Abstract
Buprenorphine is commonly used as (part of) postoperative analgesic treatment with dosage dependent side-effects such as pica behaviour. No strict consensus exists about the optimal dosing interval of buprenorphine, as its duration of action has been described as being in the range of 6–12 h. In this study, dosing intervals of 8 h (thrice-a-day) and 12 h (twice-a-day) for buprenorphine in a multimodal analgesic strategy (concurrent administration of a non-steroidal anti-inflammatory drug) were compared on food intake, weight and side-effects (gnawing on plastic Petri dishes and growth rate, indicative of pica behaviour) in rats. The food intake and weight of both intervals were comparable, as the animals from the twice-a-day group did not lose more weight or consumed less food during the analgesic period. The rats from the thrice-a-day group suffered from more side-effects, as the growth rate was decreased and more plastic was gnawed on. It is recommended to carefully evaluate analgesic and side-effects when using buprenorphine. When side-effects are observed, the possibility of increasing the dosing interval of buprenorphine should be explored. In this study, increasing the dosing interval of buprenorphine in a multimodal analgesic regimen resulted in reduced unwanted side-effects, without increasing weight loss or decreasing food intake. Although this is suggestive of provision of comparable analgesia, future studies including more pain-related readout parameters to assess the effect of the dosing interval on analgesic efficacy are recommended.
In rodents, buprenorphine has been shown to be an effective analgesic in a wide range of experimental pain models 1 and is a frequently used opioid in laboratory animals for treating postoperative pain. 2,3 In the rat, a dose of 0.05 mg/kg is recommended. 4 However, the optimal dosing scheme and treatment duration remain under debate. 5 This is due to complex pharmacodynamics, 6 pharmacokinetics 7 and inconsistent reports with respect to onset time, 6 peak effect 2,8,9 and duration of analgesia. 1,2
Besides its broad analgesic potency, buprenorphine is known to have various (dose-dependent) side-effects, including increased locomotor activity 10 and pica behaviour 2,11,12 (often seen as hoarding or ingestion of bedding material), which predispose for decreased growth rates, gastric obstruction and occasionally death. 13 Rodent strain, buprenorphine dosage, dosing interval, treatment duration, as well as presence and severity of postoperative trauma and availability of non-food substrates have all been implicated to contribute to (and interact with) pica behaviour and delayed postoperative weight gain. 2,11,12
Good pain management comprises frequent (re-)assessment cycles to evaluate both desired (i.e. adequate analgesia) and unwanted (side-)effects (i.e. pica, decreased growth rate) of the analgesic therapy. The duration of effective analgesia depends on various factors including the degree of pain (i.e. type of surgery), dose received and whether other types of analgesics are co-administered, thus providing so-called multimodal analgesia. Multimodal analgesia is an effective way of treating postoperative pain as different types of analgesics act on different sites in the pain pathway with different modes of action. 14 A great advantage of multimodal analgesia is that it may allow for lower dosages or increased dosing interval of individual drugs to obtain adequate analgesia, compared with single drug therapy (unimodal analgesia). As a consequence of a decreased dosing regimen, the side-effects of each individual drug are decreased, whereas the analgesic effect remains unaltered or even slightly improved. 15 Therefore, multimodal analgesia is preferred over unimodal analgesia (especially when the dosing scheme of a single agent is not well established).
In this study, two dosing intervals (8 and 12 h) of postoperative buprenorphine treatment (0.05 mg/kg) in the rat were compared for their efficacy in a multimodal 5 postoperative analgesic treatment (with the non-steroidal anti-inflammatory drug [NSAID] meloxicam) following neurocranial surgery with a balanced anaesthesia technique. Weight of the rats, food intake and pica behaviour were assessed on a daily basis. General animal behaviour and posture were qualitatively observed before and after handling.
Materials and methods
Animals
Adult male Wistar rats (HsdCpb:WU, Harlan Netherlands BV, Zeist, The Netherlands; 8 weeks old, body weight 250–300 g at the time of arrival, n = 38) were housed in a clear 1500 U Eurostandard Type IV S cage (48 × 37.5 × 21 cm) in pairs of two, separated from each other by a metal grid partition inserted in the middle of the cage. The rats, which are social animals, 16 were able to engage in a limited form of social interaction through sight, smell and sound without damaging each other's head-mounted SEP-measuring receptacle (for details see ‘Surgery’). The animals were provided with bedding material (Aspen chips), ad libitum access to food (CRM, Expanded, Special Diets Services, Witham, UK) and water, and paper tissues as cage enrichment. The environment was controlled (temperature, 21 ± 2°C; humidity, 47 ± 3%), with an inverse 12 h light–dark cycle (lights off from 06:00 to 18:00 h) and a radio playing constantly at a low volume as background noise. Animals were handled at least twice a day for 10 min by the experimenters.
Ethical consideration
The experimental protocol (DEC-DGK number: 2009.I.02.011) was approved by the Animal Experiments Committee of the Academic Biomedical Centre, Utrecht, The Netherlands. The Animal Experiments Committee based its decision on ‘De Wet op de Dierproeven’ (The Dutch ‘Experiments on Animals Act’, 1996) and on the ‘Dierproevenbesluit’ (The Dutch ‘Experiments on Animals Decision’, 1996). Both documents are available online at
Surgery
After an acclimatization period of two weeks, the animals underwent surgery for permanent implantation of epidural electrodes. Animals were transported individually from their housing to a separate room, where anaesthesia was induced under red lighting with 0.25 mg/kg of fentanyl (intraperitoneally; Fentanyl Janssen, Janssen-Cilag BV, Tilburg, The Netherlands; 0.05 mg/mL of fentanyl citrate) and 0.15 mg/kg of dexmedetomidine (intraperitoneally; Dexdomitor, Pfizer Animal Health BV, Capelle a/d IJssel, The Netherlands; 0.5 mg/mL of dexmedetomidine hydrochloride). As soon as the pedal reflex was absent, the animal was transported to the surgery room and, after intubation, anaesthesia was maintained with isoflurane in 100% O2. The animals received 8 mL of saline (subcutaneously) to support normal fluid balance, 5 mg/kg of enrofloxacin (intraperitoneally, Baytril 2.5%, Bayer BV, Mijdrecht, The Netherlands) and eye ointment (Ophtosan Oogzalf, Produlab Pharma Raamsdonkveer, ASTfarma BV, Oudewater, The Netherlands; 10,000 IE of vitamin A palmitate per gram).
For the surgery, the animal was positioned in the stereotactic apparatus (model 963, Ultra Precise Small Animal Stereotaxic, David Kopf Instruments, Tujunga, CA, USA). Body temperature was monitored using a rectal probe thermometer and maintained at 37–38°C with an adjustable electrically heated mattress. In addition to clinical assessment (i.e. pedal reflexes), respiratory rate, heart rate, SpO2 and inspired and expired CO2 were monitored continuously and anaesthetic administration was adjusted appropriately. After the skin incision but before detachment of the periostium from the neurocranium, 3 mg/kg of lidocaine solution (Alfacaine 2% plus adrenaline, Alfasan BV, Woerden, The Netherlands, 20 mg/mL of lidocaine hydrochloride and 0.01 mg/mL of adrenaline) was applied. Four small wired stainless steel screws (tip diameter 0.6 mm, impedance 300–350 Ω; Fabory DIN 84A-A2, Borstlap BV, Tilburg, The Netherlands) were implanted epidurally over the vertex (4.5 mm caudal to bregma, 1 mm right from midline), S1 (2.5 mm caudal to bregma, 2.5 mm right from midline) and left and right frontal sinus (10 mm rostral to bregma, 1 mm lateral from midline). An electromyography (EMG) electrode was implanted in the trapezoid muscle using suture material and the EMG wire tunnelled under the skin to the receptacle. All electrodes were wired to an eight-pin receptacle (Mecap Preci-Dip 917-93-108-41-005, Preci-Dip Durtal SA, Delémont, Switzerland) and fixed to the skull with dental cement (Simplex Rapid, Associated Dental Products, Ltd, Swindon, UK). The skin was closed in a single layer around the receptacle.
After the surgery, anaesthesia was antagonized with 0.6 mg/kg of atipamezole (intraperitoneally; Antisedan, Pfizer Animal Health BV, Capelle a/d IJssel, The Netherlands), and 0.05 mg/kg of buprenorphine (intraperitoneally; Buprecare, AST Farma BV, Oudewater, The Netherlands) in a separate room under red lighting. Rats were returned to their home cages once they regained purposeful locomotion.
Postoperative analgesia
Postoperative analgesia was provided by 0.2 mg/kg of meloxicam (subcutaneously; Metacam, Boehringer Ingelheim, Alkmaar, The Netherlands, 5 mg/mL) at 24 h intervals for two days after surgery. Additionally, the thrice-a-day group (n = 19) received 0.05 mg/kg of buprenorphine (subcutaneously) at 8 h intervals for three days after surgery, and the twice-a-day group (n = 19) received 0.05 mg/kg of buprenorphine (subcutaneously) at 12 h intervals for three days after surgery.
Measurements
Body weight and food intake of all 38 rats (n = 19 per group) were recorded daily, starting from before the start of the surgery (baseline) until all of the rats reached their preoperative weight. To obtain a representative baseline of food intake, the food intake of rats was recorded daily from six days before the surgery, by weighing the food pellets from the cage's food rack. Additionally, pica behaviour was measured from the surgery day till four days postsurgery (n = 19 in the twice-a-day group and n = 9 in the thrice-a-day group). Until four days after surgery, rats were provided with hard food pellets (CRM, Expanded, Special Diets Services) and food pallets softened by hand-warm water on a plastic Petri dish, which was refreshed every 24 h. Upon removal from the cage, the Petri dishes were cleaned and weighed to get an indication of the pica behaviour.
Results
Observation
No behavioural indication of postoperative pain or discomfort was observed in either group during any time point in the study. Although only qualitatively scored it was noted that following handling and buprenorphine administration, often increased activity (running, jumping and gnawing on the bars of the top of the cage or at hard food pallets) was observed within minutes following buprenorphine administration in both groups (data not shown).
Statistical analysis
Calculations were performed with Microsoft Excel 2003 and statistical analyses with SPSS 16.0 (SPSS Inc, Chicago, IL, USA); P values below 0.05 were considered to be significant, and a Sidak correction was applied in case of post hoc testing. Statistical graphics were generated using Sigmaplot 11 (Systat Software Inc, Chicago, IL, USA). The data were statistically analysed using a mixed model regression. A backward strategy was adopted in which all non-significant interaction terms were removed. Grand mean centering was used (i.e. values are centred at 0) for all fixed factors, so the intercept could be interpreted and collinearity was prevented. 17 The Q–Q plots of the residuals indicated a normal distribution for all variables.
For the rat's weight and food intake data, both the absolute measurements and relative measurements (relative to baseline) were analysed. The best fit was obtained by using the model with a random intercept and random slope for day. Fixed factors were group (group 1, group 2), day (day 1 till day 12) and their interaction. Simple effects were tested in a post hoc test using a nested model, estimating the effects of day 2 until day 12 compared with day 1 (baseline) per group. For the weight of the Petri dishes, the best fit was obtained by using the model with a random intercept. Fixed factors were group (twice-a-day group, thrice-a-day group), postoperative day (day 1 till day 4) and their interaction.
Weight
A significant day × group interaction was found for both the absolute and relative weight (F
12,274 = 4.45, P < 0.000 and F
11,245 = 5.33, P < 0.000, respectively, see Figure 1). Post hoc analysis showed that the baseline of the absolute weight did not differ between groups: t
41.79 = 1.51, P > 0.05. Furthermore, post hoc analysis showed that animals from the twice-a-day group significantly increased in weight (compared with baseline) at postoperative day 9, while animals from the thrice-a-day group increased in weight at postoperative day 10 (see Figure 1a). However, no differences in relative weight were found between groups on any of the postoperative days (see Figure 1b).
The absolute (a) and relative (b) weight of the rats one day before till 12 days after surgery. The grey area indicates the period of postoperative analgesia. (a) Rats from the twice-a-day group reached their baseline weight one day earlier compared with the thrice-a-day group. (b) No differences were found between relative weight of both groups on any of the days. *Sidak; P < 0.05 compared with baseline (a). †Sidak; P < 0.05 between groups (b). Data are represented as mean ± SEM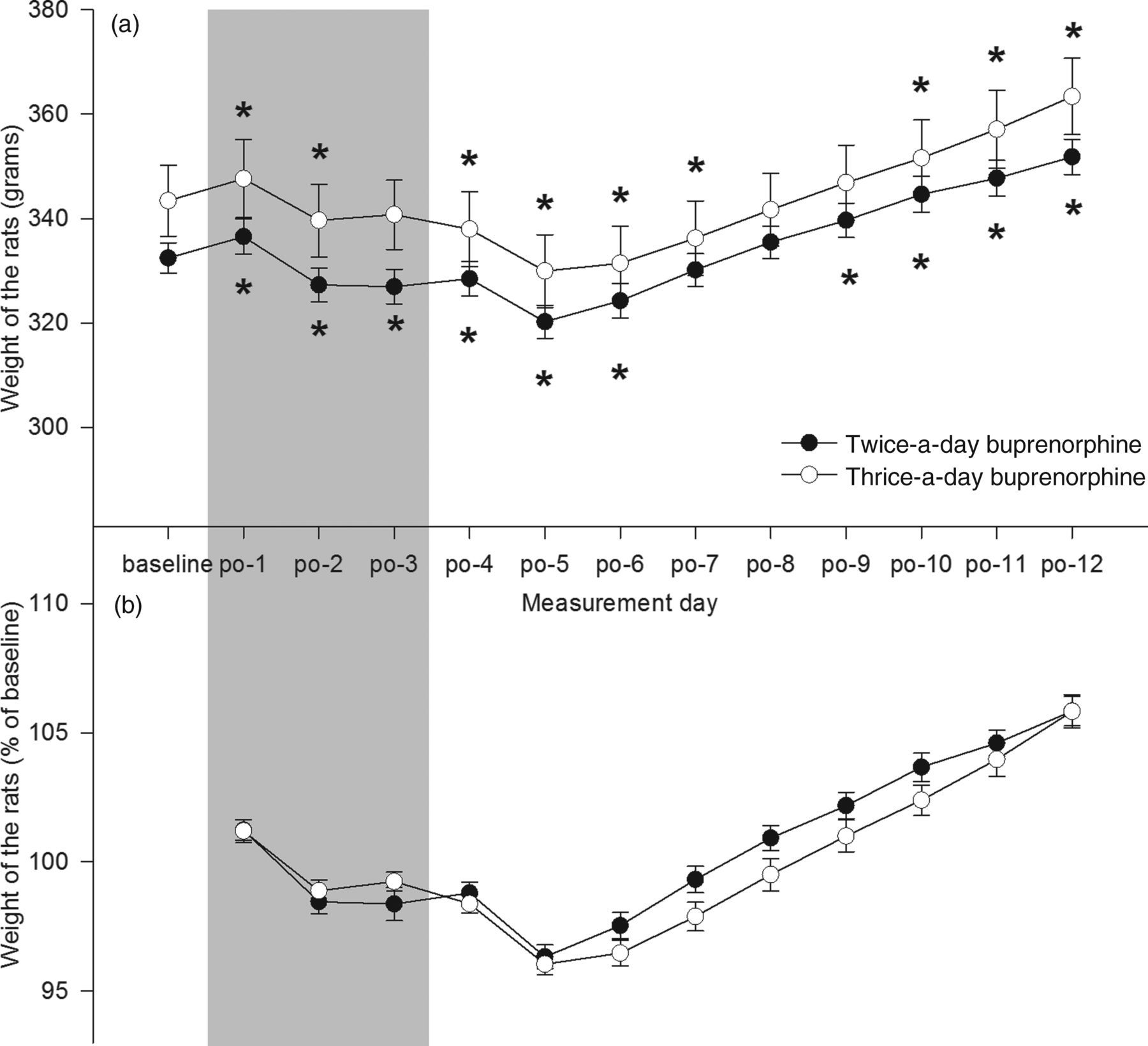
Food intake
A significant day × group interaction was found for both the absolute and relative food intake (F
12,313 = 2.65, P < 0.01 and F
11,294 = 2.58, P < 0.01, respectively, see Figure 2). Post hoc analysis showed that the baseline of the absolute food intake did not differ between groups: t
213 = −0.31, P > 0.05 (see Figure 2a). Relative food intake differed between groups on postoperative days 2 and 11 (see Figure 2b).
Absolute (a) and relative (b) food intake of the rats over time. The baseline is the average food intake during six days before the surgery. Subsequently, food intake was measured till 12 days after surgery. The grey area indicates the period of postoperative analgesia. Animals from the twice-a-day group did not consume less food during the analgesic period. *Sidak; P < 0.05 compared with baseline (a). †Sidak; P < 0.05 between groups (b). Data are represented as mean ± SEM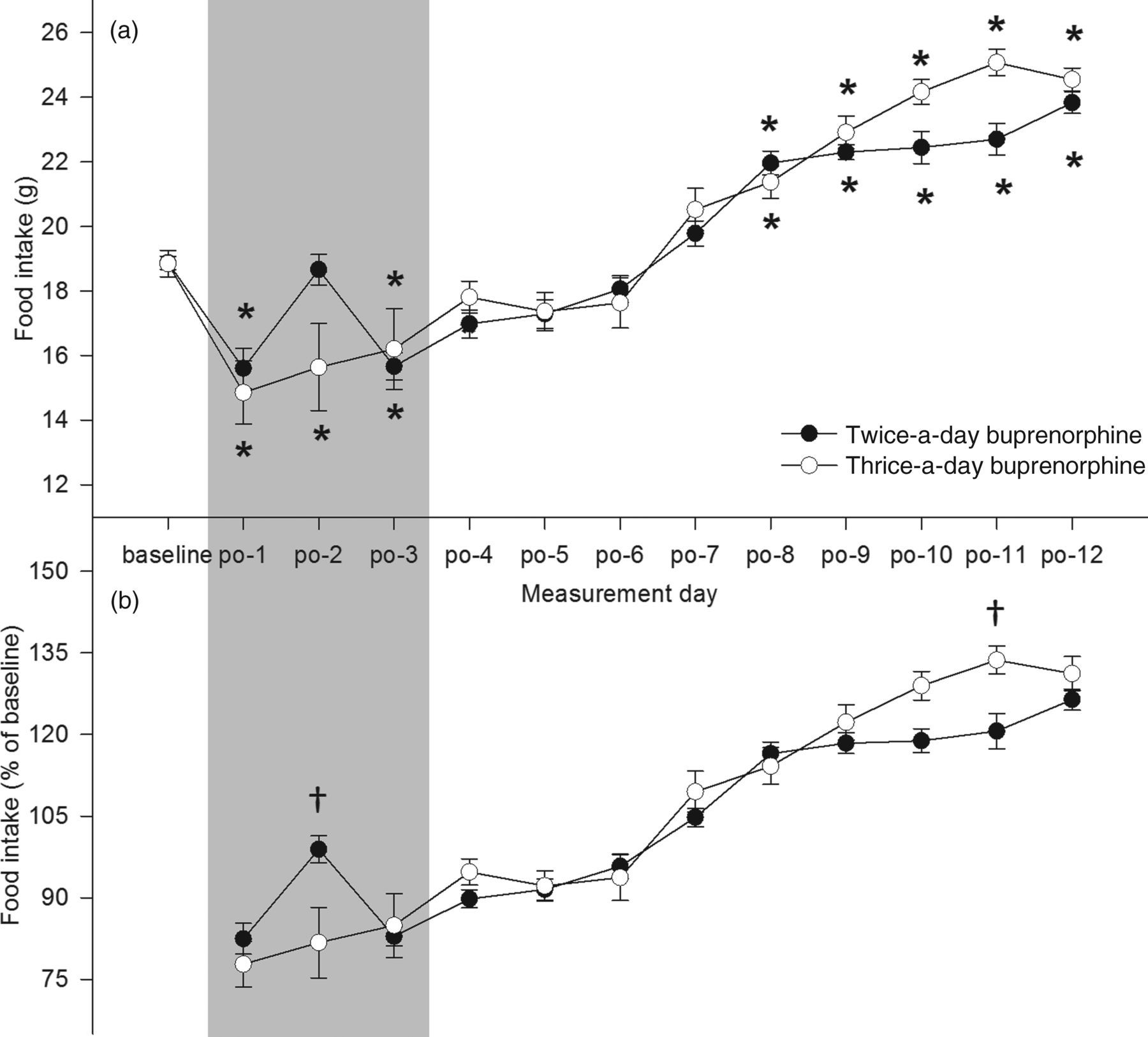
Pica behaviour
The effects of day × group and day were non-significant (F
3,87 = 0.52, P > 0.05 and F
3,87 = 1.46, P > 0.05, respectively). The weight of Petri dishes collected following the surgery showed a significant effect between groups with the dishes of the thrice-a-day group weighing less compared with those of the twice-a-day group (F
1,29 = 15.53, P < 0.000), indicating that the thrice-a-day group gnawed significantly more on the plastic dishes (see Figures 3 and 4).
The effect of the interval of buprenorphine injections (0.05 mg/kg) on pica behaviour. Animals from the twice-a-day group gnawed significantly less on the Petri dish compared with animals from the thrice-a-day group (significant main effect of P < 0.000). No significant difference over days was found. See text for details. Data are represented as mean ± SEM The effect of the interval of buprenorphine injections (0.05 mg/kg) on pica behaviour. Representative Petri dishes are shown from a rat out of the twice-a-day (left) and thrice-a-day (right) group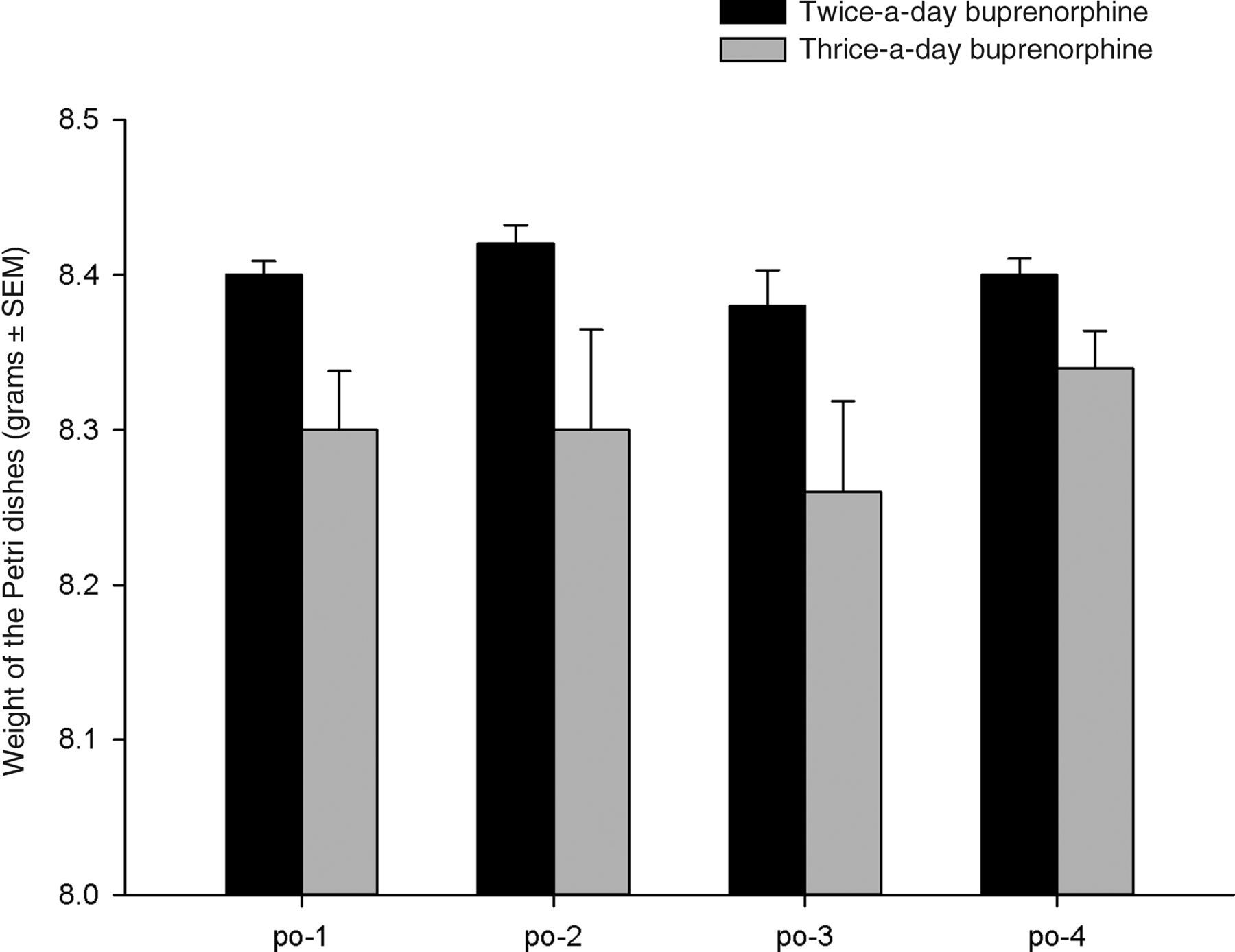

Discussion
The main finding of this study is that no difference in weight gain and overall food intake was present between rats receiving buprenorphine with a dosing interval of 12 h (twice-a-day group) and with a dosing interval of 8 h (thrice-a-day group) after neurocranial surgery, when applying a multimodal analgesic strategy. However, rats receiving buprenorphine thrice a day were more likely to suffer from side-effects such as pica, compared with the twice-a-day group.
No differences were found in the rats' postoperative weight changes between groups, until at least 12 days after surgery. Rats from the twice-a-day group did not lose more weight than rats in the thrice-a-day group. Furthermore, rats in the twice-a-day group did not consume less food than rats in the thrice-a-day group during analgesic treatment. The latter could suggest that the analgesic efficacy in both groups was comparable; however this cannot be confirmed with the studied parameters. Assessing pain in (laboratory) rodents poses considerable challenges. Traditionally, indirect and general parameters (as in this study) have been adopted to assess the efficacy of postoperative pain management. However, this allows only retrospective assessment on the ability of the provided regimen to diminish deleterious effect of postprocedural pain like excessive weight loss and food consumption. Therefore, although the comparable weight loss and food intake of the twice-a-day and thrice-a-day group may indicate comparable overall analgesia, these readout parameters are neither very sensitive nor specific for pain. Short interval changes in body weight are particularly sensitive to hydration status (i.e. water intake). However, monitoring water intake does not necessarily increase specificity for postprocedural pain and discomfort, as buprenorphine is reported to increase water intake during multimodal postprocedural pain management. 5 To better assess the analgesic efficacy of the two protocols adopted here, the measured parameters should be expanded with a wider set of indirect and direct pain-related readouts and subsequently integrated to gain specificity. Proposed additional parameters include water intake, the standardized scoring of posture, spontaneous and interactional (e.g. handling) behavioural patterns 18 and facial expression. 19
On the second postoperative day (but not other days), rats from the twice-a-day group consumed significantly more food than rats in the thrice-a-day group. Based on the current data we can only speculate on the underlying mechanism of this group specific increase in food intake on this specific day. Recovery from surgery is a dynamic process 20 characterized by a gradual decrease in, among others, caloric requirement and severity of pain. 21 Adopting a static analgesic regimen for several days increases the risk of over treatment (with respect to pain level and the cumulative dose of drugs administered). It can be speculated that at day 2 in the thrice-a-day group relative overdosing already occurred, resulting in an increase in the negative gastrointestinal side-effects of buprenorphine. 13 In the twice-a-day group relative overdosing may not have occurred until day 3, allowing an unhindered increase in food intake and catching-up on caloric need 5 giving rise to the observed group difference in food intake at day 2. A possible ceiling effect with respect to consequences of relative overdose for food intake may have occurred on day 3.
The observed increased activity following buprenorphine administration is in agreement with earlier reports 10 and has been frequently observed prior and following this study in our laboratory (data not shown). Over the period of postoperative analgesic treatment, the thrice-a-day group showed significantly more gnawing on the Petri dishes and needed one day longer to regain their baseline body weight (i.e. decreased growth rate) than the twice-a-day group. Combining these results suggest that the animals from the thrice-a-day group were more likely to suffer from side-effects such as pica behaviour without resulting in significant differences in body weight and food consumption. Although pica is regularly reported with buprenorphine, 2,11,12 detailed information concerning its clinical course (onset, peak intensity and resolution) associated with postsurgical analgesia is lacking. In this study no difference in pica behaviour was observed over time (i.e. no difference between days), when assessing pica behaviour in 24 h intervals during the three-day treatment plan. However, in future research shorter intervals should be used over a longer time period to provide more detailed knowledge about the course of pica behaviour in the postoperative period.
The dose of meloxicam used in this study is substantially lower than reported in the literature 22 and was based on multimodal pain management experience in our veterinary academic teaching hospital with client-owned rodents. The dosages in the literature are based on unimodal analgesia in experimental pain with maximum effect. However, side-effects of sequential high dose NSAIDs in rodents have been described (see e.g. Ref. 23 ).
In summary, this study demonstrates that, under the specific circumstances (i.e. multimodal analgesia strategy), administering buprenorphine thrice a day does not have apparent beneficial effects regarding food intake and weight loss, while it does increase the incidence of adverse effects (i.e. pica behaviour) compared with administering buprenorphine twice a day. Because the current data do not allow firm conclusions regarding analgesic efficacy, the authors recommend future studies including more pain-related readout parameters to assess the effect of the dosing interval of buprenorphine on analgesic efficacy in a periprocedural multimodal pain management strategy.
Conclusion
When administering analgesia (buprenorphine) to rats in the postoperative period, one should carefully evaluate both the analgesic response and the potential side-effects. When unwanted side-effects such as pica behaviour are observed, the possibility of increasing dosing intervals without decreasing the analgesic therapeutic effect should be explored.
