Abstract
Streptococcus suis type 2 (S. suis 2) is known as a major porcine pathogen worldwide and causes meningitis, septicemia, endocarditis, arthritis, and septic shock in pigs. Suilysin, a secreted protein of S. suis 2, is considered to be an important S. suis 2 virulence-associated factor. In this paper, the cerebellar lesions following experimental infection of pigs with S. suis 2 were studied. An immunohistochemical technique was applied to compare the distribution of bacteria and secreted suilysin protein in brain. The infected pigs developed histologic lesions of meningoencephalitis. Immunohistochemistry identified bacteria within the cytoplasm of neutrophils and macrophages localized in meningeal lesions. The secreted suilysin protein showed a similar localization within the cytoplasm of inflammatory cells, indicating that suilysin had high expression in vivo and may contribute to the pathogenesis of streptococcal meningoencephalitis.
Keywords
Streptococcus suis is considered one of the most important bacterial pathogens of swine. 4, 8 Among the 35 described serotypes, serotype 2 is most commonly associated with disease. As a zoonotic agent, S. suis has been isolated from human cases of meningitis, endocarditis, and recurrent toxic shock. 8 Two large-scale human outbreaks of S. suis 2 infection occurred in China in 1998 and 2005, causing extensive concerns of human health worldwide. 6 It is thought that the most important virulence factors in S. suis are the capsular polysaccharide and virulence-related proteins, including muramidase-released protein, extracellular protein factor, and hemolysin (suilysin). 4, 9
Suilysin is a member of the thiol-activated pore-forming toxin family and is secreted by S. suis into culture medium. It is toxic to various cell types and also enhances the invasion and lysis of Hep-2 cells in vitro. 7 The presence of suilysin is correlated with high virulence to pigs, and a vaccine containing purified suilysin protects pigs against experimental infection. 5, 9 These findings imply that suilysin is actively involved in S. suis infection and the host response. 1, 3, 4 This study reviews the histopathologic lesions following experimental infection with a Chinese isolate of S. suis 2 and the distribution of the whole bacteria and the secreted suilysin protein in the S. suis 2 experimental infection of piglets.
A S. suis type 2 strain named SC19 that expressed suilysin was isolated from the brain of a pig in China in 2005. The bacteria were cultured on tryptic soy agar plates containing 5% heat-inactivated sterile bovine sera at 37°C for 18 hours and then in 300 ml of tryptic soy broth (TSB, Difco, France) for 8 hours on standing. Eighteen 5-week-old healthy piglets that tested negative for S. suis serotype 2 by enzyme-linked immunosorbent assay at the beginning of the experiment were divided into 6 groups of 3 each. For experimental infection, the test groups (groups 1–5) were intranasally inoculated with 1×108 (pig Nos. 1–3), 1×107 (pig Nos. 4–6), 1×106 (pig Nos. 7–9), 1×105 (pig Nos. 10–12), 1×104 (pig Nos. 13–15) colony forming unit (CFU) strain SC19, respectively. The remaining control piglets (pig Nos. 16–18) were inoculated with TSB medium. The challenged pigs were monitored daily for clinical signs. Samples of cerebrum, cerebellum, brain stem, and cerebral dura mater were fixed in 10% neutral buffered formalin and routinely processed into paraffin. Sections of 2- to 3-μm thickness were cut for hematoxylin and eosin staining for histopathologic evaluation. At the same time, tissue sections were fixed in 2.5% glutaraldehyde and postfixed by osmication for 1 hour; the tissues were dehydrated and embedded into Epon, then processed routinely for transmission electron microscopic examination.
Pig Nos. 1–3 in the first group (1×108 CFU), pig Nos. 4–5 in the second group (1×107 CFU), and pig No. 9 in the third group (1×106 CFU) died within 3 days postinfection (dpi). Six pigs (pig Nos. 6, 7, 8, 10, 11, 12) showed severe signs of central nervous system (CNS) disease and were euthanized on 7 dpi. Pig No. 15 showed mild clinical signs and also was euthanized on 7 dpi. The remaining 2 infected pigs did not show clinical signs. Pigs (pig Nos. 16–18) in the control group did not show any clinical signs. Samples of 6 pigs (pig Nos. 6, 7, 8, 10, 11, 12) were then collected and processed for histopathology, and samples from 3 pigs (pig Nos. 6, 7, 10) were prepared for immunohistochemical analysis.
In general, affected pigs presented clinical signs of anorexia, depression, and fever (>40.5°C). The eyes of the diseased pigs were glazed, with reddening of mucous membranes. Severe nervous symptoms were observed, including incoordination, lateral prostration, paddling, opisthotonus, and convulsions. Lameness was found in all 7 pigs, apparently in posterior limbs.
Grossly, the dura mater was thickened and congested, and the inner layer of the dura mater was uneven. The surfaces of the gyri were flattened as a result of compression of the swollen brain by the dura mater and inner surface of the skull. The cerebullar sulci were shallow, and the volume of flavescent cerebrospinal fluid was increased.
Microscopically, hemorrhage and profuse exudates of neutrophils with fewer macrophages were observed in cerebral dura mater. Focal aggregates of neutrophils and a variable amount of fibrinous exudate were found in the inner layer of the dura mater (Fig. 1). Large numbers of neutrophils filled the subarachnoid and subpial spaces (Fig. 2). The meningeal vessels were very turgescent, containing intraluminal neutrophils and macrophages, and were surrounded by infiltrating leucocytes (Fig. 3).
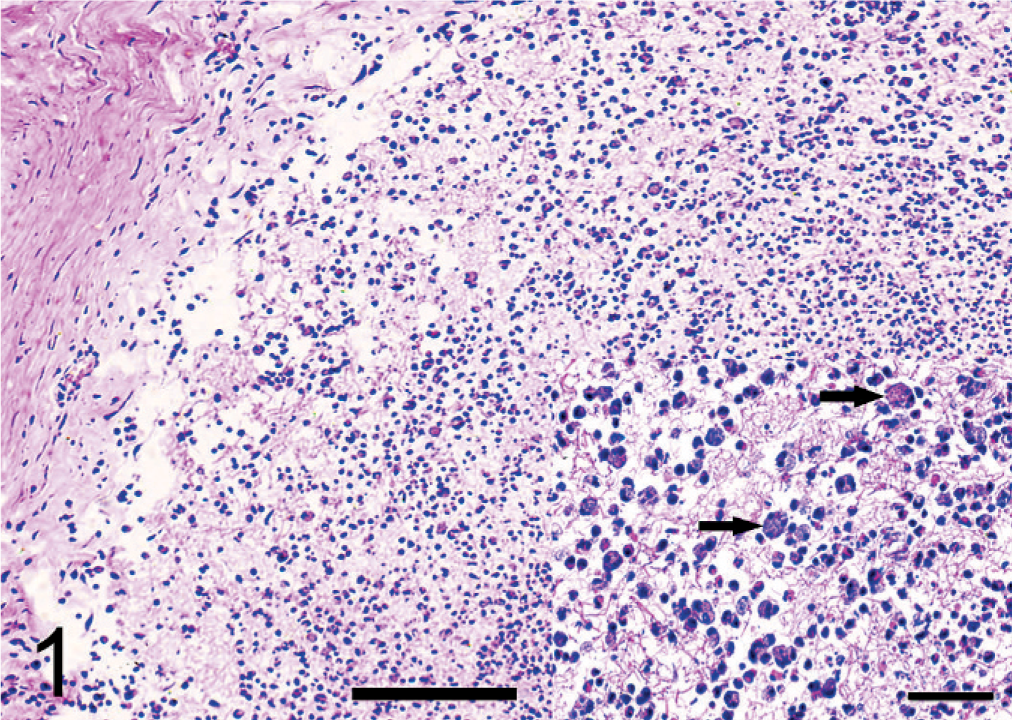
Cerebral dura mater; pig No. 6. Moderate focal aggregates of neutrophils and fibrinous exudate in the inner layer of the dura mater. The inset shows degenerating inflammatory cells (arrows) within the exudate. HE. Bar = 80 μm.
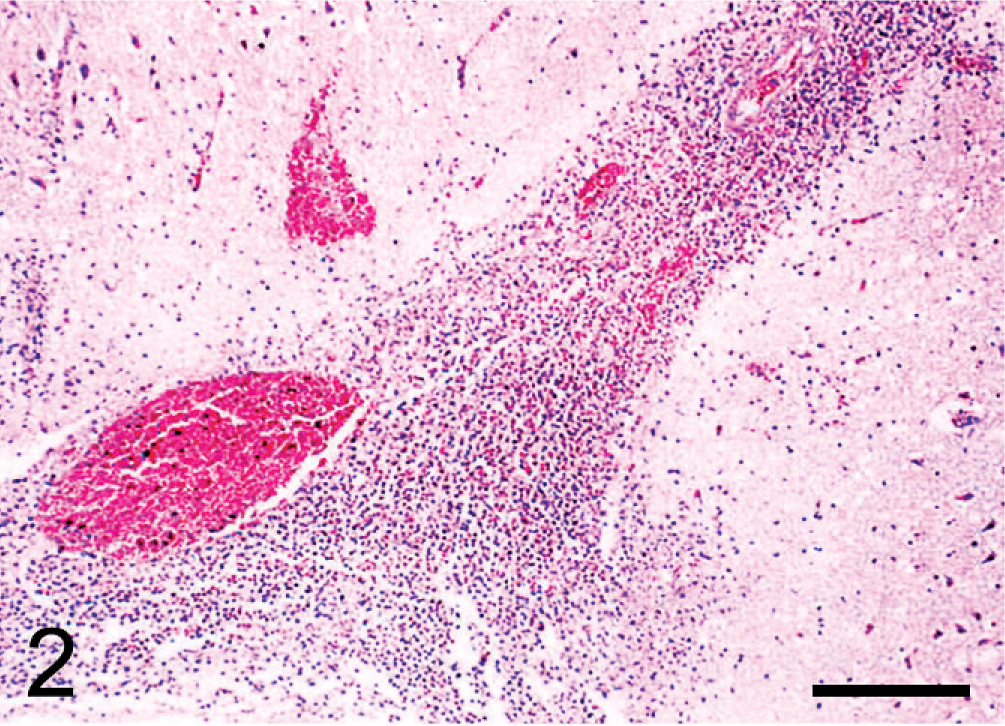
Subarachnoid space; pig No. 7. Leptomeningitis with intense inflammation consisting of neutrophils and macrophages. HE. Bar = 100 μm.
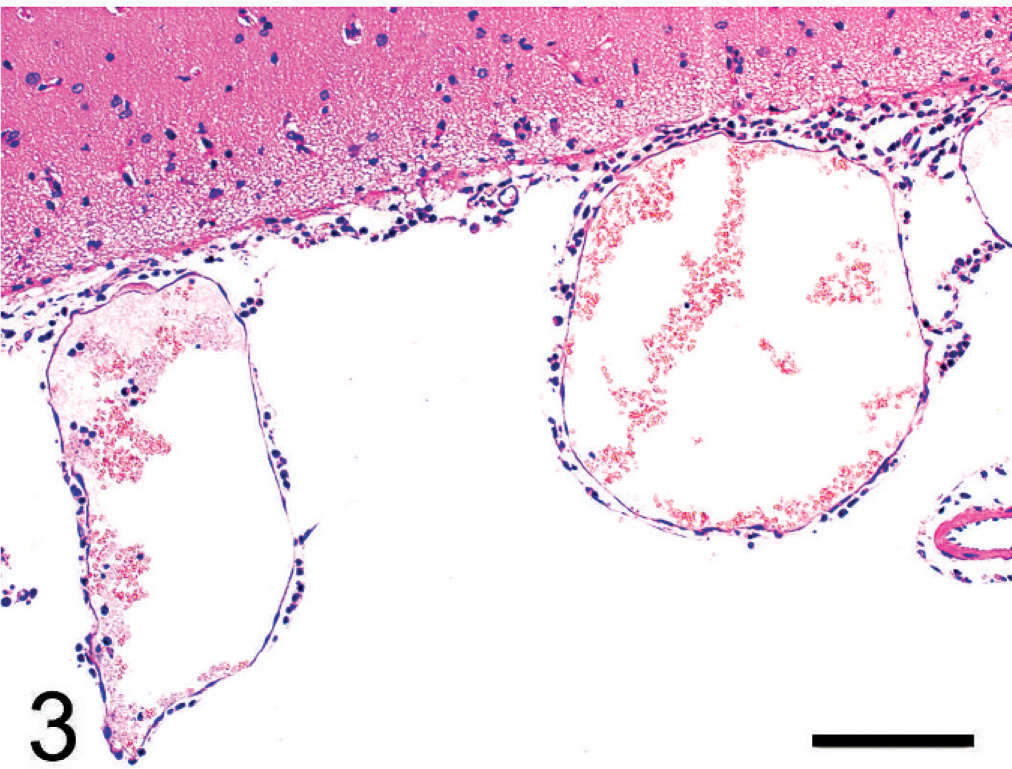
Subarachnoid space; pig No. 7. Blood vessels in the subarachnoid space also were very dilated, contained intraluminal neutrophils and macrophages, and were surrounded by extravascular leucocytes. HE. Bar = 100 μm.
The encephalitis was associated with marked cerebral edema characterized by broadening of perivascular and pericelluar spaces, massive infiltration of neutrophils, and considerable acute necrosis of neurons. In this process, the superficial layer of the cortex was infiltrated by inflammatory cells. Focal proliferation of microglial cells and scattered small glial nodules were observed. Inflammatory reactions such as perivascular neutrophil infiltrates, considerable satellitosis, and neuronophagia were seen (Figs. 4,5). Virchow-Robin spaces were distended, and the endothelial cells in capillaries were swollen. The choroid plexuses of the lateral, third, and fourth ventricles showed marked congestion associated with scant cellular exudates. The inflammation spread to the hyperplastic ependymal layer of the cerebral ventricle (Fig. 6). Subependymal tissue was edematous and infiltrated by inflammatory cells, especially neutrophils. The changes extended to the spinal cord and appeared as moderate perivascular infiltration of neutrophils, neuronophagia, and degeneration of myelin.
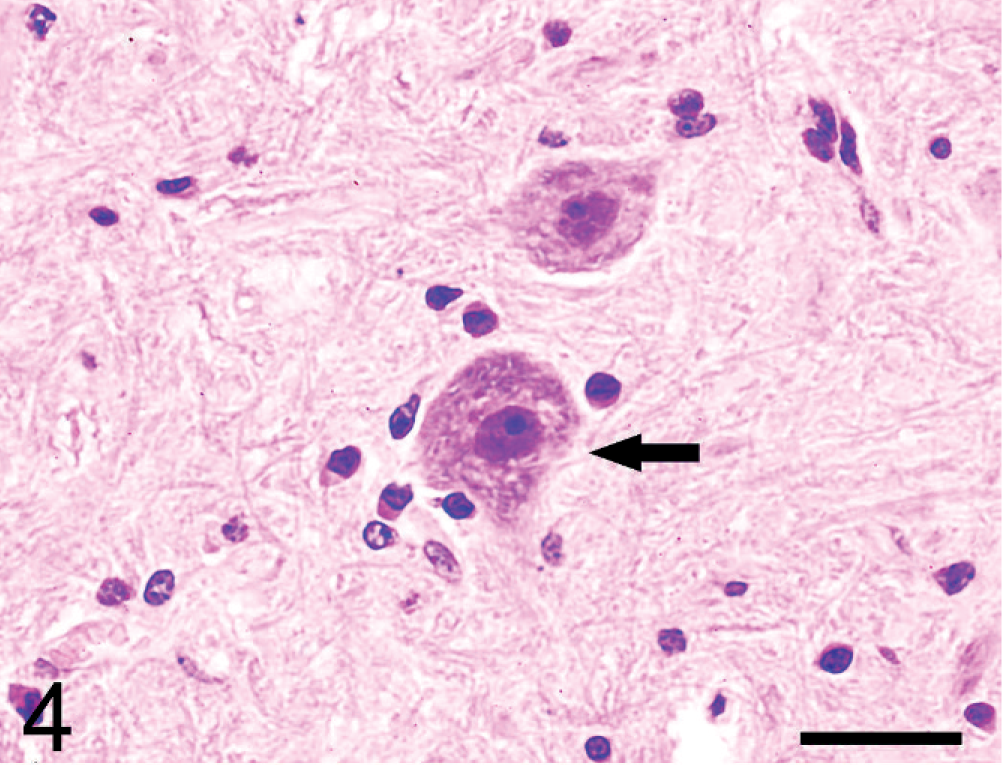
Brain; pig No. 6. Satellitosis (arrow) was observed. HE. Bar = 40 μm.
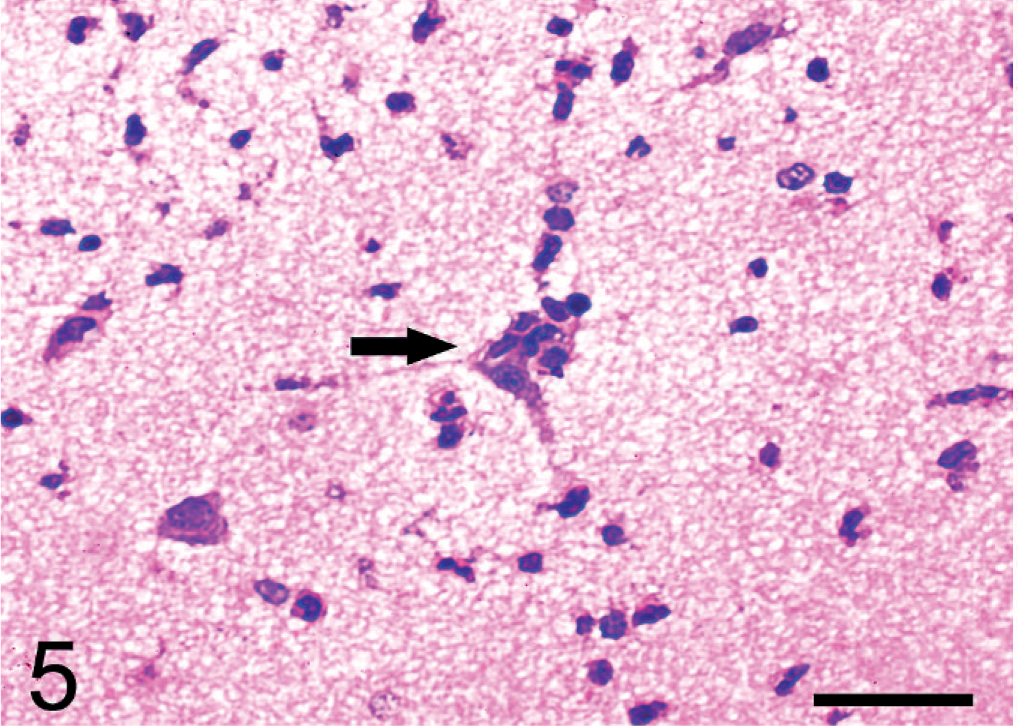
Brain; pig No. 6. Neuronophagia (arrow) was observed. HE. Bar = 40 μm.
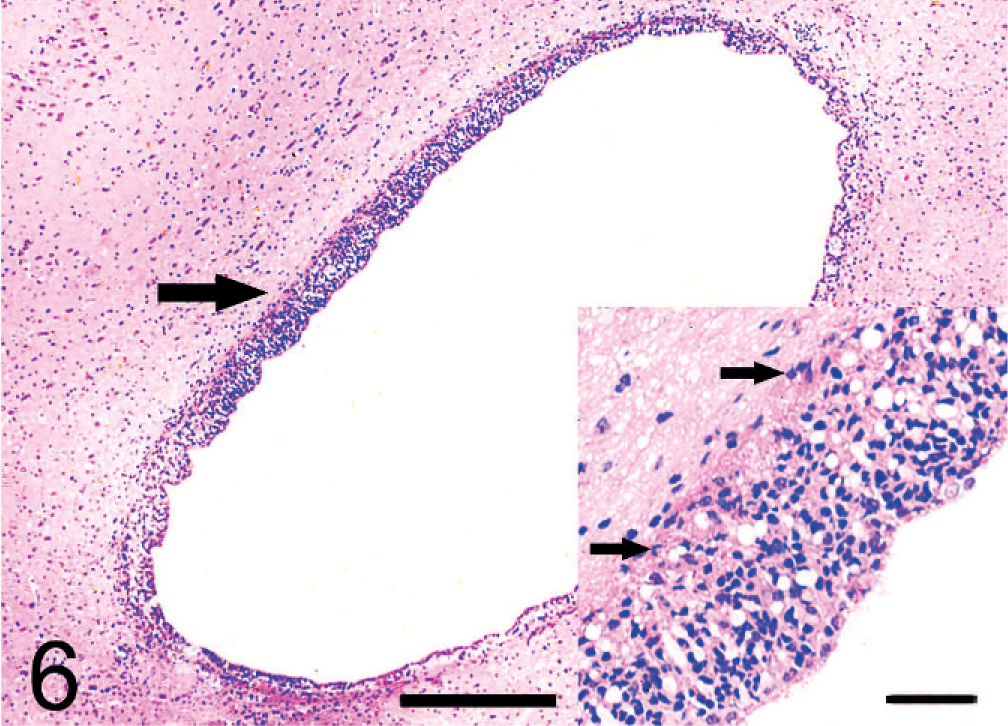
Ependyma; pig No. 10. The ependyma (arrow) was thickened and infiltrated by inflammatory cells (arrows). HE. Bar = 80 μm.
Ultrastructurally, many neurons and glial cells were degenerate or necrotic, with pyknosis, fragmentation or lysis of the nuclei and loss of organelles. Degeneration of myelin also was identified.
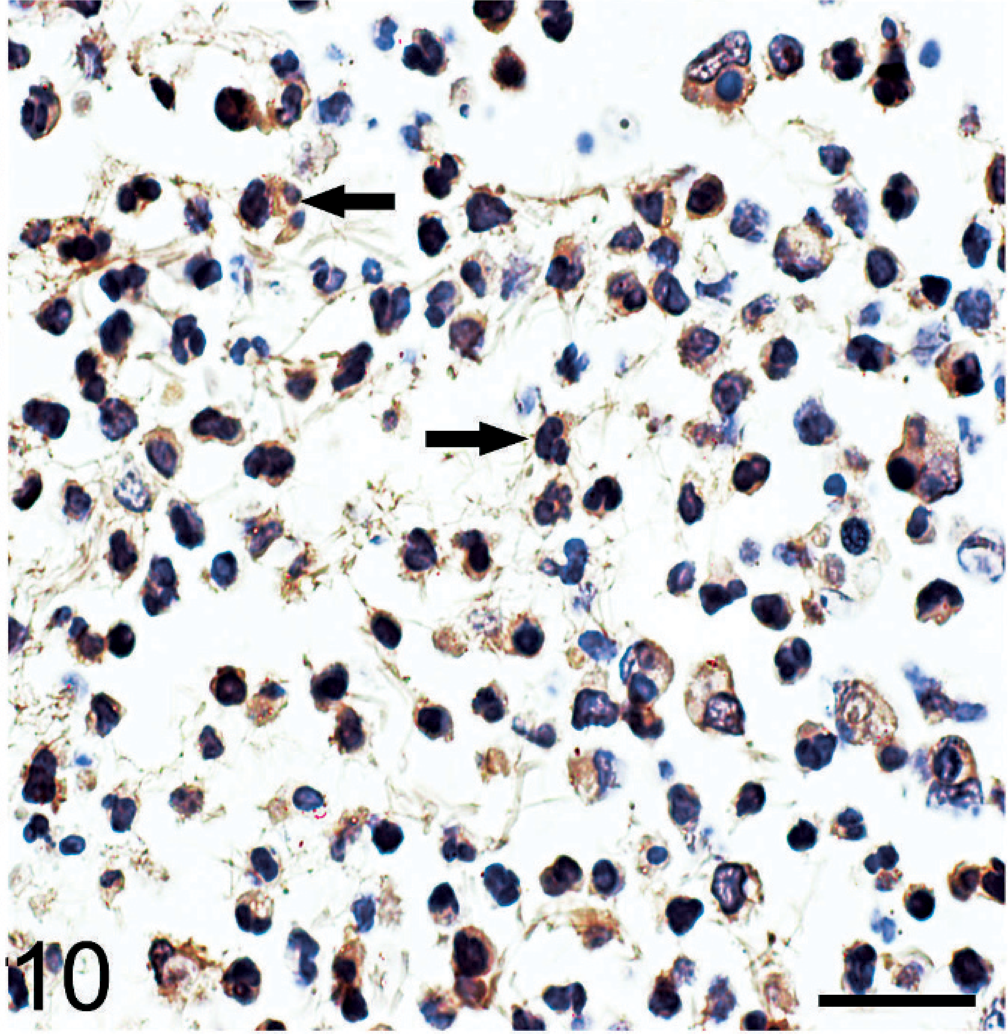
Immunohistochemical analysis for detection of S. suis 2 antigen was performed using a rabbit hyperimmune serum, and detection of suilysin was similarly performed using a monoclonal antibody, which was kindly provided by Yangying (State Key Laboratory of Agricultural Microbiology, Huazhong Agricultural University, China). The 2 antibodies (a rabbit hyperimmune serum 1 : 50, mouse monoclonal antibody 1 : 100) were diluted before being incubated with tissue sections for 1 hour at 37°C. The secondary antibody (goat anti-rabbit IgG-HRP or goat anti-mouse IgG-HRP, Southern Biotech, USA) were diluted 1 : 500 and incubated with tissue sections for another hour at 37°C and detected using a commercially available kit with streptavidin-biotin complex (Boster Company, China). Sections were counterstained with hematoxylin. Sections of brain from the control group (pig No. 18) were the negative control. For another additional negative control, the monoclonal primary antibody was omitted. Cells with brown cytoplasmic and/or membranous immunoreactivity were identified as positive. The immunoreactivity was minimal in control tissues (picture not shown) and in the infected sections without primary antibodies (Figs. 7,8). Using the polyclonal antibody to S. suis 2, immunolabelled cells were concentrated in the leptomeningeal lesions. The positive reaction was found within the entire cytoplasm of neutrophils and in macrophages (Fig. 9). A similar result was found using the monoclonal antibody to suilysin, with immunoreactivity in the cytoplasm of inflammatory cells (Fig. 10). Suilysin antigen was detected in all cerebral samples of infected pigs, although the intensity of immunolabeling varied from 50 to 85%. Immunolabeled cells were localized in the subarachnoid space, and immunolabeling of cerebral parenchyma was rare.
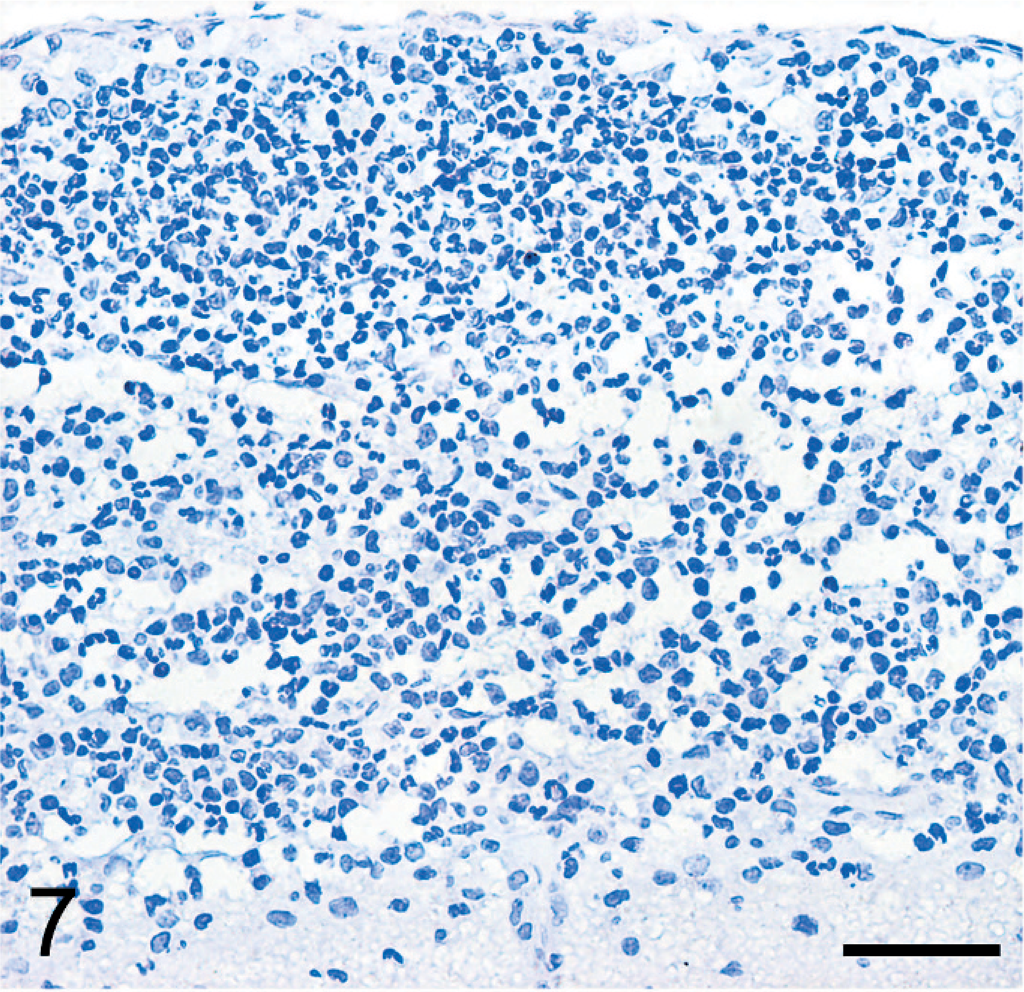
Subarachnoid space; pig No. 7. Absence of immunolabeling was observed in the infected section without S. suis 2 polyclonal antibody. Bar = 100 μm.
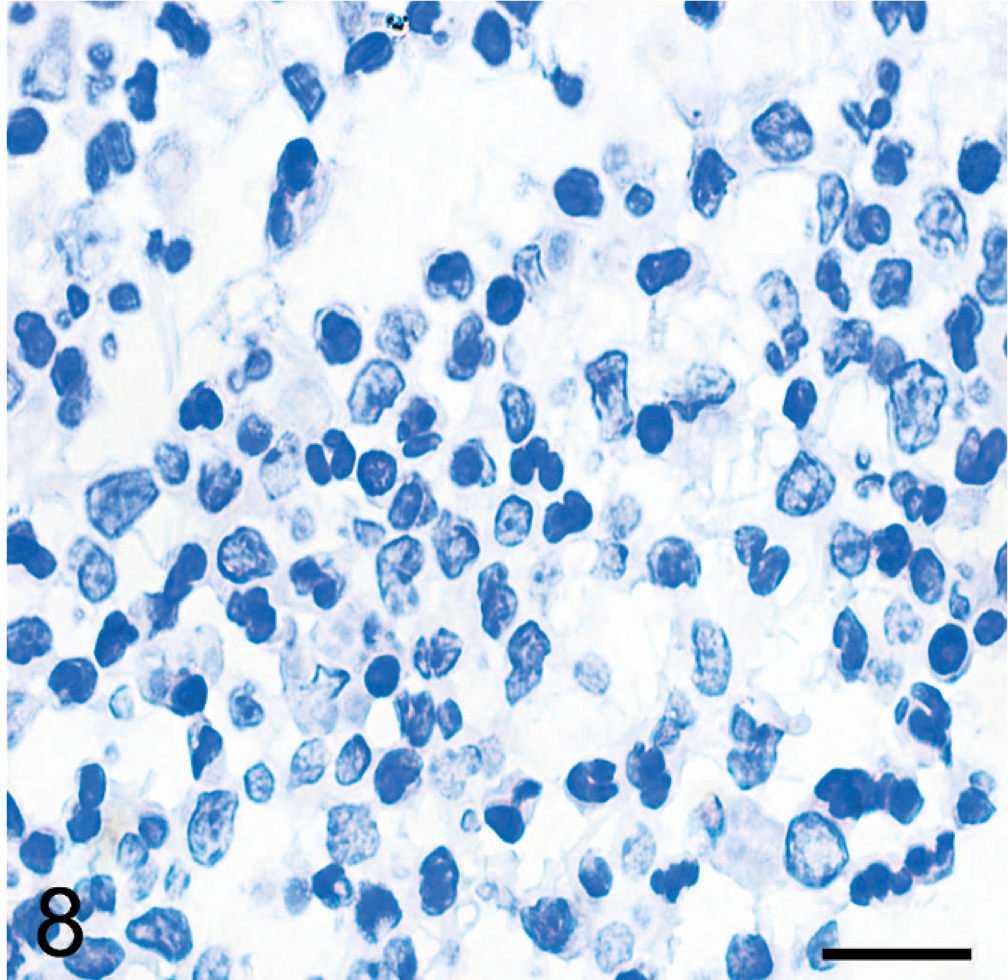
Subarachnoid space; pig No. 7. Absence of immunolabeling was observed in the infected section without suilysin monoclonal antibody. Bar = 50 μm.
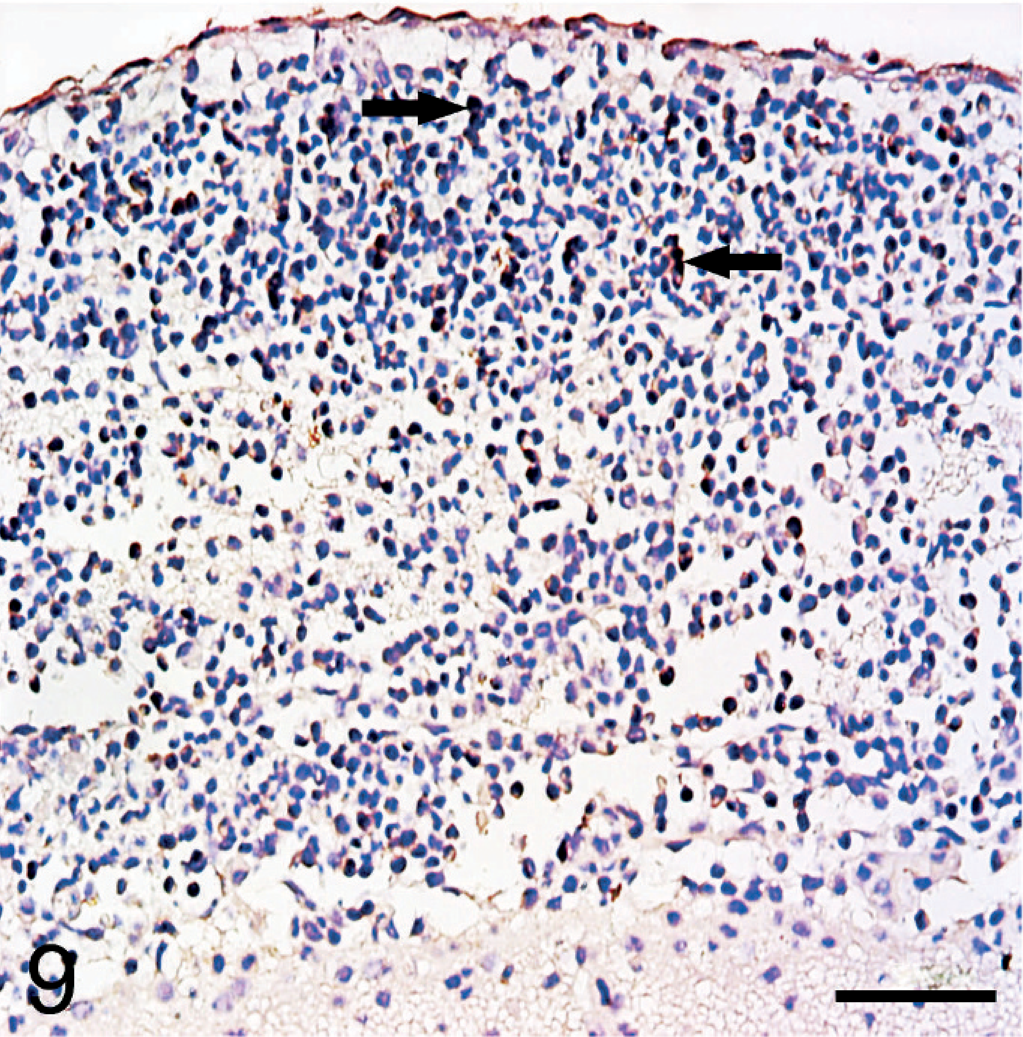
Subarachnoid space; pig No. 7. S. suis 2 antigens were observed in the cytoplasm of neutrophils (arrows) and macrophages localized in the subarachnoid space. Bar = 100 μm.
Subarachnoid space; pig No. 7. S. suis 2 antigens were observed in the cytoplasm of neutrophils (arrows) and macrophages localized in subarachnoid space. Bar = 50 μm.
In August 2005, several strains of S. suis 2, which produced β-hemolysis on sheep blood agar plates, were isolated during the outbreak of S. suis 2 in China. In the present study, experimental infection of pigs by this isolate of S. suis 2 was designed to study the disease pathologically. Typical clinical signs of CNS disease and lameness were found after challenge in 13/15 (87%) infected pigs, similar to previous reports of infection by European and North American strains. 3, 8 In present study, infection with S. suis 2 strain SC19 caused acute death of pigs. Severe lesions in the dura mater, brain, and spinal cord were observed on 7 dpi. Moreover, the meninges were estimated to be the tissue injured first and most seriously.
So far, we found that about 76% of Chinese S. suis 2 isolates had the suilysin gene (data not shown), similar with European strains for which most were suilysin- positive. 4 Moreover, suilysin-positive strains had high virulence in animals. 4 The immunohistochemical technique was used to compare the distribution of the bacteria and the secreted suilysin protein. The similar distribution suggested that suilysin had high expression in vivo and might be involved in the pathogenesis.
In conclusion, S. suis 2 reached the CNS via the blood after colonizing the nasopharynx and caused severe lesions of the dura mater, brain, and spinal cord. In this work, the disease appeared as purulent inflammation of the dura mater, subarachnoid space, and superficial layer of cerebral parenchyma. The result of immunohistochemical analysis reflected that suilysin had high expression in vivo, and there may be some interactions between suilysin and the host.
Footnotes
Acknowledgements
This work was supported by 973 program (2006CB504404), 863 program (2006AA10A206), and National Key Technology R&D Program (2006BAD06A11). We also thank Yangying for kindly providing the suilysin monoclonal antibody.
