Abstract
To estimate the glomerular filtration rate (GFR) in conscious rabbits, a single-sample method using the non-ionic contrast medium iodixanol was compared with a three-sample method using the standard agent inulin. Iodixanol and inulin were co-administered intravenously to male New Zealand White rabbits at 60 mg I/kg and 40 mg/kg, respectively, and blood was collected 30, 60, 90 and 120 min later. Serum iodixanol and inulin concentrations were separately determined by high performance liquid chromatography and colorimetry, respectively. Serum urea nitrogen (UN) and creatinine concentrations were also determined. Based on the data from healthy and cisplatin-treated rabbits, the GFR estimated by iodixanol was well consistent with that by inulin. Further, when the GFR decreased to more than 60% of the reference value, serum creatinine concentrations became elevated. However, serum UN concentrations exhibited wide fluctuations, presumably due to a difference in renal handlings. The single-sample method using iodixanol was considered to be an expedient tool in both clinical and research settings, because the stress due to a multi-sample method was reduced.
In humans, glomerular filtration rate (GFR) is the best overall index of renal function in diseases and their treatment and management. Inulin 1 and the non-ionic monomeric X-ray contrast medium iohexol 2 are considered the gold standard tracers for estimating the GFR. For experimental animals, however, inulin requires specialized technical procedures and has low solubility that needs a relatively large volume of injectable solution and blood collection. Meanwhile, iohexol was reported to possess a deteriorating potential in patients with impaired renal function. 3 Here, we focused on the isotonic non-ionic dimeric X-ray contrast medium iodixanol as a novel tracer, because little information is available on the measurement of rabbit GFR under conscious conditions without undergoing surgical manipulations. Iodixanol is rapidly excreted into urine without metabolic degradation and no or very little protein binding 4,5 as was the case in iohexol. Interestingly, iodixanol has been reported to be less nephrotoxic than the low-osmolar contrast media including iohexol in randomized double-blind, prospective multicenter studies using high-risk patients with chronic renal diseases. 3,6 Moreover, iodixanol has twice the amount of iodine in one molecule with low osmolarity compared with iohexol, presumably showing the same pharmacodynamic action as half the exposure dose of the whole body to iohexol.
In humans, the concentration of a tracer in a single plasma sample taken a few hours after injection was previously reported to correlate with renal clearance. 7 Based on this information, Jacobsson 8 devised a formula derived from a simple one-compartment model combined with the distribution volume and optimum time for taking plasma using 99Tcm-DTPA and accurately determined the GFR. To apply the above Jacobsson's formula 8 to rabbits using iodixanol, the GFR estimated by the three-sample method was first compared with that by the single-sample method in healthy and cisplatin-treated animals. Next, the GFR obtained from the single-sample method using iodixanol was compared with that from the three-sample method using the standard tracer inulin. This procedure with iodixanol was essentially the one used for rats, 9 but scaled up for rabbits.
Male New Zealand White rabbits (Oryctolagus cuniculus) weighing 2.5–3.0 kg were provided by Japan SLC (Shizuoka, Japan). The animals were housed in an individual wire cage (55 × 50 × 36 cm) in an air-conditioned facility at a temperature of 22 ± 3°C and relative humidity of 55 ± 25%, with a 12 h light/dark cycle (lights on at 8:00 h and off at 20:00 h). Rabbits were fed solid chow (Lab R Stock; Nosan, Kanagawa, Japan) and given fresh tap water ad libitum. All experimental procedures were performed in accordance with the Guidelines for Animal Experimentation issued by the Japanese Association for Laboratory Animal Science 10 and approved by the Animal Experimental Ethics Committee of Iwate University (Morioka, Japan).
Iodixanol (Visipaque 320; 320 mg I/mL, 290 mOsm/kg H2O) was purchased from Daiichi-Sankyo (Tokyo, Japan), inulin (Inulead; 100 mg/mL) from Fuji Yakuhin (Tokyo, Japan) and cisplatin injections (0.5 mg/mL) from Yakult (Tokyo, Japan). The antitumour agent cisplatin 11 was administered intravenously at 1, 2, 3 and 4 mg/kg to rabbits (n = 3) to evoke renal failure. Additional three rabbits received 0.9% saline solution (8 mL/kg) as a control. On day 5, iodixanol and inulin were co-administered intravenously at 60 mg I/kg and 40 mg/kg, respectively, into the auricular vein of rabbits, and blood (1 mL) was collected from the opposite auricular vein 30, 60, 90 and 120 min later using a 22 G needle, which was cut off from the hub (adapter). The dose regimen of iodixanol or inulin used was selected on the basis of the preliminary results. The day cisplatin was administered was regarded as day 1 in this study. At termination on day 5, all rabbits were euthanized by exsanguination under sodium pentobarbital (50–70 mg/kg, intravenously) anaesthesia, preceded by xylazine (3 mg/kg, intramuscularly) injection.
Serum iodixanol concentration was measured with reversed-phase high-performance liquid chromatography according to a previously reported procedure 5 with some modifications, 9 and serum inulin concentrations were colorimetrically determined using a commercially available kit (Diacolor Inulin; Toyobo, Osaka, Japan). It was confirmed beforehand that neither drug interference to iodixanol nor inulin level in serum was noted in the in vitro and in vivo additive studies. Serum urea nitrogen (UN) and creatinine concentrations were measured with an autoanalyser (Hitachi, Tokyo, Japan).
In the three-sample method using iodixanol or inulin, the clearance estimation is based on the one-compartment model.
12
Briefly, the area under the curve (AUC) was calculated by the linear trapezoidal rule with extrapolation using three serum samples (60, 90 and 120 min for iodixanol, and 30, 60 and 90 min for inulin). The clearance value (Cl) in each tracer was calculated from the following formula: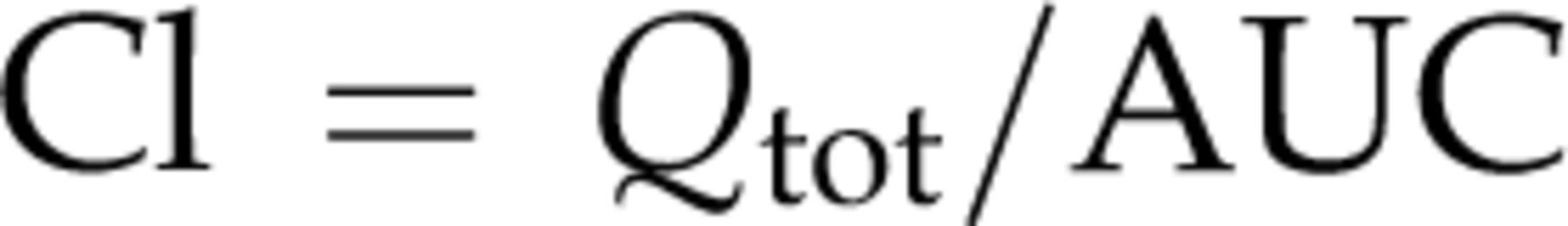
For measuring iodixanol clearance using the single-sample method, the distribution volume (V
1) of iodixanol in each animal was back-calculated by substituting Cl values and serum iodixanol concentrations (C
120 min) at 120 min (t) obtained from the three-sample method into the following Jacobsson's formula.
8
Because the V
1 value is dependent on elimination kinetics of each tracer and animal species, it is necessary to obtain it in individual animals.
9
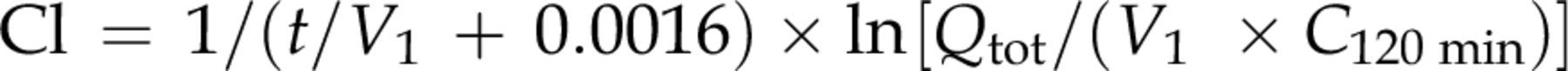
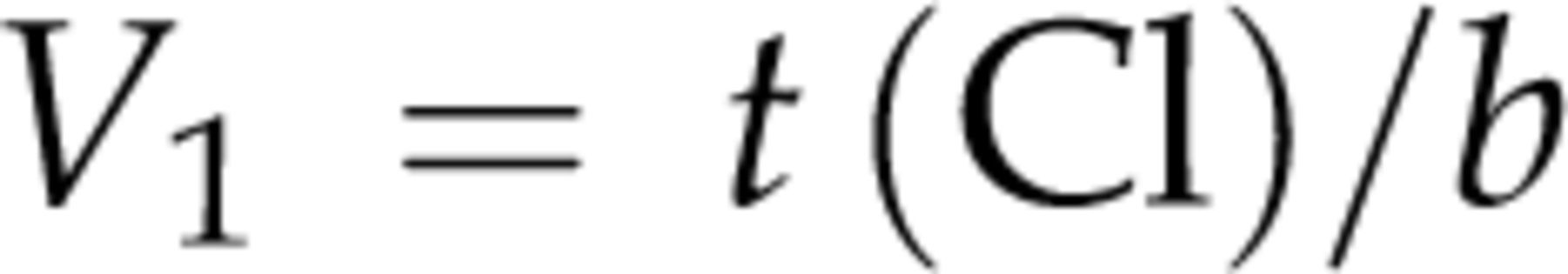
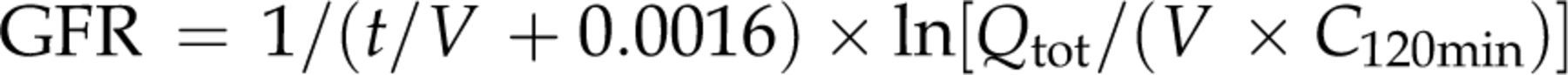
The reference GFR value (4.21 ± 0.28 mL/min/kg, n = 15) in clinically healthy male rabbits determined by the three-sample method using iodixanol were within the historical data (4.2 ± 0.6 mL/min/kg
17
and 3.5–4.7 mL/min/kg
18
), although experimental conditions were very different. Based on the data collected from healthy and cisplatin-treated rabbits, a close relationship (P = 0.001) was noted between GFR values determined by the three-sample and single-sample methods using iodixanol (data not shown). These findings resembled those reported in human patients with the single sample plasma clearance after a single injection of iohexol.
19
Similarly, there was a good correlation (P = 0.001) between GFR values obtained from the single-sample method with iodixanol and the three-sample method with inulin (Figures 1a and b), although the GFR between both methods elicited somewhat fluctuations at high values, presumably due to a difference in the qualification range (5–250 μg I/mL for iodixanol and 20–200 μg/mL for inulin), especially at a low concentration. These findings suggest that iodixanol can apply to rabbit GFR estimations.
Correlation between two methods in rabbits. (a) A scatter plot of GFR estimations by the single-sample method using iodixanol (GFR iodixanolsingle) and three-sample method using inulin (GFR inulinthree) was shown. (b) Deming's regression was y = 0.99x + 0.11. A Bland and Altman plot of the differences between two methods was shown according to GFR estimations (sample number = 28). Mean bias (solid line): 0.056. The confidence limits of agreement plots are 95%. GFR: glomerular filtration rate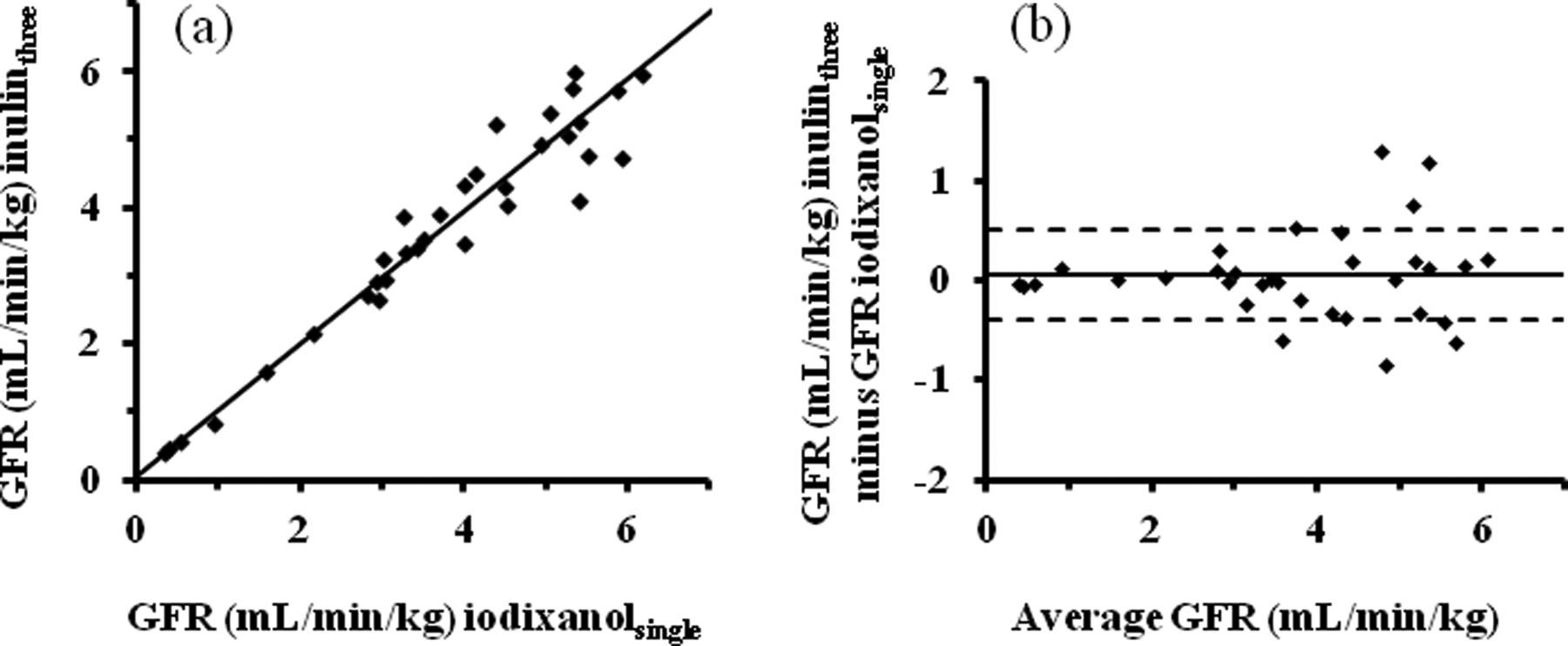
The relationship between the GFR values sought by the single-sample method with iodixanol versus serum UN and creatinine concentrations was assessed using healthy and cisplatin-treated rabbits. When the GFR value decreased to more than 60% of the reference value, serum UN (4.08 mmol/L, Figure 2a) and creatinine concentrations (0.65 mmol/L, Figure 2b) were likely to increase (reference values in our laboratory: <2.90 mmol/L for serum UN and <0.41 mmol/L for serum creatinine). However, the shape between GFR values by the single-sample method versus serum creatinine concentrations differed from that between the GFR values and serum UN concentrations. This difference may be explained by the notion that the renal handling of UN is different from that of creatinine, namely, creatinine is filtrated and only slight secretion, whereas UN is reabsorbed in the proximal tubule and flow rate in this nephron segment significantly affected its reabsorption. Thus, serum UN concentrations were considered to vary more than serum creatinine concentrations upon changes in GFR.
Relationship between GFR values determined by iodixanol versus serum urea nitrogen (UN) (a) and creatinine (b) concentrations using the data collected from healthy and cisplatin-treated rabbits. The GFR was measured by the single-sample method using iodixanol. Sample number = 28. GFR: glomerular filtration rate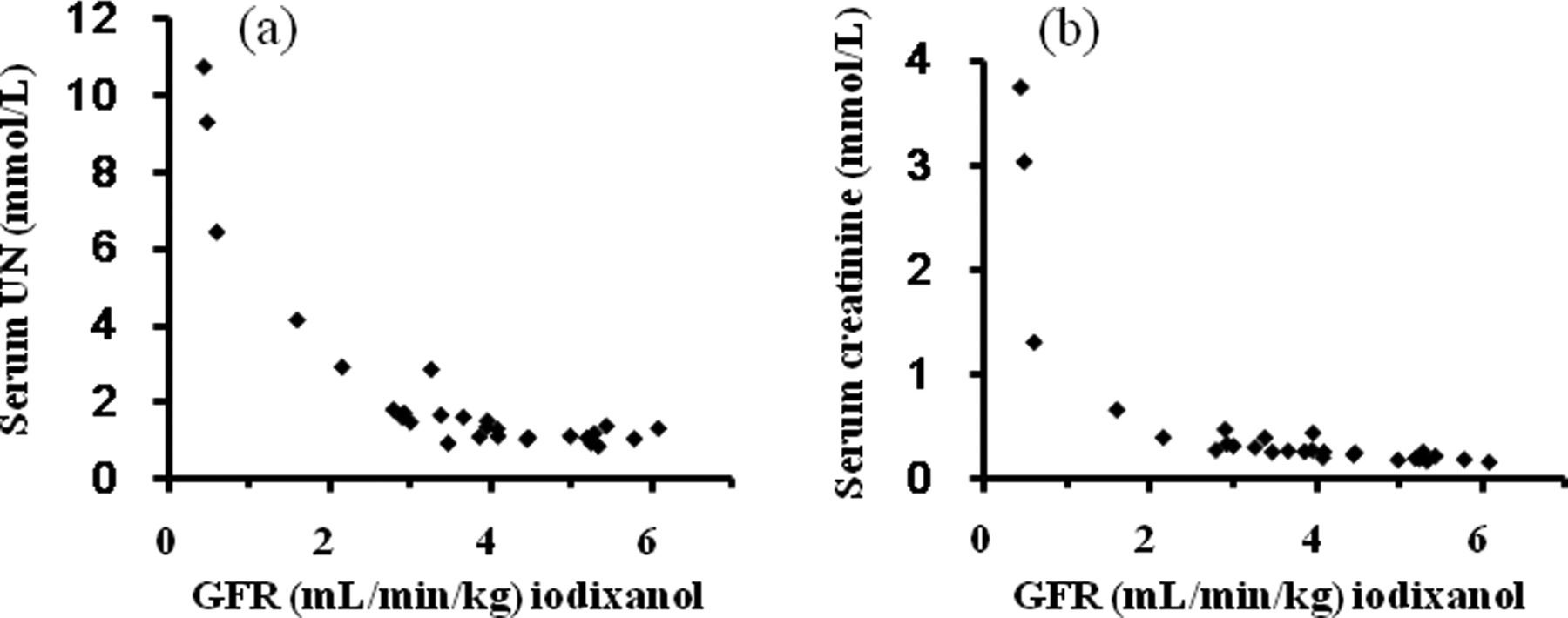
No adverse clinical signs associated with iodixanol treatments were observed throughout the experiment periods. The dose of 60 mg I/kg iodixanol corresponds to about one-ninth the human clinical dose (32 g I/60 kg body weight) as a contrast medium for angiography. The iodixanol procedure has many practical advantages such as repeated application to the same animals, 9 reduced stress and a small amount of serum specimen (80 μL), compared with the inulin method. Although the one-compartment model is a simplification and only applies after an equilibration period, this model is likely to underestimate AUC compared with the two-compartment or non-compartment model. Furthermore, the GFR estimated from calculated V values is based on many assumptions to predict the true GFR, and species differences in the Jacobsson's formula 8 between rabbits and humans have not been systematically evaluated. Therefore, further studies are required to collect cumulative background data including strain, gender and age.
In conclusion, the single-blood sample method with a bolus iodixanol injection at a low dose, which allows for repeated use in conscious rabbits, is a simplified procedure without the need for urine collection or radioisotope usage. Therefore, this assay becomes an expedient tool in both clinical and research settings, because the stress due to a multi-sample method is reduced.
Footnotes
ACKNOWLEDGEMENTS
We thank Dr Tetsuro Yamashita, Department of Food Science and Biochemistry, Faculty of Agriculture, Iwate University, for his helpful advice and suggestions on measuring serum iodixanol concentration with HPLC. We also acknowledge Dr Yoji Furuhama for providing information on the Newton method.
