Abstract
Natural eastern equine encephalitis alphavirus (EEEV) infection was diagnosed in two adult horses with anorexia and colic, changes in sensorium, hyperexcitability, and terminal severe depression. Myocardium, tunica muscularis of stomach, intestine, urinary bladder, and spleen capsule had coagulative necrosis and perivascular lymphocytic infiltrate. Central nervous system (CNS) lesions were diffuse polioencephalomyelitis with leptomeningitis characterized by perivascular T lymphocyte cuffing, marked gliosis, neuronophagia, and multifocal microabscesses. Lesions were more prominent within cerebral cortex, thalamus, hypothalamus, and mesencephalon. EEEV was identified in the cytoplasm of cardiac myocytes and smooth muscle cells of spleen, stomach, intestine, urinary bladder, blood vessels, and dendritic cells. In the CNS, EEEV-positive cells included neurons, astrocytes, oligodendrocytes, microglia, and neutrophils. EEEV was isolated from the CNS of both horses. The detailed description of the encephalic and spinal EEEV localization and the findings of EEEV in extraneural tissues contribute to the understanding of this important multisystemic zoonotic disease.
Many viruses cause encephalomyelitis in horses and other equids. Among the arthropod-borne viruses (arboviruses), which cause neurologic disease in equids, humans, and other vertebrates, there are three important diseases caused by viral agents of the family Togaviridae, genus Alphavirus: eastern, western, and Venezuelan equine encephalomyelitis (EEE, WEE and VEE, respectively). Other arboviruses able to induce encephalomyelitis in horses and humans are West Nile virus, Semliki Forest virus, louping ill virus, Murray Valley encephalitis virus, Near Eastern equine encephalomyelitis virus, Powassan virus, snowshoe hare virus, Main Drain virus, and Japanese encephalitis virus. 28 EEE, WEE, VEE viruses are single-strand RNA positive-strand viruses characterized by evolutionary stability. 2 The transmission of these alphaviruses in endemically infected areas is by wild birds, which serve as the reservoir, and mosquitoes, which are mainly vectors. 19 The EEE virus (EEEV) actively replicates within the arthropod vector. 27 EEEV infection in horses and humans, considered dead-end hosts, occurs when the virus is inoculated into susceptible hosts, usually close to wetlands where reservoirs and vectors are present in greater numbers. 22 In humans, EEEV infection may be associated with fatal encephalomyelitis and has caused epidemics of encephalitis in the eastern United States since at least the early 1800s. 12 EEE has also been described in cattle 18,23 and pigs. 24 Distribution of the EEEV in neural tissue has been reported in one human 11 and in equine and avian tissues, 20,21 as determined by both immunohistochemistry and in situ hybridization, but the descriptions were limited to some parts of the encephalon with no reported evaluation of the spinal cord or other organs. Extraneural tissue distribution of the virus, limited to the intestine, has been identified in one horse. 21 Here, we describe the clinical, pathologic, virologic findings and the distribution of virus in tissues of two horses with EEE from New Jersey that were examined at New Bolton Center, University of Pennsylvania.
Two horses, a 1-year-old intact male Paint (horse No. 1) and a 7-year-old Belgian gelding (horse No. 2), presented at different times and locations with acute onset of neurologic and abdominal clinical signs. Clinical signs included ataxia, profound changes in sensorium, hyperexcitability, and restlessness and rapidly progressed to severe depression. Horse No. 1 was recumbent and thrashed violently, resulting in significant self-injury. Following euthanasia, the two horses were subjected to postmortem examination.
Representative tissue samples of major organs and the whole central nervous system (CNS) were fixed in 10% neutral buffered formalin solution and routinely processed for histology. Eyes and trigeminal ganglia were not available for examination. Tissue sections were stained with hematoxylin and eosin, Luxol fast blue, and crystal violet for Nissl substance. An automated slide stainer (Fisher Histomatic Slide Stainer Model 172, Fisher Scientific, Indiana, PA) was used to perform the indirect immunoperoxidase staining of formalin-fixed, paraffin-embedded tissues. Serial tissue sections were mounted on Probe-On (Sigma, St. Louis, MO) glass slides coated with poly-
Both horses were in good nutritional condition. The only gross lesions observed were related to self-trauma and included moderate subcutaneous acute hemorrhages and a pelvic fracture in horse No. 1. Microscopically, lesions involved visceral organs and the CNS, where they were much more severe. Lesions in the visceral organs included multiple foci of coagulative necrosis occasionally accompanied by mild to moderate lymphocytic infiltrate with occasional macrophages. This infiltrate was also distributed around blood vessels, occasionally mildly infiltrating the outer tunica media of muscular arteries, where sometimes there were a few necrotic myocytes with nuclear pyknosis and karyolisis. Mild vascular changes were identified in the myocardium, liver (where they involved periportal sinusoids and a few hepatocytes), kidney, and the tunica muscularis of the stomach, intestine, urinary bladder, and spleen capsule. Blood vessels contained intraluminal monocytes and neutrophils, and occasional diapedesis toward the surrounding tissues was observed. Leukocyte margination and diapedesis was particularly evident in the CNS. In the CNS, lesions were diffuse polioencephalomyelitis (Fig. 1) with leptomeningitis and were more prominent within the cerebral cortex, thalamus, hypothalamus, and mesencephalon, where marked gliosis was also present. Numerous small T lymphocytes with a few macrophages and neutrophils surrounded blood vessels. Frequently, these cells were crossing the wall of congested capillaries, extending into the adjacent parenchyma, where there were clusters of segmented neutrophils. Some morphologically normal neurons were surrounded by T lymphocytes, macrophages, and neutrophils, which were more abundant around hypereosinophilic, shrunken neurons with pyknotic nuclei (neuronophagia) (Fig. 2). Neuronophagia was frequent and multifocal and randomly involved neurons of any cerebral cortical area. The white matter of cerebral hemispheres showed multifocal moderate cuffs of lymphocytes and macrophages, with mild edema. Neutrophils, macrophages, and reactive glial cells severely and diffusely infiltrated the hippocampus. The inflammatory changes were more prominent in the gyrus dentatus and relatively spared the pyramidal cells. The cerebellar cortex showed inflammatory lesions similar to those of the cerebral cortex but to a lesser extent. Sometimes there was degeneration of Purkinje cells and occasional neuronophagia with residual microglial aggregates. Neurons of the caudal brainstem, reticular substance, and the majority of the cerebral and cerebellar cortical neurons were morphologically intact, with minimal chromatolysis. The spinal cord contained severe bilateral and symmetric perivascular infiltrate composed of T lymphocytes, macrophages, and neutrophils in the dorsal, lateral, and ventral horns. There was diffuse gliosis, neuronophagia, and neuronal degeneration. Mild inflammatory cuffs were also present in the white matter, along with a few scattered microglial nodules and mild multifocal Wallerian degeneration. The lesions were more prominent in the cervical and thoracic spinal cord. Leptomeninges of cerebral hemispheres and cerebellum multifocally contained moderate numbers of T lymphocytes, macrophages, and a few neutrophils. The choroid plexus and the ependyma showed no significant lesions.
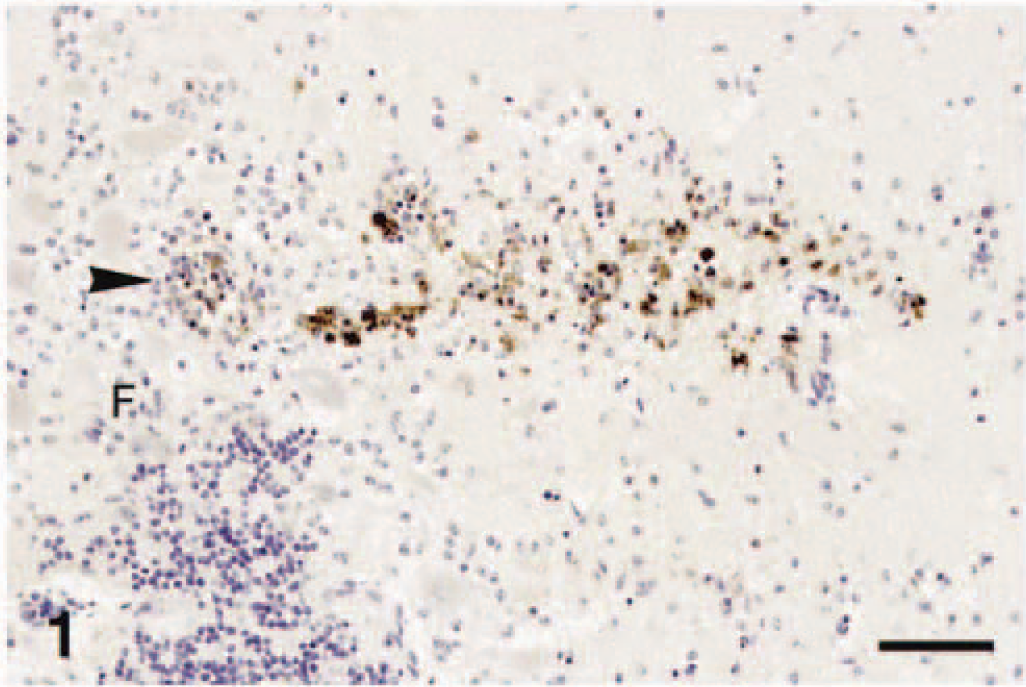
Cerebellum; horse. Numerous inflammatory cells containing EEEV around a blood vessel (arrowhead) within cerebellar folium (F). Immunoperoxidase histochemistry and hematoxylin (IPH&H). Bar = 180 µm.
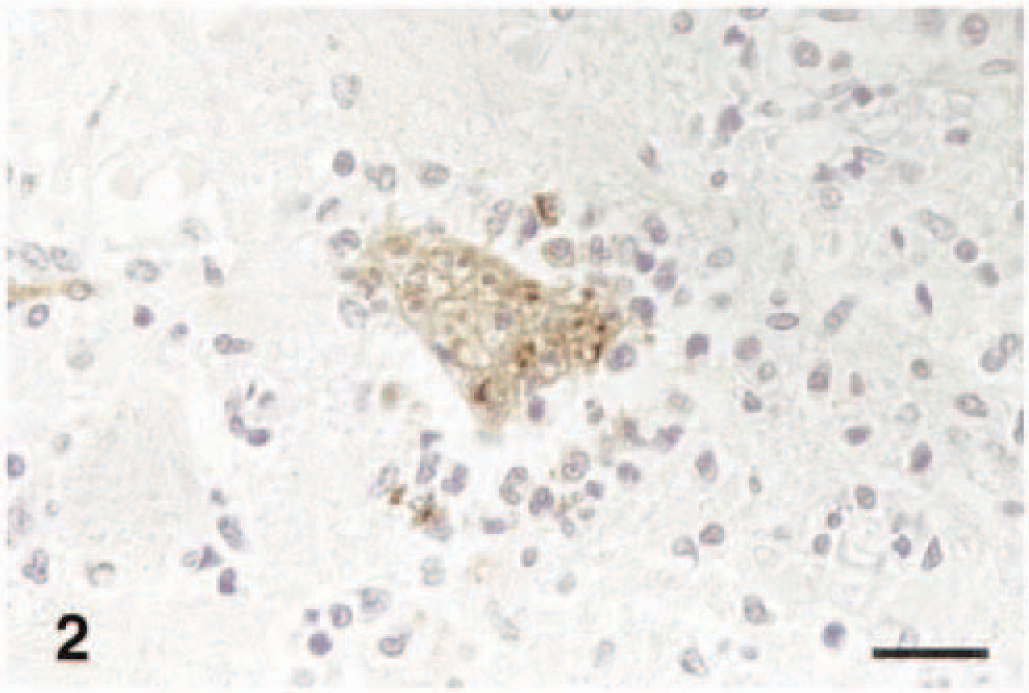
Cerebrum; horse. Neuronophagia. EEEV is contained within neuron and glial cell cytoplasm. IPH&E. Bar = 40 µm.
The EEEV antigen in the tissues of these two horses was always intracytoplasmic (Figs. 1–6). It was finely granular or formed dots or prominent clumps. EEEV was identified in the cytoplasm of morphologically intact and necrotic cells in heart (Fig. 4), lung, kidney, spleen, stomach, intestine (Fig. 5), urinary bladder, adrenal glands, and the CNS (Figs. 1, 2). More specifically, EEEV was in adventitial spindloid cells and a few tunical myocytes of blood vessels of various organs. Small to large clusters of positive smooth muscle cells were in the tunica muscularis of stomach, intestine (Fig. 5), urinary bladder, and splenic capsule. In the absence of morphologic changes there was prominent multifocal staining of renal medullar interstitial fusiform and dendritiform cells (Fig. 6) and a few fusiform cells in the loose renal pelvic connective tissue. There was staining on the surface of mesenchymal interstitial cells of the zona reticularis of the adrenal gland. In the CNS, EEEV was located in the cytoplasm of neurons (Fig. 2), extending in axons and dendrites, astrocytes, oligodendrocytes, microglial cells, and neutrophils. More frequently, the neurons of the brain stem nuclei and spinal cord were positive but slightly less so than those of the cerebral cortex. There was almost no staining in cerebellar Purkinje cells, but scattered granular cells and Golgi cells containing EEEV sometimes were observed. Occasionally, necrotic neurons did not stain. The glial cells surrounding morphologically intact and necrotic neurons often contained EEEV. In the hippocampus, in contrast to a prominent inflammatory infiltrate, EEEV was scant and infrequent in neurons and more common in neutrophils and a few microglial cells. In the cerebral cortex, there were clusters of viable and necrotic shrunken neurons with abundant staining extending in the neuropil. In these areas, the astrocytes were prominent, with common type II Alzheimer cells negative for EEEV. EEEV was also contained in the cytoplasm of intravascular neutrophils and a few monocytes (Fig. 3), some of them marginating.
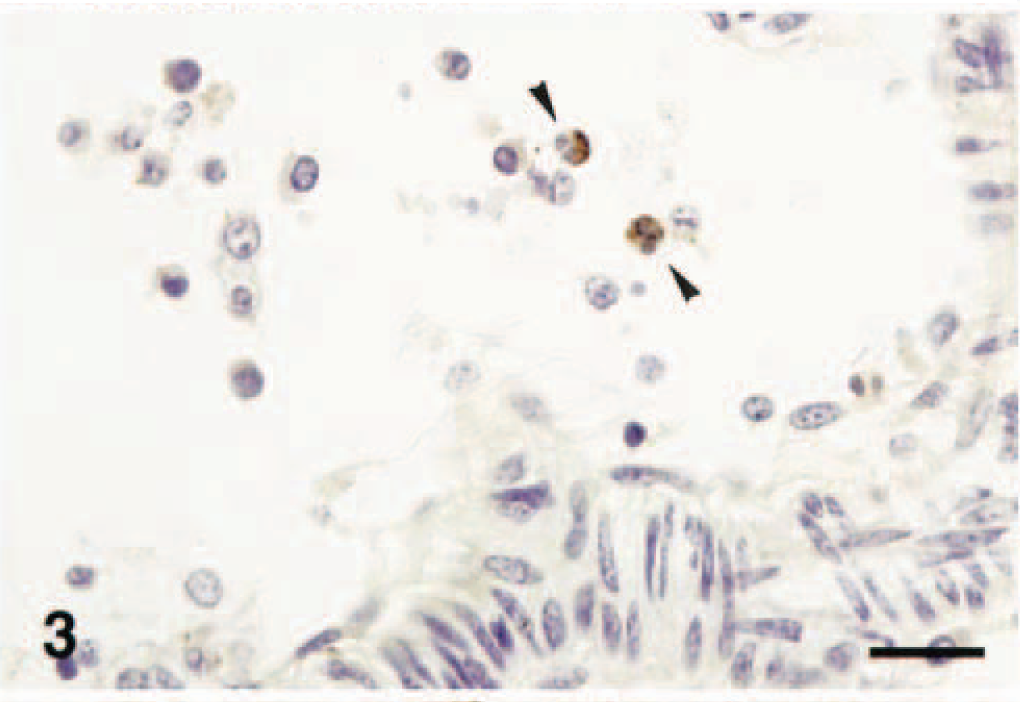
Leptomeninges; horse. In blood vessel with intravascular mononuclear cells and neutrophils, two neutrophils (arrowheads) contain intracytoplasmic EEEV. IHP&H. Bar = 25 µm.
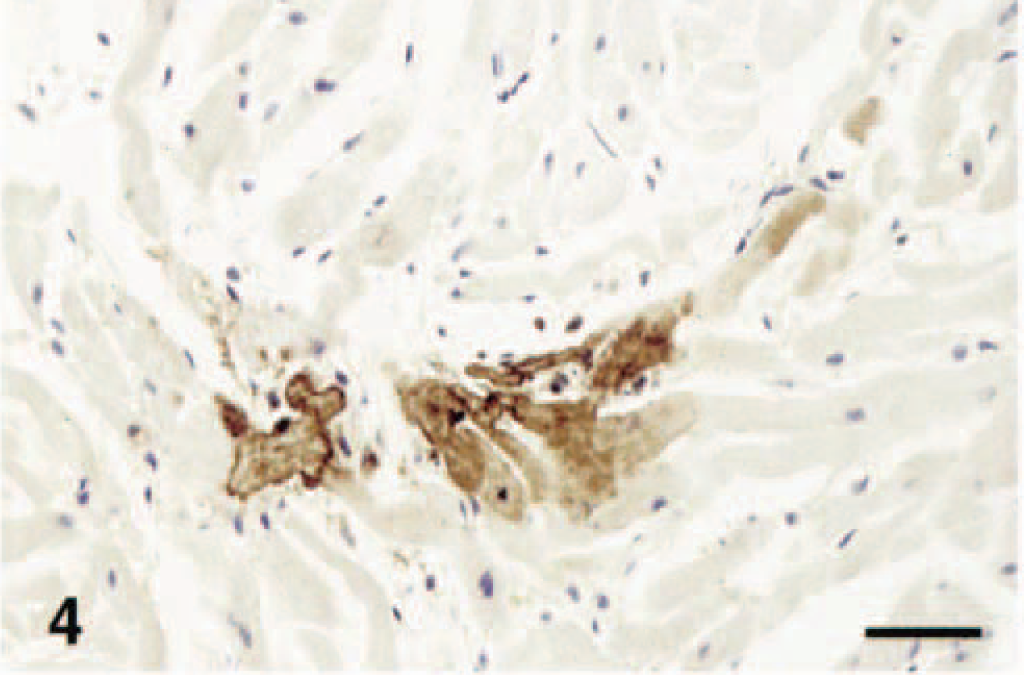
Heart; horse. Focal coagulative necrosis of EEEV-infected cardiac myocytes. IPH&H. Bar = 50 µm.
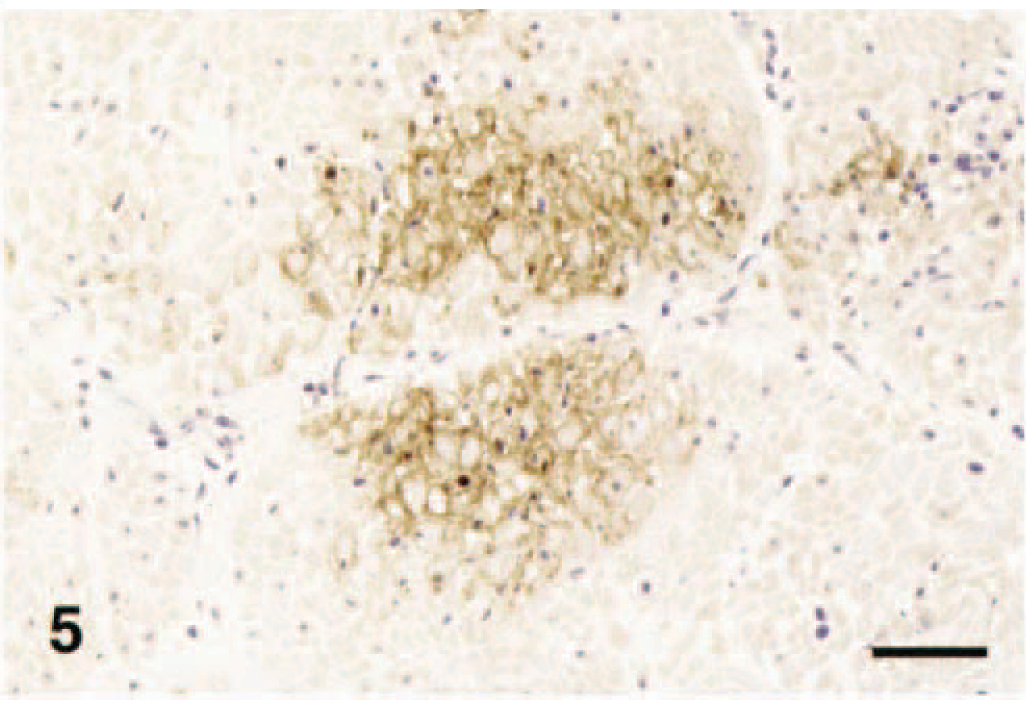
Intestinal tunica muscularis; horse. Focal coagulative necrosis of EEEV-infected smooth muscle cells. IPH&H. Bar = 120 µm.
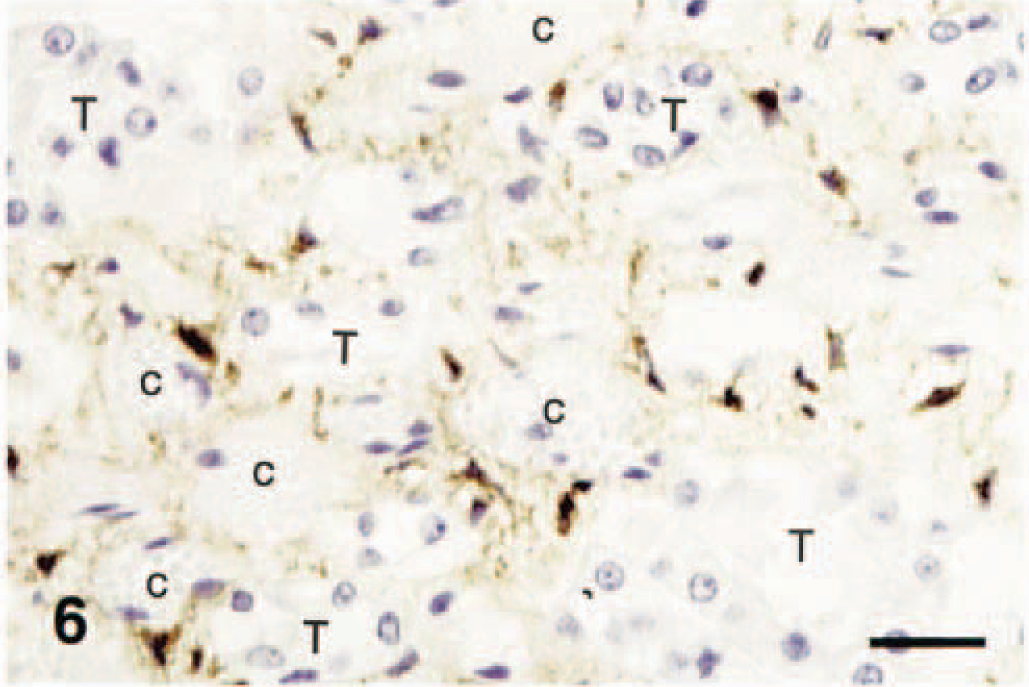
Kidney; horse. Interstitial dendritiform cells containing EEEV in contrast with negative capillaries (c) and tubules (T). IPH&H. Bar = 40 µm.
In both horses, EEEV was isolated from encephalic tissue and identified with monoclonal antibodies as EEE alphavirus on the basis of expression of the specific structural glycoprotein E1 shared by the isolates identified in the USA within the last 50 years.
EEE occurs principally along the east and gulf coasts in the United States. It is a life-threatening mosquito-borne arboviral infection of both horses and humans, and approximately one third of infected humans die. 9 Horses of any breed, sex, or age may be affected, although suckling foals under 3 months of age do not commonly develop the disease. Clinically, EEE cannot be readily differentiated from WEE and VEE in horses by physical and neurologic examination. 1 Horses affected with EEE frequently have an acute onset, and their neurologic disease reflects damage to cerebrocortical tissue. Changes in sensorium are profound, and initial hyperexcitability and restlessness rapidly progresses to severe depression. Early signs of fever and depression may be accompanied by anorexia and colic. Peracute to acute neurologic signs include but are not limited to dementia, head pressing, cortical blindness, and seizure. The prognosis for survival of EEE by equine patients is poor to guarded. Rarely, affected horses do not become recumbent and survive but have residual signs referred to as “dummy syndrome.” In some cases, the early signs may pass undetected, and apparently healthy horses are found dead. 31 Specific IgM immunoassays may be beneficial for antemortem diagnosis of EEE. 26
Cardiac irregularities may be detected in EEE and have been ascribed to brain–heart syndrome associated with profound intracranial events. 14,17 A cytopathic effect associated with EEEV in necrotic cardiac myocytes was demonstrated in these two horses and may be the primary event inciting dysrhythmias. Detailed clinical cardiovascular evaluation was not performed in these horses for reasons of practicality and examiner safety. Because the distribution of EEEV was correlated with cardiac necrosis, the observed cardiac lesions probably were not due to caudal brain stem lesions.
The CNS lesions in both the horses of this study had similar intensity and distribution and were compatible with the EEE cases previously reported. 1,21,28
There are several infectious equine diseases that have similar clinical presentations, lesions, and sometimes distribution of the infectious agent and therefore must be considered differential diagnoses of EEE. VEE and WEE lesions are rather similar to those of EEE, but in natural 25 and experimental 13 cases of VEE variable degrees of thrombosis and necrosis of the smaller arteries often occur. Rabies sometimes presents with minimal neuronophagia, is often characterized by Negri bodies, and has no neutrophilic infiltration, and the rabies rhabdovirus intracytoplasmic antigen, which is generally more abundant than EEEV antigen in the infected CNS, has not been identified within leukocytes in vivo. 8 Equine herpesvirus 1 induces ischemic myeloencephalopathy secondary to vascular necrosis with thrombosis, is not neurotropic during productive infections, and is present in small quantities within the small muscular artery endothelium and myocytes. 7 Nonsuppurative West Nile flavivirus infection has been described in horses, 3,4 where lesions primarily involve the ventral and lateral horns of the thoracic and lumbar spinal cord and the rombencephalon, and cortical cerebral involvement has not been identified. Small quantities of West Nile virus have been detected within sparse neurons and glial cells of brain stem and spinal cord in naturally infected horses. 3 Borna disease lesions are severe in the hippocampus, the spinal cord is generally not involved, and intranuclear eosinophilic Joest–Degen inclusions are sometimes observed. 12 Louping ill, 29 Japanese encephalitis, and tick-borne encephalitis 12 are characterized by striking necrosis of Purkinje cells, whereas Murray Valley encephalitis virus can be associated with extensive cerebrocortical necrosis in the horse. 10 Although the presence of microabscesses might mimic L. monocytogenes infection, the distribution of lesions is different because L. monocytogenes induces inflammation of the brain stem, and coccobacilli can be easily recognized within Gitter cells, neutrophil cytoplasm, and neuropil with histochemical and immunohistochemical staining. 32 Listeriosis is uncommon in the horse and primarily induces neonatal septicemia and pneumonia. 33 Untreated cases of equine protozoal myelitis have random lesions in the brain stem and spinal cord, no neuronophagia, and an infiltrate comprised of lymphocytes, plasma cells, macrophages, neutrophils, and a few eosinophils, with hemorrhage and malacia. 28
Natural and experimental EEE has been also reported in cattle 18,24 and pigs, 24 with lesions similar to those identified in horses, with the exception of a severe polioencephalomyelitis with low numbers of infiltrating neutrophils in a cow. 18
In contrast to most viral infections, viral antigen in these horses infrequently identified in circulation monocytes and was more common in neutrophils (Fig. 3). In previous in vitro immunohistochemical studies using fluorescent antibodies, 16 a minority of monocytes also supported alphavirus replication, although a few of these cells had virus on the surface and were able to internalize it. Further ultrastructural, immunoultrastructural, and perhaps in vitro virologic studies will be necessary to clarify the role of EEEV replication and transportation within the neutrophils. It is still unclear whether EEEV and other arboviruses use the hematogenous route to infect the CNS or use an alternative route via the nerve fibers. 3
Lesions and associated EEEV were observed in several viscera, especially within smooth muscle and cardiac myocytes. Similar lesions and EEEV localization have been previously described in the intestine of a horse. 21 Visceral involvement in addition to the CNS lesions has been previously reported in VEE alphavirus infections in equids. These changes included necrotizing lesions in lymphoid organs, pancreas, liver, and heart. 15 EEE can induce mortality in whooping cranes, 5 emus, 30 and other avian species with prominent abdominal visceral lesions such as hemorrhagic enterocolitis and splenic and hepatic necrosis.
EEEV antigen was identified within fusiform and stellate cells of the renal interstitium (Fig. 6). This distribution of antigen also has been observed with other viral infections. 6 These interstitial cells may be infected antigen-presenting dendritic cells with limited phagocytic activity, macrophages with prominent phagocytic activity, or even infected fibroblasts or myofibroblasts. The EEEV antigen detected within these renal interstitial cells could be nonviable virus or could represent virus in active replication. Further studies using in situ hybridization, reverse transcription polymerase chain reaction methods, and virus isolation will be necessary to answer these questions.
The virus was easily isolated from the CNS of both horses. Although infected cells were present within several organs, the target tissue for virus isolation and immunohistochemical evaluation remains the CNS.
Although mosquito control and immunization have significantly reduced the incidence of EEE in horses in the USA, small and large epidemics still occur, particularly in the southern states. These epidemics may be attributed to the short duration of vaccine immunity (4–6 months) and the long vector season, combined with wet weather conditions conducive for mosquito proliferation. The EEE epidemics underscore our still limited knowledge of the pathogenesis and epidemiology of this polytropic and adaptable but deadly virus. These novel findings of EEEV in extraneural tissues will contribute to the clarification of the pathogenesis of this important zoonotic disease.
Footnotes
Acknowledgements
We are grateful to Carol Nelson (New York State Diagnostic Laboratory, Cornell University) and Jackie Ferracone (New Bolton Center, University of Pennsylvania) for immunohistochemistry and to Alexis Wensky-Roberts of the Image lab® (Cornell University College of Veterinary Medicine) for iconography.
