Abstract
The phylum Microspora contains a diverse group of single-celled, obligate intracellular protozoa sharing a unique organelle, the polar filament, and parasitizing a wide variety of invertebrate and vertebrate animals, including insects, fish, birds, and mammals. Encephalitozoon cuniculi is the classic microsporidial parasite of mammals, and encephalitozoonosis in rabbits and rodents has been and continues to be recognized as a confounding variable in animal-based biomedical research. Although contemporary research colonies are screened for infection with this parasite, E. cuniculi remains a cause of morbidity and mortality in pet and conventionally raised rabbits. In addition, E. cuniculi is a potential pathogen of immature domestic dogs and farm-raised foxes. The recent discovery and identification of Encephalitozoon intestinalis, Encephalitozoon hellem, and Enterocytozoon bieneusi, in addition to E. cuniculi, as opportunistic pathogens of humans have renewed interest in the Microspora. Veterinary pathologists, trained in the comparative anatomy of multiple animal species and infectious disease processes, are in a unique position to contribute to the diagnosis and knowledge of the pathogenesis of these parasitic diseases. This review article covers the life cycle, ultrastructure, and biology of mammalian microsporaidia and the clinical disease and lesions seen in laboratory and domestic animals, particularly as they relate to Encephalitozoon species. Human microsporidial disease and animal models of human infection are also addressed. Often thought of as rabbit pathogens of historical importance, E. cuniculi and the related mammalian microsporidia are emerging as significant opportunistic pathogens of immunocompromised individuals.
Encephalitozoon cuniculi has had a colorful and confounding history in animal-based biomedical research, even after its initial identification as the etiologic agent of “infectious motor paralysis” in rabbits by Wright and Craighead in 1922. 152 The use of rodents or rabbits endemically infected with E. cuniculi with for xenodiagnoses and experimental models of human disease resulted in the clinical manifestations and histologic lesions of encephalitozoonosis being mistaken for poliomyelitis and human syphilis. 17 Endemic encephalitozoonosis in research animals has also been mistaken for animal models of herpes simplex and “human epidemic encephalitis,” B-virus infection (cercopithecine herpesvirus 1), toxoplasmosis, “human viral hepatitis,” and multiple sclerosis. 52 , 59 , 64 , 65 , 92 , 113 , 140 Although less well documented, E. cuniculi has also been mistaken for or implicated as the agent of rabies, scrub typhus, psittacosis, murine leukemia, experimental allergic encephalomyelitis, and chemical carcinogenesis. 124 In addition, E. cuniculi has been transmitted through rodent tumor lines, altering the biologic behavior of certain experimental murine tumors. 5 , 104
E. cuniculi is a member of the phylum Microspora, which includes single-celled, spore-forming, obligate intracellular parasites with a wide host distribution. 21 Microsporidia are traditionally considered protozoal organisms, although there is some genetic and molecular evidence to suggest a closer phylogenetic relationship to fungi, including the possession of a mitochondrial heat shock protein more closely related to that of fungi; α- and β-tubulins with compositions more closely resembling those of fungal tubulins; and the presence of chitin and trehalose, which are also components of fungi. 69 E. cuniculi is the most extensively studied mammalian microsporidium, and spontaneous infections have been documented in rabbits, mice, rats, muskrats, guinea pigs, hamsters, ground shrews, goats, sheep, pigs, horses, domestic dogs, wild and captive foxes, domestic cats, a variety of exotic carnivores, and nonhuman primates. 124
Interest in mammalian microsporidiosis was renewed with the identification of Enterocytozoon bieneusi as the cause of chronic diarrhea and wasting in an immunocompromised person that was positive for the human immunodeficiency virus (HIV). 35 Identification and molecular differentiation of Encephalitozoon intestinalis (originally named Septata intestinalis), Encephalitozoon hellem, and E. cuniculi in this immunocompromised population followed. 18 , 38 , 51 Vittaforma corneum, Nosema spp., Pleistophora spp., and Trachipleistophora spp. have also been diagnosed in humans, although less frequently. 24 , 61 , 128 , 138 While Enterocytozoon bieneusi remains the most frequently diagnosed microsporidial pathogen of humans, it has yet to be cultivated in vitro and, unlike the Encephalitozoon spp., lacks convenient rodent models of infection. 97 , 134 In addition, increasing reports of encephalitozoonosis in pet animals suggests that Encephalitozoon spp. may be zoonotic. 33 , 43 , 58 , 85 For these reasons, encephalitozoonosis will continue to be of interest to research-oriented veterinary pathologists and anatomic veterinary pathologists. This review article covers the life cycle of mammalian microsporidia and the basic ultrastructural and biological features shared by all members of the Microspora, and will discuss the clinical disease and lesions seen in spontaneously and experimentally infected laboratory and domestic animals, particularly as they relate to Encephalitozoon spp. Lastly, human microsporidial disease and animal models of human infection will be addressed.
Mammalian Microspora: Life Cycle, Ultrastructure, and Cellular Biology
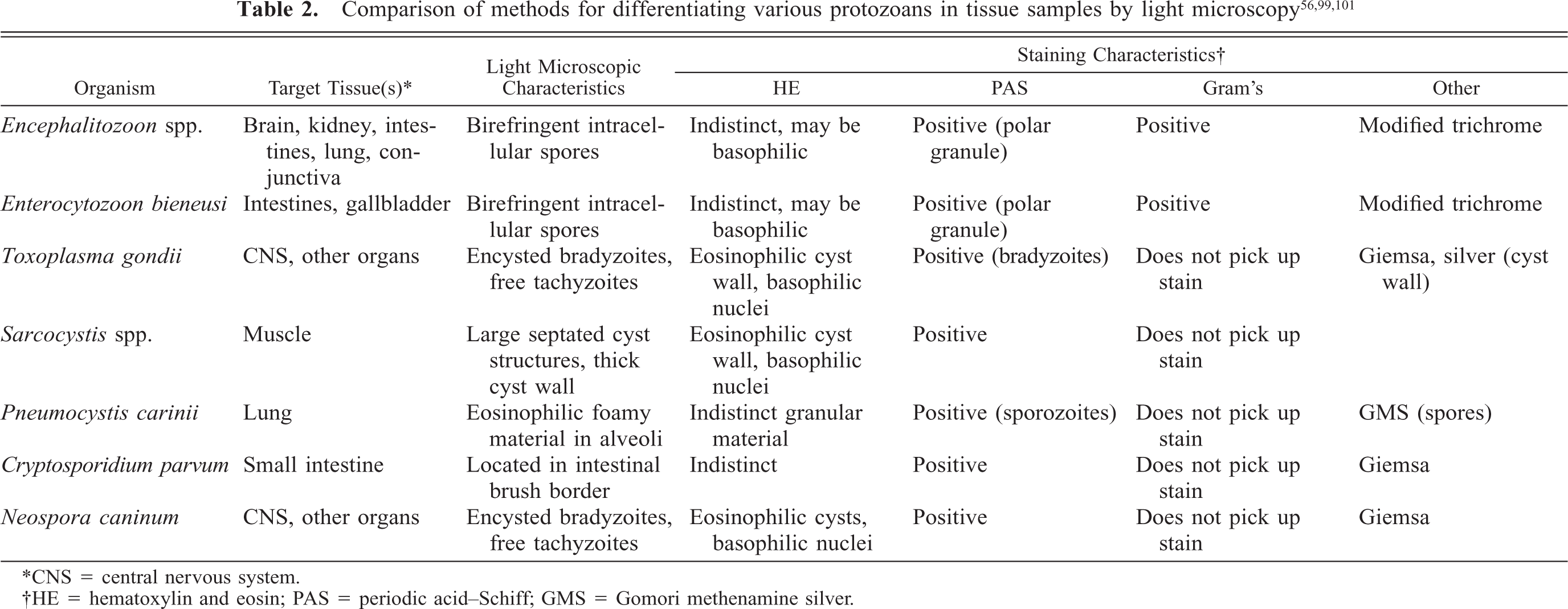
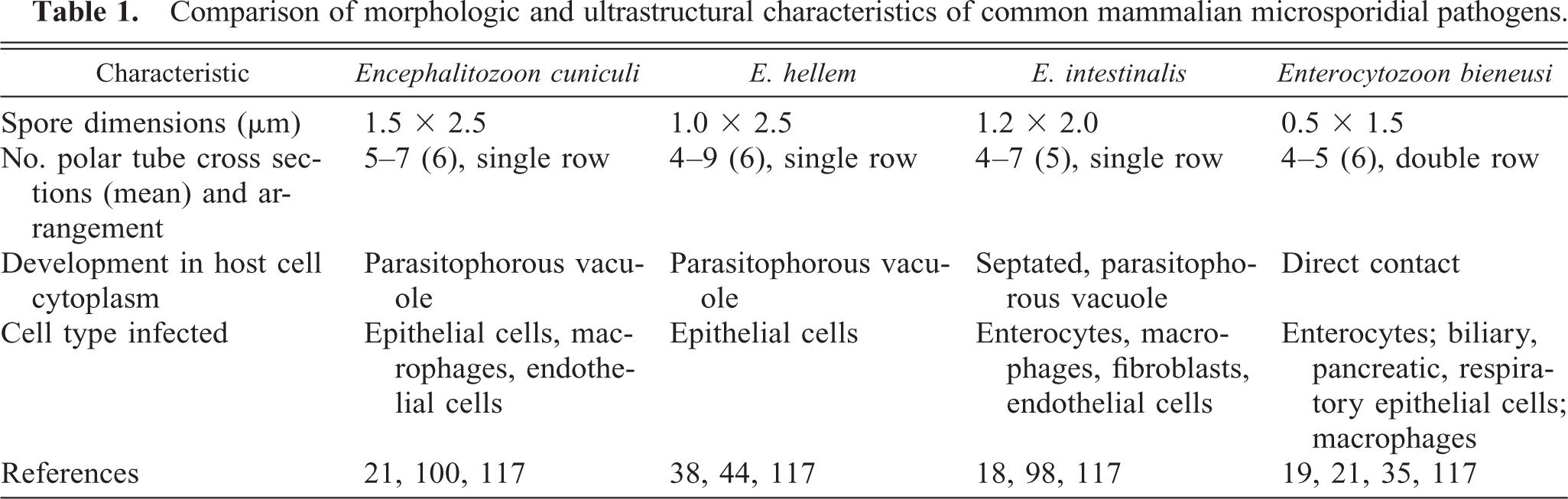
All microsporidia have an obligate intracellular life cycle, existing as environmentally resistant spores outside the host. 21 Their life cycle in mammals is postulated to be simple and direct, and most infections are acquired through ingestion or inhalation, transplacentally, or rarely, through trauma to epithelium (Fig. 1). 41 The defining criterion for inclusion in the phylum Microspora is possession of a unique organelle known as the polar filament (also referred to as a polar tube) found coiled within the spore. 21 Other ultrastructural features common to microsporidia include a corrugated, proteinaceous exospore (spore wall), a chitinous, radiolucent endospore, an anchoring disc at the anterior pole, and an electron-lucent posterior vacuole (Fig. 2). The spore nucleus can be difficult to discern from the remainder of the electron-dense sporoplasm. Stimulation by an uncharacterized environmental signal causes extrusion of the polar filament from the spore and “injection” of the infectious sporoplasm into the host cell. 148 Although polar tube extrusion is postulated to be the primary mechanism by which all microsporidia establish intracellular infection, in vitro cultivation of Encephalitozoon spp. suggests that phagocytosis of spores by host cells may also establish patent intracellular infection. 21 , 100 Once inside the host cell, the deposited sporoplasm divides to yield proliferative forms (also known as meronts) with simple plasma membranes, small prokaryotic-like ribosomes, and sparse organelles. As the proliferative forms differentiate into sporoblasts and then sporonts, a dense spore wall is laid down over the plasma membrane, the plasma membrane uniformly thickens, spore organelles (posterior vacuole, coiled polar tube, anchoring disk) are formed, and the cytoplasm becomes increasingly electron dense. Depending on the genera of mammalian microsporidia, the developing spores either become packaged within a parasitophorous vacuole (i.e., Encephalitozoon spp.) or remain in direct contact with the cytoplasm (i.e., Enterocytozoon bieneusi, Nosema spp.). There is a distinct lack of mitochondria, peroxisomes, and classical stacked Golgi membranes in all stages of parasite development. A progression of developing parasite forms is typically found in each infected cell (Fig. 3), and the appearance of an electron-lucent endospore, a clear posterior vacuole, and cross-sections of the coiled polar filament characterize mature spores. Enlargement of the parasitophorous vacuoles and/or host cell with parasites results in host cell rupture and release of spores into extracellular spaces. Infection is disseminated by direct extension of spores into surrounding cells, by introduction into the vascular system, or by both methods. 41 Parasites are difficult to identify in tissue sections by light microscopy with hematoxylin and eosin stains but may appear as clusters of refractile intracytoplasmic structures. Parasites are gram positive and rod shaped and may appear to have a clear vacuole at one pole. Although not appreciable at the light microscopic level, the presence or absence of the parasitophorous vacuole and the polar filament cross-sectional number and arrangement, in addition to spore size, aid in differentiating parasite genera at the electron microscopic level (Table 1). 18
Comparison of morphologic and ultrastructural characteristics of common mammalian microsporidial pathogens.
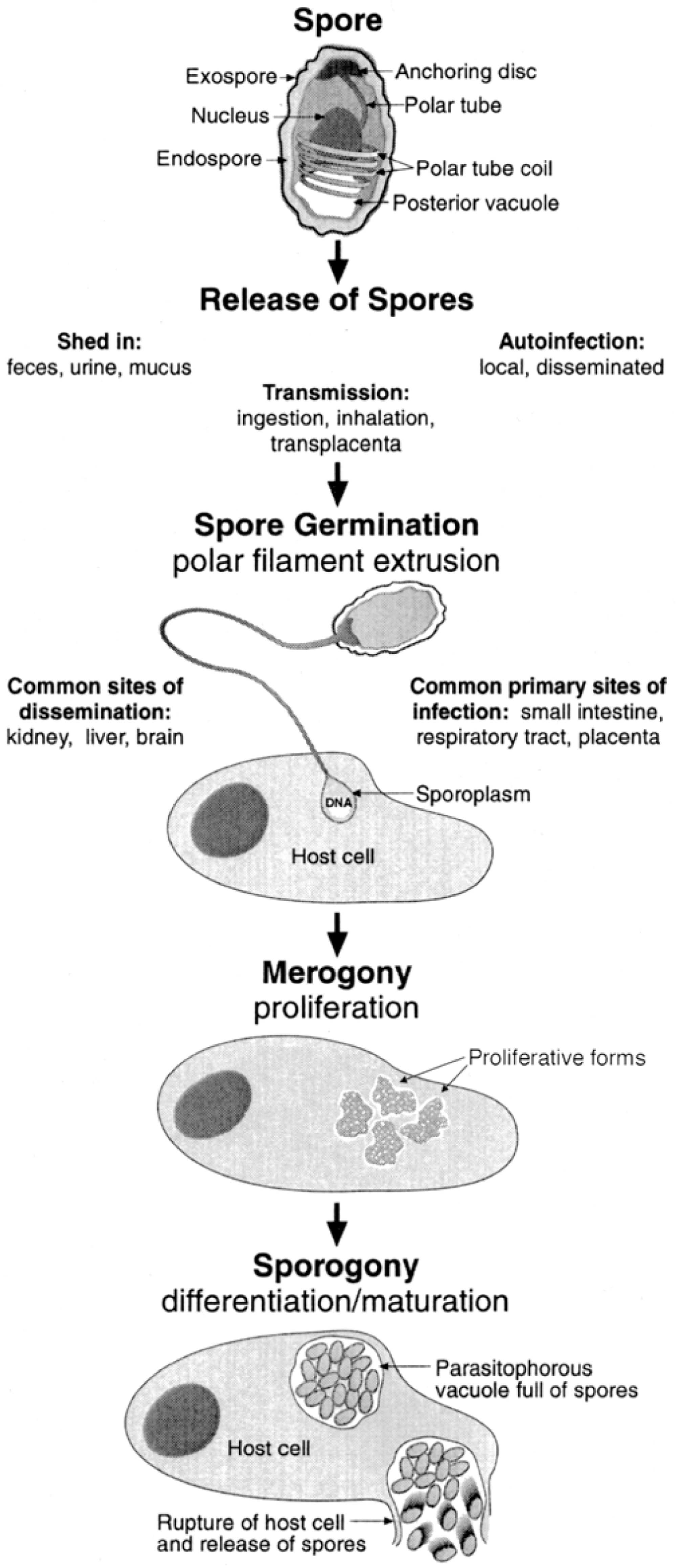
Ultrastructural features common to most mammalian microsporidia, and simplified life cycle of Encephalitozoon spp.
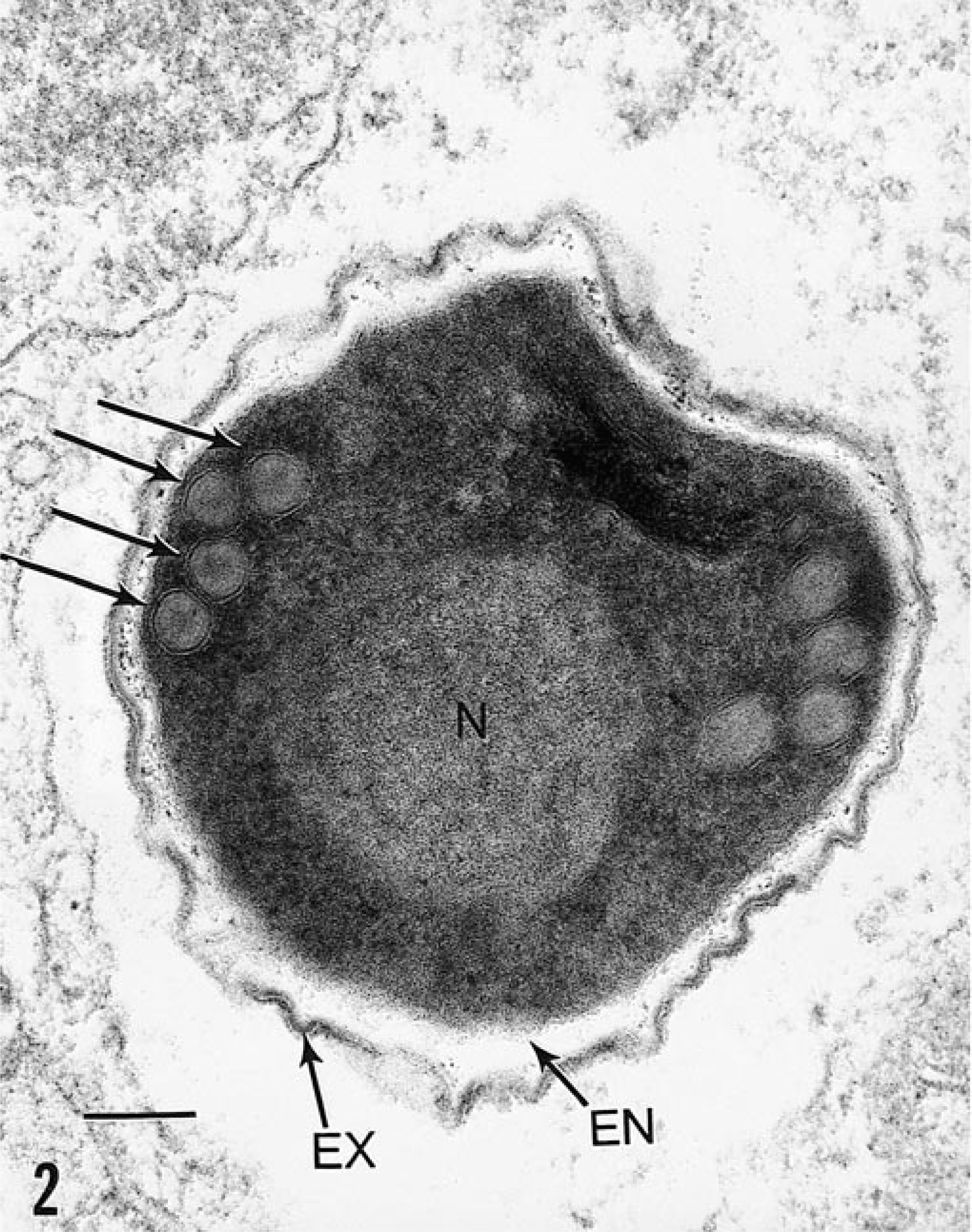
Fetal intestine; rabbit. Tissue was implanted into athymic nude mouse and experimentally inoculated with E. hellem. 143 Ultrastructural features of the spore include electron-dense exospore (EX), electron-lucent endospore (EN), a faint spore nucleus (N), and four cross-sections of the polar tubule (arrows) typical of E. hellem. Bar = 0.1 µm.
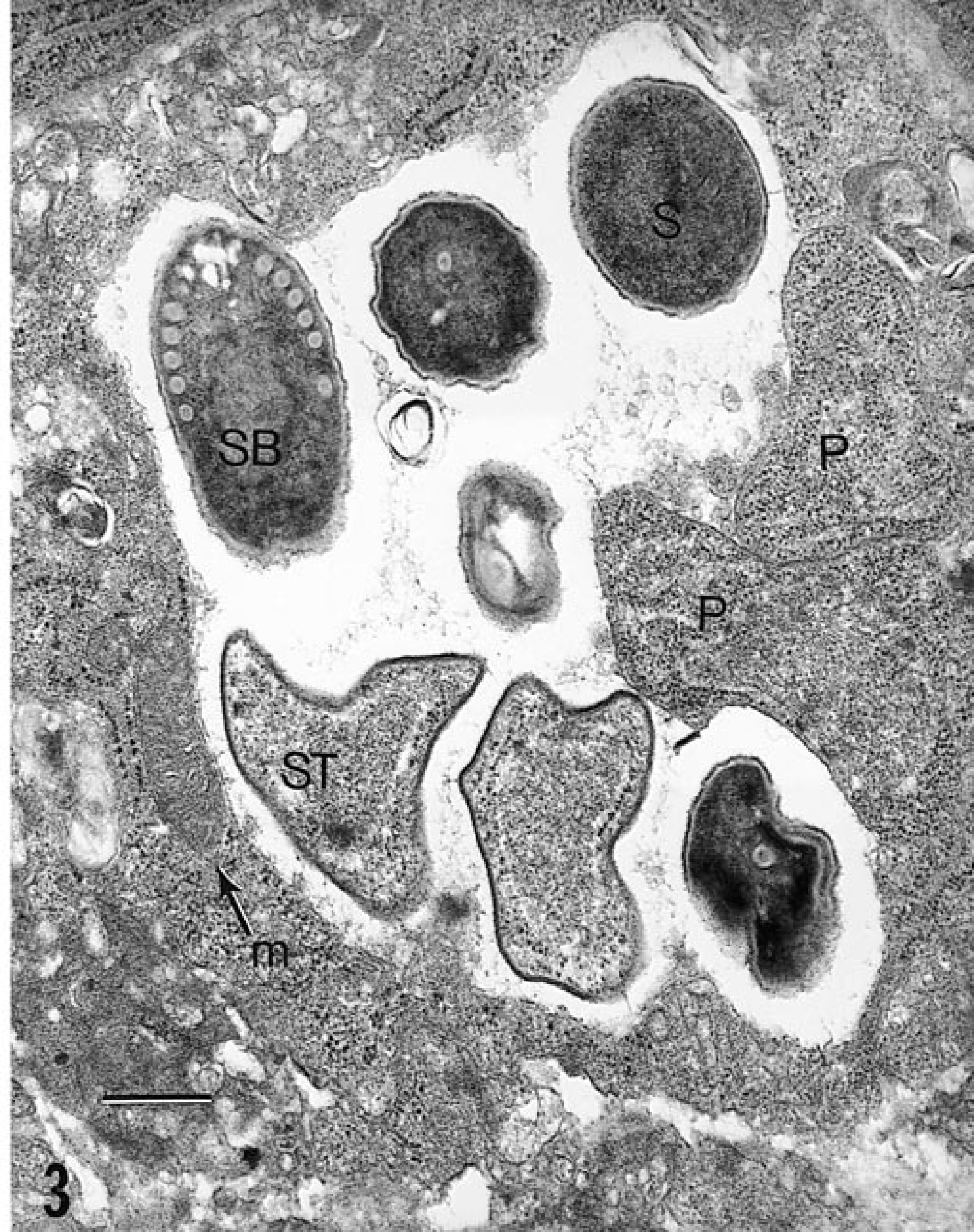
Fetal intestine; rabbit. Tissue was implanted into athymic nude mouse and experimentally inoculated with E. hellem. 143 Asynchronously developing parasite forms are present within a parasitophorous vacuole: proliferative forms (P), sporont (ST), sporoblast (SB), mature spore (S). Note host cell mitochondrion (m). Bar = 0.6 µm.
Investigation into the mechanisms of microsporidial infection has focused on the relatively unique polar filament. Extrusion of this filament, with subsequent injection of the infectious sporoplasm into a potential host cell, has been hypothesized to be due to an abrupt increase in osmotic pressure, possibly as a result of the generation of glucose metabolites from hydrolysis of stores of trehalose within spore vacuoles. 135 , 136 Trehalose is a nonreducing disaccharide of glucose used as an energy source in insects, nematodes, and fungi but not in mammals and may have potential as a metabolic target to be exploited for control of microsporidial infection. 9 , 20 , 66 In vitro polar filament extrusion has been enhanced by the presence of calcium ions in the cell medium and by the addition of H2O2 to the cultures and has been inhibited with cytochalasin D, demecolcine, nifedipine, and itraconazole, suggesting that calcium-mediated channels, reactive oxygen species, and/or cytoskeletal elements (i.e., microtubules, microfilaments) are involved in this process. 78 , 79 In addition, the in vitro and in vivo efficacy of albendazole, a microtubule inhibitor, in preventing infections due to Encephalitozoon spp. lends indirect evidence to support the role of microtubules in the extrusion process. 37 , 74 , 110 Other areas of ongoing investigation have focused on the protein composition and antigenicity of the polar filament. 31 , 32 Cell signals involved in the initiation of polar tube extrusion are unknown.
Molecular analysis of microsporidia has also revealed several interesting characteristics. The presence of genes in E. cuniculi coding for a mitochondrially expressed heat shock protein suggests that microsporidia may have evolved from a mitochondria-containing ancestor that subsequently lost this organelle through evolutionary or selective pressure. 105 The significance and function of this heat shock protein is not known. The nuclear genome of E. cuniculi is quite small, 2.9 × 106 nucleotide base pairs (as compared with 4.7 × 106 base pairs in Escherchia coli and 6.0 × 109 base pairs in human somatic cells). 11 In addition, intervening sequences (or introns) in microsporidial DNA are relatively rare. 149 Thus, microsporidia appear to encode information for infection, replication, and dissemination in an extremely efficient manner, suggesting that these parasites are highly adapted to survival in their hosts.
Diagnosis of Mammalian Microsporidiosis
The approach for diagnosing microsporidiosis in mammals will depend on the origin, use, and immune status of the animals in question. Laboratory colonies of immunocompetent rabbits and rodents are routinely screened for infection with E. cuniculi by serology; histopathology is used with selected animals to confirm positive enzyme-linked immunosorbent assays or indirect fluorescent antibody tests. Dogs suspected of being infected with E. cuniculi may also be tested initially by serology. 123 Immunodeficient animals do not mount reliable antibody responses, and microsporidial infections in these animals must be diagnosed by other methods. For animals displaying clinical microsporidial disease (e.g., seizures), examination of urine, feces, or cerebrospinal fluid may yield an antemortem diagnosis of encephalitozoonosis. These clinical samples are often screened with a calcofluor white stain for chitin, and Encephalitozoon infection is confirmed with modified trichrome stain, immunofluorescent antibodies, polymerase chain reaction (PCR), transmission electron microscopy, or some combination of methods. 23 , 39 , 81 , 93 , 145 Light microscopic examination of tissue samples and the use of special stains will aid the pathologist in detecting infection and differentiating microsporidial infection from other common protozoal infections of animals (Table 2). However, even with the use of appropriate stains light microscopy is not sufficient for species-specific identification of microsporidia, particularly in humans, who may be infected with a number of different microsporidia. PCR tests have been developed and validated for Encephalitozoon spp. and Enterocytozoon bieneusi infections in humans and animals, and although these tests are not yet routinely available to clinical veterinary diagnostic laboratories, they are specific and sensitive enough for diagnosis and identification of microsporidial infections. 14 , 68 , 83
∗CNS = central nervous system.
†HE = hematoxylin and eosin; PAS = periodic acid-Schiff; GMS = Gomori methenamine silver.
Natural and Experimental Microsporidiosis in Laboratory and Domestic Animals
Infection in rabbits
E. cuniculi was first identified in the brains and kidneys of rabbits exhibiting lethargy, tremors, and local or generalized paresis. 152 The organisms were shed in urine, and the authors speculated on their transmission and protozoal nature of these organisms. It has been suggested that E. cuniculi may spread transplacentally in rabbits. 26 Study of the pathogenesis of encephalitozoonosis in rabbits was initially hampered by the lack of non-subclinically infected rabbits and an in vitro assay to cultivate E. cuniculi. 121 These limitations have been overcome, and it has been shown that experimental infection in rabbits can be established by a variety of routes (intravenous, oral, intratracheal, rectal); serum antibodies develop by day 21 postinoculation; spores are excreted in the urine for approximately 63 days; and early infection (day 30 postinoculation) is largely confined to the lung, kidney, and liver, while later infection (day 98 postinoculation) is most commonly localized to heart, brain, and kidney. 27 , 53 , 150 The presence or absence of encephalitozoon spores in rabbit fluids or tissues may yield useful epidemiologic clues for the clinician investigating a disease outbreak.
In naturally infected rabbits, E. cuniculi is primarily acquired through ingestion of infected urine. Occasionally, an infected rabbit will manifest neurologic signs of ataxia, opisthotonos, torticollis, hyperaesthesia, or paralysis; other less specific signs may include weight loss and failure to thrive. 77 , 99 , 111 Gross lesions are usually absent in infected rabbits, although chronic infection may manifest as focal, irregular, depressed areas in the renal cortex. 50 Histologically, lesions are most frequently identified in the brain and kidney, consisting of focal nonsuppurative meningoencephalitis with astrogliosis and perivascular lymphocytic infiltration and focal to segmental lymphocytic-plasmacytic interstitial nephritis with variable amounts of fibrosis; parasites may not be evident in areas of inflammation. 25 Other sections of the same tissue may contain intracellular clusters of parasites with no associated inflammatory response (Fig. 4,5). Rarely, parasites are identified within the lens, lung, liver, and cardiac tissues of infected rabbits. 27 , 131 Parasites are best demonstrated with Gram's stain as gram-positive rod-shaped organisms free within a focus of inflammation or contained within intracellular parasitophorous vacuoles of endothelial cells, epithelial cells, or macrophages. Differential diagnoses for encephalitozoonosis in rabbits include central nervous system infection with Pasteurella multocida, Listeria monocytogenes, and Toxoplasma gondii. 52 , 77 , 103 , 144
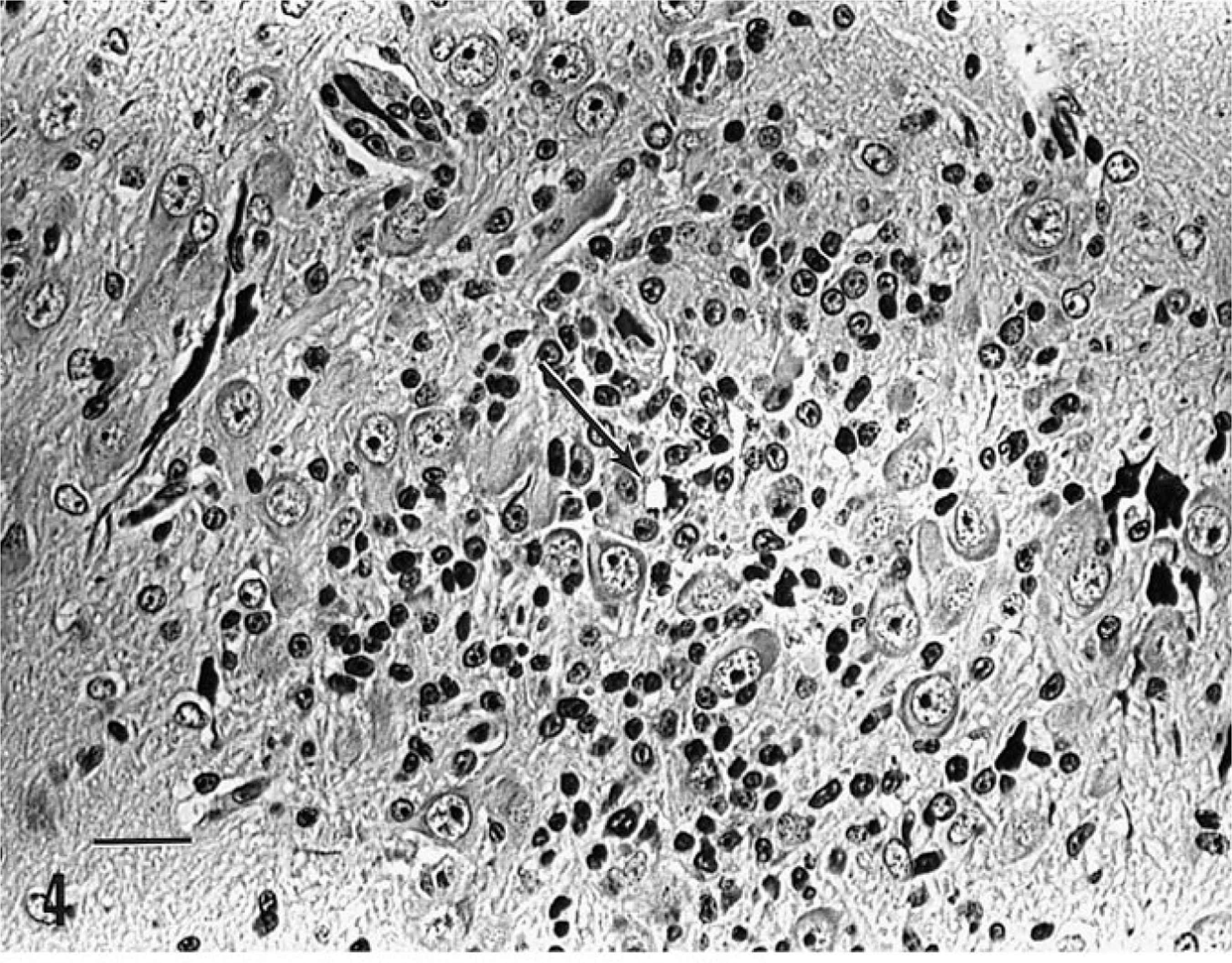
Cerebellum; domestic rabbit spontaneously infected with E. cuniculi. A focus of mononuclear cell infiltration and gliosis is present within the Purkinje cell layer. A small focus of intracellular parasites is present (arrow). Gram's stain. Bar = 50 µm.
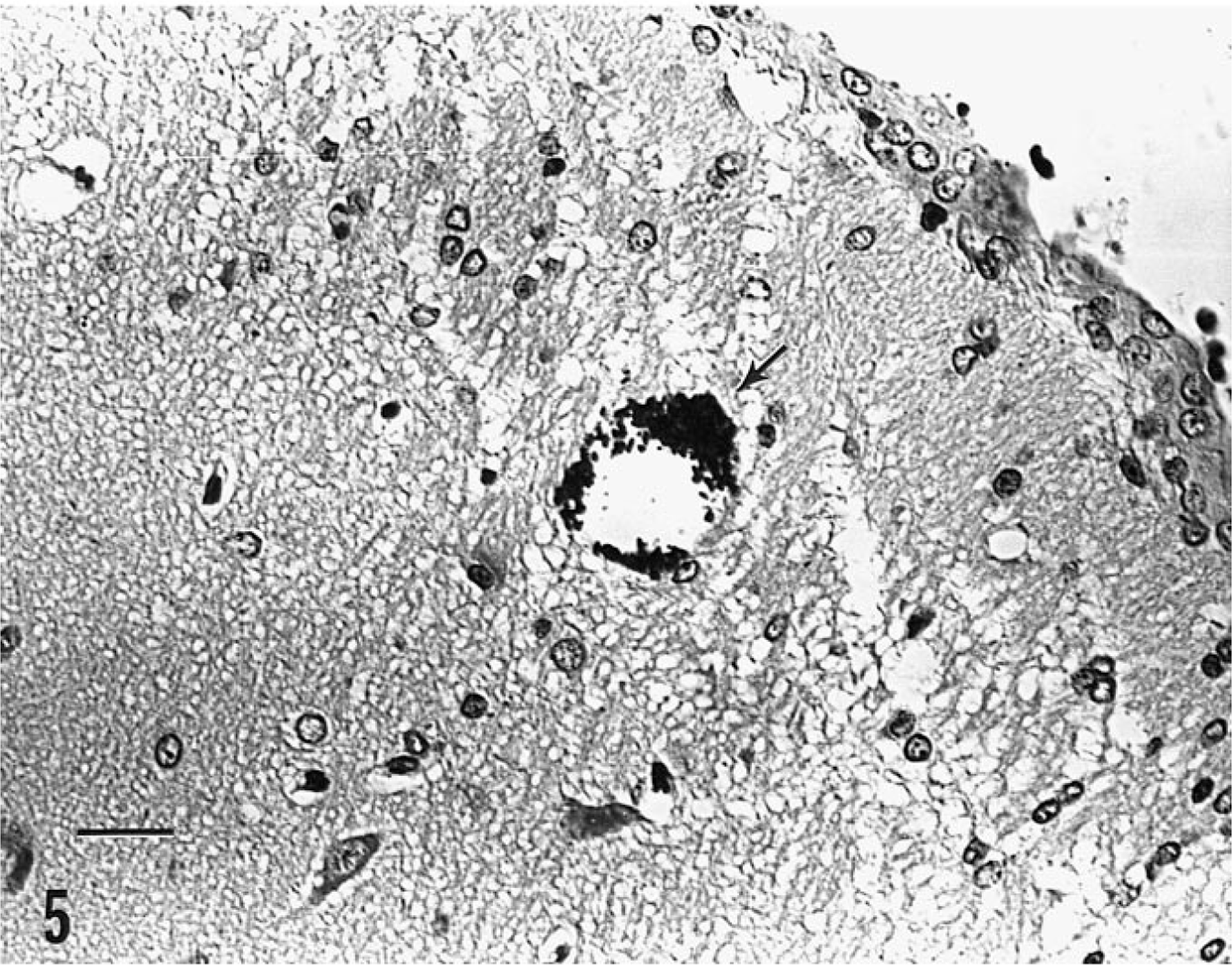
Cerebrum and meninges; domestic rabbit spontaneously infected with E. cuniculi. Parasites are present within an intracellular parasitophorous vacuole, with no associated inflammation (arrow); meninges are thickened. Gram's stain. Bar = 50 µm.
E. cuniculi is the only microsporidial organism known to cause spontaneous disease in rabbits. Experimental infections with E. intestinalis, E. hellem, and Enterocytozoon bieneusi in rabbits have not been reported.
Infection in mice
E. cuniculi infection of immunocompetent mice, as in rabbits, is usually subclinical. 55 , 102 Histologic lesions of mononuclear inflammatory foci in the liver, lungs, and brain have been attributed to subclinical E. cuniculi infection. 63 Experimental infection in athymic nude or severe combined immunodeficient (SCID) mice however, is typically widespread, with parasites disseminating by direct extension or hematogenously, depending on the route of inoculation. Clinical signs include lethargy, wasting, and death. Gross necropsy lesions include hepatosplenomegaly and multifocal miliary white spots throughout the liver, spleen, lung, and cardiac tissue. 42 , 60 , 76 Infection of the liver, spleen, pancreas, lungs, heart, kidneys, brain, peritoneum, and pleura is characterized by random, multifocal miliary granulomas with various amounts of cell debris and suppurative necrosis. 42 , 76 The gram-positive parasites may be found free within foci of inflammation or intracellularly in macrophages, epithelium, or endothelium. Mice inoculated orally may have focal ulcerative intestinal mucosal lesions that often extend to the submucosa and tunica muscularis. 76 Differential diagnoses for E. cuniculi infection in mice include infection with Clostridium piliforme (Tyzzer's disease), Corynebacterium kutscheri (pseudotuberculosis), Pseudomonas aeruginosa, salmonellosis, mouse hepatitis virus, ectromelia virus (mousepox), and hematopoietic/lymphoreticular neoplasms. 3 , 36 , 46 , 54 , 57 , 62 , 127
In addition to E. cuniculi infection, experimental E. intestinalis and E. hellem, but not Enterocytozoon bieneusi, infections have been documented in immunocompromised mice. Intestinal and systemic E. intestinalis infections have been documented in several strains of immunosuppressed mice. 1 , 49 , 143 E. hellem administered intraperitoneally to athymic nude mice results in similar gross and histologic lesions as seen in E. cuniculi-infected immunoincompetent mice. 42 Spontaneous infections with E. intestinalis, E. hellem, or Enterocytozoon bieneusi have not been described in mice.
Infection in guinea pigs
As with immunocompetent mice, E. cuniculi infection in guinea pigs is subclinical. Vertical transmission of the organism is speculated to occur; E. cuniculi infection was detected serologically in hysterectomy-derived gnotobiotic guinea pigs. 13 A recent, small serologic survey of inbred and outbred guinea pigs from a variety of sources revealed a seroprevalence of almost 50% to E. cuniculi. Most of these animals lacked gross and histologic evidence of infection. 142 Multifocal necrosis of the cerebrum with granulomatous encephalitis and interstitial nephritis with tubular necrosis, regeneration, and fibrosis have been described in infected guinea pigs. 142 Spontaneous and experimental infections with other Encephalitozoon species or other microsporidia have not been reported in guinea pigs.
Infection in carnivores
Natural infection of carnivores with E. cuniculi has been most thoroughly described in farm-raised foxes and domestic dogs. High mortality among blue fox kits, resulting in economic losses for the fur industry in Scandinavian countries, occurred as a result of endemic encephalitozoonosis. 95 E. cuniculi infection in domestic dogs has been reported from South Africa and the United States. 8 , 106 , 123 Puppies in these outbreaks had histologic and serologic evidence of encephalitozoonosis, and organisms were successfully reisolated in vitro from infected tissues. Serologic results in the parents were suggestive of recent E. cuniculi exposure. 123
Unlike the subclinical asymptomatic infection typically seen in rabbits and immunocompetent rodents, encephalitozoonosis is a sporadic, fulminating disease in neonatal carnivores. 123 In utero infection is suspected as the mode of transmission in fox kits. 89 Clinical signs in pups and kits include reduced appetite and stunted growth, ataxia, tremors, posterior weakness, and blindness with progression to circling, aggressive behavior, and convulsions. 87 , 132 Gross lesions include pale radial streaks extending from the cortex to the renal pelvis on cross sections of kidney and edematous, distended meninges. Thickened, necrotic, and tortuous medium-size to small arteries (consistent with polyarteritis nodosa) are often observed at necropsy in cardiac, intestinal, and central nervous system tissues in blue foxes. 95 Histologic lesions in dogs and foxes include multifocal microgranulomatous hepatitis, interstitial lymphocytic-plasmacytic nephritis, interstitial pneumonia, and lymphocytic-plasmacytic meningoencephalitis with perivascular cuffing and occasional intracellular parasitic cysts containing gram-positive spores. 132 Surviving animals shed parasites in the urine. 87 Morbidity and mortality vary within a litter of pups or kits, and the parents typically remain unaffected. Differential diagnoses for neonatal canine encephalitozoonosis include canine herpesvirus and canine distemper virus infections, Neospora caninum, gram-negative septicemia, and hydrocephalus or other intracranial abnormalities. 4 , 6 , 94 , 112 , 126 , 133 Infection of carnivores with other species of Encephalitozoon or other microsporidia has not been reported.
Infection in nonhuman primates
Reports of spontaneous infection with E. cuniculi in nonhuman primates have been best documented in squirrel monkeys (Saimiri sciureus). 122 , 154 In these studies, affected monkeys were classified as either neonates (<4 weeks old) or infants (<9 months old) suspected of acquiring the infection in utero. Antemortem signs were usually absent. There is a single case report of spontaneous intestinal microsporidiosis, presumed due to E. cuniculi, in an adult, wild-caught dusky titi monkey (Callicebus moloch) that was likely immunocompromised. 120
E. cuniculi infection in squirrel monkeys is typically subclinical, and gross lesions are not apparent. 7 Neonatal, and presumably immunosuppressed, squirrel monkeys most frequently have microscopic lesions in the central nervous system. These lesions include multifocal granulomas within the white and gray matter, nonsuppurative meningitis, and vasculitis. 7 , 154 Parasites are gram-positive, rod-shaped organisms free within foci of inflammation or contained within parasitophorous vacuoles. Nonsuppurative interstitial nephritis and pneumonia with intralesional parasites also occur. 7 , 154 Placentitis, characterized by a mononuclear inflammatory infiltrate and organizing granulomas, has been described. 7 The primary differential diagnosis for encephalitozoonosis in squirrel monkeys is infection with Toxoplasma gondii. 7 Subclinical encephalitozoonosis has also been demonstrated histologically in vervet monkeys (Cercopithecus pygerythrus) experimentally infected with E. cuniculi isolated from domestic dogs. 137
There is a single report of experimental infection of immunocompetent and immunosuppressed rhesus macaques (Macaca mulatta) with E. cuniculi and E. hellem. 42 Immunosuppressed macaques that received E. hellem intraveneously demonstrated spores in suppurative lesions of the liver and kidneys, whereas macaques inoculated orally with E. hellem or E. cuniculi shed spores periodically in the urine and feces but lacked gross and histologic evidence of parasite infection. Experimental and spontaneous Enterocytozoon bieneusi infections have recently been reported in several macaques (Macaca mulatta, M. nemestrina, and M. cyclopsis) infected with simian immunodeficiency virus (SIV). 22 , 83 , 134 Infection was associated with chronic hepatobiliary disease in these macaques and was characterized histologically by nonsuppurative and proliferative cholecystitis. 83 Parasites were also identified in the upper gastrointestinal tract mucosa. 83 E. bieneusi has also been diagnosed in SIV-negative, clinically healthy captive rhesus macaques, suggesting that this parasite may be a common or transient inhabitant of the digestive system of otherwise healthy macaques. 82 Spontaneous infection with Encephalitozoon intestinalis or E. hellem infections have not been reported in macaques, squirrel monkeys, or other Old or New World primates.
Infection in other animals
There are relatively few reports in the literature describing natural or experimental infection with E. cuniculi in rats; most of these reports are limited to historical surveys of rodent populations. 70 , 102 , 104 Infections with other microsporidia have not been reported in rats. However, there is an increasing number of reports of spontaneous microsporidial infection in psittacines, including lovebirds (Agapornis spp.), budgerigars (Melopsittacus undulatus), and eclectus parrots (Eclectus roratus). 12 , 71 , 96 , 108 In many of the early reports, the etiologic agent was identified as E. cuniculi, but molecular analysis has revealed that the currently reported psittacine infections are caused by E. hellem. 12 Infected birds are young and frequently are coinfected with other pathogens. Clinical signs include anorexia, lethargy, and failure to thrive. 12 Lesions include focal hepatic necrosis with intralesional parasites. Parasites can also be identified in intestinal mucosa, crypts, and lamina propria and in renal tubular and bile duct epithelial cells, without an associated inflammatory response. 12 There are no published reports of experimental infections of psittacines with E. cuniculi or E. hellem nor are there reports of spontaneous or experimental infections with E. intestinalis or Enterocytozoon bieneusi. Although less widely reported, spontaneous infection of pigs with E. bieneusi and Encephalitozoon intestinalis and infection of a donkey, dog, cow, and goat with E. intestinalis have been described. 14 , 34 Infection in these animals was determined by PCR identification of spores from fecal samples. Additional diagnostic and epidemiologic work is needed to determine whether infection in these mammals results in clinical disease and whether they serve as potential zoonotic reservoirs of Microspora.
Microsporidiosis as an Emerging Human Disease
Until the mid-1980s, human encephalitozoonosis was infrequently reported and was usually associated with immunoincompetent children manifesting neurologic signs. 10 , 84 , 86 The etiologic agent in these cases was presumed to be E. cuniculi. With the advent of HIV, acquired immunodeficiency disease (AIDS), and the development of more sophisticated diagnostic techniques, the spectrum of microsporidia capable of infecting humans has broadened and reports of human infections have increased. The majority of these infections result in disorders of the digestive tract (Table 3); Enterocytozoon bieneusi is most frequently identified. 97 E. bieneusi was first described in 1985 as the cause of chronic diarrhea and wasting in an AIDS patient. 35 It is primarily a pathogen of the small intestine and has been implicated as the etiologic agent responsible for 30–70% of the cases of AIDS-related diarrhea and wasting. 15 More recent epidemiologic surveys of AIDS patients with chronic diarrhea suggest that the prevalence of microsporidiosis in this patient population is closer to 15%. 16 This organism is also recognized as a cause of self-limiting “traveler's diarrhea” in immunocompetent individuals. 2 E. bieneusi infects mucosal enterocytes, resulting in duodenitis, but has also been implicated as the cause of sclerosing cholangitis and cholecystitis. 97 , 107 There is also some suggestion that E. bieneusi can infect the upper respiratory tract through aspiration of intestinal contents, resulting in sinusitis. 30 Clinical signs of diarrhea and wasting associated with E. bieneusi are reported to improve with albendazole, thalidomide, fumagillin, or furazolidone therapy, but elimination of infection has not been confirmed with these compounds. 45 , 47 , 90 , 125 In a more recent study, clindamycin was suggested to be efficacious against E. bieneusi infection. 72
Microsporidia recognized as pathogens in immunocompromised humans and lesions associated with infection.

Encephalitozoon intestinalis (originally named Septata intestinalis) was differentiated from Enterocytozoon bieneusi in 1993 as yet another agent of chronic diarrhea and weight loss in AIDS patients. 18 Since 1993, Encephalitozoon intestinalis has been identified infecting the small and large intestines, liver, gallbladder, respiratory and urinary tracts, and ocular tissues. 80 , 91 , 129 , 130 , 151 Lesions attributed to E. intestinalis infection include ulcerative enteritis, granulomatous colitis, granulomatous hepatitis, necrotizing cholangitis and cholecystitis, sinusitis, bronchitis, ulcerative cystitis, and keratoconjunctivitis. Eradication of parasite infection has been reported with albendazole therapy. 48 , 67
E. hellem was isolated in 1990 and was first described as the cause of keratoconjunctivitis in HIV-positive patients. 38 , 153 Although morphologically and ultrastructurally identical to E. cuniculi, E. hellem was identified by immunologic and molecular analysis as a separate species. 38 , 44 , 141 E. hellem has since been shown to infect the respiratory and urogenital tracts of immunocompromised individuals, causing lesions of necrotizing tracheobronchitis, necrosuppurative prostatitis, lymphocytic-plasmacytic interstitial nephritis, and ulcerative cystitis. 114 , 119 , 147 Ocular infections with E. hellem have been eradicated with topical fumagillin or parental itraconazole, and disseminated infections have responded to treatment with albendazole. 40 , 153
It was not until 1995 that E. cuniculi infection, differentiated from E. hellem with monoclonal antibodies, was identified in an HIV-positive patient with clinical signs of chronic sinusitis, rhinitis, and keratoconjunctivitis. 51 E. cuniculi has since been shown to have a tissue distribution in immunocompromised humans similar to that in rabbits, infecting renal endothelial and epithelial cells, microglia, and myocytes. 88 , 146 Electron microscopy remains the gold standard for diagnosing and differentiating all human microsporidial infections, although PCR primers have been developed and hold promise of a faster, less invasive means of detection that can be adapted to large scale epidemiologic studies. 149 Reports of human microsporidiosis will continue to increase as recognition of these parasites and the spectrum of diseases they cause increase.
Animal Models of Human Microsporidiosis
The ease with which the Encephalitozoon spp. can be cultivated in vitro has facilitated the development of several experimental animal models of infection for E. cuniculi, E. intestinalis, and E. hellem (Table 4). Mice are most frequently used because of their small size, ease of handling and infecting, and simple housing requirements, and because they are available in a variety of genetically defined immunocompromised strains. None of these murine models reproduces the clinical signs of diarrhea commonly observed in humans infected with E. intestinalis nor the respiratory signs most frequently associated with human E. hellem infection (see Table 2). Tissue distribution of E. cuniculi in the brain and kidneys in infected mice (and rabbits) appears similar to that seen in infected humans. 88 , 146 Systemic disease can be reproduced in a number of immunosuppressed mouse strains by a variety of inoculation routes with E. cuniculi, E. hellem, and E. intestinalis, and these models will aid in examining the mechanisms of microsporidial dissemination. 42 , 76 , 143 Mouse models have also been valuable in elucidating the immunologic response to infection with Encephalitozoon spp. 73 Foreshadowing the diagnosis of microsporidial infection in T-cell-deficient AIDS patients, early work with E. cuniculi demonstrated that antibody production and T lymphocytes contribute to the control of encephalitozoonosis in mice and that lymphokine-activated macrophages were responsible for killing the parasite, as occurs with other intracellular parasites. 55 , 115 , 116 The course of E. cuniculi infection in immunocompetent mice also represents a model that will aid in answering questions regarding the host-parasite relationship. 42 This information may be important as reports of transient microsporidial infection in healthy humans increase. 109 Despite the number of case reports documenting treatment of human microsporidial infection with various therapeutic agents, there are relatively few reports of drug efficacy against this disease in animals. 28 , 131 Future work with the mouse models should address host stimuli that initiate polar tube extrusion, identification of potential host cell receptors for parasite invasion, mechanisms of intracellular survival and immune system evasion, and drug screening and efficacy testing. Mouse models will continue to be valuable for filling the gaps in our knowledge of human and mammalian encephalitozoonosis.
Animal models of Encephalitozoon spp. and Enterocytozoon bieneusi infection. ∗
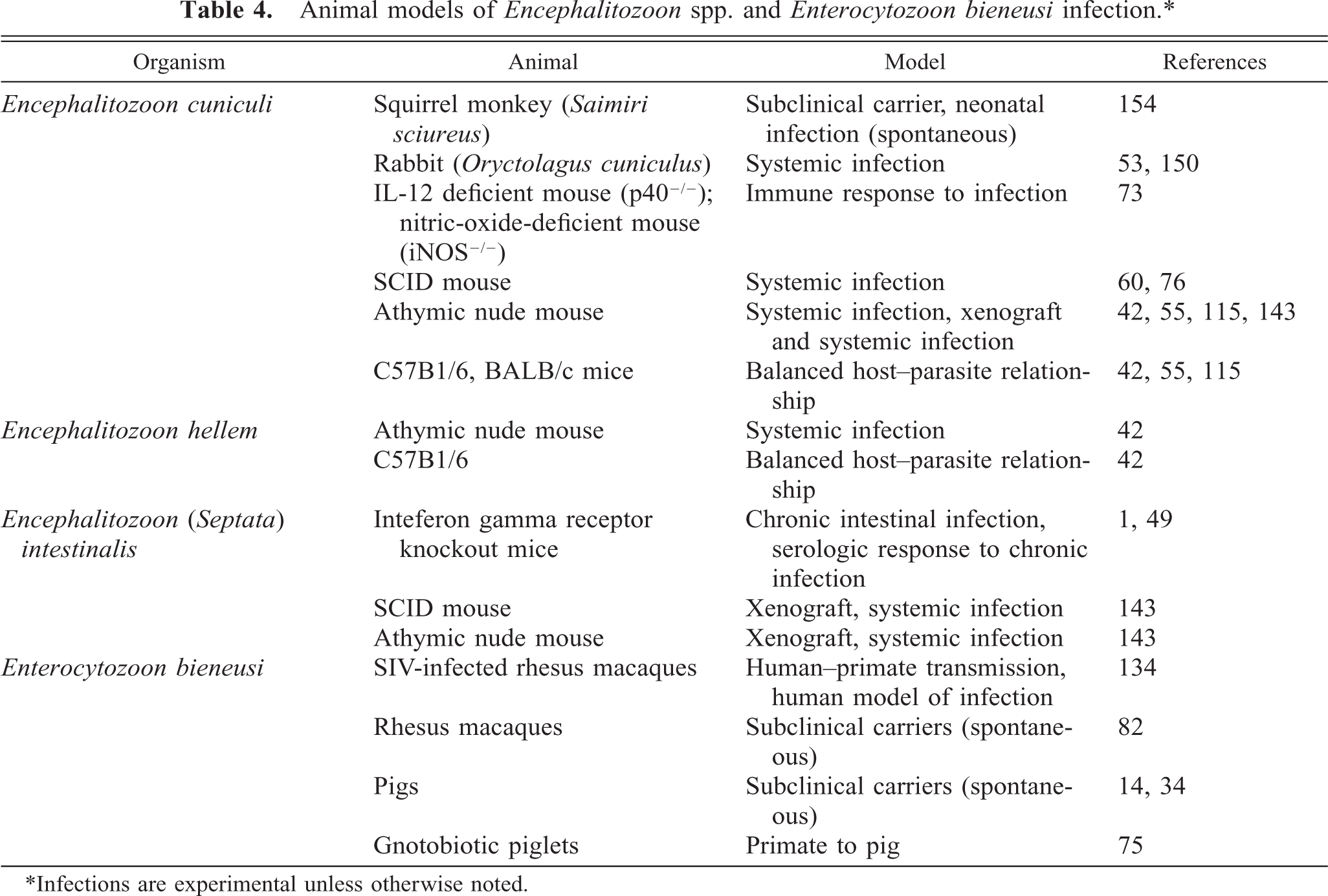
∗Infections are experimental unless otherwise noted.
Unlike the relatively promiscuous Encephalitozoon spp., there have been no reports of sustained in vitro cultivation of Enterocytozoon bieneusi, and until recently there were no animal models for studying human infection with this parasite. The inability to continuously propagate this organism in vitro and the limited clinical material from which to purify these parasites have contributed to the difficulty in developing a convenient rodent model of infection. 139 A recent report documented patent infection in gnotobiotically reared piglets. 75 Demonstration of experimental E. bieneusi infection in SIV-infected macaques, identification of spontaneous subclinical infection in immunocompetent macaques, and the similarity of target organs for E. bieneusi between humans and macaques offers promise of an animal model with which to further examine the pathogenesis, disease course, and potential drug therapies for E. bieneusi infection in humans. 82 , 83 , 134
Comments
Recognized as a mammalian protozoal parasite for nearly 80 years, Encephalitozoon cuniculi and several additional microsporidia have been identified as opportunistic pathogens in AIDS patients only in the last 15 years. Very little is understood regarding the cellular and molecular biology, pathogenicity, and epidemiology of these organisms, particularly in comparison with other human parasites such as Plasmodia, Trypanosoma, Leishmania, and Toxoplasma gondii. Although not causing the same global morbidity and mortality in the human population as some other protozoal pathogens, microsporidia are recognized as significant secondary invaders in immunosuppressed people. Additional information is needed regarding animal and environmental reservoirs of the microsporidia and the zoonotic potential of microsporidiosis for humans. Methods of preventing infection and identifying effective therapeutic agents against Encephalitozoon and other microsporidia will also be important for immunocompromised humans and for affected companion and production animals. A variety of animal models for human E. cuniculi, E. hellem, and E. intestinalis infection exist, but there are still gaps in our knowledge regarding parasite transmission and life cycle, the host–parasite relationship, modes of cellular infection and dissemination, and the molecular signals that govern these events. Although nonhuman primates are capable of sustaining infection with Enterocytozoon bieneusi, development of a reproducible rodent model of infection would facilitate study of this microsporidium. Future work should be directed at refining current models and developing new animal models of human encephalitozoonosis and other microsporidial infections. In addition, the differences in pathogenicity and disease mechanisms among Encephalitozoon cuniculi, E. hellem, E. intestinalis, and Enterocytozoon bieneusi have yet to be determined. Veterinary pathologists are uniquely situated to assist in answering these questions because of their exposure to a diverse number of animal species and their knowledge of infectious disease processes. Considered perhaps a disease of historical importance in the field of laboratory animal medicine, encephalitozoonosis has reemerged as a parasitic infection to be added to the expanding list of human microsporidial diseases and other HIV-associated opportunistic infections. Microsporidiosis of humans and animals will continue to be of scientific interest well into the next century.
Footnotes
Acknowledgements
The editorial comments of Dr. Howard B. Gelberg are gratefully acknowledged.