Abstract
To delineate the anomaly and frequency of their occurrence in a pig model, we reported the topography of the vagus laryngeal branches and compared the differences with humans. Thirty sides of cervical vagus nerve in 15 fresh cadavers (Sus scrofa) were microdissected. We measured the branch diameters and lengths of the laryngeal branches using a Vernier caliper with a resolution of 0.01 mm. Two patterns of the vagus laryngeal branches were shown: 56.7% with the cranial laryngeal nerve (CLN) and 43.3% without the CLN. The diameters and the length of the CLN were not affected by the side of the neck (P > 0.05), but the diameters of the recurrent laryngeal nerve (RLN) and the nodose ganglion were significantly different between left and right sides (P < 0.05). The left RLN was thinner than the right side in diameter (P < 0.05). Four of the 30 sides had anastomoses between the vagus and the cervical sympathetic chain. There were some differences between the pig anatomy and human anatomy, but the patterns were largely similar. The similarities support the utility of this model, which is closer in size to humans than the standard rodent models.
The cranial laryngeal nerve (CLN), and its two branches the internal laryngeal nerve (ILN) and the external laryngeal nerve (ELN), together with the recurrent laryngeal nerve (RLN) are critical for swallowing, speech and respiration. 1 Due to their anatomical positions, these nerves are vulnerable to iatrogenic injury during various surgeries, such as thyroidectomy, 2,3 spine surgery 4 and endarterectomy. 5 Therefore, a thorough understanding of the laryngeal anatomy and the possible variations of these nerves in pigs is important for successful outcomes of studies using this animal model.
The measured variables
There were two patterns of laryngeal branches of the vagus: with CLN and without CLN as shown in Figure 1. In all, 56.7% (17 of 30 cases) showed the CLN trunk arising from the nodose ganglion of the cervical vagus, just caudal to the jugular foramen within the carotid sheath. In the other 13 cases, the ILN and the ELN branched from the cervical vagus nerve directly without the CLN truck. The bifurcation of the CLN into the ILN and the ELN from its origin of the nodose ganglion was diverse in distance. The distance of the CLN from the origination to the upper point of the bifurcation of the common carotid artery was 4.84 ± 0.52 mm. All of the ILNs originated from the nodose ganglion directly. In some cases the course of the ILN was straight but in others it twisted before reaching the larynx. The ILNs were all dorsal to the cranial laryngeal artery, and then combined with it parallelly or caudally to pierce the lateral aspect of the thyrohyoid membrane laterally.
Two anatomical patterns of the vagus laryngeal branches: (a) with cranial laryngeal nerve (CLN): the CLN trunk arising from the nodose ganglion (NG) of the cervical vagus, bifurcating into the internal laryngeal nerve (ILN) and the external laryngeal nerve (ELN); (b) without CLN: the ILN and the ELN branched from the cervical vagus nerve directly without the CLN truck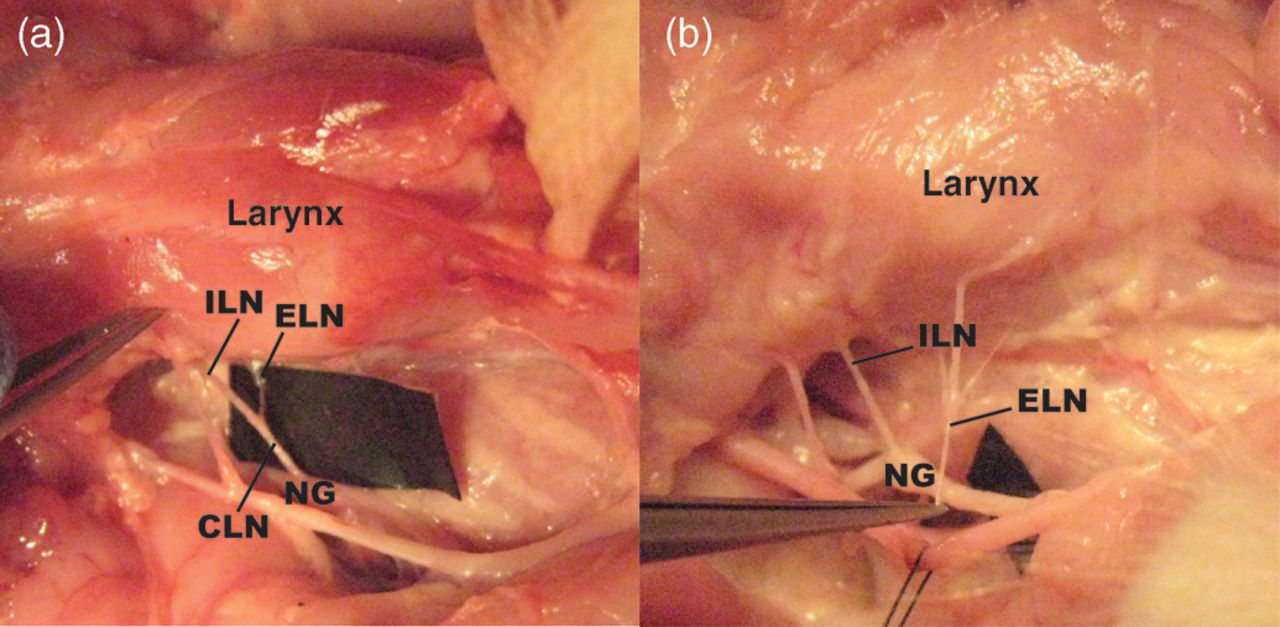
In the 13 cases without the CLN, nine ELNs came from the nodose ganglion, three from the vagus and one from a combination of two small branches coming from the nodose ganglion and the ILN separately. Among the three cases where the ELNs came from the cervical vagus, one originated rostrally to the nodose ganglion, extending caudally to the inferior pharyngeal constrictor muscle and the upper oesophagus. No branch was found to innervate the cricothyroid muscle. The other two ELNs came from the vagus just caudal to the nodose ganglion. Twenty-two of the 30 ELNs (73%) had no branch before reaching the cricothyroid muscle, while the other 27% had two to three branches innervating the inferior pharyngeal constrictor muscle and the cricothyroid muscle.
The left RLN from the cervical vagus recurred around the arch of the aorta before ascending, whereas the right looped around the right subclavian artery. Then all RLNs travelled rostrally along the oesophageal–tracheal groove and entered the larynx dorsally to the cricothyroid joint. In all cases, this nerve had no branches before entering the larynx. Four of the 30 sides had anastomoses between the cervical vagus and the cervical sympathetic chain or the cranial cervical ganglion. One occurred between the vagus and the cervical sympathetic chain. One was between the CLN and the cranial cervical ganglion. The other two were between the ELN and the cranial cervical ganglion. No anastomosis was found with the RLN outside the larynx.
Lengths and diameters of the measured variables in pigs with and without cranial laryngeal nerve (CLN)
*Right side thicker
†Left side thicker
The classic description of the superior laryngeal nerve in humans is of a constant and short trunk that divides into the internal and external branches. This is also true of some animals, such as dogs, cats, rats and pigs. 6 However, in our study, less than 57% of cases had this normal CLN configuration. This pattern has not previously been reported in pigs, although it has been in humans. Kambic et al. 7 reported that in 5% of their dissections the ILN and the ELN arose from the lower pole of the nodose ganglion of the cervical vagus. Another report documented 6% of cases missing the trunk of the superior laryngeal nerve in 40 fresh cadavers, with the bifurcation occurring just below the nodose ganglion. 8 In humans the bifurcation of the superior laryngeal nerve into internal and external branches usually occurs with the bifurcation of the common carotid into the internal and external carotid arteries (75%), but sometimes the superior laryngeal nerve divides alongside the bifurcation of the carotid artery (15%) or more caudally (10%). 9 In pigs the bifurcation of the CLN in the pig model was outside the bifurcation of the common carotid artery caudally and medially. In humans, the ILN was largely observed in a parallel and medial position to the superior laryngeal artery (89%), and in 11% of the dissections it was located inferiorly and medially to that artery. Then both went downwards and medially piercing into the thyrohyoid membrane in humans. 8 In pigs, the ILN is always located dorsal to the cranial laryngeal artery parallelly or caudally. In humans the ELN runs either superficially or deeply to the inferior pharyngeal constrictor muscle, after which the terminal branches of the ELN penetrate the horizontal and the oblique bellies of the cricothyriod muscle and the inferior pharyngeal constrictor muscle. 10 In our study, all the ELNs except one innervated the cricothyriod muscle and the caudal pharyngeal constrictor muscle. Most of the ELNs (73.3%) only had one branch arriving in the cricothyriod muscle. In humans 17.3% of the RLNs had a communicating branch with a similar diameter to the RLN connecting with the sympathetic trunk. 11 We did not find any variations in pigs. An anastomosis connecting the cervical sympathetic chain with the CLN and its branches is called the CLN loop (in humans it is called the superior laryngeal nerve loop) with the incidence of 98.3% in humans, 12 which is significantly higher than our findings in pigs.
Our sample provided statistical details as to the pattern of the laryngeal branches of the cervical vagus nerve in the pig, with details on the topography and anatomical variations. There were some differences between the pig anatomy and human anatomy, but the patterns were largely similar. These results should be useful to researchers who are carrying out accurate experiments and studies on laryngeal function and surgery in a pig model. The similarities support the utility of this model, which is closer in size to humans than the standard rodent models.
Footnotes
ACKNOWLEDGEMENTS
We thank Dr Charley C Della Santina for providing the microscope and Stacey Lukasik for her assistance in the various phases of the present research. We appreciate Regina Campbell-Malone and Shaina Holman for helpful comments on the manuscript. This work was supported, in part, by NIH DC9980 to RZG.
