Abstract
During the review of a rat carcinogenicity study, a spectrum of granular cell lesions was recognized in the distal female reproductive tract. To verify the diagnoses, cell populations of nine granular cell alterations of the cervix or vagina were characterized immunohistochemically and four were evaluated ultrastructurally. Immunoreactivity was demonstrated in 8/9 cases with $100 protein, 6/9 cases with neuron-specific enolase, and 7/9 cases with Leu-7. Granular cells were negative for smooth muscle-specific actin and calretinin. The immunohistochemical profile of these lesions was similar to that previously reported in other species, including humans. Ultrastructurally, as expected many lysosomal bodies were present in the cytoplasm of granular cells in all specimens evaluated. Based on the detailed evaluation of a series of lesions, we adopted the following diagnostic criteria and nomenclature for the granular cell changes of the female reproductive tract of rats. Granular cell aggregates were non-space-occupying lesions composed of clusters of typical granular cells. Benign granular cell tumors were space occupying lesions that typically contained prominent interstitial collagen and were either discrete masses or were difficult to discern from the surrounding tissues. Some benign tumors also contained foci of spindle cells with decreased granularity. Malignant tumors exhibited pleomorphism and an increased nucleus: cytoplasm ratio morphologically but had the same biologic behavior as the benign tumors. We applied these diagnostic criteria during the review of controls from 9 carcinogenicity studies. Up to 23% of control females in those carcinogenicity studies had granular cell lesions that could be classified into one of the three categories. Granular cell lesions appear to be common in the cervix/vagina of the Sprague-Dawley rat, and tumors may develop from granular cell aggregates.
Granular cell tumors in several species have been described from different anatomic locations, including the reproductive tract. 4 7–9, 11 17 20 22 On the light microscopic level, regardless of the species of origin and anatomic site, granular cell tumors are characterized by a uniform population of cells with abundant granular eosinophilic cytoplasm that is periodic acid–Schiff (PAS) positive and diastase resistant and typically with small basophilic nuclei. 17 20 22 Most granular cell tumors are positive with immunohistochemical markers for neuroectodermal origin such as S100 protein and neuron-specific enolase (NSE). 2 3 4 15 16 18 Ultrastructurally, the hallmark of the tumor is the many lysosomal bodies. 6 20 Granular cell tumors are an enigma because no normal cell counterpart is known. Over the years, several cells of origin have been proposed, from muscle cells to fibroblastic and histiocytic cells. 6 22 Generally, most granular cell tumors are considered to be of Schwann cell origin, although there is also evidence of other lineages, such as primitive meningeal arachnoid cells in the case of rat meningeal granular cell tumor. 6 9 18
Another interesting feature of granular cell tumors is that although they may occur in a wide range of sites among different species, their distribution is species specific. In dogs, granular cell tumors most frequently occur in the oral cavity, in horses in the lungs, and in rats in the meninges and brain. 9 12 17 19 22 Granular cell tumors of the rodent reproductive tract have been primarily reported in mice following treatments with estrogens. 7 8 Single cases of spontaneous cervical or vaginal granular cell tumors have been reported in rats. 4 14 In one rat carcinogenicity study, a spectrum of lesions composed of granular cells occurred. Granular cell alterations of control and treated rats of this original study were examined microscopically with special stains and were evaluated ultrastructurally. The initial finding of several granular cell lesions in control and treated rats stimulated our subsequent work to characterize the spectrum of lesions in control rats of eight additional carcinogenicity studies. Here, we summarize this work and present the newly adopted diagnostic criteria and nomenclature for granular cell lesions of the distal female reproductive tract of aged Sprague-Dawley rats.
Materials and Methods
Tissue samples
Samples for the prospective segment of the study originated from control and treated animals of a 104-week carcinogenicity study of Sprague-Dawley rats (Charles River, Raleigh, NC). For the retrospective segment of the study, control animals from archived rat carcinogenicity studies were selected if the vagina was a protocol-required tissue. The in-life phase of these 104-week carcinogenicity studies, with Sprague-Dawley rats originating from several Charles River facilities within the USA, took place at the same site between 1983 and 1997. In this retrospective and prospective study, the female distal reproductive tract of control animals from a total of 9 studies was reviewed.
Light microscopy and immunohistochemistry
Tissue samples were obtained longitudinally from the cervix and vagina, routinely processed, embedded in paraffin, and stained with hematoxylin and eosin (HE); a few samples were also stained with PAS (with or without diastase digestion) and Masson's trichrome. The immunocytochemical reaction was carried out with the streptavidin-biotin complex method using alkaline phosphatase as substrate and new fuchsin as chromogen. Positive controls included multiorgan blocks and normal structures within sections containing the granular cell lesions. Negative control sections were incubated with nonimmune serum instead of the primary antibody. The immunohistochemical methods are summarized in Table 1.
Immunohistochemistry reagents and procedures for evaluation of granular cell lesions in rats.
Electron microscopy
Specimens were collected from remnants of four tumors maintained in formalin and were transferred to modified Karnovsky's fixative, postfixed in 0.1 M sodium cacodylate–buffered 1% osmium tetroxide, and stained en bloc in 2% uranyl acetate in 10% ethanol. Samples were embedded in EMbed (Epon) 812, and ultrathin sections cut for transmission electron microscopy were double stained with uranyl acetate and lead citrate and examined using a Zeiss EM-902 electron microscope.
Results
Diagnostic criteria
Granular cell lesions were typically not recognized grossly and were often difficult to distinguish from the surrounding tissues microscopically. They either appeared as typical tumors or contained only aggregates of a few cells similar to those seen in tumors. Because published diagnostic criteria were not available, we developed a nomenclature and diagnostic criteria for use in these rats (Table 2).
Criteria for the diagnosis of granular cell lesions of the female distal reproductive tract of rats.

Granular cell aggregates
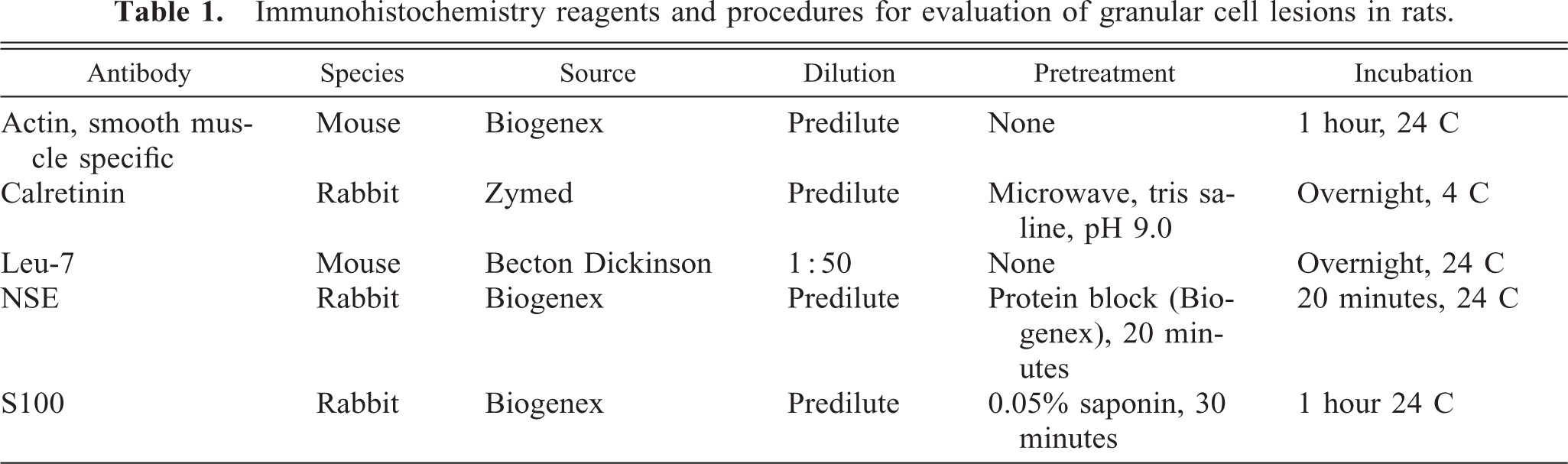
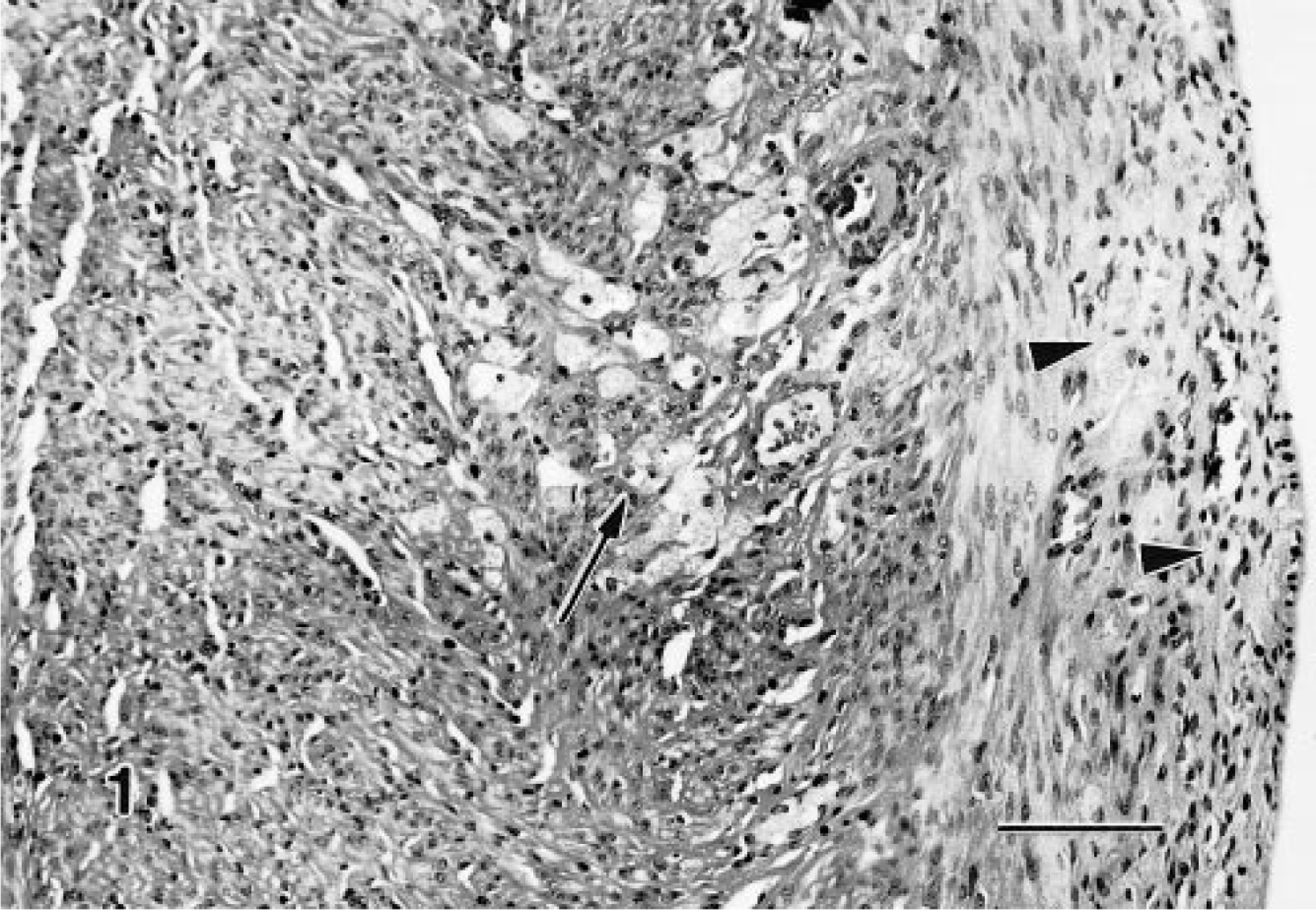
Lesions that typically lacked collagen, did not appear to be space-occupying, and did not efface the normal tissue architecture were recognized as granular cell aggregates (Fig. 1). Many of the aggregates were within the wall of the cervix/vagina, but they were also found in the adventitia. Single scattered granular cells were thought to be below the diagnostic limits and were excluded from consideration (Fig. 2). Although the number of cells forming aggregates varied, aggregates of minimal severity were composed of a cluster of at least three to five cells. In severe lesions, minimal amounts of collagen could be present.
Granular cell aggregate, distal female reproductive tract; Sprague-Dawley rat. Granular cells appear to infiltrate the inner layer of the muscularis and are obvious (arrow), whereas those in the outer layer are difficult to discern (arrowheads). HE. Bar = 80 μm.
Granular cells, distal female reproductive tract; Sprague-Dawley rat. The two granular cells (arrowhead) within the outer muscle layer are considered below the diagnostic limits. HE. Bar = 50 μm.
Benign granular cell tumors
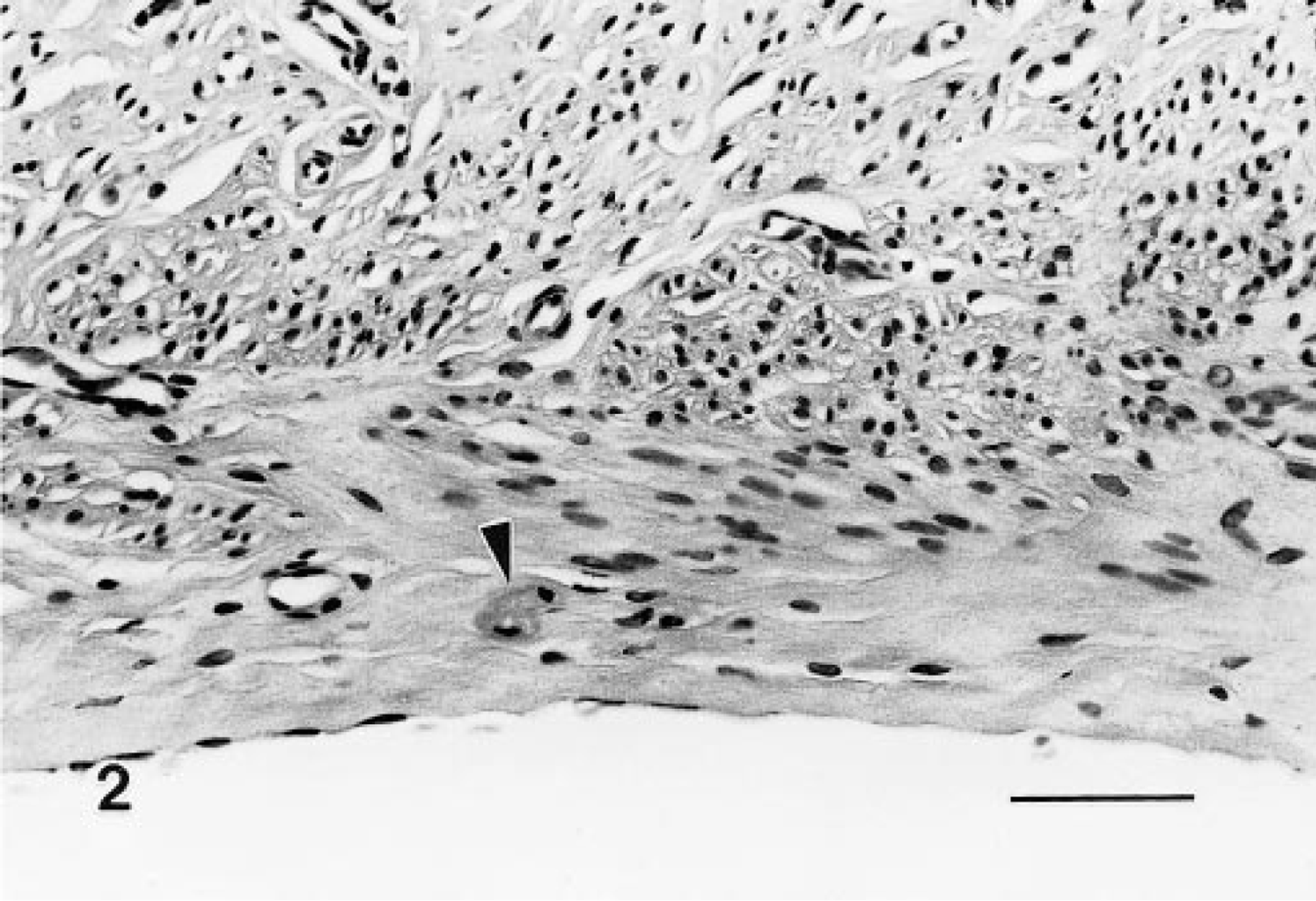
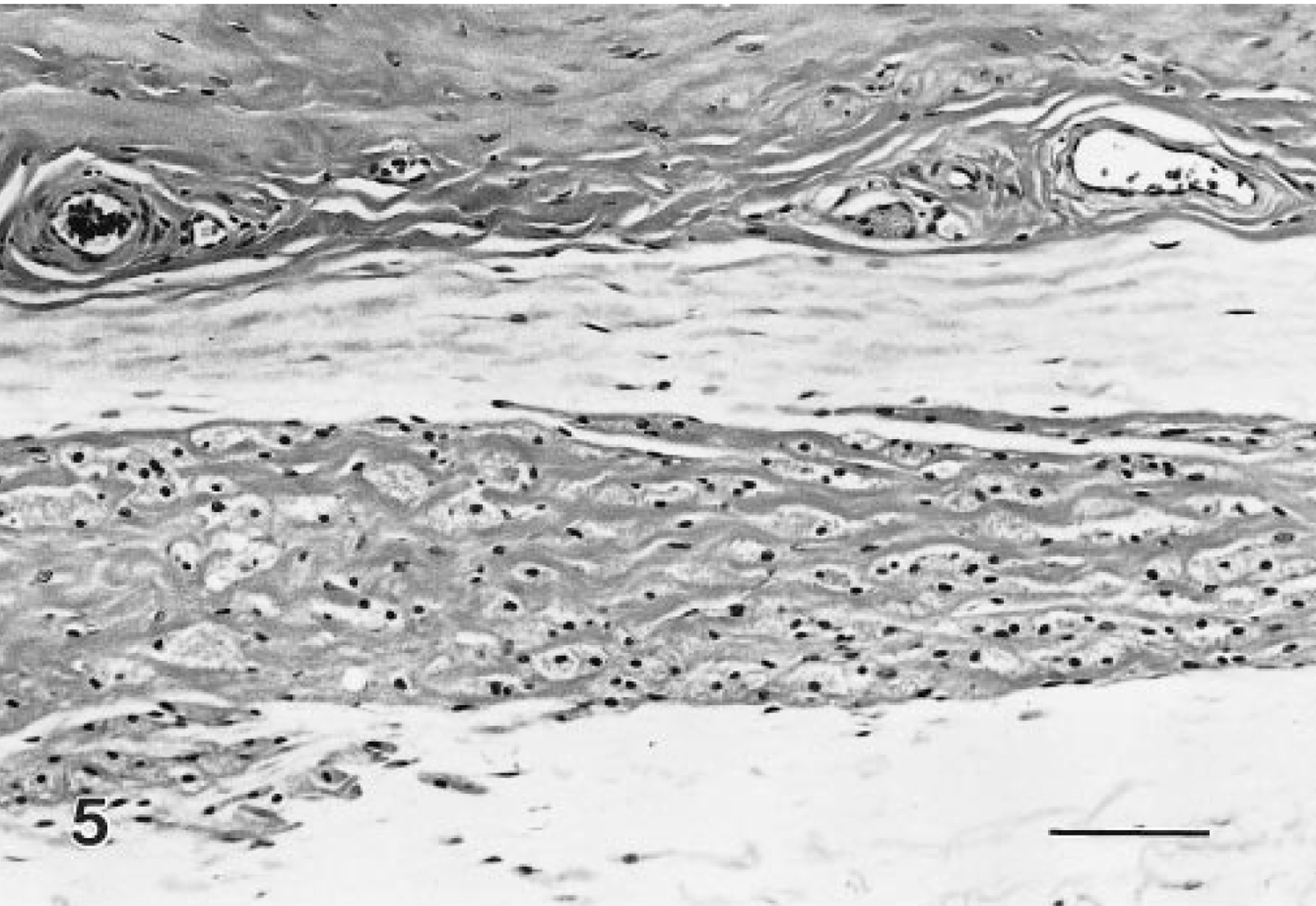
These lesions were space occupying and expansile, and although often microscopic, they were composed of typical granular cells and variable but frequently abundant amounts of interstitial collagen. Many of the tumors were distinct nodules within the muscularis or extended into the adventitia; however, others had indistinct borders and expanded along tissue planes and ligaments (Figs. 3–6). A variant of the benign tumor contained foci of cells with decreased granularity and was identified as a granular cell tumor with spindle-cell component. In benign tumors, mitotic figures were not noted.
Benign granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Granular cells form symmetrical cervical nodules and are also present along advential surface of the vagina (arrowheads). HE. Bar = 3 mm.
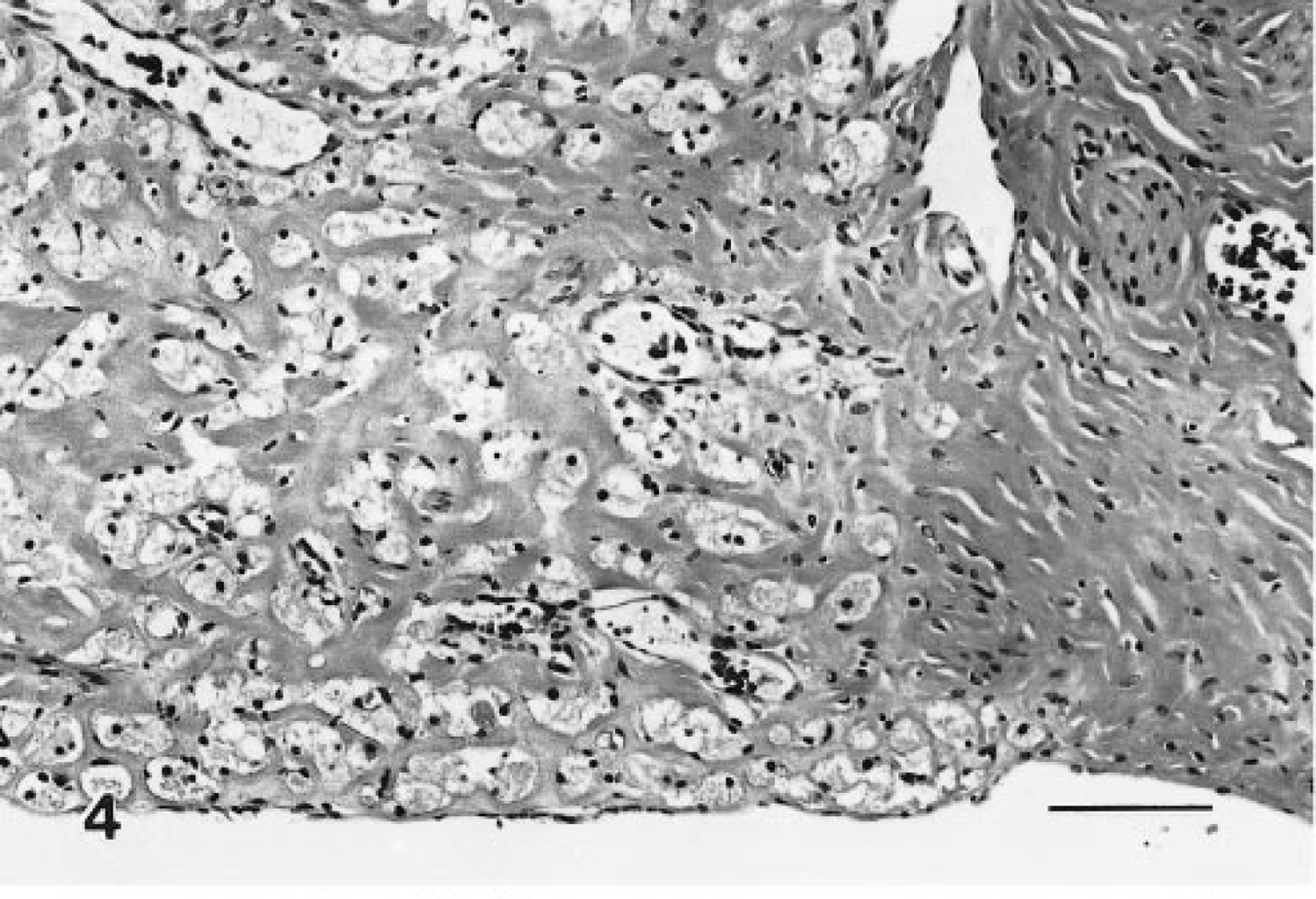
Benign granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Detail of Fig. 3, edge of a nodule. Moderate amounts of collagen separate granular cells. HE. Bar = 80 μm.
Benign granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Detail of Fig. 3, adventitial surface. The lesion is more densely cellular containing less collagen in this region than in the cervical nodules. HE. Bar = 80 μm.
Benign granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. A wide band of closely apposed granular cells are along the adventitial surface. HE. Bar = 80 μm.
Malignant granular cell tumors
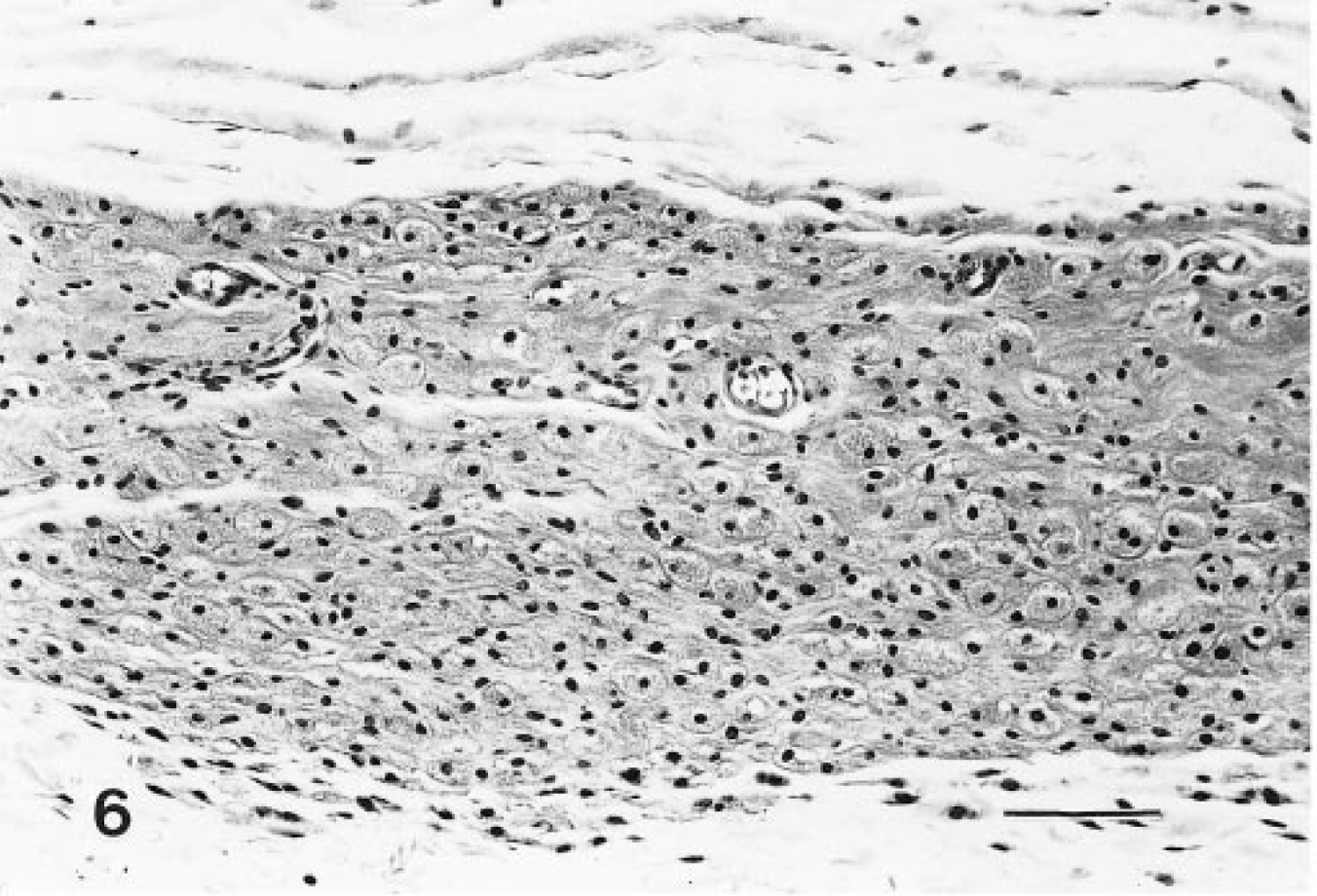
These lesions were generally larger nodules, although often they were not noted grossly. Microscopically, they were composed of a pleomorphic cell population. Typically, the periphery of the tumors contained well-differentiated granular cells, whereas cells with decreased granularity, an increased nucleus:cytoplasm ratio, and spindle-cell morphology were more centrally located (Figs. 7, 8). Mitotic figures were not common, and necrosis was occasionally observed. Tumors were classified as malignant based only on their morphologic features; however, there was no evidence that their biologic behavior was different from that of their benign counterparts. There were no regional or distant metastases.

Malignant granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. A large mass has a central area of necrosis that is surrounded by typical granular cells and spindle cells. HE. Bar = 300 μm.
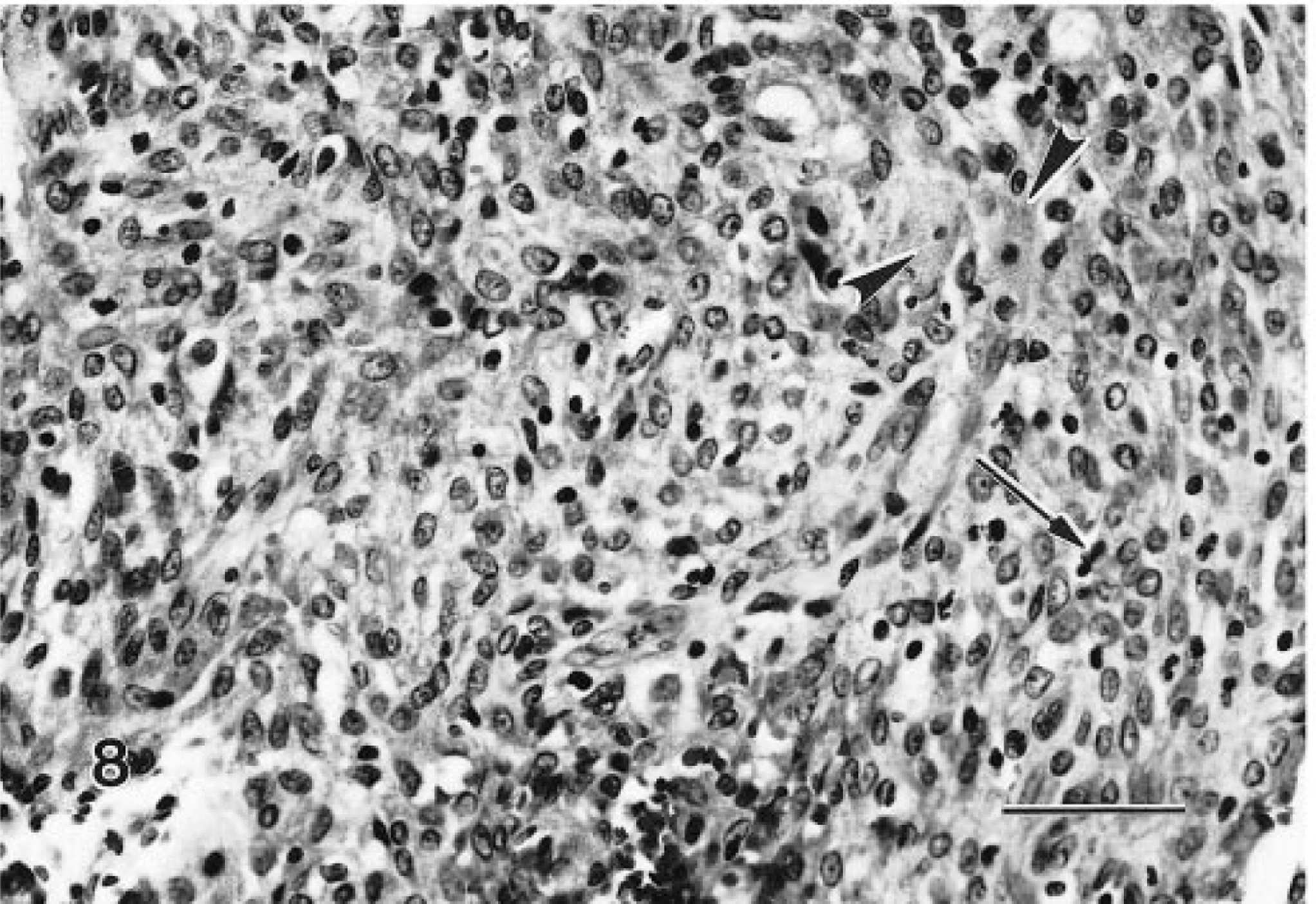
Malignant granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Higher magnification of Fig. 7. Pleomorphic spindle cells are admixed with occasional well-differentiated granular cells (arrowheads). Mitotic figures (arrow) are not common. HE. Bar = 50 μm.
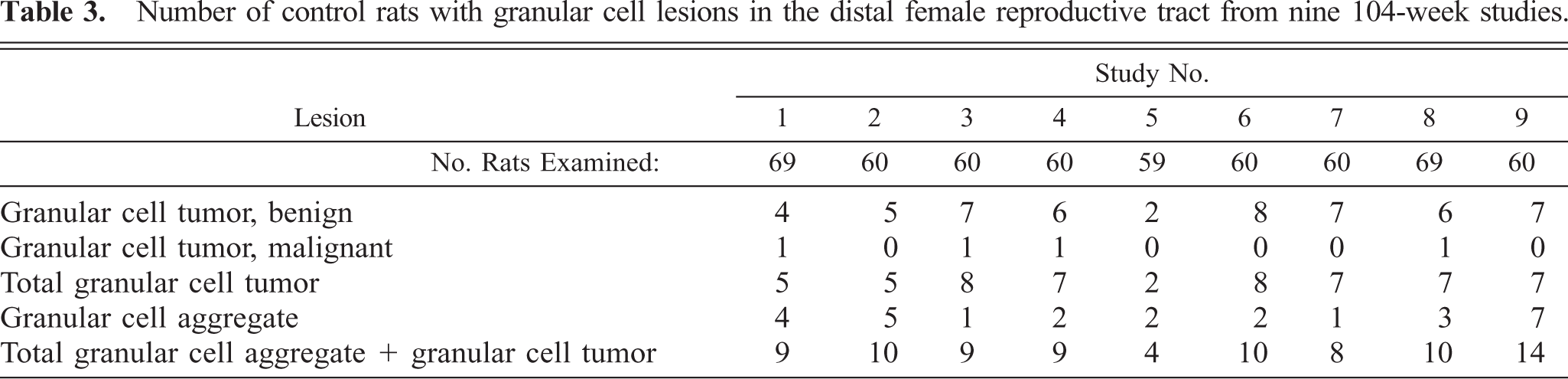
Granular cell lesions in control animals from carcinogenicity studies
The incidence of granular cell lesions in the distal female reproductive tract of control Sprague-Dawley rats in nine carcinogenicity studies is summarized in Table 3. Typically, a single longitudinal section of the cervix/vagina was evaluated from each animal of the group. At least one animal in each control group had a granular cell aggregate, and up to seven animals had benign granular cell tumors. Morphologically malignant granular cell tumors occurred in no more than one animal per group. Based on a single representative longitudinal section of the distal reproductive tract, up to 23% of the animals had some type of granular cell lesions. A small number of animals were evaluated from 13- and 26-week studies. In rats from the shorter studies, no granular cells were recognized. A few rats from the 26-week studies had typical granular cells; however, these cells were too few to meet the diagnostic criteria for aggregates (data not shown).
Number of control rats with granular cell lesions in the distal female reproductive tract from nine 104-week studies.
In 25–50% of the sections, at least portions of the paracervical ganglia were included. One such ganglion contained a minimal granular cell aggregate (Fig. 9), and another ganglion was effaced by a benign granular cell tumor. The numerous paracervical ganglia, however, were not predilection sites for granular cells. In contrast, nerves were easily recognized within or near granular cell tumors, and granular cells were present occasionally within nerves.
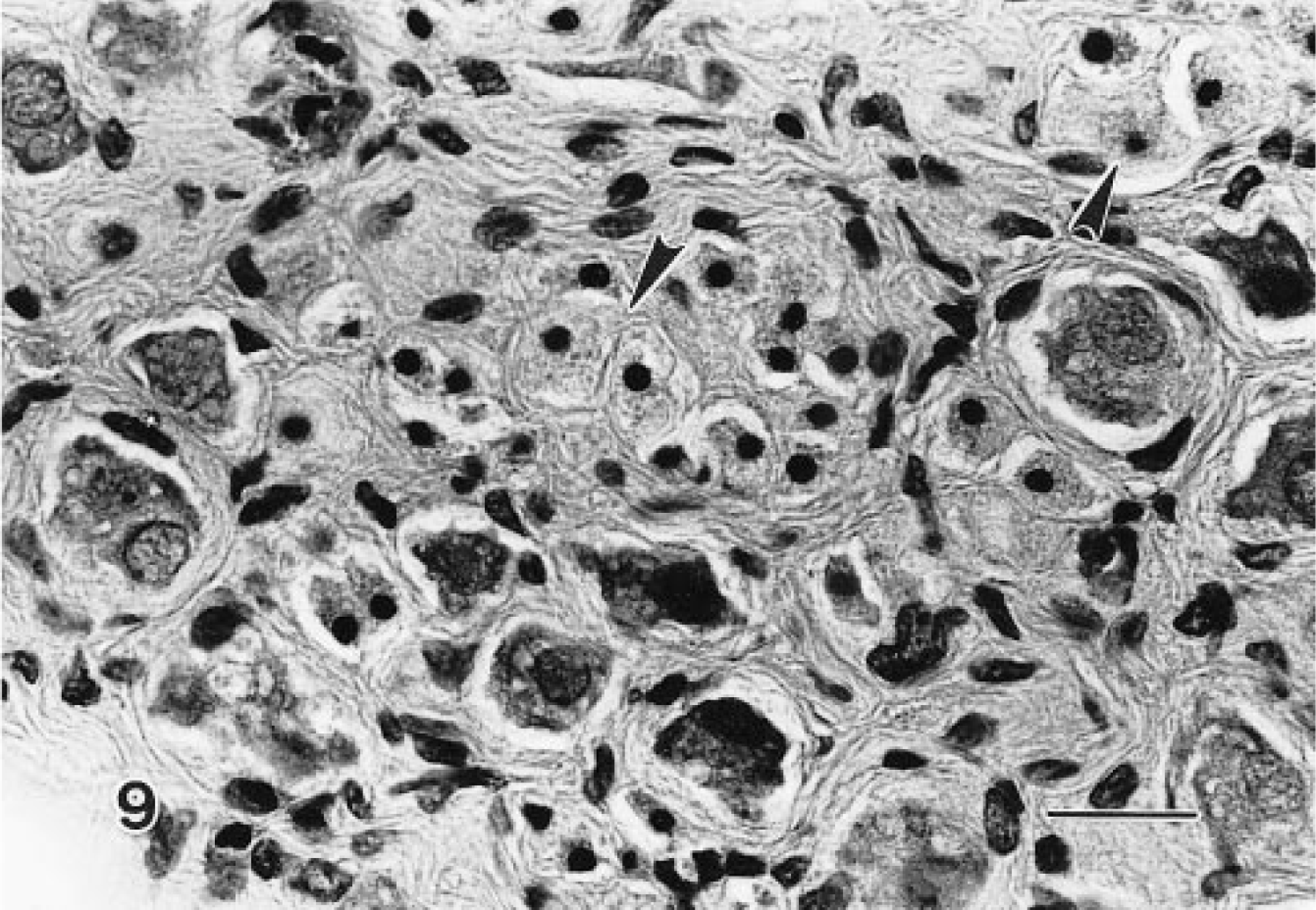
Granular cell aggregate, paracervical ganglion; Sprague-Dawley rat. Typical granular cells (arrowheads) are between neurons of the ganglion. HE. Bar = 25 μm.
Immunohistochemistry and electron microscopy
The staining characteristics of granular cell lesions observed using a panel of immunohistochemical markers are presented in Table 4. Immunoreactivity of various intensities was demonstrated with S100, NSE, and Leu-7 (Figs. 10–12). In this cohort of granular cell lesions, S100 was the most useful marker, with the most consistently strong signal for the demonstration of granular cells. Moreover, nerves were visualized with ease within or in the vicinity of lesions in S100-stained sections (Fig. 10). Immunoreactivity with S100 was primarily cytoplasmic in granular cells, although nuclear staining was also detected in somewhat vesicular nuclei. Immunoreactivity to smooth muscle–specific actin was interpreted as negative because only single cells of two tumors were stained. In the majority of the lesions, only vaginal and vascular smooth muscle exhibited positive staining with this marker. Calretinin did not stain granular cells in any of the lesions, although nerves were stained within sections of the female distal reproductive tract. Some specimens had somewhat weak immunoreactivity to one or more of the markers primarily because of the poor preservation of granular cellular cytoplasm, which was a feature of many of the lesions. Interpretation of immunohistochemical sections was somewhat further hampered by the nonspecific staining reaction of the often abundant interstitial collagen.
Summary of immunohistochemical staining with various antibodies in granular cell tumors of the rat distal female reproductive tract.
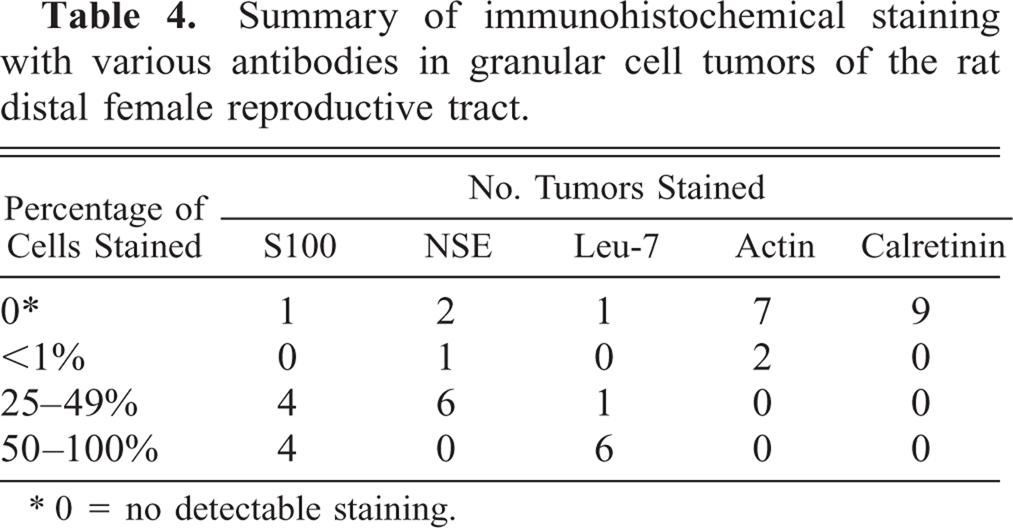
0 = no detectable staining.
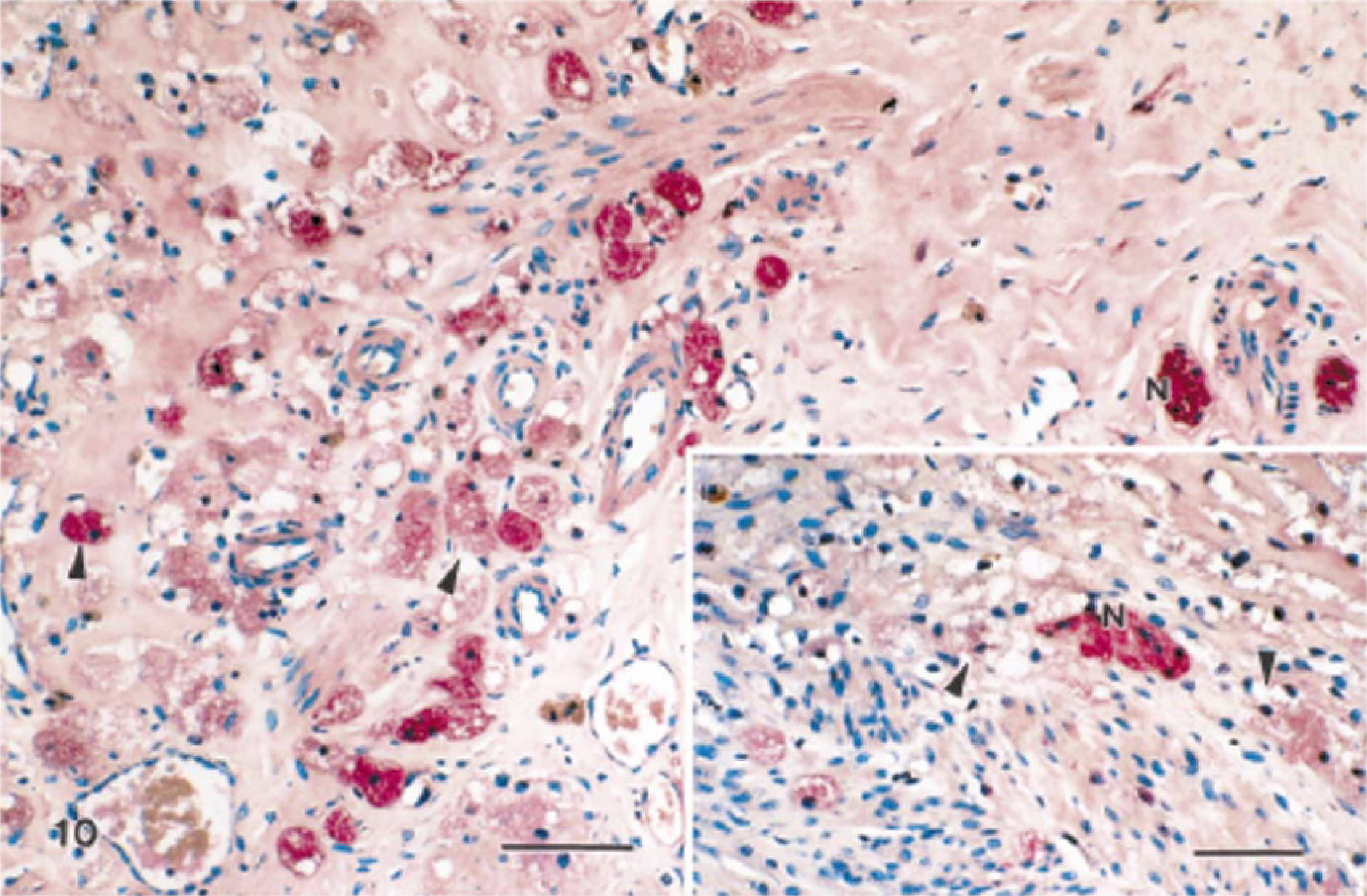
Granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Most granular cells exhibit strong immunoreactivity to S100 (arrowheads). Streptavidin–biotin complex, alkaline phosphatase substrate, new fuchsin chromogen, Mayer's hematoxylin counterstain. Bar = 50 μm. Inset: Nerves (N) are surrounded by poorly preserved granular cells that are S100 positive. Bar = 25 μm.
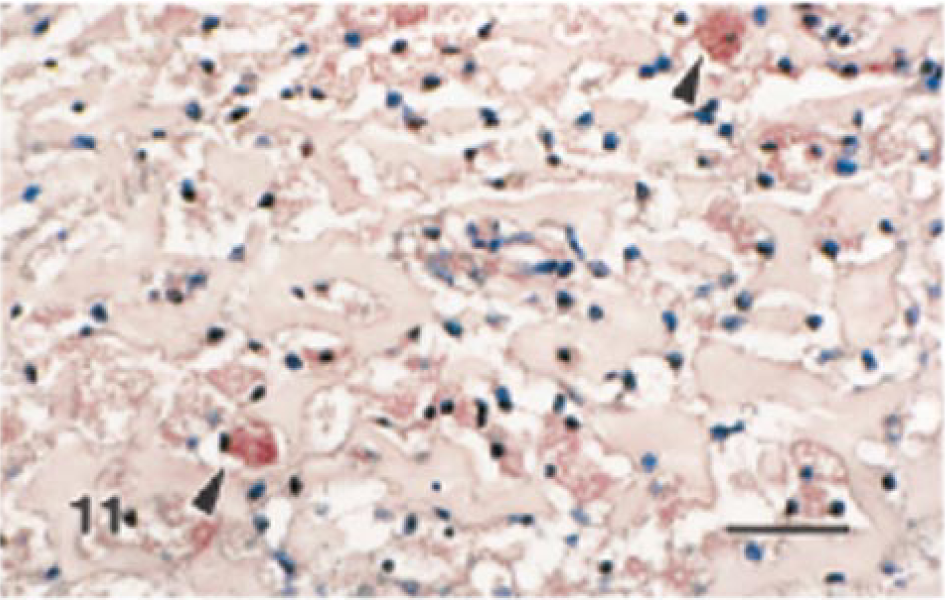
Granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Many granular cells exhibit immunoreactivity to NSE (arrowheads). Streptavidin–biotin complex, alkaline phosphatase substrate, new fuchsin chromogen, Mayer's hematoxylin counterstain. Bar = 50 μm.

Granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Many granular cells exhibit immunoreactivity to Leu-7 (arrowheads). Streptavidin–biotin complex, alkaline phosphatase substrate, new fuchsin chromogen, Mayer's hematoxylin counterstain. Bar = 50 μm.
Ultrastructurally, granular cells were slender, contained prominent cytoplasmic granules, had long cell processes, and were surrounded by abundant interstitial collagen (Fig. 13). The interstitium appeared to contain only typical collagen fibers and lacked fibrous long-spacing collagen or Luce bodies. The most prominent features were the many single membrane-bound bodies with electron-dense granules, which were considered to be lysosomes of various sizes and shapes containing granular, amorphous, or myelin figures (Fig. 14). The contents of granules did not resemble disintegrating cell organelles. The cell processes were often interwoven and had closely apposed plasma membranes. Widely scattered cell junctions could be identified in all specimens examined, but no basal laminae were visible. Typically, the cell junctions appeared as rows of three or four desmosome-like structures that contained an intercellular gap surrounded by fine filaments appearing as electron-dense plaques but that had no tonofilaments (Fig. 14, inset). Most of the cells had oval nuclei that contained finely dispersed chromatin and were slightly indented and occasionally contained a single nucleolus. Additional common ultrastructural features of the granular cellular cytoplasm were the slightly dilated rough endoplasmic reticulum, multivesicular bodies, and mitochondria.
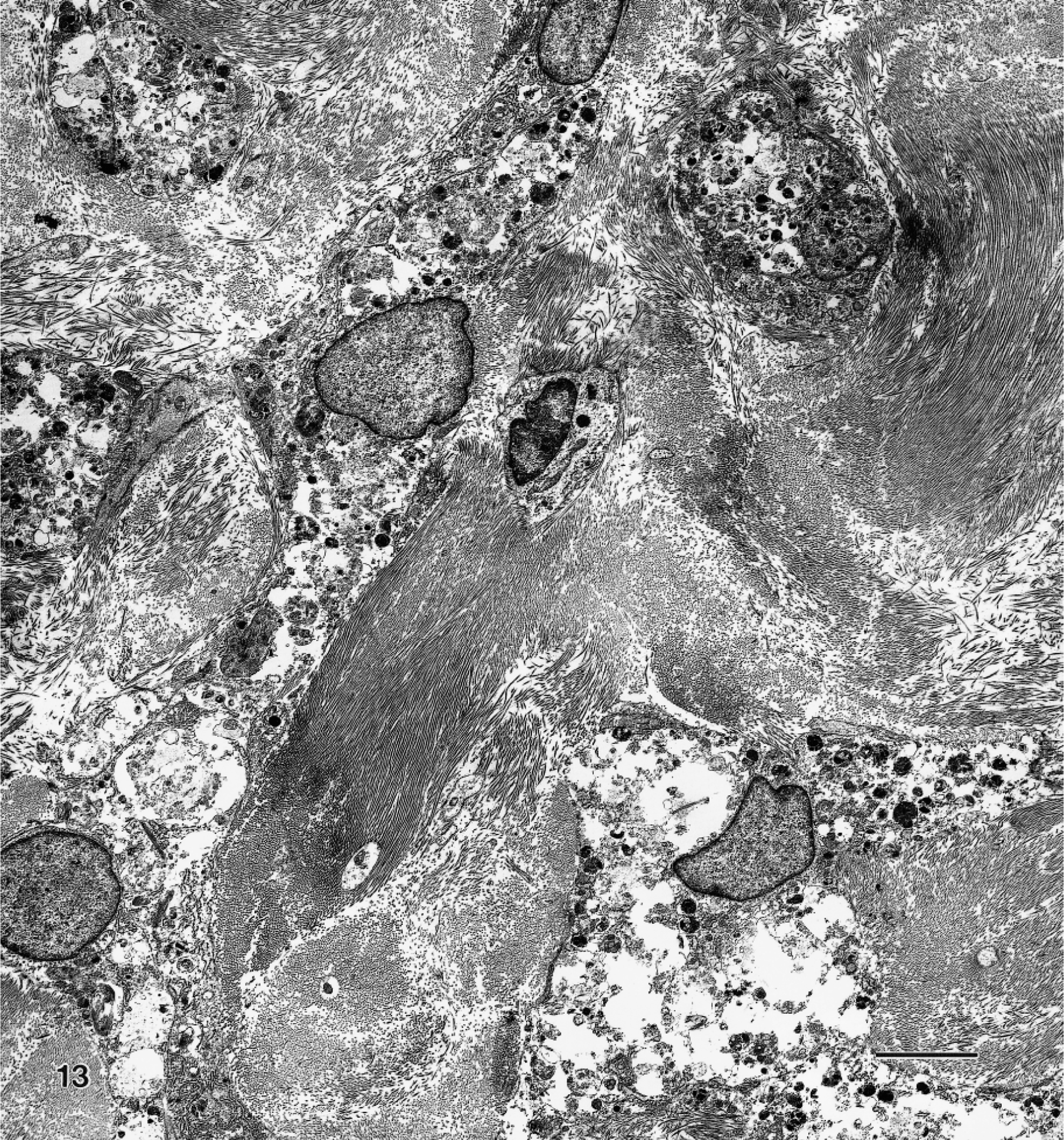
Transmission electron micrograph. Granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. Neoplastic cells with long slender cytoplasmic processes are surrounded by collagen. The electron-dense cytoplasmic granules are prominent. Uranyl acetate and lead citrate. Bar = 7 μm.
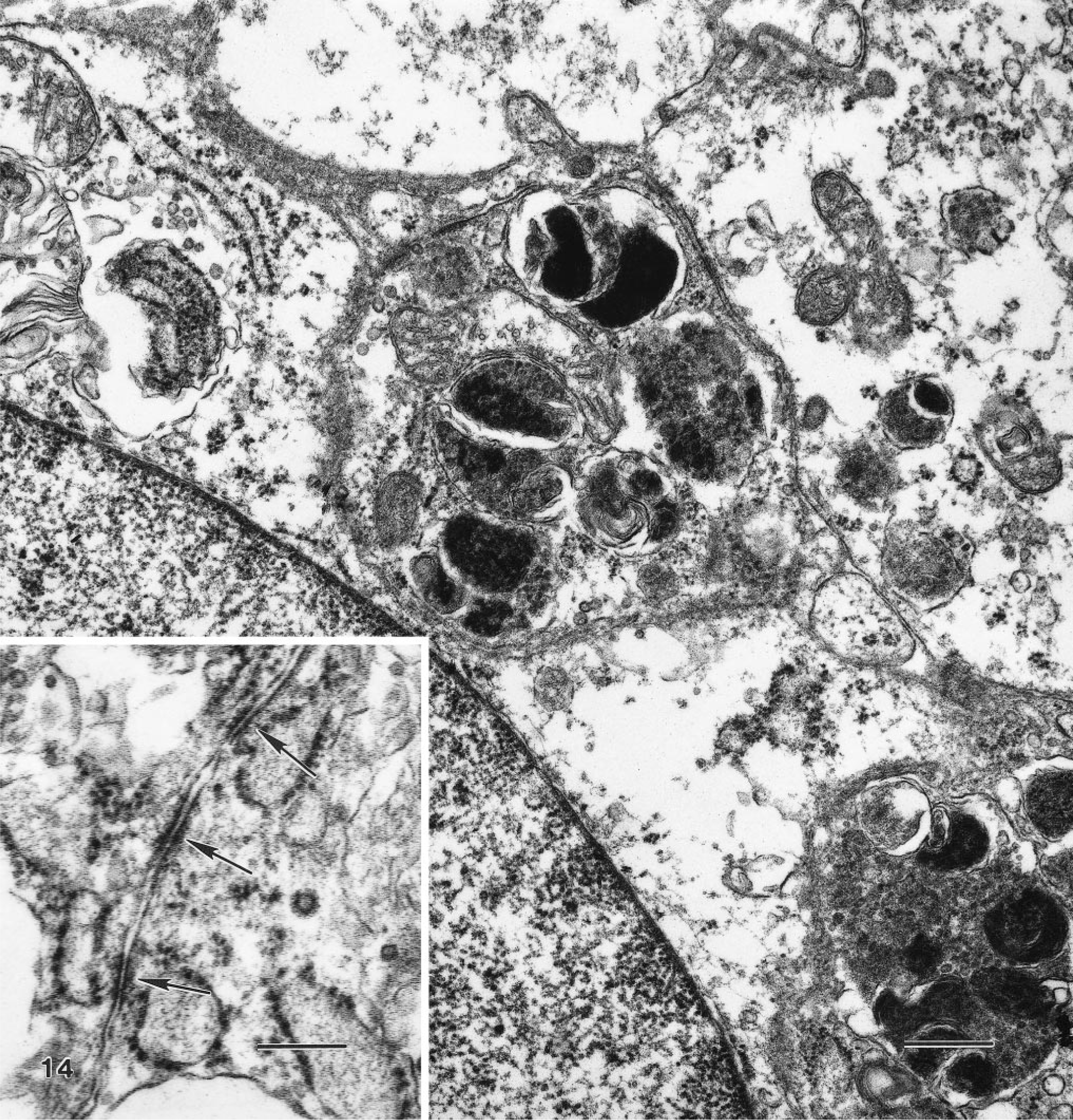
Transmission electron micrograph. Granular cell tumor, distal female reproductive tract; Sprague-Dawley rat. The contents of the electron-dense cytoplasmic granules cannot be identified as cytoplasmic organelles. A mitochondrion is between granules. Uranyl acetate and lead citrate. Bar = 0.5 μm. Inset: Several cell junctions (arrows) are present between cells. Bar = 0.25 μm.
Discussion
Granular cell tumors have been sporadically encountered in a wide range of species in many anatomic locations. 17 19 22 In humans, granular cell tumors are somewhat more common in women than in men and have been reported most often in the head and neck region, especially in the tongue. However, other sites, including breast, gastrointestinal tract, and vulva, also have been reported. 15 21 23 In animals, granular cell tumors are also known to occur in many anatomic locations in several species, with apparent species-specific distribution of anatomic sites. Granular cell tumors are the most common lung tumors in the horse, are not uncommon in the meninges and brain of the rat, and have been reported in the tongue of dogs. 12 17 22 Regardless of species and anatomic location, the majority of these tumors apparently have a homogeneous light microscopic appearance and a benign clinical course. 15 17 21 Malignancy was associated with local recurrence in a cat and recurrence or metastasis in humans. 15 17 21 In our study, we based the diagnosis of malignant granular cell tumor on morphologic features that were reported to be associated with malignant behavior in humans, although these rat tumors behaved in the same way as their benign counterparts. 15 21 Some of the features in humans that were associated with malignancy were size, necrosis, pleomorphism, and mitotic activity. These tumors were originally reported as granular cell myoblastomas and were believed to arise from skeletal muscle; subsequently, histiocytic, fibroblastic, and Schwann cell origins, among others were proposed. 6 13
Ultrastructurally and immunohistochemically, granular cell tumors appear to be more heterogeneous than they do when examined using conventional microscopic methods, although they share key features. The granularity of the cytoplasm is due to abundant lysosomes in all granular cell tumors. 4 6 22 24 Other ultrastructural features, such as angulate body cells, basal lamina, and desmosomes, may or may not be present. Ultrastructurally, the four granular cell lesions of the present report contained many lysosomes but no angulate body cells, and in these features they were similar to the reported single lesion from the reproductive tract of a Wistar rat. 4 Widely scattered and poorly developed cell junctions were reported in this Wistar rat but were not described in detail, whereas in the Sprague-Dawley rats of the present study, all specimens contained desmosome-like structures. Poorly formed desmosome-like junctions have been described in granular cell tumors of humans. 16 None of the rat tumors whose ultrastructural features have been published, including those of the present report, contained basal lamina, regardless of whether they were meningeal or from the female reproductive tract. 4 12
Human granular cell tumors have been characterized in more detail immunohistochemically than have those of animals. Most human and animal granular cell tumors display immunoreactivity to S100. 23 Many human granular cell tumors are also positive for NSE and Leu-7. 3 11 Although these markers are often viewed as nonspecific and are either widely distributed or have nonspecific immunoreactivity with tissues such as muscle, positive immunoreactivity to several of these markers supports the proposed neuroectodermal or Schwann cell origin of granular cells. Recently, three human granular cell tumors were positive with calretinin, a calcium-binding protein that is related to S100. 5 In the present study of granular cell lesions in the rat reproductive tract, S100, NSE, and Leu-7 all produced positive reactions, but calretinin did not. This finding does not contradict the proposed cell of origin but rather suggests that the cells expressed different epitopes or that the calretinin immunoreactivity in the granular cells of these rats was below the detection limit of our methods. Similar species differences were found earlier, when nerves and neurons of rats had positive immunoreactivity for calretinin, but rat normal or neoplastic mesothelial cells, unlike those of humans, were negative. 10 Such heterogeneity is further suggested by occasional immunoreactivity of human tumors to α-1 antichymotrypsin and α-1 antitrypsin; immunoreactivity to α-1 antitrypsin was also demonstrated in canine tumors. 13 17
The immunohistochemical and ultrastructural characterization of the series of granular cell lesions from the distal reproductive tract of female rats did not result in new insights as to the pathogenesis of the lesion but did provide additional data in support of these lesions as similar to those of humans and probably of neural origin. The frequent finding of nerves within or around the many granular cell lesions is circumstancial evidence of “guilt by association” for a neural origin.
Because there is no known counterpart to granular cells in normal tissues, hyperplasia as a preneoplastic lesion does not occur. Because of the extremely small size of many of the lesions in the rat reproductive tract, only those that were clearly space-occupying were diagnosed as tumors, whereas smaller, nonexpansile lesions were identified as aggregates. Based on our limited experience with female rats from 13- and 26-week studies and the review of more than 540 female rats from nine carcinogenicity studies, it appears that scattered granular cells occur in the distal female reproductive tract with increasing frequency as age increases. It was suggested that small groups of cells in the mouse cervix might become granular cell tumors after certain stimuli, such as estrogen treatment. 8 Such small clusters of cells similar to granular cells have been identified only in the mouse and rat female reproductive tract. In mice, a modest incidence of granular cell tumors was associated with treatment with diethylstilbestrol or 17β estradiol, and these mice had other evidence of estrogenic effect. 7 8 There are no published data for mice on the incidence of spontaneous cervical granular cell tumors, but these tumors are known to occur. 8 There was no evidence of an estrogenic effect in the rats of our study; however, we observed remarkable reduction in the incidence of granular cell aggregates and tumors following treatment with aromatase inhibitors (unpublished). An unexpected finding was the apparent high incidence of granular cell lesions in a single longitudinal section of the cervix/vagina. The lack of familiarity with the lesion and the infrequent reports are probably due to the very small, typically microscopic, size of these lesions. Moreover, in toxicity and carcinogenicity studies, the cervix/vagina is either not routinely evaluated microscopically or is represented by only a single cross section. 1 Granular cell lesions are probably even more common than the 23% incidence reported here; no step or serial sections were examined. In addition to sampling problems, underreporting was further compounded by the innocuous appearance of many of the lesions, making them difficult to distinguish from the surrounding tissues. The chances for correct diagnosis in the rat are improved when the predilection sites of the lesion, i.e., the serosal surface and supportive ligaments of the cervix/vagina, are carefully evaluated.
Footnotes
Acknowledgements
We thank the following pathologists for reviewing selected slides and participating in discussions about granular cells: C. Fletcher (Brigham and Women's Hospital, Harvard Medical School, Boston, MA), D. Goodman (Covance, Vienna, VA), R. Hess (Dornatch, Switzerland), F. McConnell (Consulting Pathology Services, Penn Valley, CA), D. Prentice (PCS Consultants, Ettingen, Switzerland), and G. Williams (Department of Pathology, New York Medical College, Valhalla, NY). We also thank G. Argentieri, M. Arnella-Venezia, K. Killary, and J. Zimmerman for technical assistance.
