Abstract
Histologic examination of eyes and brains of 27 dogs experimentally infected with either Ehrlichia canis, E. ewingii, E. chaffeensis, or human granulocytic ehrlichia (HGE) was conducted in the course of several experiments, the primary objectives of which were to investigate the susceptibility of the domestic dog to infection with various ehrlichial species and to assess the ability of ixodid tick species to acquire and transmit those infections. Uveitis and meningitis occurred in each of the dogs infected with E. canis but was not observed in dogs infected with the other Ehrlichia species. The inflammatory infiltrate was predominantly lymphocytic, monocytic, and plasmacytic; granulocytes were notably few. Ocular inflammation was most common and most intense in the ciliary body, becoming less intense in the choroid, iris, and retina, respectively. Meningitis was often accompanied by mild neuroparenchymal vascular cuffing and gliosis. The meningeal inflammatory cell infiltrate included a prominent monocyte population. Ocular and meningeal lesions were present in all E. canis-infected dogs from 22 through 200 days postexposure. Neither ocular nor brain lesions were observed with any of the other ehrlichial infections.
Keywords
Ehrlichieae are tick-transmitted rickettsiae that infect leukocytes of specific mammalian hosts. 9 Ehrlichia canis, E. ewingii, and E. platys are well-established as canine parasites and pathogens, 1 16 whereas E. chaffeensis, a zoonotic agent commonly thought to be endemic in white-tailed deer (Odocoileus virginianus), causes monocytic ehrlichial infection in humans, and an unnamed ehrlichial species causes human granulocytic ehrlichiosis (HGE). 18 Recently, E. ewingii has also been recognized to cause infection in humans. 3 There is morphologic, molecular, and serologic evidence that dogs are susceptible to infection with E. chaffeensis and the agent of HGE. 2 6 10 15
Disease caused by E. canis typically occurs in three phases. The initial acute phase is characterized by fever, malaise, lymphadenomegaly, splenomegaly, thrombocytopenia, leukopenia, and nonregenerative anemia. Abatement of signs occurs in 2–4 weeks, followed by a subclinical phase that persists for 2–3 months to years, during which the carrier state exists. 16 Some dogs subsequently enter a chronic phase, a period when severe clinical ehrlichiosis occurs. E. canis causes ocular disease and meningitis, 4 13 and E. ewingii causes relatively mild systemic illness in dogs. 16 Existing descriptions of the histopathology of ehrlichial uveitis are based on careful examination of only three dogs. 8 17 Although a number of cases of synovitis and an association with eosinophilic meningitis have been reported in dogs with E. ewingii infection, ocular disease has not been recorded. 5 12 14 There is a single case report of uveitis in a dog infected with E. platys, the cause of cyclic thrombocytopenia in dogs. 11
In recent years ehrlichial infections have been recognized with some frequency in humans. 18 Such infections range from subclinical to fatal. Disease is usually undifferentiated but often includes leukopenia, thrombocytopenia, and elevations of serum hepatic transaminase activity. 7 Cerebrospinal fluid pleocytosis, increased protein, and cerebral lesions have occurred in association with disease resulting from E. chaffeensis infection. The HGE agent and E. ewingii both induce undifferentiated disease in humans, but neither ocular nor central nervous system disease have been associated with these two agents. 3 18
The tissues used in the present study were obtained during the course of several experiments whose primary objectives were to investigate the susceptibility of the domestic dog to infection by E. chaffeensis and the agent of HGE and the ability of various ixodid tick species to acquire and transmit those and other ehrlichial agents to the domestic dog. In the present study, we compared the relative ability of experimentally induced infection with E. canis, E. ewingii, E. chaffeensis, and the agent of HGE to induce inflammation in the canine eye and cerebral meninges and characterized the cellular composition and magnitude (intensity) and distribution of inflammation at several postexposure time points.
Methods
The 27 dogs utilized in these experiments were housed in tick-free facilities of the Laboratory Animal Resource Unit (Oklahoma State University, Stillwater, OK) and maintained under standard laboratory conditions. Several methods of experimental exposure were utilized. Various exposure methods were utilized for E. canis. Exposure to E. ewingii was by intravenous administration of infective dog blood. Transmission of E. chaffeensis was either by exposure to infected ticks (two dogs) or by intravenous administration of infected cell culture (two dogs). The agent of HGE was successfully transmitted by intravenous injection of infective dog blood (three dogs), by exposure to infected Ixodes scapularis (two dogs), and by intravenous injection of infected mouse blood (one dog). Susceptibility/transmission was confirmed in each of the dogs by demonstration of Ehrlichia morulae in Wright-Giemsa–stained peripheral blood leukocytes of exposed dogs. In some instances, infection was further documented by polymerase chain reaction assay and seroconversion. 6 10 After the desired transmission/susceptibility data were obtained or after a period of convalescence, dogs were euthanatized by intravenous injection of a barbiturate solution, and necropsies were performed. Tissues obtained at necropsy were fixed in neutral buffered formalin, processed by routine methods for paraffin sectioning, and stained with hematoxylin and eosin (HE). Among the many organs and tissues examined histologically by light microscopy were one or both eyes and the brain. The intensity of ocular and meningeal inflammation was subjectively quantitated; + = minimal inflammatory cell infiltrate, ++ = infiltrates of intermediate density, and +++ = lesions with extensive and densely cellular infiltrates. Fourteen dogs were infected with E. canis; the postexposure interval until euthanasia was 22–23 days for two dogs, 26–34 days for nine dogs, and 49, 163, and 200 days for three dogs. Three dogs were infected with E. ewingii and euthanatized 23, 115, and 134 days later. Four dogs were infected with E. chaffeensis 29–69 days prior to euthanasia. Six dogs were infected with the agent of HGE; three were subjected to necropsy at <20 days postexposure, two others at 33 days postexposure, and 1 at 48 days postexposure.
Results
Whereas each of the 14 dogs infected with E. canis had ocular lesions and meningitis, neither ocular lesions nor meningitis were observed in any of the dogs infected with E. ewingii, E. chaffeensis, or the agent of HGE. The E. canis-infected dogs were in either the acute (11 dogs) or subclinical (three dogs) phase of infection when euthanatized. Inflammatory changes in the eye typically involved the ciliary body, iris, choroid, and retina and consisted of rather diffuse or aggregated populations of infiltrating inflammatory cells. Responding cells were predominantly lymphocytic but included monocytes and plasma cells. In infections of longer duration, the proportion of lymphocytes and plasma cells increased. Granulocytic cells were few. Infiltrates were most frequently present, most intense, and generally diffusely distributed in the ciliary body (Table 1, Fig. 1). Occasionally there were extensions of the infiltrate into the adjacent sclera. Only in the most severe infections were ciliary processes involved. The iris was somewhat less severely affected; cellular infiltrates usually existed as aggregates of inflammatory cells, but diffusely distributed infiltrates did sometimes occur. Choroiditis was present and expressed as diffuse or somewhat aggregated clusters of inflammatory cells (Fig. 2). Retinitis, indicated by relatively mild but occasionally intense perivascular cuffs, was the least common of the ocular lesions observed.
Location and intensity of inflammation at specified intervals following experimental exposure of 14 dogs to Ehrlichia canis.

DB = whole blood from carrier dog; administered intravenously.
TICK = Rhipicephalus sanguineus; feeding by adult ticks experimentally exposed as nymphs.
DH82 = infected mammalian cell culture; administered intravenously.
IDE8 = infected tick cell culture; administered intravenously.
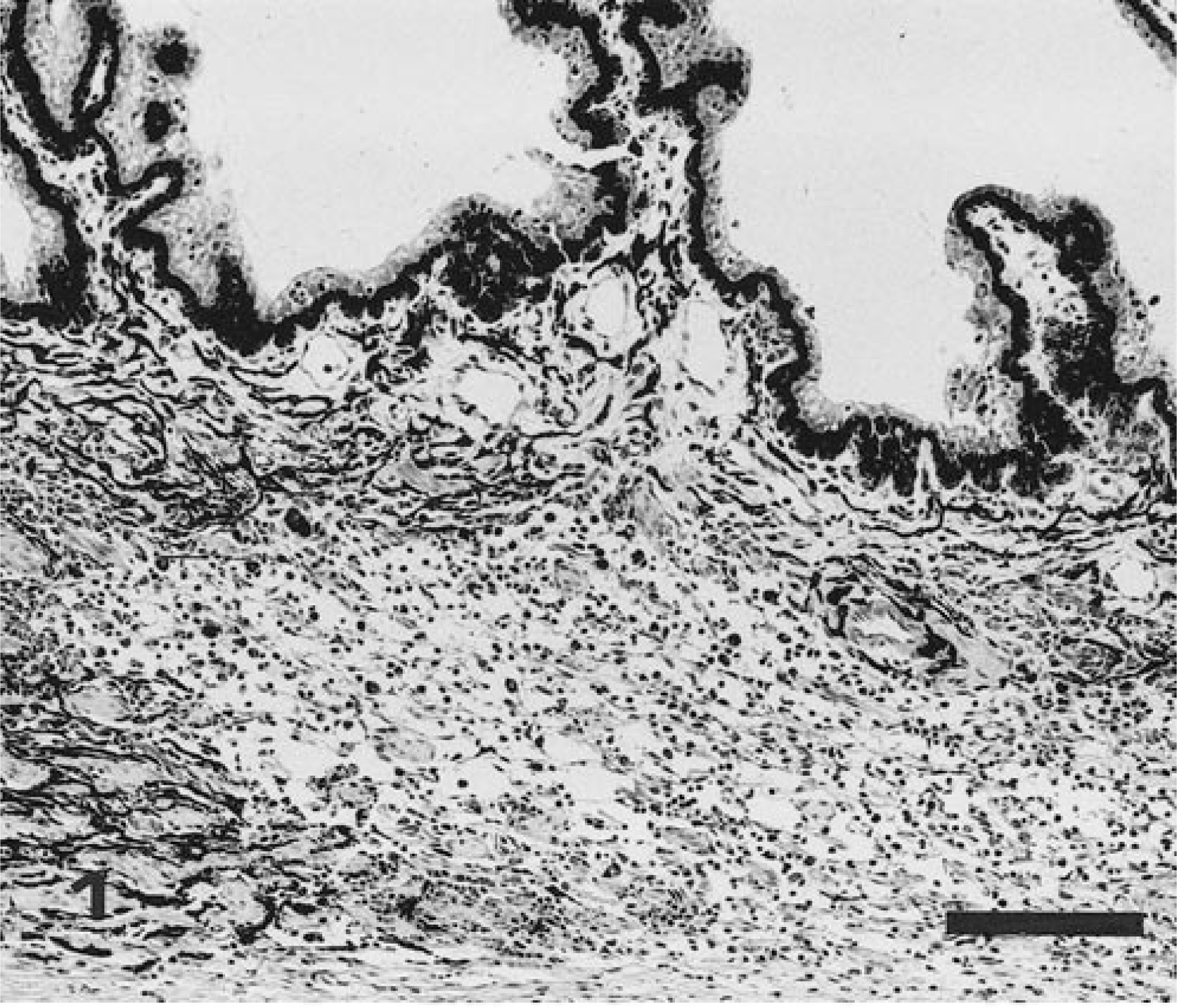
Eye; dog. Lymphoplasmacytic infiltrate and edema of ciliary body of dog exposed to E. canis 28 days prior to necropsy. Intensity of inflammatory infiltrate = +++. HE. Bar = 150 µm.

Eye; dog. Nonsuppurative choroiditis characterized by moderately severe (+++) cellular infiltrate composed predominantly of monocytes. E. canis postexposure interval = 26 days. HE. Bar = 50 µm.
Meningitis was present in each of the 14 dogs infected with E. canis but was not present in any dogs infected with E. ewingii, E. chaffeenesis, or the agent of HGE. Eight of the 14 dogs with meningitis had encephalitis, as indicated by perivascular cuffing and/or foci of gliosis in the neuroparenchyma. Meningitis was generally characterized by monocytic/lymphocytic/plasmacytic infiltrates either widely or focally distributed in the piaarachnoid (Fig. 3). When infiltrates were mild, there was clear orientation around vessels. The more densely cellular inflammatory cell infiltrates had a prominent population of monocytes; the more sparsely cellular, older lesions contained proportionally greater numbers of lymphocytes and plasma cells. Neuroparenchymal lesions generally consisted of vascular cuffing; foci of gliosis were occasionally present in E. canis-infected dogs but not in dogs infected with other ehrlichial agents.
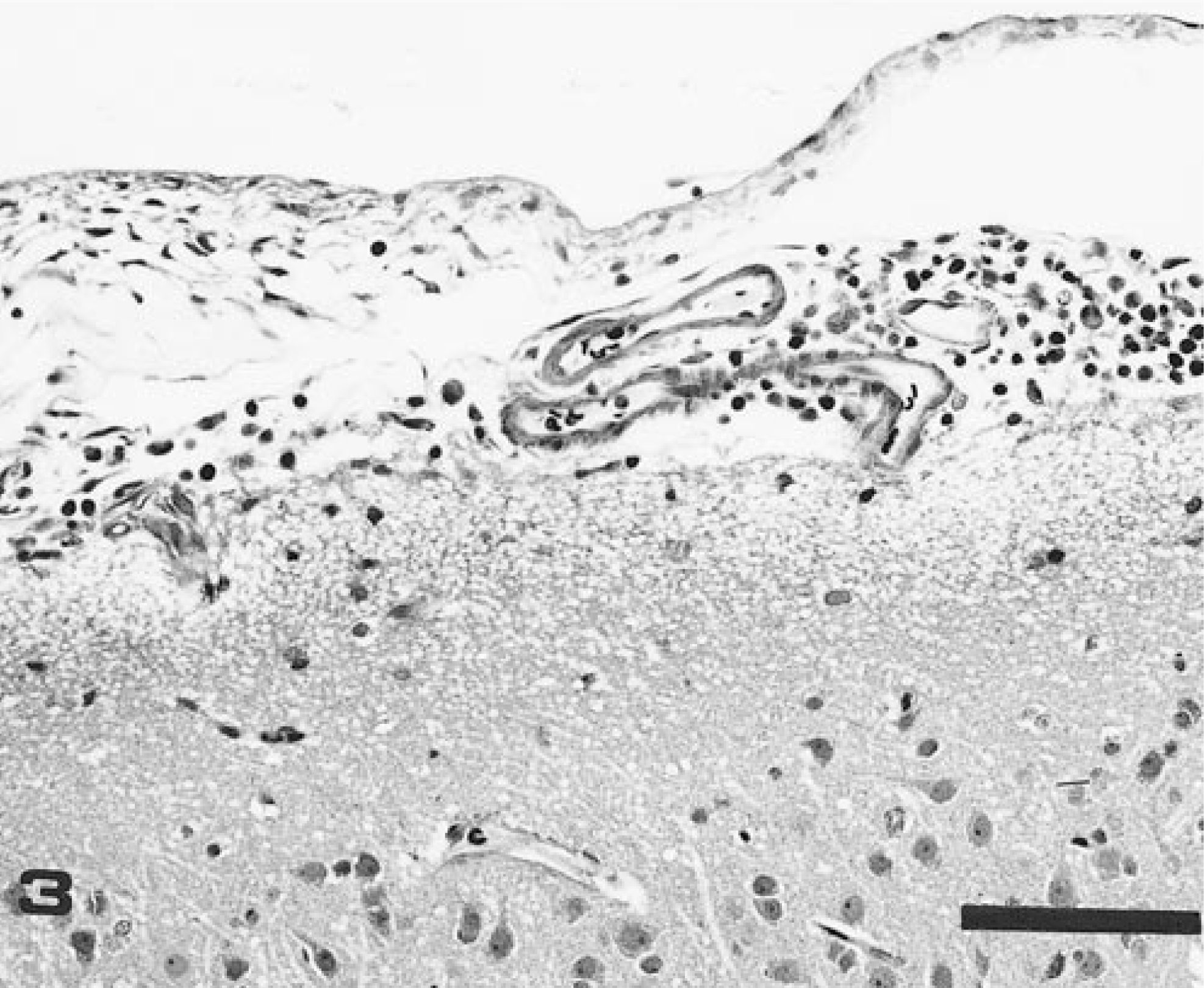
Meninges; dog. Lymphoplasmacytic leptomeningitis in dog exposed to E. canis 49 days prior to necropsy. Intensity of inflammatory infiltrate = ++. HE. Bar = 100 µm.
Discussion
Whereas other researchers have reported ophthalmoscopic abnormalities in dogs at 20, 8 28, and 33 17 days postexposure and histologic lesions at 35, 45, and ≥74 days postexposure, 8 17 ocular inflammation associated with E. canis infection was histologically evident in the present study in 2 dogs as early as 22 days postexposure and demonstrable in a mild degree in two infected dogs at 163 and 200 days postexposure. Neither of the two dogs euthanatized at ≥163 days postexposure were clinically ill at the time of necropsy, yet ocular lesions were clearly present.
These observations regarding the distribution and relative intensity of the inflammatory infiltrate within segments of the eye of dogs infected with E. canis are comparable to previous observations, 17 i.e., lesions are most frequent and intense in the ciliary body and iris and of decreasing intensity and frequency in the choroid and retina. Uveal inflammation and meningitis associated with E. canis infection consistently coexisted in the 14 dogs studied. The intensity of intraocular and meningeal inflammation was generally quantitatively related. Meningeal inflammation occurs with greater frequency and probably persists longer than neuroparenchymal inflammation. No special attempt was made to observe or discover ehrlichial morulae within reacting cells of the eye or meninges. Although organisms are quite readily demonstrable in air dried, alcohol-fixed Romanovsky-stained imprints of certain tissues, organisms are exceedingly difficult to identify with confidence in formalin-fixed histologic sections. 9 Although the domestic dog is susceptible to natural and/or experimental infections with E. ewingii, E. chaffeensis, and the agent of HGE, neither ocular disease nor meningitis accompanied those infections in the 13 dogs examined in this study.
