Abstract
The pig represents a useful, large experimental model for biomedical research. Recently, it has been used in different areas of biomedical research. The aim of this study was to review the basic anatomical structures of the head region in the pig in relation to their use in current research. Attention was focused on the areas that are frequently affected by pathological processes in humans: the oral cavity with teeth, salivary gland, orbit, nasal cavity and paranasal sinuses, maxilla, mandible and temporomandibular joint. Not all of the structures have an equal morphology in the pig and human, and these morphological dissimilarities must be taken into account before choosing the pig as an experimental model for regenerative medicine.
History of using the pig as an experimental model
The pig, one of the first species to be domesticated, represents one of the most important livestock species with nearly 500 different breeds with a worldwide distribution. 1 Recently, the pig became a very frequent and favourite biomedical model. 2–11 It could possibly represent a significant source of organs for future transplantations. Minipigs are considered as an experimental model in many biomedical fields due to their apparent similarity to the human in terms of anatomy, as well as for economic advantages and ethical reasons. Because of the physiological similarities, the transfer of results acquired in pigs to human conditions is more exact compared with other experimental animals such as the mouse, rat or rabbit. The history of using pigs as an animal model suitable for research is also much older. The first symposium devoted exclusively to the basis for and the extent of the utilization of pigs in biomedical research was held at the Pacific Northwest Laboratory, Richland, Washington, in 1965. 5
Many different pig breeds carrying human diseases and symptoms such as diabetes mellitus and melanomas have been established. 8,12,13 Pigs and minipigs have become firmly established as the main research models in some areas of biomedical and pharmacological research because of their anatomical similarities to humans (e.g. body size, skin, cardiovascular system and urinary system), their functional similarities (gastrointestinal system and immune system) and because of the availability of disease models (e.g. arteriosclerosis, metabolic syndrome, gastric ulcer and wound healing). 14 Recently, the minipig was used as a model for testing the toxicity of new medicines and chemicals. It is necessary to test new pharmaceuticals intended for use in humans on non-rodent species. The most common choices are dogs or, in limited numbers, primates. Pigs and minipigs have been identified as being suitable to take the role of non-rodent species in the toxicity testing of pharmaceutical products because of their haematological and cardiovascular similarities to humans. The RETHINK project recently focused on the evaluation of the potential impact of toxicity testing in the minipig as an alternative approach. 15,16 Therefore, it is evident that the pig can be a more useful experimental animal model in many aspects compared to the other animals (mice, rats, rabbits or dogs) that are routinely used.
This study aimed to summarize the clinically most important anatomical structures of the craniofacial region, to compare their morphology in the pig with corresponding structures in the human and to review a possible use of the pig model in craniofacial research.
Clinical relevance of the pig model to human oral cavity diseases
In contrast to humans, where the oral cavity is nearly oval-shaped, the oral cavity of the pig is narrow and long, and does not differ between breeds (Figure 1). The labia are short and less movable, which reduces the ability of the pig to fully open the oral cavity compared to other species. Such an anatomical layout of the labia and oral cavity makes intubation for anaesthesia complicated. For clinicians, it is important to notice that the pig cannot breathe with an open mouth and the nasal airways have to be left free during intubation for respiration. The intubation technique requires training and experience as the long oral cavity makes this difficult.
The external nose of the pig, supported by rostral bone, is fused with the upper lip to form the rostrum (Figures 1 and 2). A thick layer of fat –
The hard palate is covered by the oral mucosa with prominent palatal ridges –
The oral mucosa of the pig provides a suitable model for studying the biological processes that regulate scarless wound healing in order to find novel approaches for preventing scar formation. The histological structure of the palatal mucosa shows the same pattern as that of humans. 18 Furthermore, oral scarless healing was shown to resemble fetal skin, with rapid and transient inflammatory reactions in contrast to the adult skin, and it showed molecular responses during healing. 19,20 Uncovering the molecular pathways involved in scar formation and processes of the pig oral mucosa healing without their formation might allow them to be used in the development of new clinical techniques and in the discovery of molecules suppressing scar formation after surgical treatments.
The oral mucosa of the pig can also be used to improve clinically effective delivery systems for DNA and RNAi technologies. 3 These days, interest in delivering drugs through the buccal mucosa has increased, but a major limitation in buccal drug delivery is the low permeability of the epithelium. Therefore, knowing the effect of drug applications on individual layers of the oral mucosa is very critical. 21–23 The porcine snout and buccal mucosa have been successfully used as a model for human treatment in penetration studies. 7,21 Finally, there is a need in reconstructive surgery for flaps lined by non-keratizing stratified squamous epithelium or mucous membrane in human medicine. 24 Pig buccal mucosa flaps were prefabricated in the skin and found to be significantly enlarged after 1 week of incubation. These procedures can be used for nasal or oral reconstructions. 24
The porcine tongue, which is long and firmly attached to the floor of the oral cavity by a double frenulum, possesses a similar histological structure as in humans. It was recently used as a model for investigating surgical techniques for reducing tongue volume by cold ablation (coblation). 25–27 Coblation is a radiofrequency method used for the volumetric reduction of soft tissues in patients with obstructive sleep apnoea syndrome. 28 As significant lesions often develop after this procedure, surgical approaches were tested on the porcine tongue in order to determine the process of lesion/scar formation and to develop further improvements in this surgical technique.
Clinical relevance of the pig model to human salivary gland diseases
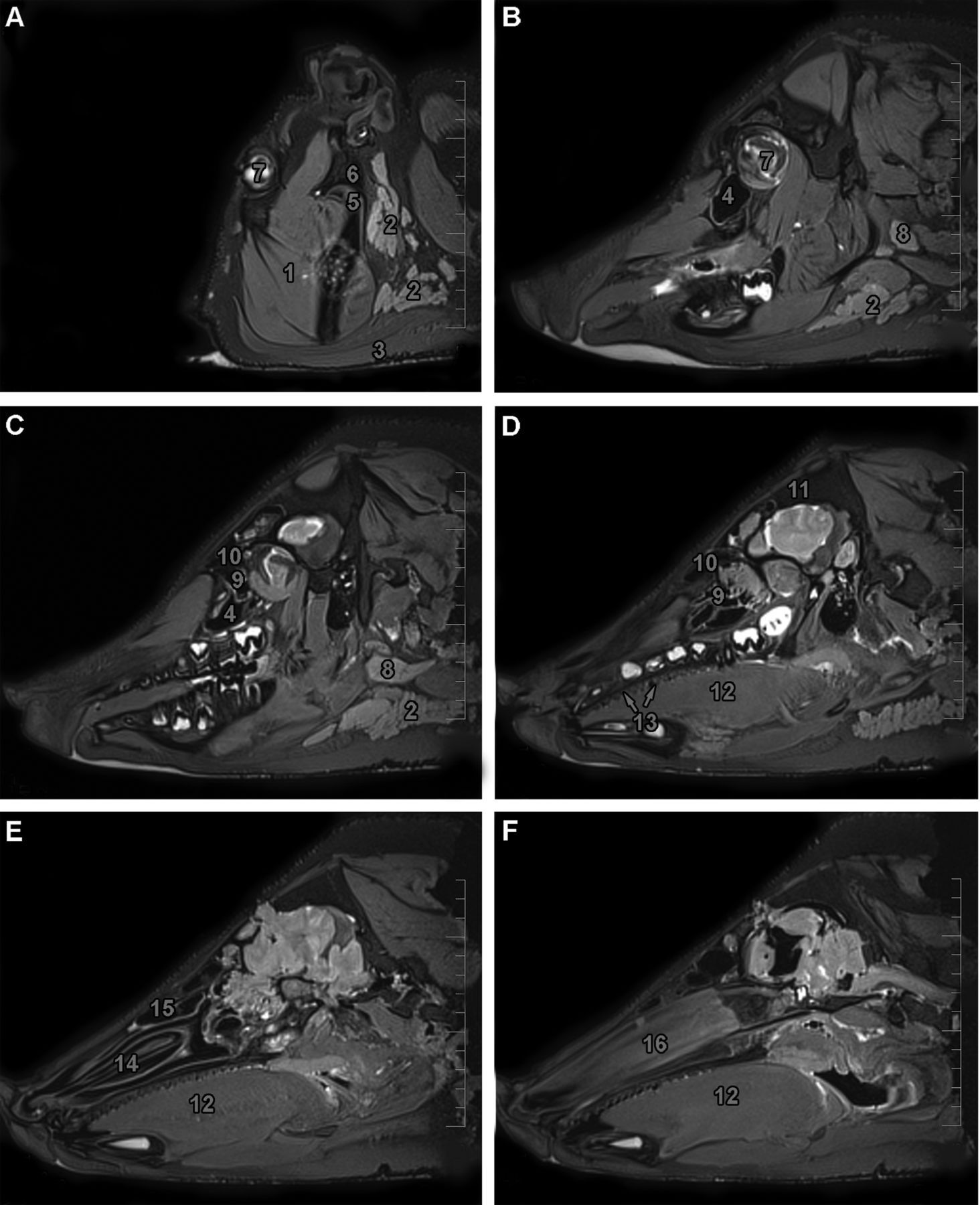
The same types of major salivary glands are found in humans as in the pig: the parotid gland and the submandibular and sublingual glands (Figure 4). Minor salivary glands are localized throughout the oral cavity in the buccal, labial, palatal and lingual regions. In contrast to humans, pig buccal glands are arranged into two lines: dorsal and ventral.
The parotid gland (
The mandibular gland (
There are two sublingual salivary glands in the pig. The monostomatic sublingual gland (
The salivary glands in rodent models are well described and often used. However, they exhibit several specifics that hinder their use by many researchers. These limitations include the small volume of the salivary glands, the thin diameters of the gland ducts and also the short lifespan of these animals. 29 Therefore, the pig is a more suitable model for these studies due to the similar size of their salivary glands and because pigs share many morphological and physiological characteristics with human glands, including the ductal system and their structure. Pigs are also widely used for salivary gland research because their saliva flow rate is very similar to that in humans. 29,30 Furthermore, the histological structure of the submandibular gland is characterized by a parenchyma of mixed acini and serous acini in both the human and pig. 31 Typical serous acini of the parotid gland have also been found to be microscopically and histochemically similar. 32
Investigators and clinicians have been paying attention to salivary glands due to the fact that these structures are very often affected in patients with head and neck carcinoma, which is treated by ionizing radiation. 33 Studying radiation-induced structural and functional changes in salivary glands is important for human oncology treatment. 34,35 The similarity between porcine and human physiology and the availability of slaughterhouse tissues suggests the use of porcine parotid cells as a model for amylase secretion. 36 Also, localized gene transfer to salivary glands has great potential for the treatment of salivary gland, systemic, oral and upper gastrointestinal tract diseases. Numerous studies on rodents have shown that salivary glands can secrete transgenic secretory proteins into either the saliva or bloodstream. 30,37 However, there are still many unsolved questions regarding this problem, so minipig salivary glands, given their volume and morphological similarities to human salivary glands, may be useful as a large animal model for preclinical gene transfer experiments. 38,39
Clinical relevance of the pig model to human dental diseases
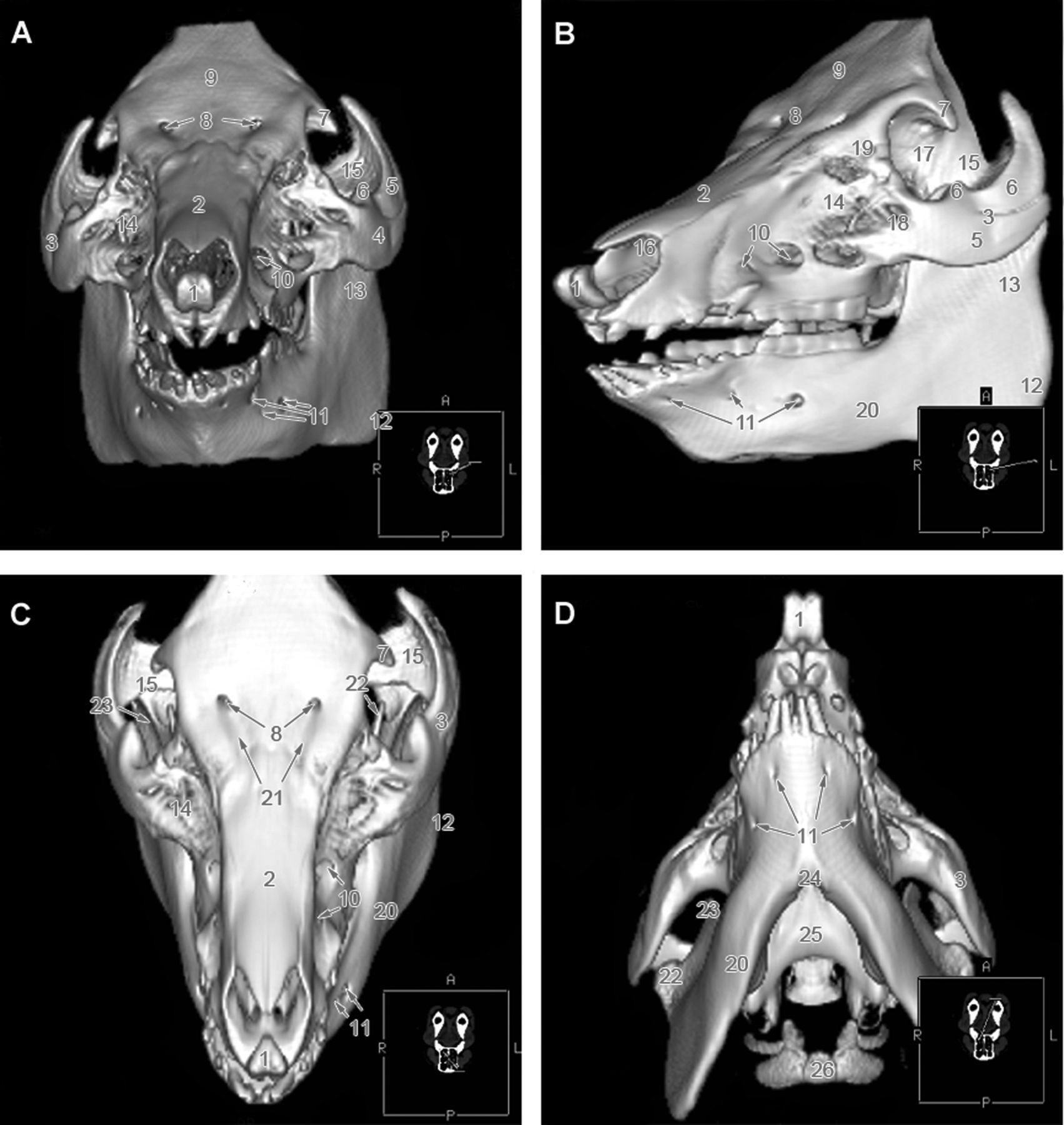
The pig has been used as a dental model for a long time as they share the bunodont and brachyodont type of dentition with the same pattern of enamel mineralization. 40,41 Furthermore, both humans and pigs have diphyodont and normodont dentition with four types of teeth in the permanent dentition, each with a specific size and shape (Figures 2 and 4). The dental formula of deciduous dentition in the pig is 3i 1c 3p in all dental quadrants and 3I 1C 4(3)P 3M for permanent dentition; 29 in some cases, the first premolar is markedly smaller than the other teeth or it can be absent, as also seen in some other domestic animals where the presence of these teeth is rather rare. This tooth is often called ‘dens lupinus’.
While humans are born edentulous, and the first deciduous teeth erupt at approximately 6 to 10 months after birth, newborn piglets usually have eight erupted teeth: 42 they are the third incisor and canine in all dental quadrants. These teeth are called ‘needle teeth’ and are often cut off because they can injure the mammary gland of the sow. Deciduous dentition in the pig is complete at 6 to 8 months of age and the permanent teeth erupt from 4 to 24 months after birth.
Recently, significant attention was paid to the possibilities of tooth replacement in humans. The regeneration of a functional tooth is one of the most promising therapeutic strategies for the replacement of lost or damaged teeth.
43
With the enhancement in tissue engineering and stem cell biology, several possible options for tooth replacement have been developed. Current research is focused on improvements in artificial dental implantations followed by periodontal apparatus recovery or the
From a clinical perspective, the most important part of the tooth is the root, which forms the support for the (natural or artificial) crown, as the crown alone cannot fulfil normal tooth functions without a viable root. 49 Stem cells from human apical papillae or periodontal ligament stem cells have been successfully used to form the root/periodontal complex of porcelain crowns in the minipig. 49
Dental implants were tested for their stability and healing process in minipig maxillae using different types of dental implants, coatings and rate of osseointegration in different age groups. 50–52 Recently, growth factors such as bone morphogenetic protein (BMP) and collagen were shown to support the healing and osseointegration process after dental implantation. 53–55 The pig mandible was shown to be less suitable than the maxilla for testing dental implants for two main reasons. Firstly, there is the superficial position of the large inferior alveolar canal. During insertion of the implant, penetration of the superior wall of the inferior alveolar canal is likely in pigs and must be taken into account. Secondly, the canines (especially in male pigs) fill a major part of the mandibular bone. In cases where there is a problem with these teeth, it is too complicated to extract them without ruining the complex structure of the bone. Therefore, proper implantation in the pig mandible is more difficult and the rate of successfully implanted teeth was only reported to be about 60%. 54
Tooth components of pigs on normal and low phosphorus diets were compared 56 and the effect of tooth extraction on bone mineralization apposition was analysed. 57,58 As porcine alveolar bone shows a similar bone mineral density and bone mineral content to human alveolar bone, 59 it can be used as a model for human implantation techniques or dietary effects on tooth mineralization.
Periodontal diseases are ranked among the most frequent health problems in humans. At present, there is no ideal therapeutic approach for the management of periodontitis or for achieving optimal periodontal tissue regeneration.
60
From the clinical aspect, the loss of periodontal supporting tissue caused by inflammatory periodontal disease is the main complication.
61
Similar symptoms were observed in a pig after the age of 6 months – swollen gingiva, plaque and calculus formation, 1–2 mm red collarettes on marginal gingiva and bleeding on probing. The inflammatory process in periodontal tissues in pigs was similar to that seen in human periodontal diseases;
29
thus, the ability of culture cells replanted from alveolar bone and the periodontium to form new periodontal tissues was investigated in pigs.
61
Cultured cells were found to contribute to the formation of new cementum, bone and attachment tissues and they prevented epithelial downgrowth during wound healing.
61
Furthermore, the possibility of using autologous periodontal ligament stem cells (PDLSCs) to treat periodontal defects in a porcine model of periodontitis was studied.
60
Autologous PDLSCs were obtained from teeth extracted from minipigs. Cells were cultured
Clinical relevance of the pig model to human nasal cavity diseases
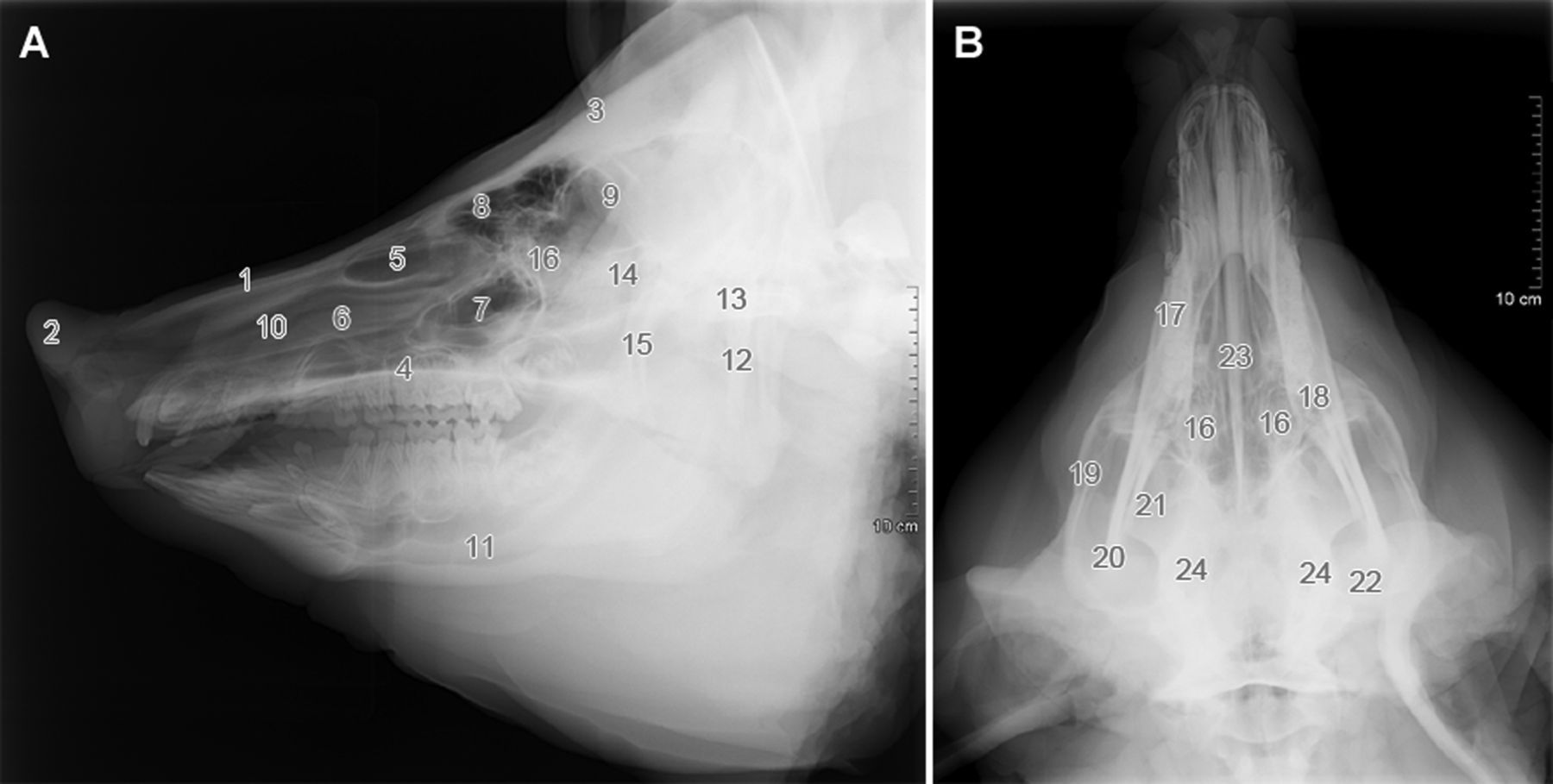
The nasal cavity of the pig is very long and narrow (Figure 4). The bony roof of the nasal cavity is almost complete rostrally on account of the long nasal and rostral bones.
Paranasal sinuses in the pig are separated into two main complexes (maxillary and frontal) and another two small sinuses (sphenoid and lacrimal). The maxillary sinus projects into the lacrimal and zygomatic bones (Figure 4). Caudally, the maxillary sinus extends to a transverse level, passing through the last molar tooth. The frontal sinus (
Discovering the pharmacokinetics of drugs administered to the nasal cavity is the aim of many experimental studies. The interest in delivering drugs through the nasal mucosa (mucus and mucociliary clearance, enzymatic degradation, immunological factors, blood flow and the deposition of drugs in the nasal cavity) of experimental animals is important for researching non-invasive treatments in humans. 64,65 The pig model was used to study the effects of the systemic administration of the nitric oxide synthesis inhibitor on the vasculature of the pig nasal mucosa, 66 and to test different vasodilators (capsaicin, resisiferatoxin) on the nasal mucosa and superficial skin. 67 Furthermore, the airway cell biology of the pig is similar to that of humans, therefore recombinant adeno-associated virus (rAAV)-mediated gene therapy was used in the lungs for the treatment of cystic fibrosis. 68,69
Nasal polyps develop from the respiratory mucosa. However, the inflammatory conditions in the nasal mucosa that may play an important role in the aetiology and pathogenesis of nasal polyp formation are not fully understood.
70
The nasal mucosa of the pig can be used as an experimental model in the study of
The pig was tested and found to be a suitable animal model for creating and closing oronasal communications.
80
Furthermore, whether or not these defects could be closed using biodegradable materials was also analysed.
81
Although bone formation in maxillary sinus rafting or dental implants is routinely used in humans, surgeons cannot take histological samples to gain a better understanding.
82
However, a detailed analysis is possible in the pig, which offers new opportunities for studying these processes in
Clinical relevance of the pig model to human maxillary bone diseases
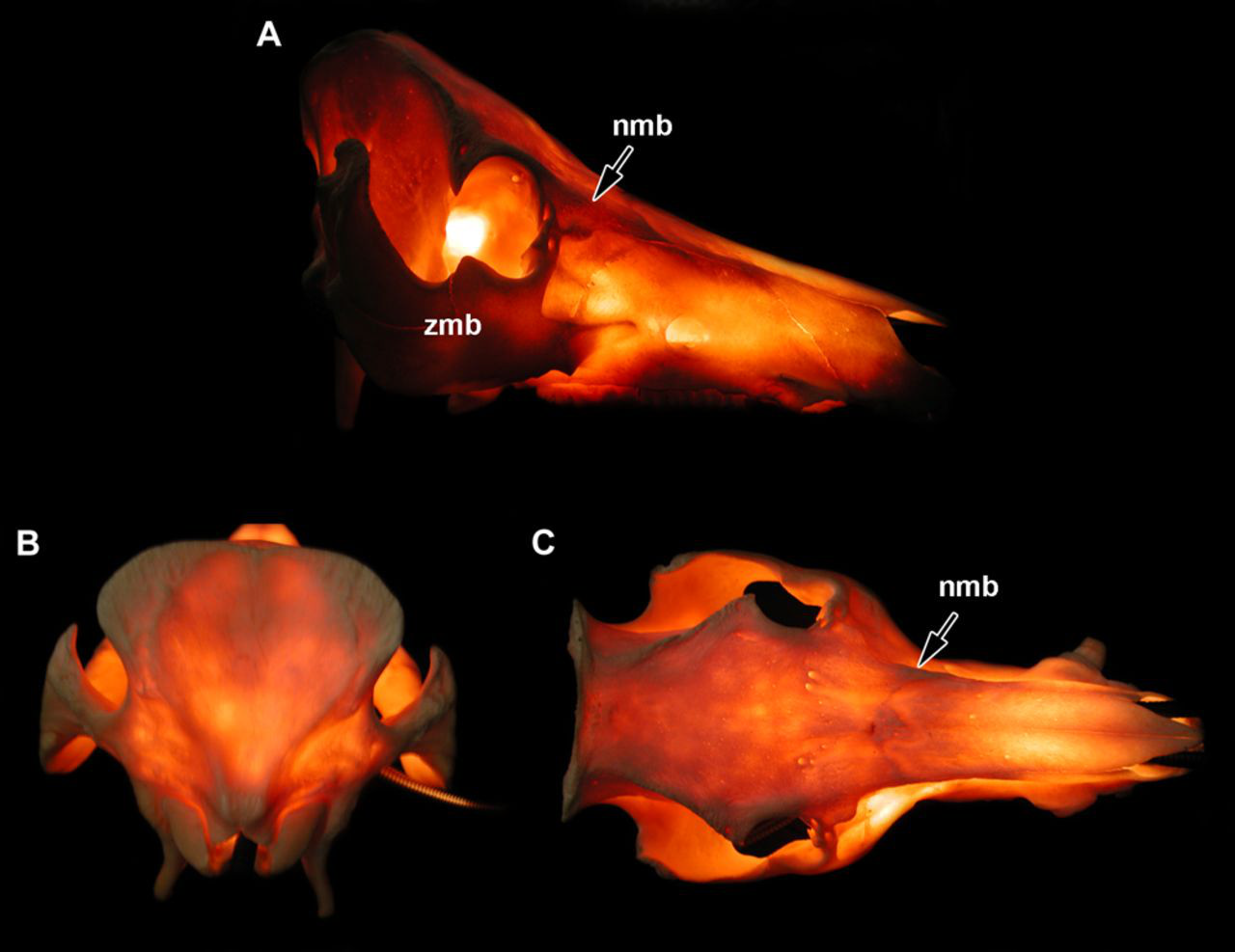
The maxilla is the main bone of the upper jaw that carries the upper premolar and molar teeth (Figure 2). The facial surface is smoothly concave. The infraorbital opening (
Bone pillars or buttresses are areas with thicker bone tissue supporting the maxillofacial region. In both humans and dogs, they represent clinically important structures regarding the management of craniofacial fractures. 85 The buttresses can be identified by transillumination of the skull (Figure 5). In humans, they are divided into three main areas: the medial (nasomaxillary) buttress, the lateral buttress (zygomaticomaxillary) and the posterior (pterygomaxillary) buttress, similar to the pig (Figure 5). The lateral buttress is the most prominent and seems to play a major role in supporting the upper jaw of the pig. Similar to the dog and in contrast to humans, the pig does not have a bony support around its entire orbit, but its caudal part is supported by the orbital ligament. Therefore, the zygomatic arch bridges and transmits all forces and, therefore, it is reinforced by thick and dorsally protruding bony tissue.
Regarding bone anatomy, morphology, healing and remodelling, the pig is considered to be closely representative of humans and therefore a suitable species of choice. 86 Pigs have a denser trabecular network 87 but the structure of the lamellar bone is similar to that in humans. 88 When compared to the bone composition of various different species, porcine bone has been shown to have similarities to human bone in terms of bone mineral density and bone mineral concentration. 59 Also, the bone remodelling processes are similar, as well as the mechanical parameters during trabecular and intracortical BMU-based remodelling. 87,88 A comparison of the regeneration rate of bone in dogs, pigs and humans 89 revealed that pigs have a more similar rate of bone regeneration to humans than to dogs (dog, 1.5–2.0 mm/day; pig, 1.2–1.5 mm/day; human, 1.0–1.5 mm/day). In addition, in a study of the effects of fluoride on cortical bone remodelling in growing pigs, the results showed that pigs have a similar cortical bone mineralization rate to humans. 90
Due to these similarities, a pig model was recently used to study bone formation after maxillary distraction. 9 This surgical procedure is mainly recommended for children with cleft palates where the upper teeth are situated behind the lower teeth. The Le Fort I device was tested for its stability and the pig was established as a useful model for studying the healing mechanism during distraction osteogenesis. 9
Experiments in the minipig may also help in designing internal non-bioresorbable and bioresorbable distraction devices. The pig maxilla can be used in tissue-engineering approaches for coordinated autologous tooth and bone reconstruction, and it also provides a basis for future improvements in this technique for eventual clinical use in humans. 91,92
As the maxillary bone is a very thick and massive bone, recent studies of distraction osteogenesis used a more easily accessible bone: the frontal bone.
92–94
Temporal and parietal bone segments were used to evaluate the optimal timing and long-term effects of fixation techniques on a growing cranium
Clinical relevance of the pig model to human orbital diseases
The orbit of the pig is relatively small compared with humans (Figures 2 and 3). The infraorbital margin is formed by the lacrimal and zygomatic bone (
The adult pig can effectively serve as a model for resident training in lateral canthotomy or as a dummy orbit for the teaching and training of diagnostic and eye surgery procedures. 98,99 To date, there is no precise method for controlling and monitoring expansion to induce normal growth in the developing facial skeleton, but the pig orbit is suitable for stimulating normal orbital growth in the neonatal facial skeleton. 100 Another application of the pig orbit is in laser surgery for the detection of temperature distributions in orbital tissues during and immediately after the application of CO2 and Nd:YAG laser irradiation to muscle tissue adjacent to the optic nerve. 99,101
Moreover, the pig orbit is also an excellent experimental model for the development and testing of alloplastic materials that could be used in orbit reconstruction after trauma, tumours or developmental abnormalities. These materials would degrade slowly and have osteoconductive properties to allow their replacement and remodelling by osseous tissue. These properties should be tested in a pig model in order to determine their uses in human medicine. 102–104
Clinical relevance of the pig model to human mandible diseases
The mandible or the lower jaw consists of the body (
It must also be pointed out that the presence of multiple mental foramens in pigs makes local anaesthesia more difficult than with humans. Only large, local infiltration anaesthesia is possible, with no possibility of selectively anaesthetizing specific nerves, therefore making this an imprecise procedure. Furthermore, the thick layer of fatty tissue covering the ramus mandible can make access to this area difficult.
Current strategies for jaw reconstruction require multiple surgeries to replace bone and teeth. The pig mandibles serve to help improve tissue engineering approaches for coordinated autologous tooth and mandible reconstructions and as a model for providing a basis for future improvements in these techniques for eventual clinical use in humans. 105–107 Pigs are suitable experimental animal models for studying the blood supply in the mandibular cortex and the design of osteosynthetic plates and screws. 108,109 The pig mandible is also necessary for understanding the mechanisms and molecular events that regulate bone formation during distraction osteogenesis. 110–112 Moreover, the pig mandible helps in testing endoscopic procedures for the placement and activation of a distraction device for mandibular advancement, 113 and for the endoscopic resection of mandibular angles. 114
Stem-cell-based tissue engineering is currently viewed as a very promising method for bone regeneration, 115 and the pig can be a very useful model. Stem cells of miniature pigs were isolated from deciduous teeth, the ilium or bone marrow and engrafted on to the critically sized bone defects generated in pig mandible models. 11,105,106,116–119 The results indicated that these cells were able to engraft and regenerate bone in order to repair mandibular defects. A further step will be to transfer the acquired results to human clinical trials.
Clinical relevance of the pig model to human temporomandibular joint diseases
The morphology of the pig temporomandibular joint (TMJ) closely resembles that of humans,
120
including its internal structures, such as the articular disc, and its attachments.
121
The pig TMJ is a simple incongruent joint, similar to that of humans (Figure 4). Except for the small mastoid eminence (
Human disorders of the TMJ are represented by a wide spectrum of morphological and functional changes that can affect not only the TMJ but also masticators and other areas of the face. This disorder is showing an increasing trend in humans, perhaps due to the influence of psychological stress in the present population. The presence of long-term tenseness and emotional stress is considered to be the main aetiological factor. These tissues are difficult to visualize dynamically and therefore the
The pig was recently proposed as the best non-primate model for human TMJ disorders; it has been used for direct measurements of TMJ tissue deformation and load during biting. 122,123 The pig can also be useful in tissue engineering of the articular disc as the topographical biochemical and biomechanical parameters of its disc are most similar to the disc in humans 124,125 or in the development of new therapies for degenerative TMJ diseases and post-traumatic conditions. 126
Furthermore, the invasive arthroscopic surgery of the TMJ is technically demanding and requires the acquisition of adequate arthroscopic skills that can hardly be obtained from patients alone. Thus, the pig TMJ serves as a reliable educational model for arthroscopic surgery and its further refinement in the TMJ. 127
Current applications for clinicians
The completion of pig genome sequencing has opened up the pig model for use with modern molecular methods. 1 In recent times, the pig has not just been used as an animal model for surgical treatment, but also for the possibility of targeting diseases via gene therapy. 3 Bone regeneration was found to be enhanced in minipigs after BMP-2 gene delivery using liposomal vectors, 128,129 adenovirus-mediated transfer 130 or the gene delivery was combined with collagen carrier. 128 Moreover, the protein can be also directly provided on hyaluronan-based hydrogel mixed with hydroxyapatite nanoparticles 131 to improve the healing of cranial defects. As the formation of new bone is also accompanied by angiogenesis, 132 the using of growth factors represent an ideal method for the cranial reconstruction and the possibility to repair a large-scale skull defect 133 Furthermore, mesenchymal stem cell transplantation can be used to reconstruct orofacial tissue. 134 Therefore, we hope that our recent study will open up the pig model to further applications by surgeons, as well as veterinary researchers.
Footnotes
ACKNOWLEDGEMENTS
This work was supported by the Czech Science Foundation (grant 304/08/P289). The lab runs under IRP IPAG No. AVOZ 5045015.
