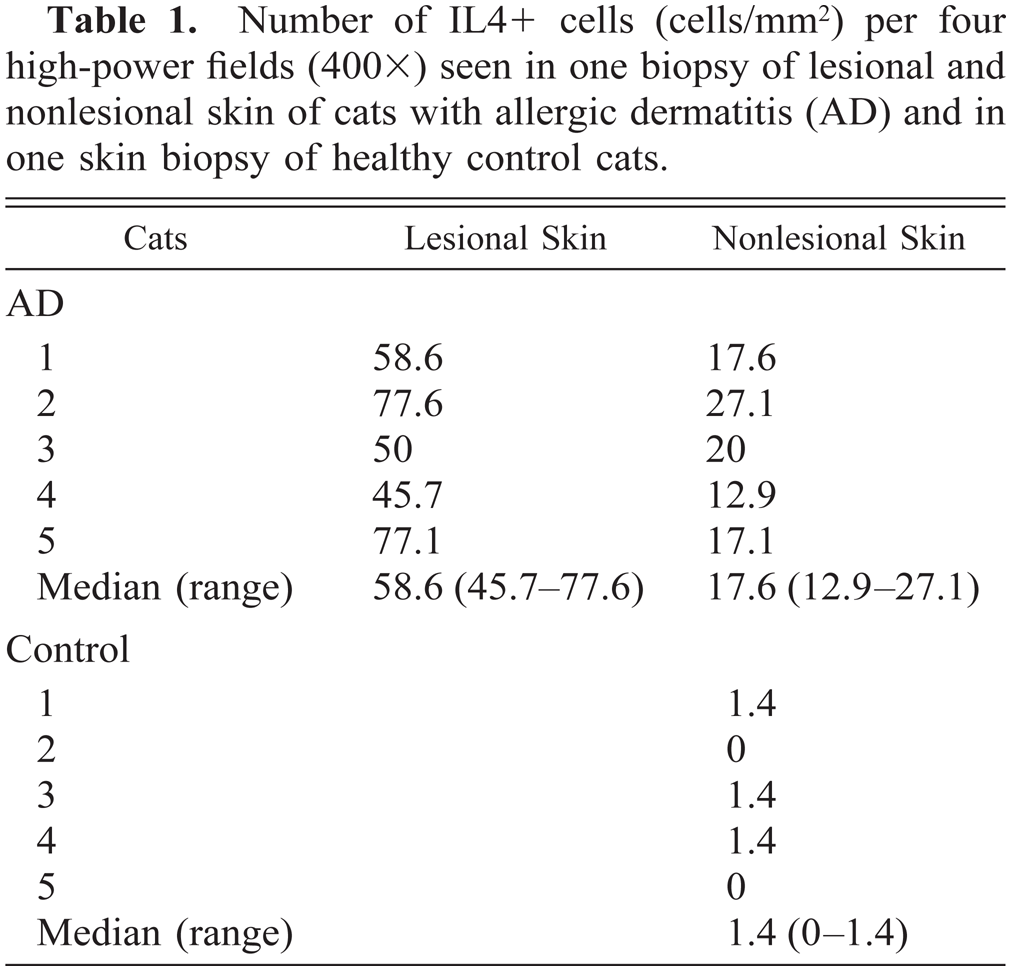
Exocytosis of IL4 + cells was found in the lower part of the epidermis in lesional skin. In lesional and nonlesional skin, predominantly in the superficial dermis, a perivascular infiltration with IL4+ cells was found (Figs. 1, 3). The isotype-matched control antibody resulted in no staining. In lesional skin of the allergic cats 58.6 (45.7–77.6) (median and range) IL4+ cells/mm2 were found, whereas in the nonlesional skin of the same animals 17.6 (12.9–27.1) IL4+ cells/mm2 were present (Table 1). A very limited number of cells stained positively for IL4 in the superficial dermis of the control animals (1.4, 0–1.4) (Table 1, Fig. 2). In addition, no intraepidermal positively staining cells were found. These differences were significant (P = 0.001).
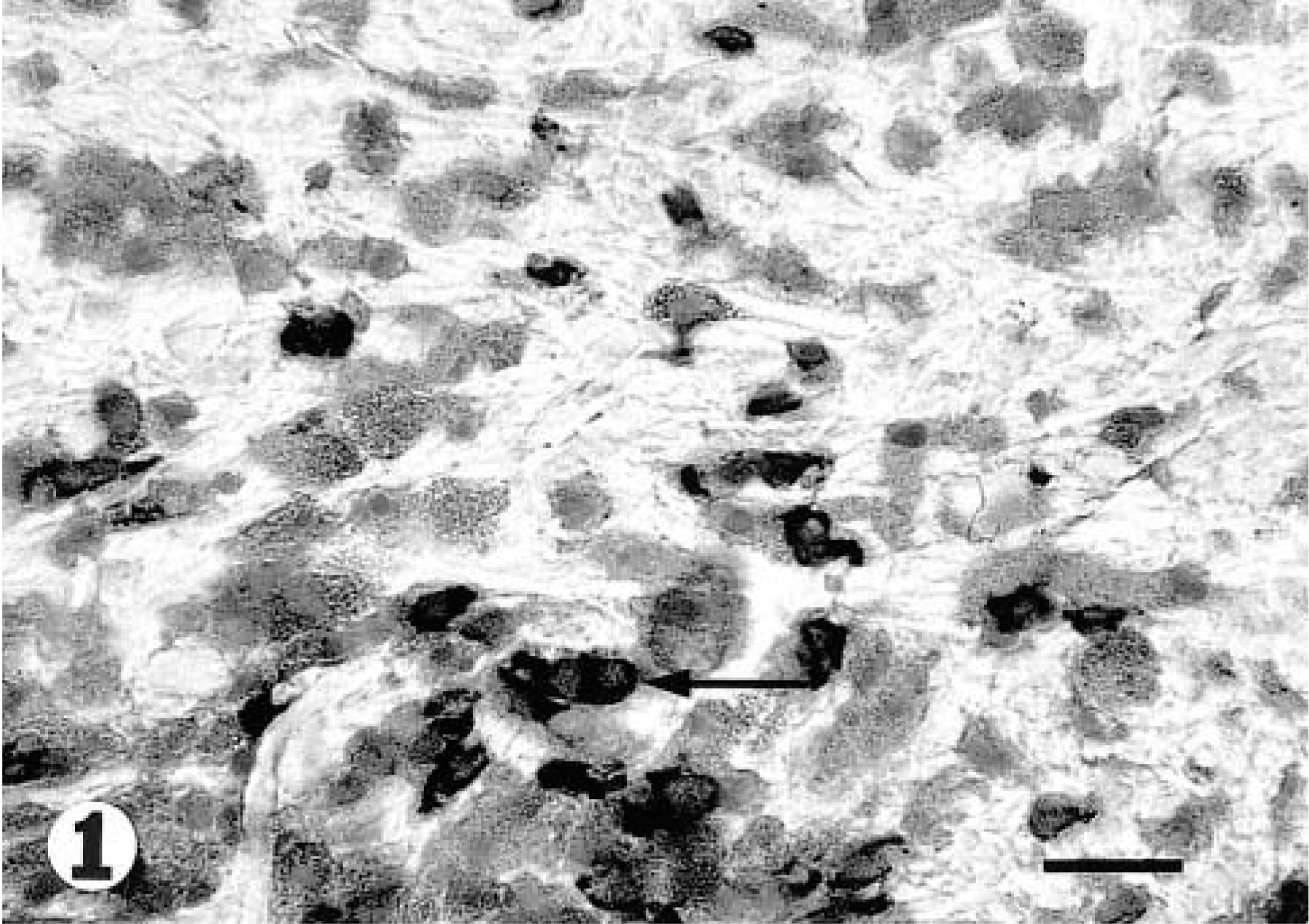
Lesional skin; cat with allergic dermatitis. Cryostat section stained for IL4 with a shows a number of positively staining cells (arrow). Biotin–streptavidin–alkaline phospate complex method. 40×. Bar = 30 μm.

Skin; healthy cat. Section lacks positively stained cells. 40×. Bar = 30 μm.
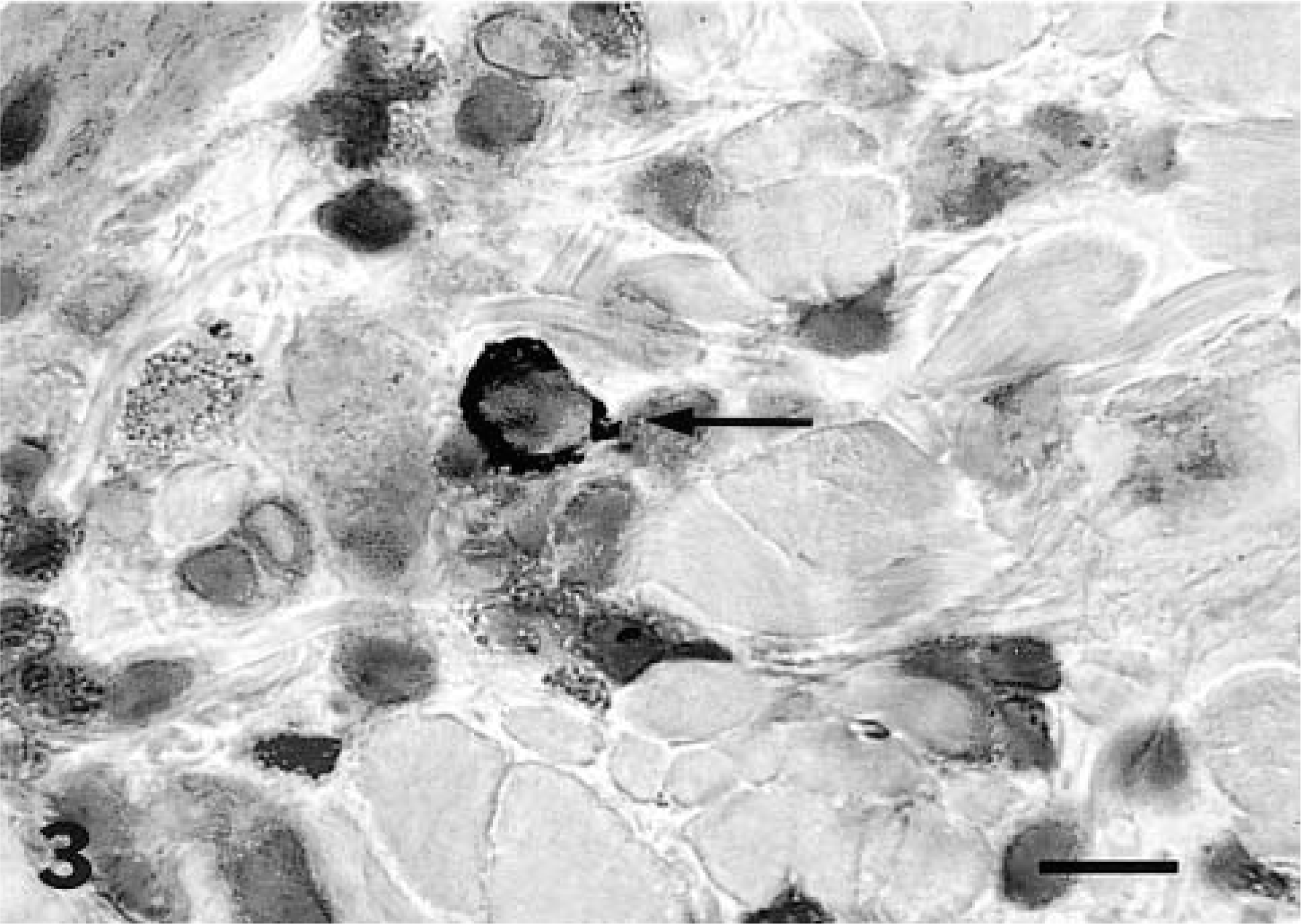
IL4+ cell (arrow); cat. The mast cells to the left of the IL4+ cell did not stain with this antibody. 100×. Bar = 90 μm.
Number of IL4+ cells (cells/mm2) per four high-power fields (400×) seen in one biopsy of lesional and nonlesional skin of cats with allergic dermatitis (AD) and in one skin biopsy of healthy control cats.
To determine whether the IL4+ cells were T cells, sections were double stained for either pan-T or CD4 in combination with anti-IL4. All IL4+ cells also stained positively for pan-T and CD4 within the epidermis and dermis. Single IL4+ cells were not observed, indicating that in cat skin all IL4+ cells were indeed CD4+ T cells. In the double labeling experiment for IL4 and mast cell chymase, there was only occasional minimal double staining for IL4 and mast cell chymase.
Western blotting of recombinant feline IL4
On western blots (Fig. 4), recombinant feline IL4 (rf-IL4) and recombinant feline IL2 (rf-IL2) reacted with a polyclonal antibody against the fusion protein (GST). The rf-IL4 but not the rf-IL2 cross-reacted with the anti-human IL4 mAb 1-41-1 (Fig. 5).
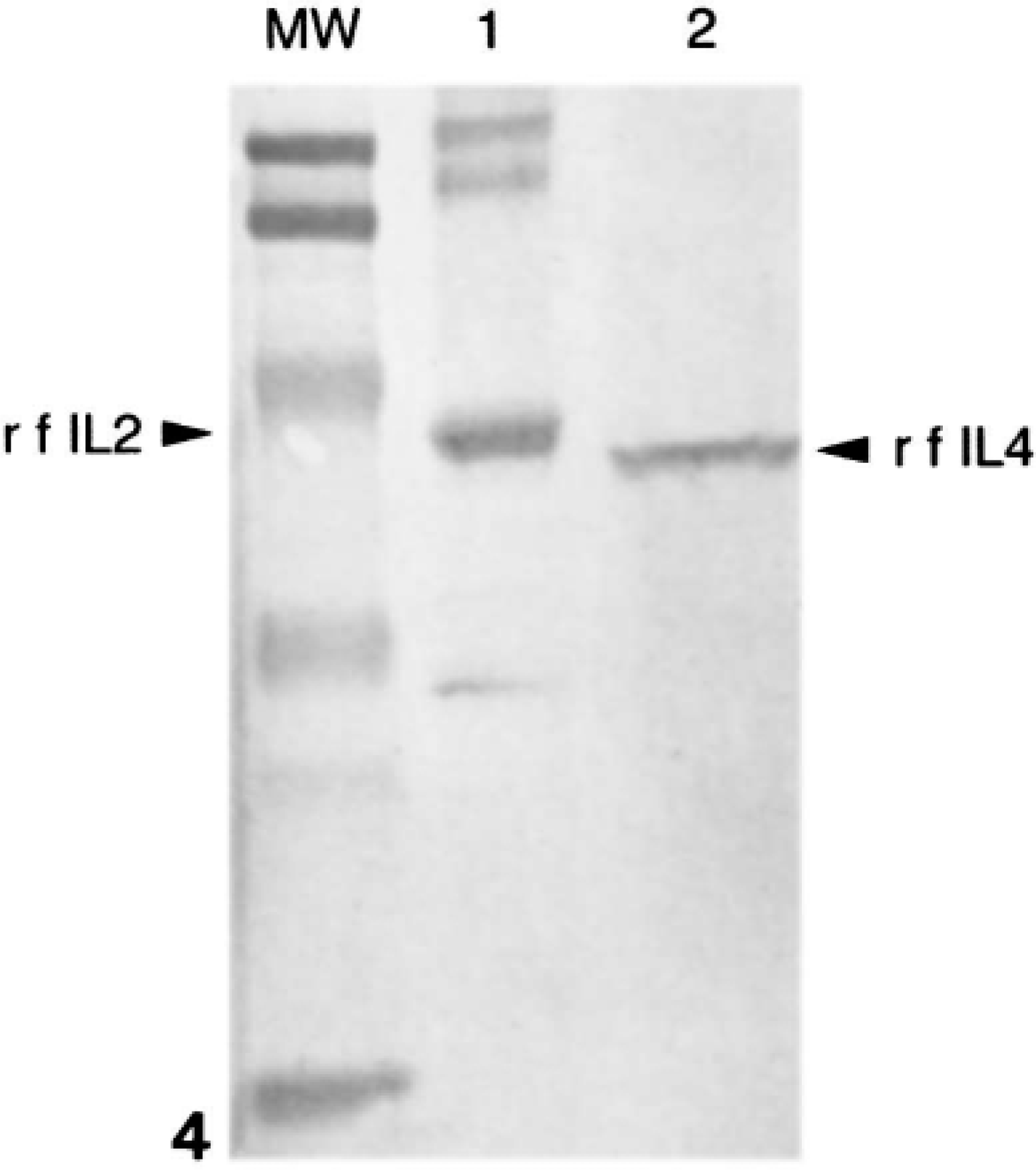
Expression and immunoreactivity of recombinant feline (rf) IL4. rfIL2 (lane 1) and rfIL4 (lane 2) were expressed as fusion proteins with glutathione-S-transferase (GST), electrophoresed on a 15% polyacrylamide gel, and transferred to a nitrocellulose membrane. Membranes were then probed with anti-GST polyclonal antibody.
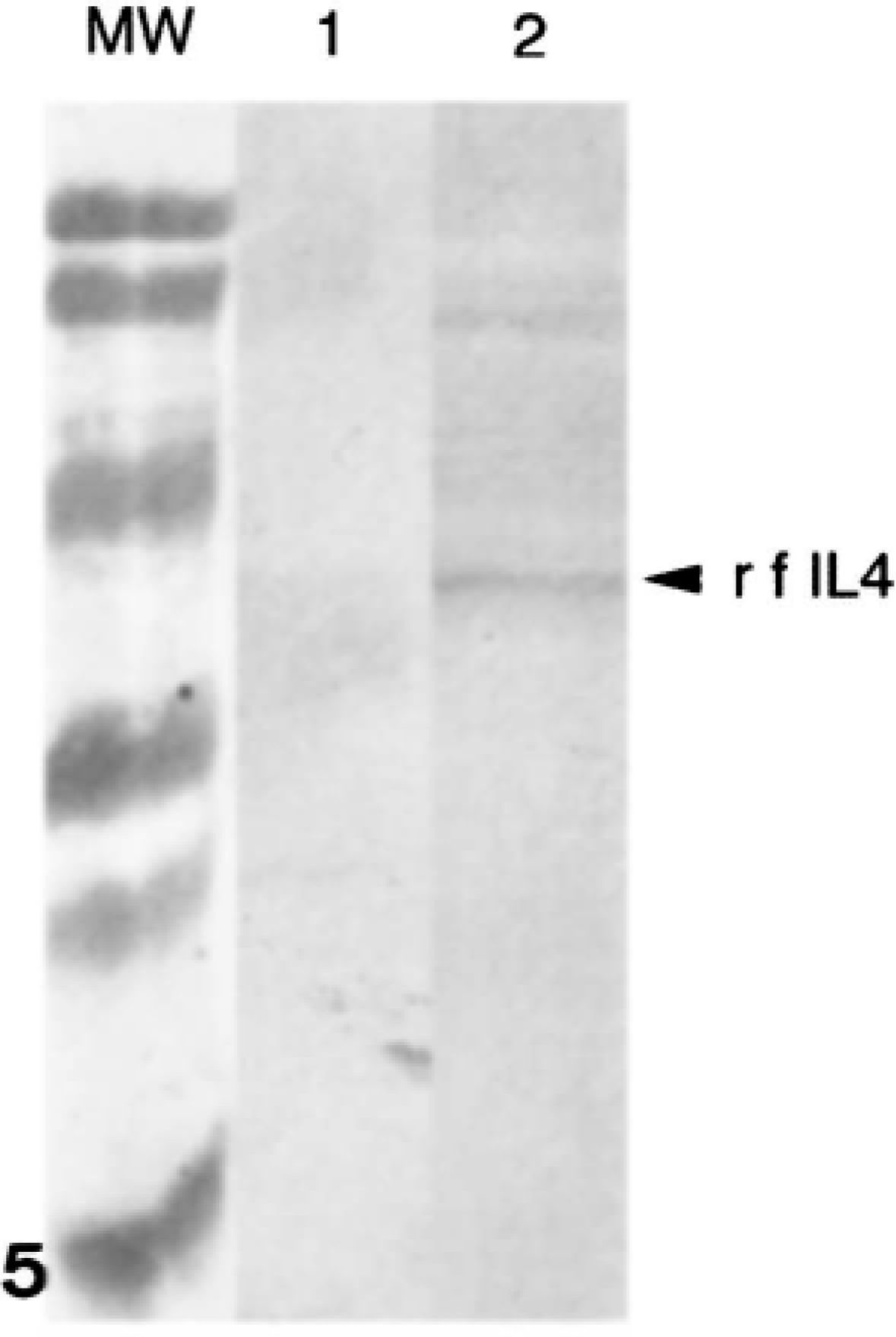
Expression and immunoreactivity of recombinant feline (rf) IL4. rfIL2 (lane 1) and rfIL4 (lane 2) were expressed as fusion proteins with glutathione-S-transferase (GST), electrophoresed on a 15% polyacrylamide gel, and transferred to a nitrocellulose membrane. Membranes were then probed with anti-GST polyclonal antibody.
Discussion
Atopic dermatitis in humans is considered to be a T-cell-mediated disorder, and an important role has been ascribed to CD4+ T cells, especially the Th2 subset.5 In this study, T cells of cats with a history and clinical signs of pruritic miliary dermatitis compatible with AD were investigated. AD in cats is associated with a predominant increase of CD4+ T cells in lesional skin.16 As the next step in the characterization of regulatory mechanisms, the cytokine production by these cells was investigated.
Cats with negative intradermal skin test results (n = 2) were included in this study because negative skin test results do not exclude atopic dermatitis and because these skin tests are considered less reliable in cats than in dogs.13,26 In 20% of humans with atopic dermatitis (those who have the so called intrinsic type), skin tests are negative, and these patients have no measurable allergen-specific IgE in serum.24
Our findings that AD cats had a significant increase in IL4+ cells in lesional and clinically noninvolved skin compared with skin of healthy control cats is compatible to what has been described in humans with atopic dermatitis.11 Double staining for IL4 and CD4 or IL4 and pan-T revealed that all cells staining positively for IL4 also stained positively for CD4 or pan-T. This finding is in agreement with findings in humans with atopic dermatitis using the same antibody against human IL4.20 In that study staining for IL4 was only present in the CD4+ population; no mast cells or eosinophils were positive.20
We did not find a correlation between positive skin test results and number of IL4+ cells. In humans with atopic dermatitis, a relationship between skin-infiltrating Th2-like cells and allergen-specific IgE response could not be demonstrated.23 In addition, the expression of IL4 mRNA changes with chronicity of the lesion in patients with atopic dermatitis.11 Although clinically similar lesions were biopsied, the chronicity of the skin lesions in these cats may have varied.
It is not known whether cats display distinct polarized Th cell subsets as do mice or whether feline Th cell subsets are more comparable to those of humans, where many of the Th1 and Th2 cells produce a mixed pattern of cytokines.15 It remains unclear whether these feline IL4+ T cells are Th2-like cells or Th0 cells, because staining with anti-human IFN-γ antibodies was unsuccessful and staining for other cytokines was not performed.
In humans, mast cells and eosinophils at mucosal sites and mast cells in skin are able to produce IL4.3,4,12 In the present study, the putative feline IL4 was produced primarily by T cells; no single cells staining positively for IL4 were found in a double staining for CD4 or pan-T and IL4. These findings were confirmed by the double labeling experiment for IL4 and mast cell chymase, in which different cells stained positively. These results are similar to those of a study in humans with atopic dermatitis using the same antibody reactive with IL4. In that study, only CD4+ cells and no mast cells or eosinophils stained positively for IL4.20
Immunohistochemistry and western blots both indicated that CD4+ T cells in lesional and nonlesional skin of cats with AD produce IL4 and that the anti-human IL4 antibody cross-reacts with the feline IL4 molecule. This observation contributes to the characterization of the immunopathogenesis of feline atopic dermatitis.
