Abstract
Streptozocin (STZ) induces diabetes mellitus in sheep and pigs. To test the effect of STZ in cattle, cows were given 75–150 mg STZ per kilogram of body weight. Cows receiving 150 mg/kg required euthanasia within 24 hours after infusion because of the severe systemic effects of STZ. Seven cows receiving doses of ≤100 mg/kg had mild to marked decrease in islet immunoreactivity for insulin and in pancreatic islet density and mild to severe tubulointerstitial nephritis. Two cows receiving 75 and 85 mg/kg STZ regained their ability to produce insulin and return blood glucose to basal levels. One cow given 100 mg/kg STZ developed insulin insufficiency consistent with type I diabetes mellitus. These findings demonstrate the susceptibility of the bovine pancreas to STZ; however, severe systemic complications were encountered. Alternative dosages and methodologies should be considered in future attempts to induce diabetes in cattle using STZ.
Insulin has been implicated in the control of ovarian function in vivo and in vitro in rats, 9 pigs, 13 beef heifers, 5 and humans. 2 Researchers have used an energy-restricted cow model to investigate mechanisms of reduced fertility rates in beef cattle. 1,12,17 In these studies, insulin was reduced in the estrous cycle immediately before nutritionally induced anestrus. However, researchers postulate that both over- and underfed animals may have altered mixed-function oxidase activities in the liver that influence the metabolic clearance rate of reproductive hormones. 21 To offset postulated problems associated with endocrine and metabolic events in nutritionally altered animals, our laboratory designed and implemented studies to develop a chemically induced diabetic cow model conceived as a result of the apparent success observed in manipulating the reproductive events in streptozocin (STZ)-induced diabetic pigs and sheep. 3,13
Streptozocin (2-deoxy-2[[(methyl-nitroamino) carbonyl] amino] α (and β)-
Nine 5–7-year-old Angus cows ranging in weight from 550 to 600 kg were administered STZ (Pharmacia & Upjohn, Kalamazoo, MI) on a mg/kg body weight basis according to the following treatment regimen: cow No. 1, 75 mg/kg; cow No. 2, 85 mg/kg; cow No. 3, 100 mg/kg; cow Nos. 4 and 5, 150 mg/kg. Cow Nos. 6–9 were monitored for 35 days to act as their own controls and then received a two-infusion regime of 75 mg/kg infusion on day 1 and a 35 mg/kg infusion on day 7. For cow Nos. 1–5, cows of similar age and size were paired with each treated animal to serve as controls. Only one control cow (cow No. 10) was necropsied for gross and microscopic comparisons.
All treatments were initiated between days 8 and 14 of the estrous cycle. Approximately 12 hours before STZ infusion, animals were fitted with indwelling teflon jugular catheters (Milacath, Medical Instrumentation for Animals, MILA International, Erlanger, KY). Catheters were maintained for 36 hours after STZ infusion. Two blood samples were collected 2 hours apart before STZ treatment. After STZ treatment, blood samples were collected hourly for the first 24 hours, every 2 hours from 24 through 36 hours, and daily before the morning feeding throughout the remainder of the observation period (210 days or until euthanasia). Daily blood samples were standardized to minimize reported variations in endocrine and metabolite levels related to circadian rhythms, environmental factors, physiological state, and nutrition. 22
STZ was dissolved in a 0.1 M sodium citrate and 0.1 M citric acid buffer solution (vehicle), pH 4.5, and was administered intravenously within 5 minutes of solution preparation as a rapid bolus at a concentration of 100 mg/ml of vehicle. 14 Control cows received vehicle at identical dose volumes. The STZ dosage for cow Nos. 4 and 5 (150 mg/kg) was based on published reports of dosages that produce experimental diabetes mellitus in swine. 4,13 Because of the complications in these cows (Table 1), we refocused our attention in cow Nos. 1–3 and 6–9 on determining a more appropriate dose. Crude estimates were determined from calculations based on the weights of pancreatic tissue harvested from cows and pigs whose live weights were known. These crude estimates indicated that the swine pancreas is approximately twice as large as the bovine pancreas (0.001 vs. 0.0005%, respectively) in terms of overall percentage of body weight (BW). Using the swine pancreas weight by BW percentage of 0.001, we calculated that pigs rendered diabetic at 150 mg/kg BW received STZ to the pancreas in amounts of approximately 150 mg/g of pancreas weight. Given this information, we attempted to use STZ to induce diabetes in the cows by giving doses of 75–100 mg/kg BW.
Characterizations of cows administered streptozocin.

∗ Cow Nos. 4 and 5 were euthanatized within 24 hours of STZ treatment; cow Nos. 6-9 acted as their own controls for 45 days prior to STZ injection.
† Tissues evaluated by immunohistochemical methods using anti-insulin antibody conjugate. A minimum of 30 mm2 of pancreatic tissue was evaluated from each cow. Any level of insulin immunoreactivity was considered positive in calculations of islet density. Islet staining intensity: A = weak, individualized cell reactivity in <25% of islet; B = mild to moderate insulin reactivity in 25–50% of islet; C = moderate insulin reactivity in 50–75% of islet; D = strong reactivity in 75–100% of islet. Average staining intensity was calculated as [(A × 1) + (B × 2) + (C × 3) + (D × 4)]/(A + B + C + D); × = 0.00–1.00; ++ = 1.01–2.00; +++ = 2.01–3.00; ++++ = 3.01–4.00.
Exogenous insulin administration was withheld 7 days posttreatment to determine initial effects of STZ on beta islet cell function. After evaluation of daily blood glucose concentrations over this period of time, exogenous insulin replacement therapy was initiated in an attempt to control blood glucose concentration to within 50–100 mg/dl before the morning feeding. Protamine zinc insulin (Protamine Zinc & Anilin; Anpro Pharmaceutical, Arcadia, CA) was subcutaneously injected daily (prefeeding) at a level of 0.25–0.5 IU/kg BW. 24
Severity of the induced diabetic state was assessed by daily monitoring of blood glucose levels and by intravenous glucose tolerance testing (IVGTT). For cow Nos. 1–3, cows of similar age and size were paired with each treated cow to serve as controls. Exogenous insulin treatment ceased on the day before an IVGTT began and recommenced immediately after its conclusion. A baseline blood sample (time 0) was collected before rapid infusion of intravenous glucose (0.5 g/kg BW; 50% dextrose solution). Blood was collected at 20, 40, 60, 90, 120, 180, and 240 minutes postinfusion.
Complete necropsy examinations were performed on all treated animals and one control (cow No. 10). Representative sections from all organs were expediently collected and fixed in 10% neutral buffered formalin. Tissues for microscopic evaluation were routinely processed, embedded in paraffin, sectioned at 4–5 µm, and stained with hematoxylin and eosin (HE). Immunohistochemical staining to determine the presence of insulin or glucagon was performed on pancreatic tissue from the distal duodenal limb using standard procedures as described by the manufacturer (Biogenex Laboratories, San Ramon, CA).
For morphometric evaluation of pancreatic islets, any level of insulin immunoreactivity was considered positive in calculations of islet density. Islet-staining intensity was assessed as follows: A = weak; individualized cell reactivity in < 25% of islet; B = mild to moderate insulin reactivity in 25–50% of islet; C = moderate insulin reactivity in 50–75% of islet; D = strong reactivity in 75–100% of islet. Average staining intensity was calculated as [(A × 1) + (B × 2) + (C × 3) + (D × 4)]/(A + B + C + D) and reported as follows: + = 0.00–1.00; ++ = 1.01–2.00; +++ = 2.01–3.00; ++++ = 3.01–4.00.
Results are summarized in Table 1. Tissues subjected to routine microscopic evaluation (brain, heart, liver, intestine, forestomachs, abomasum, or thyroid gland) were normal unless otherwise stated. No clinical signs of illness were observed among control cows. Cow No. 10, the control cow, exhibited no histologic lesions of the pancreas (Fig. 1) or alterations in insulin or glucagon immunoreactivity. Insulin immunoreactivity was diffuse and intense throughout the central islet cells (Fig. 2; cow No. 10). Islets with positive insulin immunoreactivity at any level were present at a density of 1.93 islets/mm2 of total pancreatic tissue. Glucagon immunoreactivity was also intense, although it was confined to the periphery of islets (Fig. 3; cow No. 10).
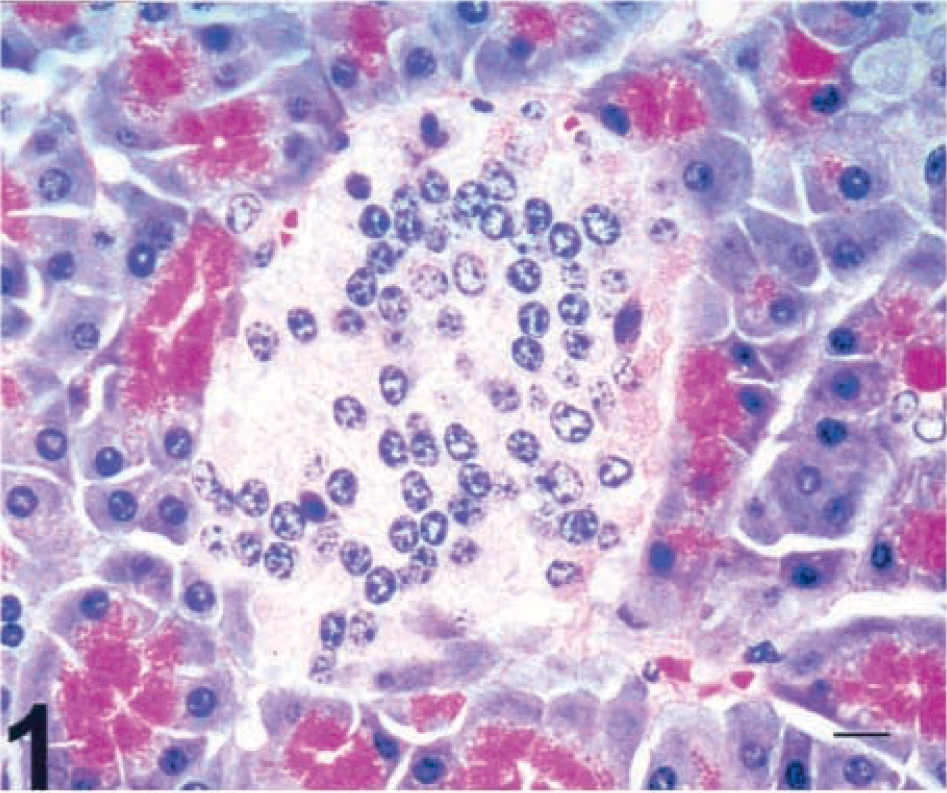
Pancreas; control cow No. 10. HE. Bar = 15 µm.
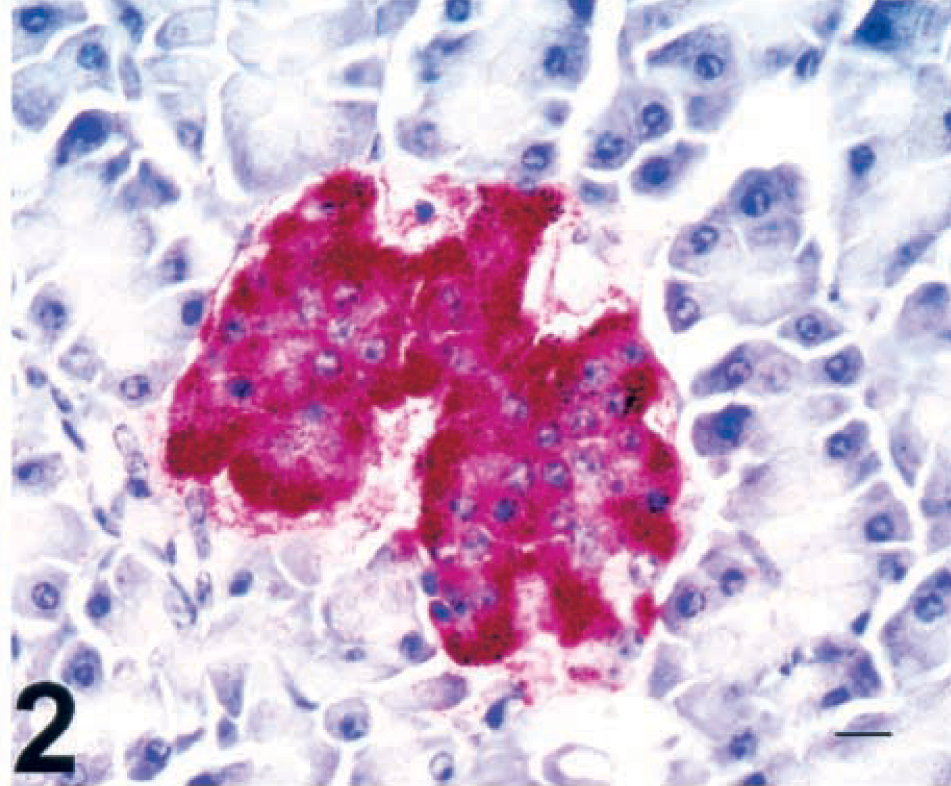
Pancreas; control cow No. 10. Intense diffuse central islet immunoreactivity of insulin antigen. SAAP immunostain, Mayer's hematoxylin counterstain. Bar = 15 µm.
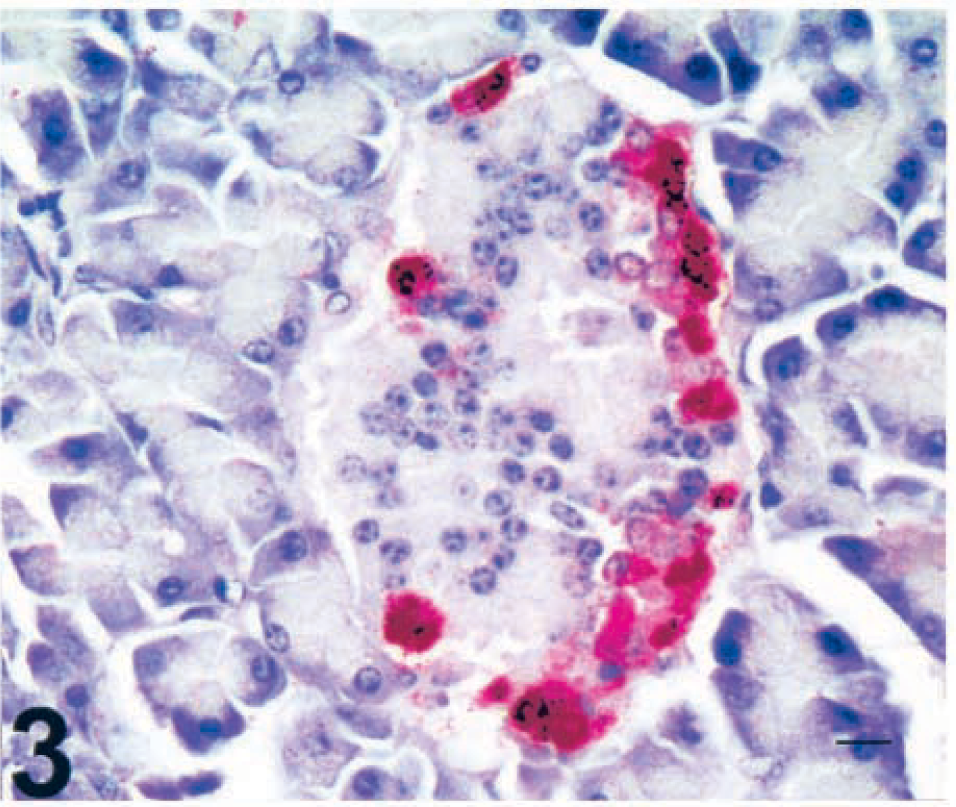
Pancreas; control cow No. 10. Intense peripheral islet immunoreactivity of glucagon antigen. SAAP immunostain, Mayer's hematoxylin counterstain. Bar = 15 µm.
Cow Nos. 4 and 5, which received 150 mg/kg STZ, were euthanatized within 24 hours postinfusion. Beginning 8–10 hours after STZ infusion and continuing until death, these cows had abnormal vocalization, reduced rumen motility, bloating, and abdominal discomfort, which they exhibited by frequently striking their abdomens and altering position. Gross lesions in both cows consisted of marked peripancreatic edema of the mesentery. A small volume of clear yellow ascitic fluid and slight thoracic and pericardial effusion were observed. Histologically, pancreatic islet necrosis was diffuse, but exocrine pancreatic acinar epithelium and ductal and connective tissues appeared normal (Fig. 4; cow No. 5). Immunohistochemical staining revealed abundant insulin immunoreactivity throughout the necrotic islets (Fig. 5; cow No. 5) and prominent glucagon staining limited to the periphery of necrotic islets.
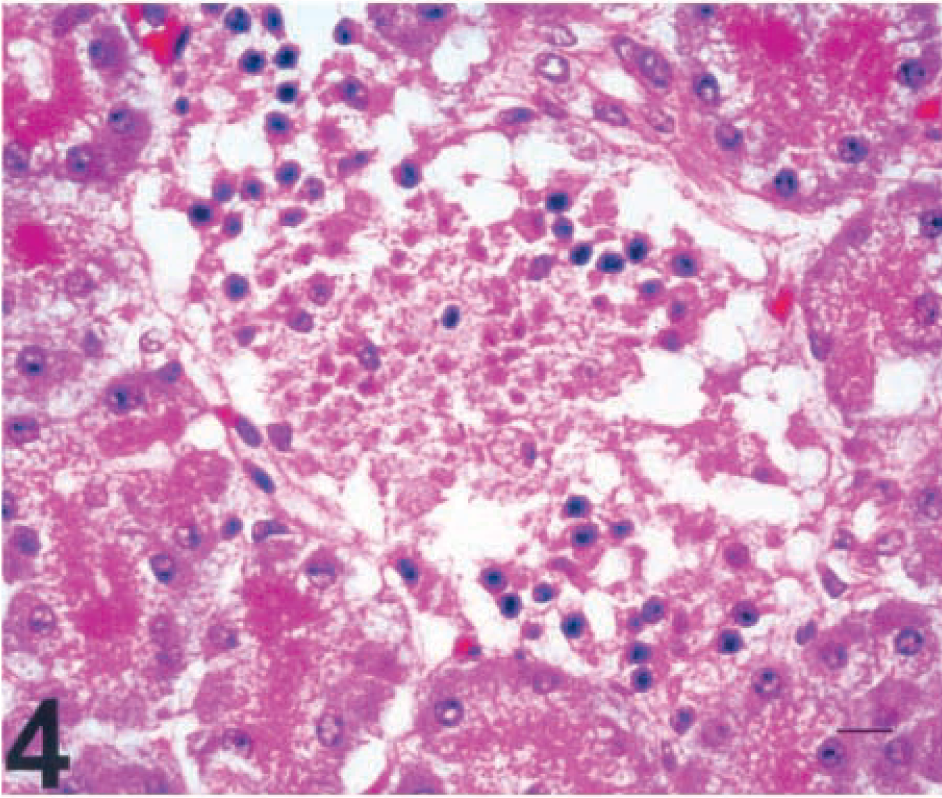
Pancreas; cow No. 5, 24 hours after infusion of 150 mg/kg STZ. Diffuse global necrosis of pancreatic islet cells with sparing of the exocrine pancreatic acinar epithelium. HE. Bar = 15 µm.
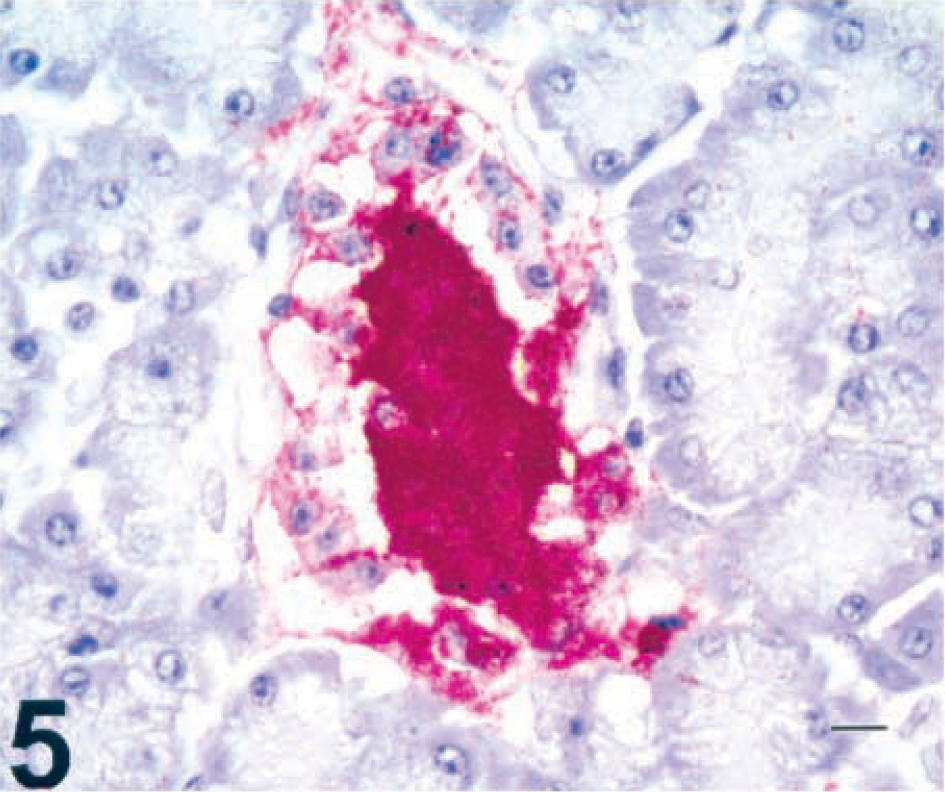
Pancreas; cow No. 5, 24 hours after infusion of 150 mg/kg STZ. Intense central islet immunoreactivity of insulin antigen. SAAP immunostain, Mayer's hematoxylin counterstain. Bar = 15 µm.
Although acute morbidity and mortality did not occur at other doses, cow No. 3 (100 mg/kg) had many drug-related complications. Beginning 5 days after STZ infusion, feed and water intake was depressed for 7 days. During this period, the cow lost 100 kg of body weight and required supplementation with oral electrolytes. Blood glucose levels for this cow were difficult to maintain throughout the study, and no weight was regained. The cow was euthanatized 3 months postinfusion because of elevated blood urea nitrogen (81 mg/dl), weight loss (115 kg), and reduced water and feed intake. At necropsy, no gross lesions were observed in visceral organs. Histologically, identifiable pancreatic islets were of adequate cellularity, with no evidence of inflammatory cell infiltration or fibrosis. Islet insulin immunoreactivity was present at a level of 0.61 islets/mm2 of total pancreatic tissue but was limited to cytoplasmic staining of individual scattered beta cells (Fig. 6; cow No. 3). Glucagon immunoreactivity was limited to peripheral alpha cells. Microscopic examination of renal tissue revealed marked variability in tubular diameter, and tubular epithelium was attenuated to hypertrophic. Periglomerular fibrosis was also moderate and diffuse. Aggregates of small lymphocytes were present multifocally within the cortical interstitium.
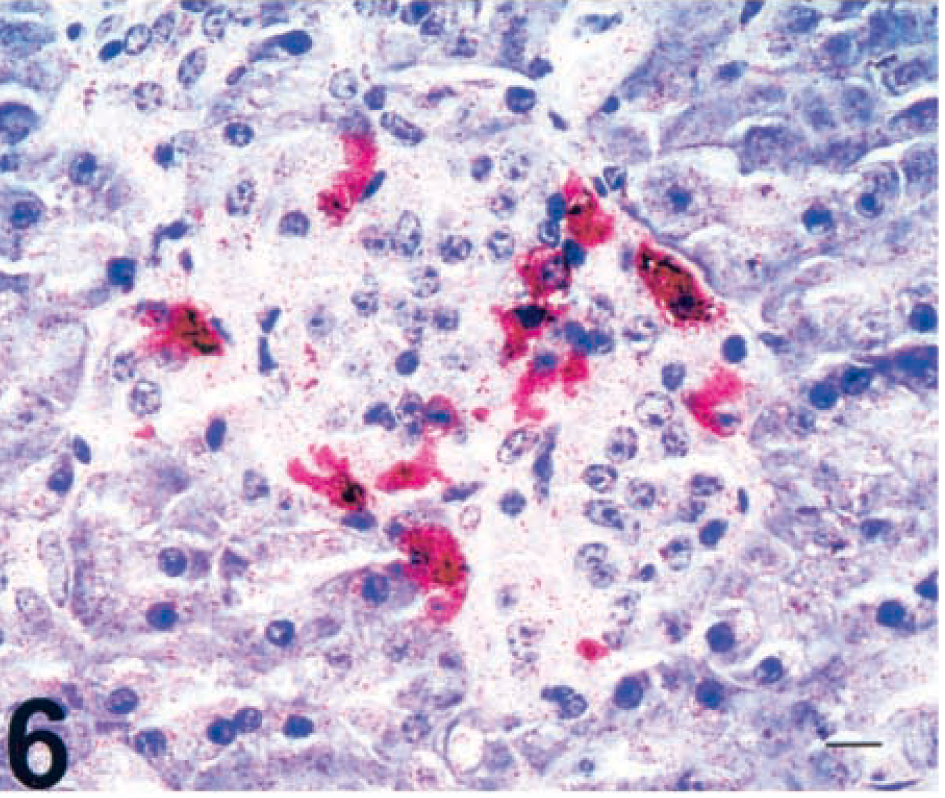
Pancreas; cow No. 3, 3 months after infusion of 100 mg/kg STZ. Scattered individualized beta-cell immunoreactivity for insulin antigen. SAAP immunostain, Mayer's hematoxylin counterstain. Bar = 15 µm.
Cow Nos. 6–9, given two infusions of STZ, had various outcomes. After the first infusion, cows were given oral electrolytes to offset reduced feed and water intake and weight loss. Oral electrolyte supplements ceased within 1 week of the second STZ infusion. Cow No. 6 was euthanatized 6 days after the second STZ infusion because of severe omasal ulcerations of unknown etiology. Cow No. 7 was euthanatized at the same time because of pulmonary aspiration of oral electrolyte supplements. Cow No. 8 was euthanatized 23 days after the second STZ infusion because of renal disease. Cow No. 9 completed the planned observation period. This cow was in good condition but failed to regain 90 kg of body weight lost because of STZ treatment.
Histologically, no evidence of inflammation, necrosis, or fibrosis was observed in the islets of these cows. Insulin immunoreactivity was reduced in islets from all sections examined; and staining was limited to the cytoplasm of individual beta cells. Islets exhibiting any level of insulin-positive staining varied in number from 0.84 islets/mm2 to 1.68 islets/mm2 of total pancreatic tissue.
Cow Nos. 1 and 2 (75 and 85 mg/kg, respectively) survived the experimental procedures and planned observation time in good condition. Depressed feed and water intake and weight loss were not observed in these cows, and no oral electrolyte supplements were given. Histologic and immunohistochemical alterations in pancreatic and renal tissues from cow Nos. 1 and 2 were similar to those in cow Nos. 6–9.
During the initial IVGTT after STZ infusion, cow Nos. 1–3 and 6–9 were unable to mount an endogenous insulin response to a rapid intravenous bolus of 0.5 g dextrose/kg BW. For subsequent IVGTT, cow Nos. 1, 2, and 9 were able to produce sufficient endogenous insulin within the IVGTT period to bring glucose levels to basal values. Cow No. 3 exhibited virtually no insulin response to the IVGTTs and required daily intramuscular injections of exogenous insulin (40–120 IU) to control blood glucose levels. In contrast, cow Nos. 1, 2, and 9 produced enough endogenous insulin by 30 days after treatment to be removed from exogenous insulin therapy.
This research represents the first attempt to develop a diabetic cow model through the use of STZ. Intravenously administered STZ led to physiologic alterations consistent with reports of spontaneous and chemically induced diabetes in other animals. Features described by others, 8,10 such as rough, dry hair coat, dehydration, poor appetite, weight loss, poor muscle tone, polyuria, and polydypsia, were also evident among the treated cows. Alterations in serum glucose and insulin concentrations revealed a dose–response relationship between STZ and the severity of diabetes in this and other studies. 4,7,19 Decreased immunohistochemical staining for insulin in pancreatic islet tissue in the treated cows was associated with increased levels of intravenously administered STZ.
Cow Nos. 1, 2, 3, and 6–9 had decreased immunohistochemical staining for insulin within islets and a decreased density of islets relative to the reference cow No. 10. Cow No. 3, who exhibited the greatest resemblance to a type 1 diabetic state, showed a lack of insulin response and increased blood glucose levels (> 400 mg/dl) when challenged by an IVGTT. The islet density of this cow was reduced approximately 68% compared with the control (0.61 islets/mm2 versus 1.93 islets/mm2, respectively). Similar results have been reported when STZ was used to induce diabetes mellitus in mice and rats. 11,15
Cow Nos. 1, 2, 3, and 6–9 had mild to severe tubulointerstitial nephritis. These findings differ from those reported with STZ administration in sheep, 3 goats, 19 and swine, 4 in which no systemic complications or gross lesions indicative of renal toxicity were reported. Renal disease has been reported in experiments of STZ-induced diabetes mellitus in lab animals and following chemotherapeutic administration of STZ for the treatment of several forms of human carcinoma. 23 Many human patients treated with STZ experience renal toxicity, as evidenced by azotemia, anuria, hypophosphatemia, glycosuria, and renal tubular acidosis. 14 The chronic nephrotoxic effects of STZ are well documented in the literature for mice and humans and may help to explain the variation in chronic tubulointerstitial nephritis observed in these cows. 6,16 No pathogenic bacteria were isolated from affected kidneys, and special stains failed to indicate the presence of spirochetes in affected renal tubules.
Gastrointestinal ulcerations were noted in cow No. 6. Gastrointestinal ulcerations have not been recognized as an effect of STZ administration in sheep, goats, or swine. However, increased susceptibility of gastric mucosa to ulcergenic stimulation has been noted in STZ-induced diabetic rats. 20 In the present study, gastrointestinal ulceration may have been indirectly associated with STZ secondary to uremia and stress.
Results of these studies demonstrate the susceptibility of the bovine pancreas to the effects of STZ. Although STZ may be used in an animal model to help investigate insulin's role in bovine reproduction, the systemic complications noted in these cows precluded accomplishment of the study's original objective. In the future, alternative dosages and methodologies should be employed with any attempt to develop STZ-induced diabetes in cattle.
Footnotes
Acknowledgements
This work was supported in part by Technical Contribution 4484 of the South Carolina Agriculture Experiment Station, Clemson University. We recognize Dr. J. W. Lauderdale (Pharmacia & Upjohn, Kalamazoo, MI) for providing streptozocin for these experiments. We thank C. Bright, A. Burdette, and J. Fisher for technical assistance with blood collection and B. Walsh for assistance in preparation of histology and immunohistochemical specimens. We also thank Dr. D. Henricks, S. Gray, and B. Lackey for the preparation and administration of the STZ and for laboratory assistance.
