Abstract
Erythema multiforme major was diagnosed in a dog with necrotizing parvoviral enteritis. Skin lesions consisted of ulceration of the footpads, pressure points, mouth, and vaginal mucosa; vesicles in the oral cavity; and erythematous patches on the abdomen and perivulvar skin. Microscopic examination of mucosal and haired skin specimens revealed lymphocyte-associated keratinocyte apoptosis at various levels of the epidermis. Basophilic cytoplasmic inclusions were seen in basal and suprabasal keratinocytes. Immunohistochemical staining, performed with canine parvovirus-2-specific monoclonal antibodies, confirmed the parvovirus nature of the inclusions in the nucleus and cytoplasm of oral and skin epithelial cells. This is the first case of canine erythema multiforme reported to be caused by a viral infection of keratinocytes. This case study indicates that the search for epitheliotropic viruses should be attempted in cases of erythema multiforme in which a drug cause cannot be identified.
In humans, the classification of erythema multiforme (EM) variants recently has been revised with an emphasis on clinical manifestations. 2 The relevance of this modified clinical nosology has been supported by subsequent epidemiologic and pathologic case studies. Dermatoses described clinically as EM minor and major most commonly seem to be caused by viral infections leading to lymphocyte-mediated keratinocyte apoptosis. 1 3 6 7 Human EM generally is caused by herpes simplex virus, 3 6 7 but it also can be triggered by other infectious agents such as parvovirus B19. 8
In 1998, the consensus clinical classification used for human EM was adapted to the canine species. 4 That case study established that, in contrast to previous reports, 10 canine cases of EM (minor or major) rarely were associated with previous drug exposure. 4 In non–drug-related cases, offending causes could not be determined but a viral etiology was considered plausible. The purpose of the present paper is to describe a canine case of EM major in which parvovirus infection of epidermal and mucosal keratinocytes led to lymphocyte-associated apoptosis and clinical signs of EM major.
A 2-month-old female Great Dane puppy was presented, 3 days after adoption, with acute-onset diarrhea, vomiting, dehydration, and skin lesions. Because parvovirus enteritis had been diagnosed recently at the facility of the dog's breeder, parvovirus was suspected as the cause of diarrhea. However, 6 days before the initial presentation, the dog had received a tetravalent vaccine (distemper, parvovirus, parainfluenza, and hepatitis). Dermatologic examination revealed well-demarcated ulceration of the footpads (Fig. 1) and pressure points, as well as mouth and vaginal mucosae. Vesicles were seen in the oral cavity. Erythematous patches were present on the abdomen and chin. In spite of fluid therapy and intravenous cephalexin and metoclopramide, the dog died 2 days after presentation.
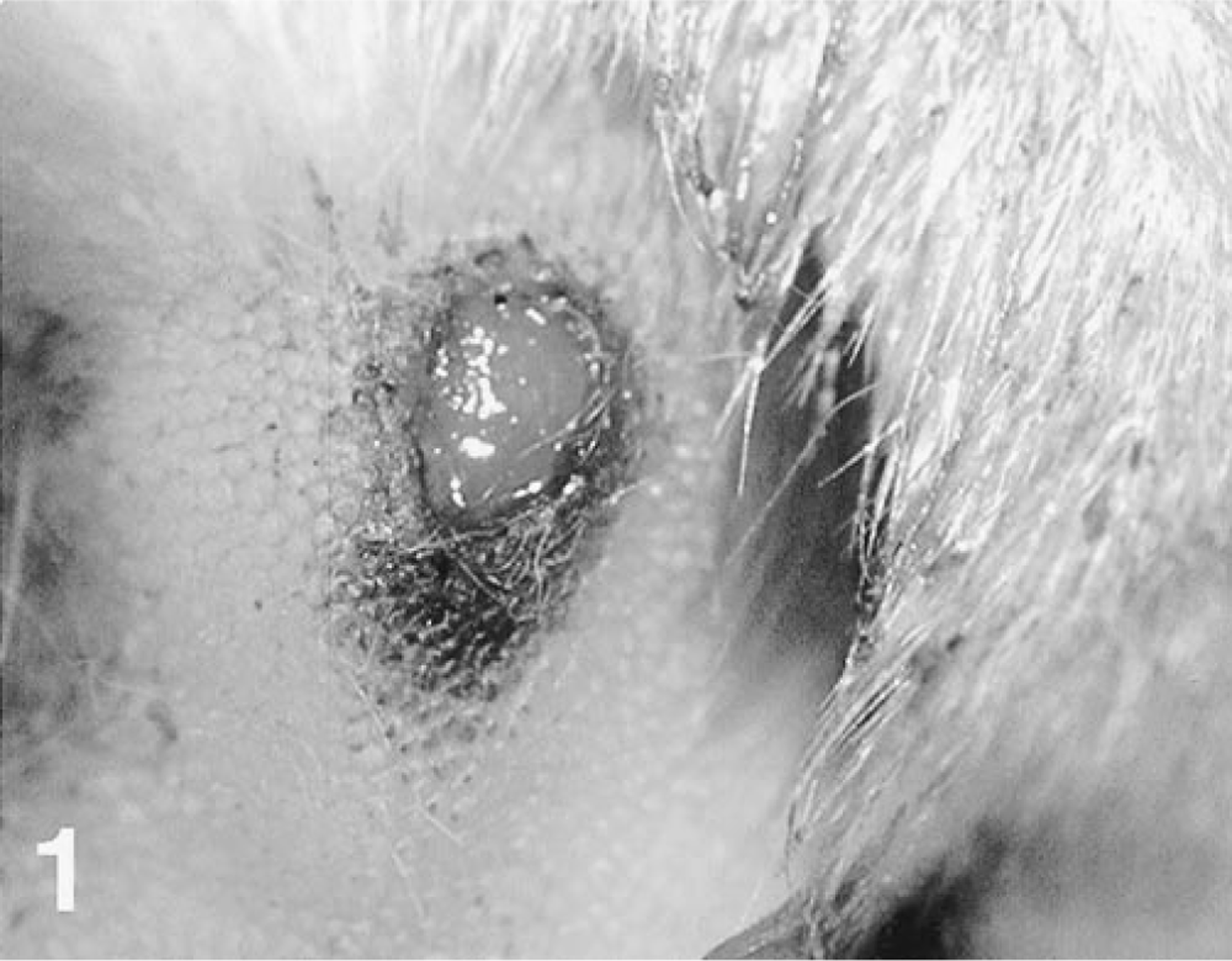
Skin, footpad; dog. A sharp-edged ulcer is present.
A necropsy was performed and necrotic lesions were seen throughout all intestinal sections. Histopathologic analysis of small intestine specimens consisted of severe segmental necrotizing enteritis suggestive of an acute infection due to canine parvovirus-2 (CPV-2). Viral inclusions were not identified in the intestinal specimens, presumably because of the severe necrosis of the digestive epithelium. Skin biopsy specimens were obtained from lesional skin and oral mucosa. Focal mononuclear interface gingivitis was identified in biopsy samples collected from the gum. Additionally, confluent basal keratinocyte vacuolation progressing to vesiculation with epithelial ulceration and neutrophil accumulation was observed. Prominent lymphocyte exocytosis was present in preblistered mucosal epithelium. Keratinocyte apoptosis, often in close contact with lymphocytes (e.g., satellitosis), was observed at all levels of the epithelium. In some specimens, basophilic inclusions were observed in the cytoplasm of basal and suprabasal keratinocytes. Examination of haired skin specimens revealed varying degrees of the same pathologic process. The epidermis exhibited focal hyperplasia, crusting, and erosion. Lymphocyte exocytosis and keratinocyte apoptosis with satellitosis were restricted to sites of epidermal hyperplasia (Figs. 2, 3). Numerous basophilic cytoplasmic inclusions were seen in the lower third of the hyperplastic epidermis (Figs. 2, 3).
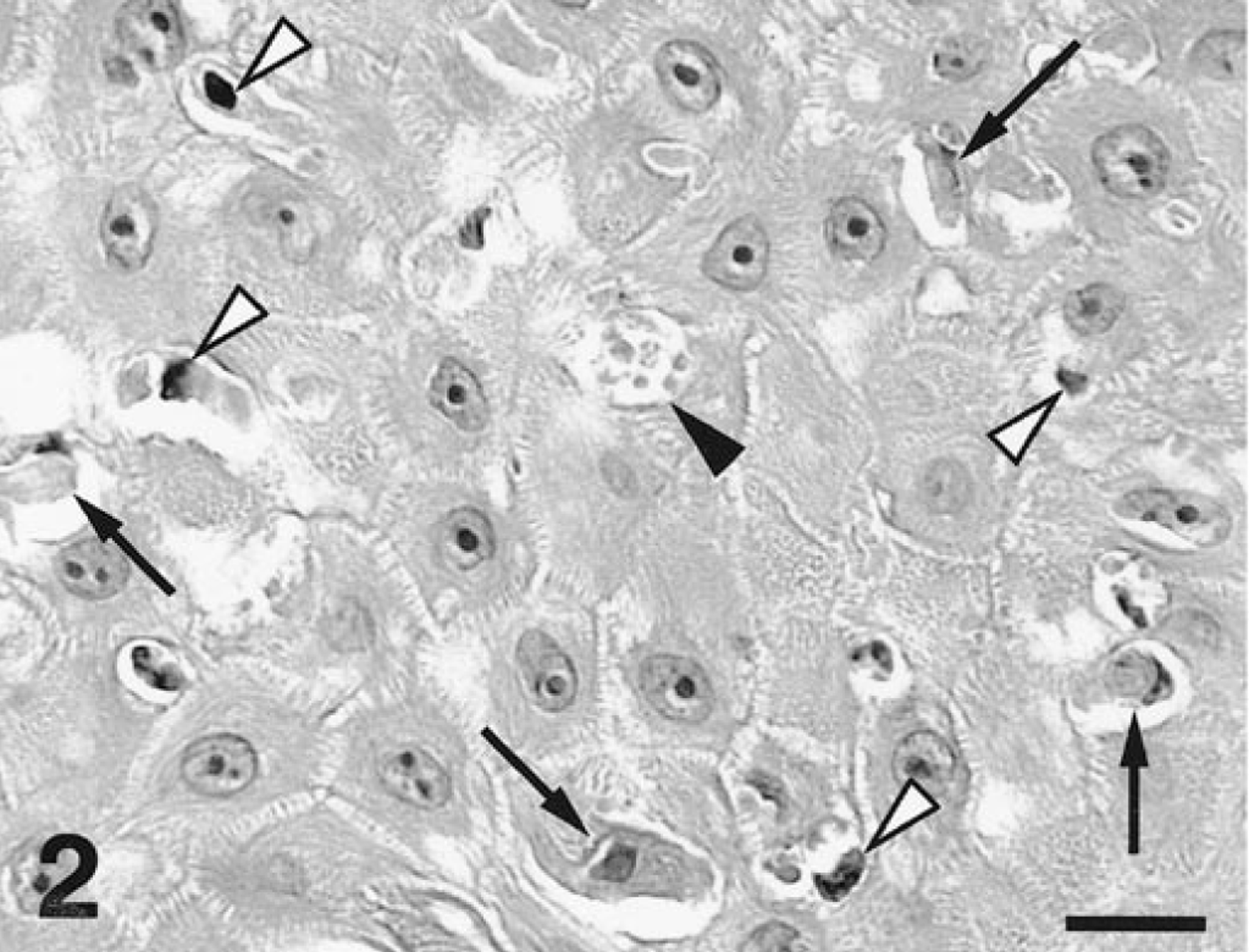
Haired skin; dog. Clusters of lymphocytes (white arrowheads) are located in the immediate vicinity of apoptotic keratinocytes (black arrows). Viral inclusions are visible in an intracellular vacuole (black arrowhead). H.E. Scale bar = 18 µm.
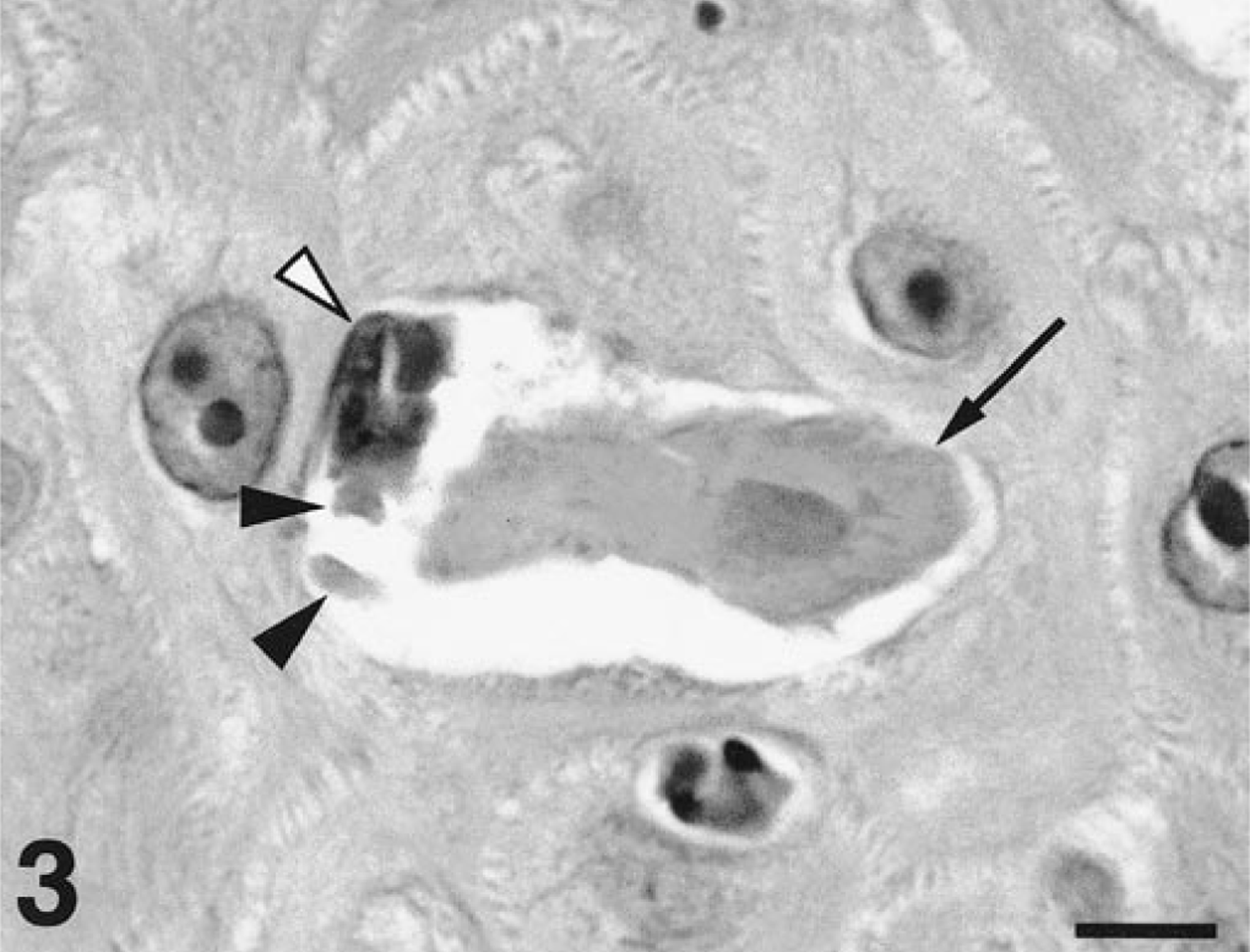
Haired skin; dog. A lymphocyte (white arrowhead) is situated near an apoptotic keratinocyte (black arrow) that contained viral inclusions (black arrowheads). H.E. Scale bar = 4 µm.
To verify the viral origin of cytoplasmic inclusions, a three-step avidin–biotin–peroxidase immunohistochemical technique was performed as previously described. 9 Immunostaining of paraffin-embedded sections was done with two monoclonal antibodies specific for CPV-2 (CPV2c2A and CPV103B10A, 12,000 dilution, Mérial, Lyon, France). Examination of negative controls, consisting of sections immunostained with irrelevant monoclonal antibodies, was unremarkable. In mucosal specimens, multiple intracellular parvovirus inclusions were seen throughout the epithelium. In haired skin samples, parvovirus inclusions were seen most commonly coalescing in basal and juxtabasal keratinocytes of hyperplastic epidermis (Fig. 4). The smallest viral inclusions were identified in the keratinocyte's nucleus (Fig. 4). Inclusions further aggregated and filled-up the cytoplasm of epithelial cells leading to displacement of the nucleus to the cell's periphery and subsequent cell degeneration. Immunostaining of digestive specimens similarly demonstrated CPV-2 particles in the epithelial crypts of the small intestine. Furthermore, viral inclusions of skin and mucosal sections were negative when immunohistochemistry was performed using monoclonal antibodies specific for either distemper virus (1:50, Mérial, Lyon, France) or papillomavirus-group–specific antigens (AR087–5R, undiluted, Biogenex, San Ramon, CA).
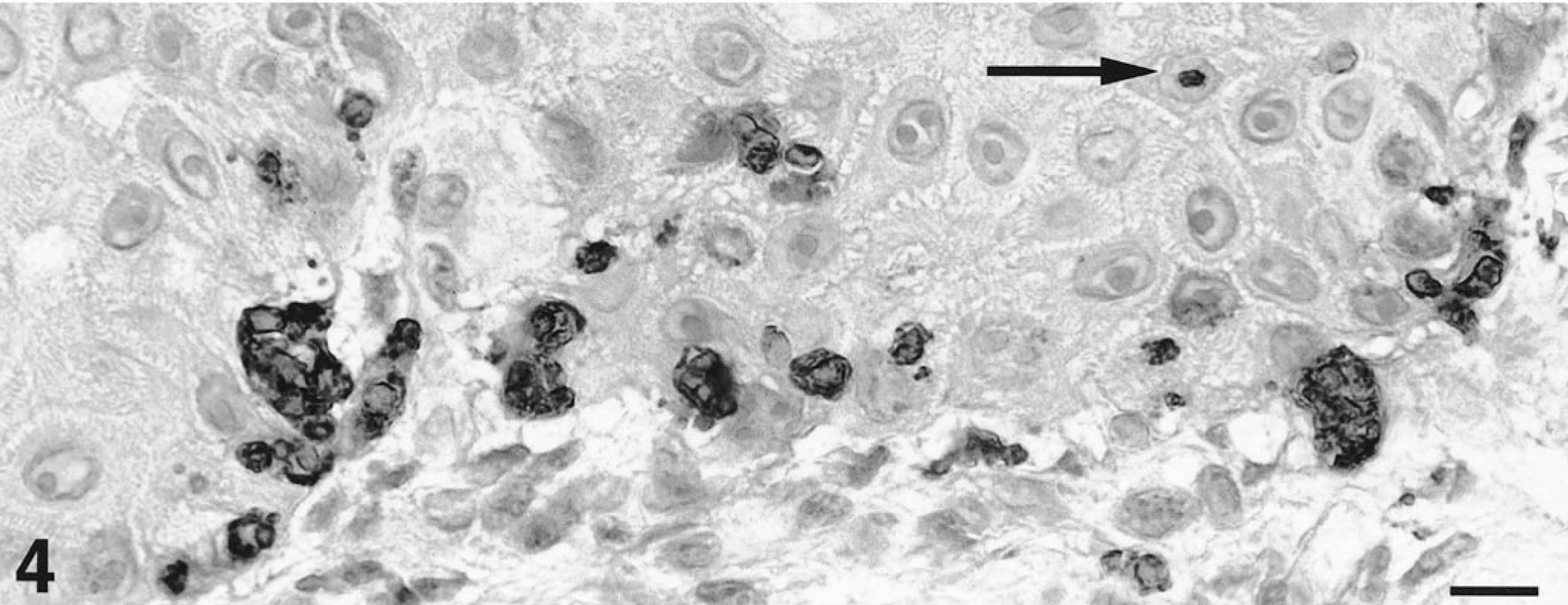
Haired skin, abdomen; dog. Dark-staining viral inclusions fill the cytoplasm of basal and juxtabasal keratinocytes. Viral aggregates are occasionally present in the superficial dermis. Small inclusions are visible within keratinocyte nuclei (black arrow). Avidin–biotin–peroxidase immunohistochemistry, aminoethylcarbazole chromogen, hematoxylin counterstain, CPV2c2A parvovirus-specific monoclonal antibodies. Scale bar = 11µm.
According to the recently proposed classification of canine EM, the skin lesions exhibited by this patient fit the criteria for a clinical diagnosis of EM major (erythematous patchy lesions with ulcerations on less than 10% of the body surface and with more than one mucosa affected). 4 Our histologic and immunohistochemical investigations suggested CPV-2 infection of mucosal and epidermal keratinocytes as the primary cause of EM in this dog. Remarkably, most parvoviral inclusions were identified in the cytoplasm of keratinocytes, whereas only rare viral particles were seen in cell nuclei. However, these observations are identical to those described in glossal specimens of dogs naturally infected with CPV-2. 5 Indeed, viral replication initially occurs in the nucleus but large virion clusters appear as cytoplasmic aggregates. However, these inclusions still are surrounded by the nuclear membrane and should be referred to as pseudocytoplasmic. 5 A viral infection of keratinocytes suggests a logical pathogenesis of EM lesions in this dog. We hypothesize that an infection of stem cells and transient amplifying keratinocytes most likely occurred following hematogenic dissemination of the parvovirus. Viral peptides could be presented by class I major histocompatibility complex molecules at the surface of epithelial cells. Recognition of viral antigens by T-lymphocytes, possibly sensitized by the previous parvovirus vaccination, would trigger these cytotoxic cells to induce the apoptosis of virus-infected keratinocytes.
The present case study supports the concept that a viral etiology is possible in some forms of canine EM. We propose that a search for epitheliotropic viruses (e.g., distemper, papilloma-viruses, parvoviruses, and herpesviruses) should be attempted in cases of canine EM in which a causative drug cannot be clearly established.
Footnotes
Acknowledgements
We thank Dr. Guaguère for his comments in the initial phase of the study and Drs. Latour and Soulier (Mérial, Lyon, France) for providing distemper- and parvovirus-specific monoclonal antibodies.
