Abstract
A large variation in spontaneous tumour development in the multiple intestinal neoplasia (Min) mouse model between laboratories has been reported. The composition of the diet might be an important factor. We examined the impact of five commercial rodent diets: the natural ingredient breeding diet Harlan Teklad 2018 (HT), the purified breeding diet AIN93G, the natural ingredient maintenance diet RM1, and the purified maintenance diets AIN93M and AIN76A, on the spontaneous intestinal tumorigenesis in the Min mouse model. The Min mice were fed one of two breeding diets during gestation and until four weeks of age, thereafter one of the three maintenance diets. Min mice bred on the breeding diet HT had significantly higher numbers and incidences of tumours in the colon, but fewer tumours in the small intestine than the breeding diet AIN93G. The maintenance diet RM1 gave a significantly higher number of small intestinal and colonic tumours and precancerous lesions called flat aberrant crypt foci (ACF) compared with the maintenance diets AIN93M and AIN76A. These findings show the importance of defining the type of diet used in experimental intestinal carcinogenesis studies, and that the diet should be taken into consideration when comparing results from different studies with Min mice.
The multiple intestinal neoplasia (Min) mouse was discovered by Moser and co-workers in 1990 as a result of random mutagenesis with ethylnitrosourea, 1 and has since then been extensively used as an experimental tool in the intestinal cancer research field. 1,2 The Min mouse is heterozygous for a mutation in the tumour suppressor gene Adenomatous polyposis coli (Apc), which leads to spontaneous development of several tumours throughout the intestine. This mouse model also develops flat aberrant crypt foci (ACF) in the colon which is believed to be precancerous lesions. 3
It has been estimated that as much as 80% of colon cancer in humans is diet-related. 4 Hence, the rodent diet used in intestinal carcinogenesis studies is a crucial factor. Several studies have shown that changes in diet composition affects the tumour development in the Min mouse 5–7 Rodent diets are classified after the degree of refinement of the ingredients, and the three terms commonly used are natural ingredient, purified and chemically-defined diets. 8 The natural ingredient diets contain unrefined ingredients such as corn, wheat, oats, beet pulp, fish meal, soybean meal, wheat bran and vitamin and mineral supplements. These diets are the most widely used, highly palatable and cheaper than the purified diets, 9 and are often referred to as lab chow, chow, stock, cereal-based or grain-based diets. Contaminants, such as heavy metals, pesticides and mycotoxins, as well as nutrient levels may vary from batch to batch due to seasonal changes and the source of ingredients. 10 A list of ingredients and proximate analysis of the macronutrients is usually available.
Purified diets are often used in experimental studies and are also referred to as semi-purified, semi-synthetic or synthetic diets. These diets are made from refined ingredients such as casein, sugar, starch, vegetable/animal fat, cellulose, vitamins and minerals, and the quantities of the different ingredients are given by the manufacturer. These provide a more consistent nutrient and contaminant levels, and should have minimal variations from batch to batch. 9 The downside is that they are often less palatable, and for long-term studies the diet may not contain adequate amounts of trace nutrients. 11 The last group consists of chemically-defined diets that are formulated entirely with chemically pure compounds such as amino acids, sugars, triglycerides, minerals and vitamins, and are expensive and even less palatable than the purified diets, and are therefore rarely used. Additionally, it is difficult to formulate a chemically-defined diet that contains all the essential nutrients for growth and maintenance. 10
The aim of this study was to examine how different commercial rodent breeding and maintenance diets influence intestinal tumorigenesis in Min mice. The rodent diets examined in the present study were chosen because they are easily accessible and are widely used in studies with Min mice and other rodent models.
Materials and methods
Animals
The animal experiment was approved by the Norwegian Animal Research Authority (NARA). Twenty C57BL/6J (strain: 000664) wild-type (wt) females were purchased from Charles River Laboratories International (Margate, UK). The mice from Charles River UK is bred in accordance with the Jackson Laboratory Genetic Management System. The rest of the C57BL/6J wt females and the C57BL/6J Min males (strain: 002020) were bred at the Norwegian Institute of Public Health, Oslo, Norway, and were originally purchased from the Jackson Laboratory (Bar Harbor, ME, USA). C57BL/6J MinApc+/− (Min) males were mated with wt females and produced litters that were used in the study. Housing details: microbiological status: conventional. Housed during experiment in air-flow Thorens IV rack, in Makrolon cages, size 28 × 28 × 11.5 cm, 9016 cm3 (1–6 animals per cage) on sterilized Nestpak Aspen 4 HK bedding. Room temperature and humidity: 21 ± 2°C; 64 ± 18%. Daylight lighting 100–300 Lux, 1 m above floor, 12 h day/night cycle. Drinking water: municipal tap water, untreated, ad libitum. Feed: ad libitum, for more details see below. The experiment was performed from October 2006 to April 2008. Only Min mice were used in the study. The mice were euthanized by cervical dislocation at eight weeks of age. Scoring of flat ACF and tumours was done as reported previously. 12
Genotype analyses of Min mice
The Min mice produced were identified by allele-specific polymerase chain reaction (PCR). Ear cartilage was used for DNA isolation. The biopsy was transferred to a sterile 1.5 mL microcentrifuge tube (Brand GmbH, Wertheim, Germany), on ice, suspended in 60 μL TE-buffer with SDS (10 mmol/L Tris pH 7.4, 0.1 mmol/L EDTA pH 8, 0.05% SDS) and incubated at 95°C for 10 min. Aliquots of 6 μL 10 mg/mL proteinase K were added before incubation at 55°C, overnight. Finally, the samples were incubated at 95°C for 10 min to inactivate the proteinase K, and stored at −20°C until PCR amplification. PCR was carried out with a BIO-RAD iCycler Thermal cycler (Bio-Rad, Hercules, CA, USA) as follows. Genomic DNA (5 μL of 1:50 dilution of the isolated DNA) was amplified in a 10 μL reaction volume, which contained final concentrations of 0.1 μmol/L MAPC-9 (5′-GCCATCCCTTCACGTTAG-3′), 1.61 μmol/L MAPC-MT (5′-TGAGAAAGACAGAAGTTA-3′) and 0.8 μmol/L MAPC-15 (5′-TTCCACTTTGGCATAAGGC-3′) (primers purchased from Eurogentec SA, Seraing, Belgium), 200 μmol/L each of dCTP, dGTP, dTTP and dATP, 2.5 mmol/L MgCl2, 1 × buffer II (10 mmol/L Tris-HCl, pH 8.3, 50 mmol/L KCl) (both MgCl2 and buffer II were purchased from Applied Biosystems, Foster City, CA, USA) and 0.0165 U GoTaq polymerase. The amplification conditions were 3 min at 94°C before 36 cycles at 94°C for 15 s, 54.5°C for 15 s and 72°C for 20 s, followed by a final extension at 72°C for 7 min. The PCR products were visualized by electrophoresis through 2% agarose gel followed by staining with SYBR Green I (Cambrex BioScience, Rockland, ME, USA). The wt allele was identified as a 600 bp PCR product; the Min allele was identified as a 300 bp product. The reagents were purchased from Sigma-Aldrich Corp (St Louis, MO, USA), Fluka (Buchs SG, Switzerland) or Promega (Madison, WI, USA), if not stated otherwise.
Rodent diets
The Min mice received one of two breeding diets, Harlan Teklad 2018 (E) Global (HT) (Harlan Teklad UK, Bicester, Oxfordshire, UK) or AIN93G (Product D10012G, Research Diets, New Brunswick, NJ, USA), during gestation and until four weeks of age. Then the pups were separated from the dam and were given one of three maintenance diets: RM1 (SDS RM1 (E), Special Diets Services, Witham, UK), AIN93M (Product D10012M, Research Diets, New Brunswick, NJ, USA) or AIN76A (Product D10001, Research Diets, New Brunswick, NJ, USA). This results in six different diet combinations: HT/RM1, HT/AIN93M, HT/AIN76A, AIN93G/RM1, AIN93G/AIN93M and AIN93G/AIN76A (Figure 1).
A schematic presentation of the six different diet combinations in the study is shown. Min mice were given one of two breeding diets during gestation and until four weeks of age, then the feed was changed to one of the three maintenance diets. The mice were sacrificed at eight weeks of age. Dark grey: natural ingredient diets; light grey: purified diets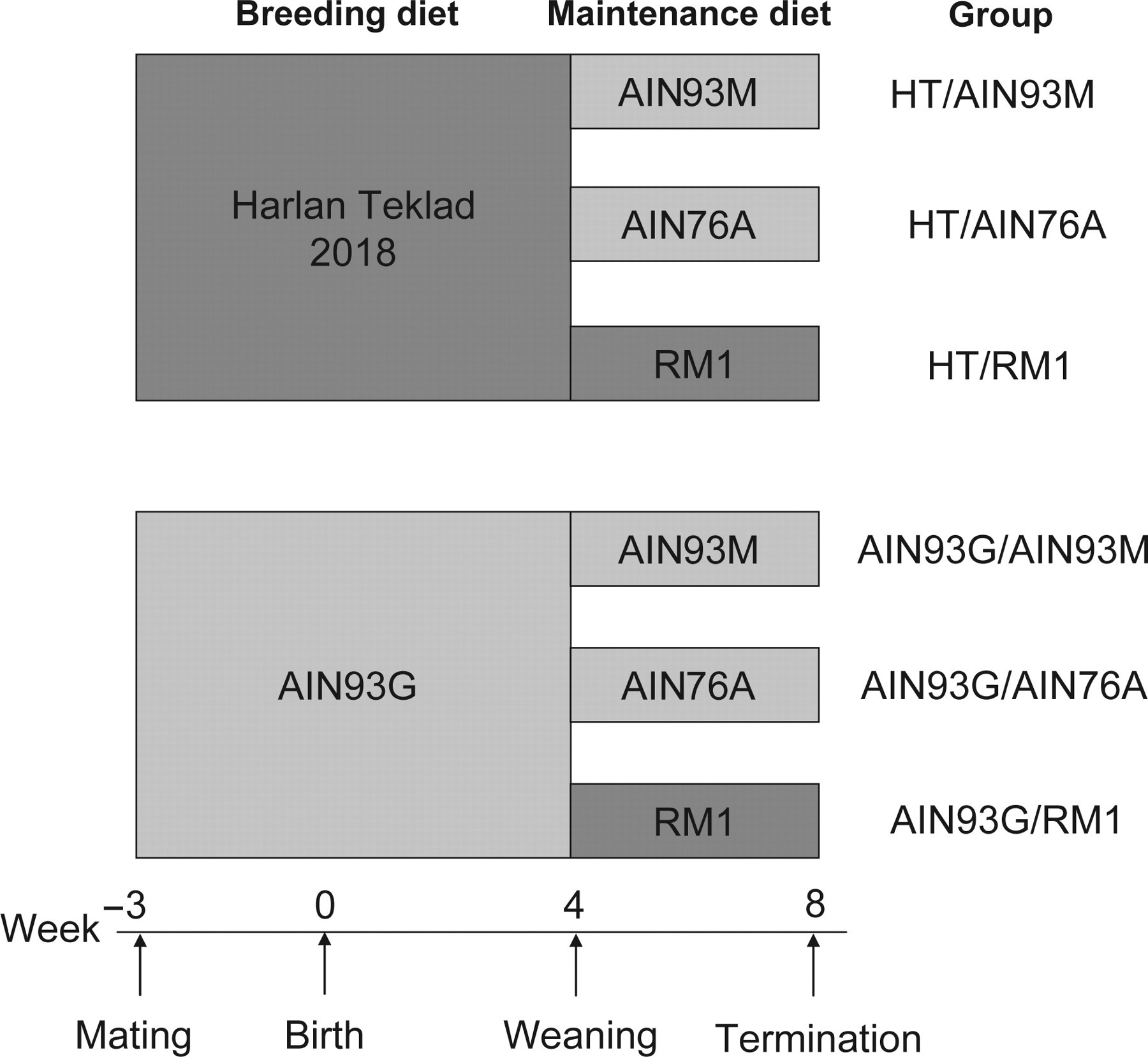
The ingredient list for the natural ingredient breeding diet HT: wheat, maize, wheat middlings, soybean oil, soybean meal, corn gluten meal and dried brewers yeast. The natural ingredient maintenance diet RM1 contains wheat, barley, wheat middlings, soybean oil, soybean protein concentrate, dehulled extracted toasted soybean and whey powder. Both of the natural ingredient diets are also supplemented with vitamins and minerals.
The ingredient list for AIN76A: casein, corn starch, sucrose, cellulose, corn oil, and a mineral and vitamin mix. The ingredient list for the AIN93 diets is similar to that of AIN76A, except for the substitution of corn oil with soybean oil to increase the content of linolenic acid. Additionally, the mineral and vitamin concentrations were adjusted and a portion of the sucrose in the AIN76A diet was replaced with cornstarch and dextrin.
13
An overview of the macronutrient content of the five different commercial diets based on the stated values provided by the manufacturers is illustrated in Figure 2.
An overview of the macronutrient content of the five different commercial diets used in this study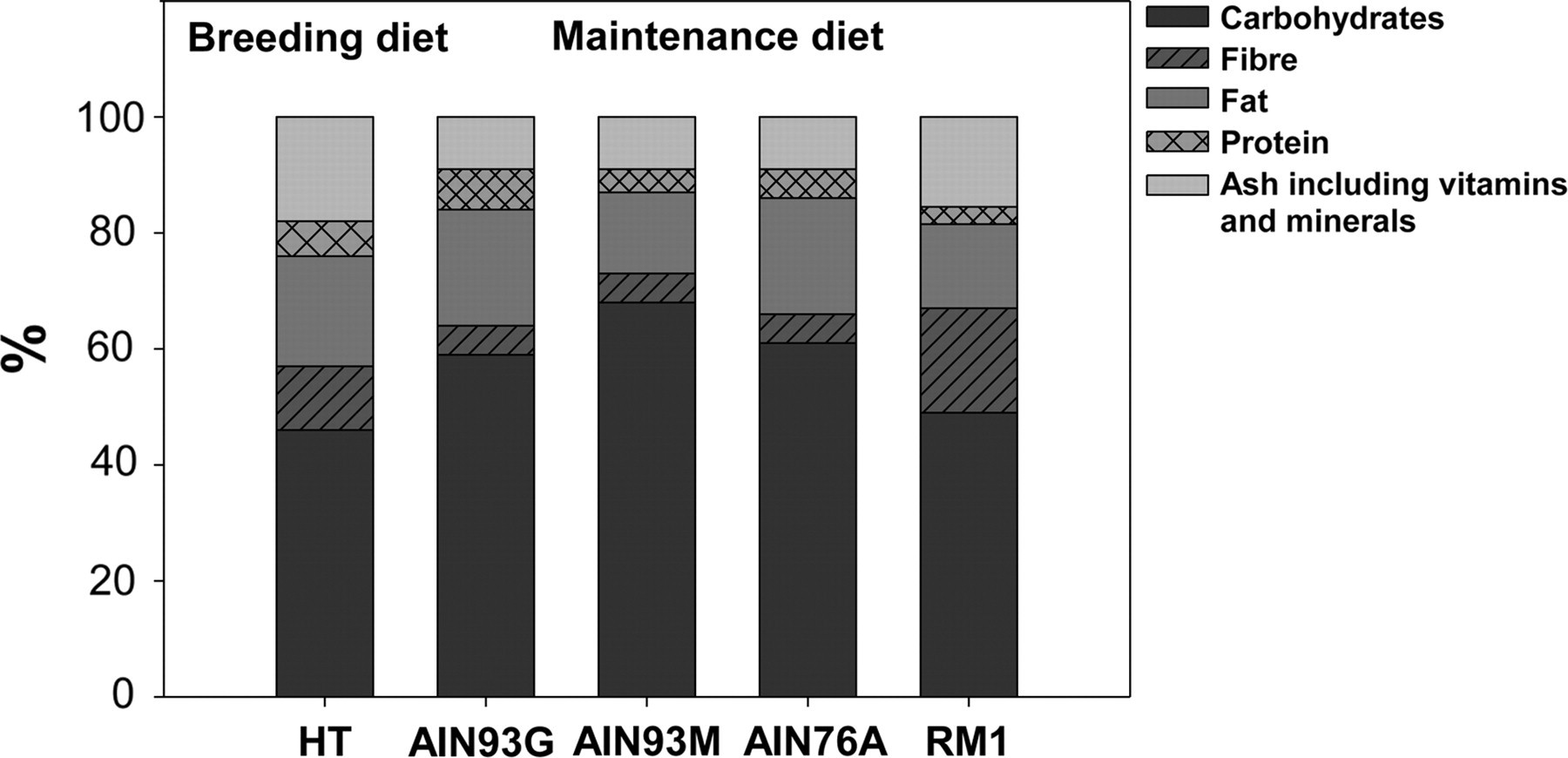
Statistical analysis
Data for the number and size of tumours and number of flat ACF were analysed by non-parametric tests as data were not normally distributed. The tests used were two-way analysis of variance (ANOVA) on Ranks or Kruskal–Wallis one-way ANOVA followed by pair-wise comparison with Tukey's or Dunn's method, respectively, to identify which groups differed from one another. Differences in incidence between the groups were tested by Z-test (SigmaStat software; Jandel Scientific, Erkrath, Germany). A P value of <0.05 was considered significant.
Results
Maintenance and breeding feed
The combination of breeding and maintenance feed that gave the lowest and highest number of small intestinal tumours was HT/AIN76A, yielding a median of 12 tumours and AIN93G/RM1 giving a median of 63 tumours, which is a five-fold difference in tumour development (Table 1). Even though the combination AIN93G/RM1 gave the highest number of small intestinal tumours, it did not differ significantly from the two other maintenance diets in combination with the same breeding diet (AIN93G/AIN76A and AIN93G/AIN93M); this was probably due to the large variation in this group (Figure 3b and Table 1). However, AIN93G/RM1 had a significantly higher small intestinal tumour number than both the combinations HT/AIN93M and HT/AIN76A (Table 1). Additionally, we observed a significantly higher tumour number for the feed combination HT/RM1 in comparison with HT/AIN76A (Figure 3a and Table 1). The Min mice which received HT/AIN76A had a significantly higher average size of the small intestinal tumours compared with those given AIN93G/RM1, which were the same groups that had the lowest and highest tumour number, respectively (Table 1).
A line plot showing the average (mean) number of tumours per animal along the small intestine of Min mice given different breeding and maintenance diets. (a) The breeding diet Harlan Teklad within the different types of maintenance diets: AIN93M/AIN76A/RM1. (b) The breeding diet AIN93G within the different types of maintenance diets: AIN93M/AIN76A/RM1 An overview of the number, size and incidence of tumours and flat aberrant crypt foci (ACF) in the intestine of Min mice fed six different diet combinations
a–gSimilar letters in one row indicate a significant difference between the groups with a P value <0.05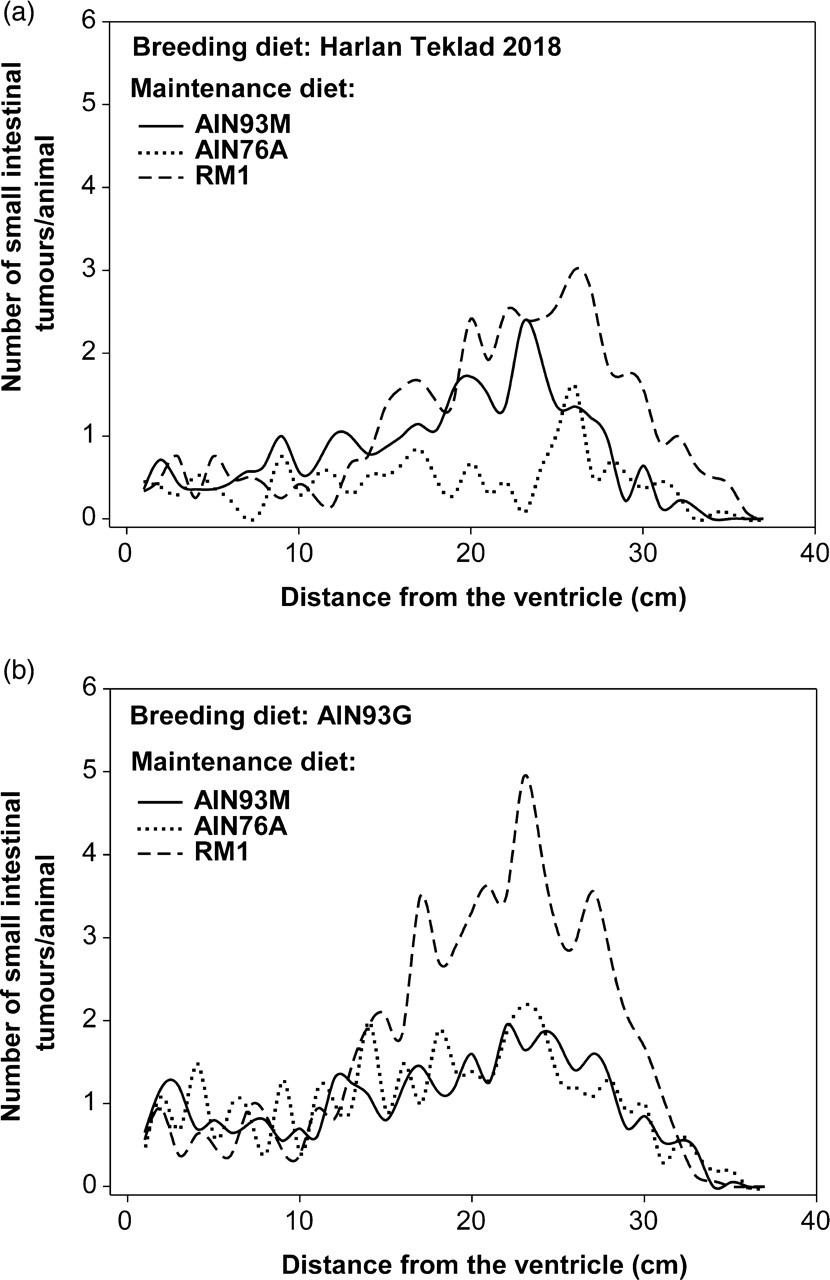
In the colon, there were large differences between the combinations of breeding and maintenance feed that gave the lowest and highest number of flat ACF, with HT/AIN76A and AIN93G/AIN76A yielding a median of zero compared with HT/RM1 giving a median of 14. The feed combination HT/RM1 induced a higher number of flat ACF in the colon in comparison with the numbers in those receiving HT/AIN93M and HT/AIN76A (Figure 4a and Table 1). Min mice fed AIN93G/RM1 had a higher number of flat ACF in comparison with those receiving AIN93G/AIN93M or AIN93G/AIN76A (Figure 4b and Table 1). The same combinations, HT/RM1 and AIN93G/RM1, also had a higher incidence of flat ACF compared with their respective feed combinations (Table 1). All the mice fed the diet combination HT/RM1 had one or more flat ACF in the colon (100% incidence), compared with mice fed the diet combination AIN93G/AIN76A which had only 10% incidence (Table 1). Several other diet combinations also differed from one another with respect to both number and incidence of flat ACF (Table 1).
A line plot showing the average (mean) number of flat aberrant crypt foci (ACF) per animal along the colon of Min mice given different breeding and maintenance diets. (a) The breeding diet Harlan Teklad within the different types of maintenance diets: AIN93M/AIN76A/ RM1. (b) The breeding diet AIN93G within the different types of maintenance diets: AIN93M/AIN76A/RM1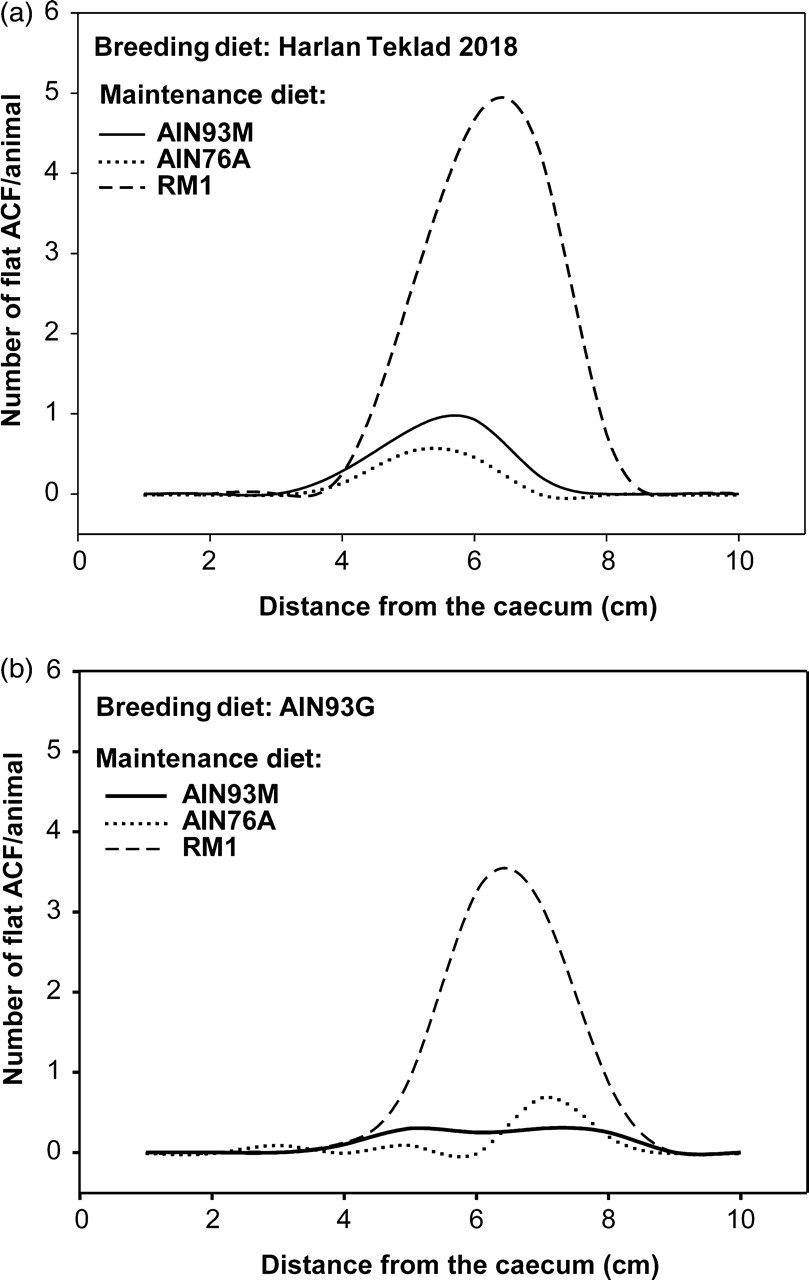
Min mice fed the breeding and maintenance combinations HT/RM1 and HT/AIN76A had significantly higher incidence of colonic tumours in comparison with those receiving AIN93G/AIN93M (Table 1). With regard to the number of colonic tumours, there were only two combinations of breeding and maintenance feed that differed; HT/RM1 gave a significantly higher number of colonic tumours than AIN93G/AIN93M (Table 1). With regard to the size of the colonic tumours, the diet combination AIN93G/RM1 increased the average size compared with HT/AIN76A (Table 1). None of the other diet combinations differed from each other concerning the size of the colonic tumours, yet the median value for AIN93G/AIN76A was as high as AIN93G/RM1. The reason for this was that the data for the size of colonic tumours were normally distributed and that the mean values were slightly different than the median values in Table 1.
There were no significant differences between the groups given different breeding and maintenance for final body weight, and no significant differences between males and females for the number, size or incidence of tumours and flat ACF (data not shown).
Maintenance diet
The effect of the three different maintenance diets RM1, AIN76A and AIN93M as such were compared while controlling for the two different breeding diets. There were no significant differences between the two purified AIN diets for number, size, or incidence of tumours and flat ACF. However, the natural ingredient diet RM1 differed significantly on several of the parameters compared with the two AIN diets.
The maintenance diet RM1 gave a significantly higher number of small intestinal tumours compared with the AIN maintenance diets, with a median number of 49 tumours for RM1 and 28 and 22 for AIN93M and AIN76A, respectively (Figure 5a). The choice of maintenance feed also showed a tendency to affect the average size of the small intestinal tumours, with RM1 giving a non-significant lower average size of the small intestinal tumours compared with the AIN diets (Figure 5b).
A box-plot and whisker graph showing the difference between the three different maintenance diets, the natural ingredient diet RM1 and the purified diets AIN93M and AIN76A. (a) The number of small intestinal tumours. (b) The average size of small intestinal tumours. (c) The number of colonic tumours. (d) The average size of colonic tumours. (e) The number of flat aberrant crypt foci (ACF). The box represents 25th and 75th percentiles and illustrates the median (solid line) and the mean (dashed line). The whiskers show the 10th and 90th percentiles and outliers are indicated as dots. Similar letters indicate a significant difference between the groups with a P value <0.05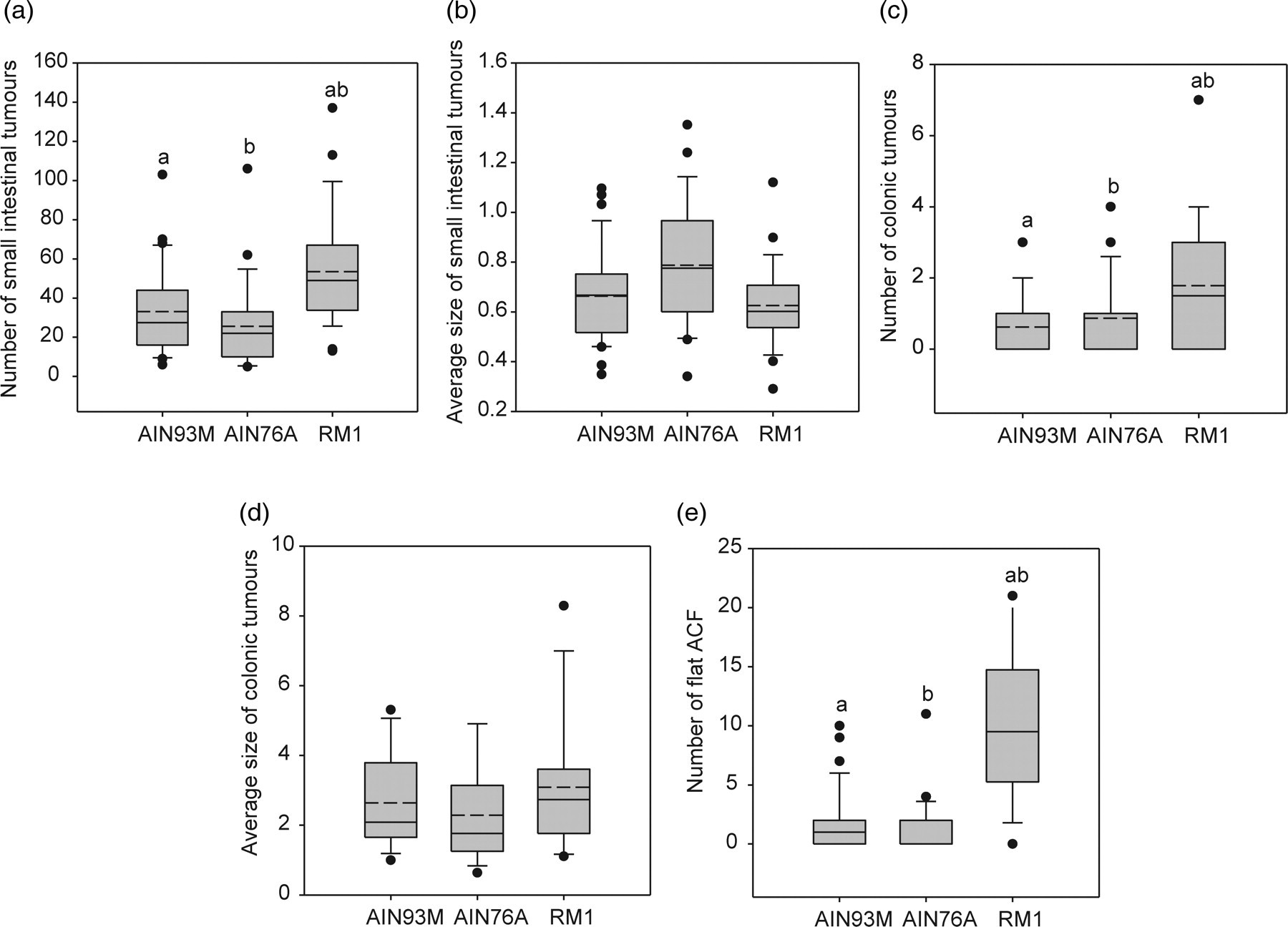
The diet RM1 gave a significantly higher number of colonic tumours compared with the two AIN diets (Figure 5c). No significant difference was seen between the average sizes of the colonic tumours (Figure 5d), nor were there any differences between the incidences of colonic tumours of Min mice given the different maintenance diets. Although there was a strong tendency that Min mice receiving RM1 had a higher incidence of one or more colonic tumours compared with those given the AIN diets (data not shown).
The Min mice fed the maintenance diet RM1 had a higher incidence of flat ACF in comparison with those on the AIN maintenance diets, with a flat ACF incidence of 93%, 53% and 30% for RM1, AIN93M and AIN76A, respectively (data not shown). For the number of flat ACF the diet RM1 also gave a significant higher number compared with the two AIN diets, with an average flat ACF number for RM1, AIN93M and AIN76A of 9.5, 1.0 and 0, respectively (Figure 5e).
Breeding diet
The two different breeding diets, the natural ingredient diet HT and the purified diet AIN93G, were compared as such, while controlling for the effect of the different maintenance diets.
The purified breeding feed AIN93G induced significantly more tumours in the small intestine than the natural ingredient feed HT, with a median number of 38 and 26, respectively (Figure 6a). Regarding the average size of the tumours, the opposite occurred, as Min mice bred on HT had a significantly higher average size of the small intestinal tumours compared with those fed AIN93G (Figure 6b).
A box-plot and whisker graph showing the difference between the two different breeding diets, the natural ingredient diet HT (Harlan Teklad 2018) and the purified diet AIN93G. (a) The number of small intestinal tumours. (b) The average size of small intestinal tumours. (c) The number of colonic tumours. (d) The average size of colonic tumours. (e) The number of flat aberrant crypt foci (ACF). The box represents 25th and 75th percentiles and illustrates the median (solid line) and the mean (dashed line). The whiskers show the 10th and 90th percentiles and outliers are indicated as dots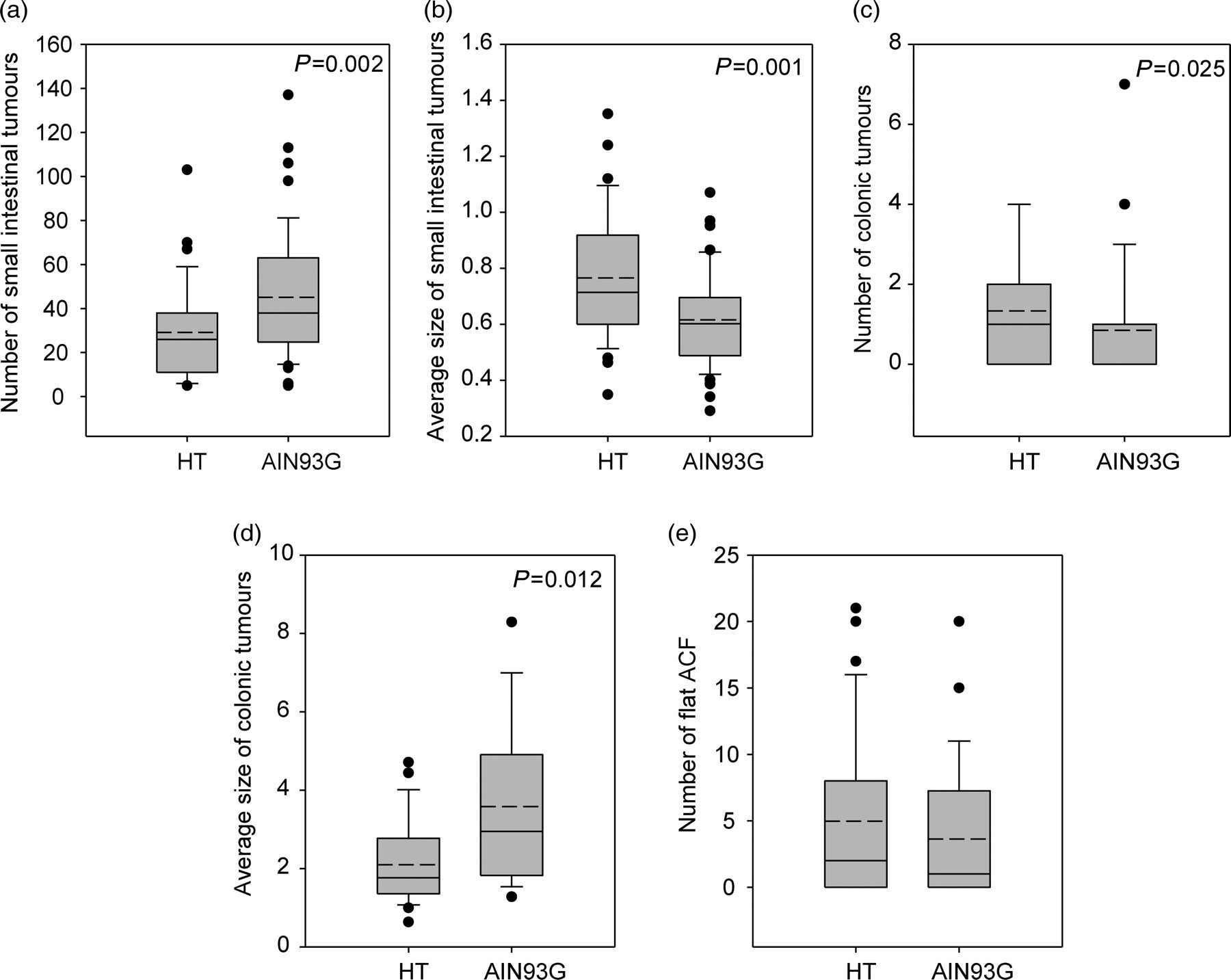
Min mice receiving HT had a higher colonic tumour number compared with mice on AIN93G, with a median number of 1.0 and 0, respectively (Figure 6c). The colonic tumour incidence of mice given the breeding feed HT and AIN93G were 67% and 41%, respectively (P = 0.020) (data not shown). However, the breeding feed AIN93G significantly increased the average size of the tumours in the colon compared with HT (Figure 6d). There was no difference either for the number (Figure 6e) or the incidence (data not shown) of flat ACF in mice fed the two breeding diets.
Differences in the length of the small intestine and colon
The length of the small intestine and colon was measured for each mouse. The mice fed the natural ingredient breeding diet HT had significantly longer small intestines (3%) in comparison with the mice fed the purified breeding diet AIN93G (not shown). No difference in the length of the colon was observed for mice fed the two breeding diets. The natural ingredient maintenance diet RM1 significantly increased both the length of the small intestine and colon compared with the two AIN diets, with 5% and 11%, respectively. Adjusting for the different lengths of the small intestine and the colon did not affect the statistical significant differences among the groups, although the differences somewhat decreased (not shown).
Discussion
We found that both the breeding and maintenance diets strongly influenced the number and incidence of intestinal lesions in Min mice. The natural ingredient breeding diet HT induced a greater number and incidence of tumours in the colon, but fewer tumours in the small intestine than the purified breeding diet AIN93G. The natural ingredient maintenance diet RM1 gave a higher number of tumours in the small intestine and colon and increased both the number and incidence of flat ACF compared with the two purified AIN maintenance diets.
Both of the natural ingredient diets, used for breeding and maintenance (HT and RM1), had the highest number of colonic tumours in the Min mice, and the combination of HT/RM1 also had the highest number and incidence of both colonic tumours and flat ACF. Other studies have shown that natural ingredient diets induce more lesions and DNA adducts in rodents than purified diets. 14–17 In another study, there were no differences in the number of tumours in the intestine between the Min mice fed the natural ingredient diet RM1 or a purified diet. 18 However, these authors found that the natural ingredient diet RM1 significantly increased the length and weight of both the small intestine and colon compared with the length of the intestine of the mice fed the purified diet. 18 This is in accordance with our results. However, the differences in tumour and flat ACF number did persist even when adjusting for the differences in the length of the intestine. Thus, the increase in length of the intestine can therefore not alone explain the differences regarding tumorigenesis between the groups.
We found that the breeding diet, which the mice were exposed to during gestation and lactation, had an impact on the induction of tumours in the intestine of Min mice. In an experiment with transgenic LacZ mice, the purified diet AIN93G fed to mice during gestation and lactation induced fewer mutations in the offspring at three weeks of age than the natural ingredient diet. 16 When pregnant rats were fed a natural ingredient diet (Purina-5001), it was shown that the fetus had higher levels of DNA adducts in the liver at birth, 24 h and nine days after birth compared with the fetus of rats fed the purified diet AIN93G. 15 This indicates that the dietary effects were transmitted both during gestation and lactation.
Interestingly, the two breeding diets affected tumorigenesis in the small intestine and colon differently; the natural ingredient breeding diet HT induced a greater number and incidence of tumours in the colon, but fewer tumours in the small intestine than the purified breeding diet AIN93G; this might indicate that there are different mechanisms for the induction of tumours in the small intestine and the colon. 19 Another possible biological explanation for the observed differences in flat ACF and tumour formation in the colon and small intestine for the two breeding diets in this study may be through modification of the intestinal microflora. Dietary fibres are fermented by bacteria in the colon to short-chain fatty acids, mainly acetate, propionate and butyrate which interact with the immune system through cell differentiation and apoptosis. 20 In a study, increased dietary fibre content reduced the number of tumours in the small intestine, but not in the colon. 14
When comparing the stated values for macronutrients provided by the manufacturers, it shows that RM1 and HT differ greatly in both the type and amount of dietary fibre compared with the three AIN diets, which all contain 5% dietary fibre contributed by cellulose. It is stated by the manufacturer that HT contains 3.8% crude fibre. However, dietary fibre is a complex entity, consisting of many non-digestible components and the fibre content is not uniformly declared by the manufacturers. The crude fibre content, which is the residue left after chemical treatment, is not synonymous with dietary fibre, which is a term used to define the total fibre content in food. The crude fibre analysis fails to recover a large part of what is considered to be total dietary fibre. 21 Additionally, a similar natural ingredient diet from HT stated to have a crude fibre content of 5% was shown to contain 18% dietary fibre. 14 As the carbohydrate content is stated to be 57% for HT, with the starch and sugar comprising 46%, it was estimated that the total dietary fibre content of HT was 11%. Therefore, the maintenance diet RM1 contains three times and HT two times the amount of dietary fibre compared with the AIN diets. The dietary fibre in the maintenance diet RM1 is contributed by cellulose, hemicellulose, lignin and pectin, and the breeding diet HT most likely also contains these four types of dietary fibre, although this information is not provided by the manufacturers. Wise and Gilburt 22 analysed 35 natural ingredient animal diets for dietary fibre content, and it was shown that all of the diets contained cellulose, hemicellulose, lignin and pectin. In a study, mice fed Harlan Teklad had more tumours in both the small intestine and colon compared with the AIN93M fed mice. 14 Min mice fed purified diets supplemented with apple pomace (which has a high pectin content) or pectin as such had increased size of colonic tumours and increased number of small intestinal tumours. 18,20 We hypothesize that the type and amount of dietary fibre is partly responsible for the increased intestinal tumorigenesis in Min mice fed the two natural ingredient diets.
Contaminants and non-nutrients that are not reported by the manufacturers might also have an impact on the differences observed between the different diet groups. The two natural ingredient diets used in this study, especially in the case of RM1, most likely contains some biologically active factors that affects the tumorigenesis in Min mice. Food consumption may be another factor; as it has been shown previously that food consumption is higher for the natural ingredient diets than purified diets. 9,17,18 One explanation for the higher food consumption of natural ingredient diets is that they contain fewer calories per gram feed than the purified diets. Data collected from 82 experiments show that the adult body weight and the daily caloric intake are highly correlated in mice. 23 In the present study, there were no differences in adult body weight for any of the diet groups, which might suggest that the daily caloric intake was similar for all groups. However, in another study, Harlan Teklad-fed mice had a higher intake of calories compared with AIN93M-fed mice, yet there were no difference in adult body weight. 14
This study shows that widely used commercial rodent diets have a strong impact on the spontaneous intestinal tumorigenesis in Min mice. This may be due to several different factors, as a possible higher daily caloric intake for the Min mice fed the natural ingredient diets and the amount and type of dietary fibre, but several other unknown factors are also likely to be involved. The present study shows that the commercial diets used in studies with Min mice should be carefully selected and that the feed used should be taken into consideration when comparing results from studies with Min mice.
Footnotes
ACKNOWLEDGEMENTS
We thank Andre Larsen, Tone Rasmussen, Radah Isabel Sheik Zain and Victor Labay Ong for excellent technical assistance. This work was supported by the Norwegian Research Council (175157/V40).
