Abstract
The life-threatening effects of intracranial hypertension on brain perfusion and cerebral metabolism are the subject of current research in different animal models. The purpose of this study was to describe an efficient, reliable and inexpensive surgical method for temporary elevation of intracranial pressure (ICP) in acutely instrumented pigs in a research setting. Therefore, a balloon catheter was inserted into the left lateral ventricle and an ICP sensor was placed in the parenchyma of the right cerebral hemisphere. Ten acutely instrumented pigs were studied while under deep terminal general anaesthesia. The step-by-step inflation of the intraventricular balloon allows one to achieve the desired ICP up to 46 mmHg and maintain it at this level. ICP values ranged from a median of 2 (1–2) mmHg to 43 (29–45) mmHg. To the authors’ knowledge, this is the first detailed description of a minimally invasive surgical technique for temporary ICP elevation in pigs via stepwise inflation of an intraventricular balloon.
The treatment of patients with primary neuronal injury such as severe traumatic brain injury (TBI), intracranial haemorrhage or ischaemic insults in the central nervous system presents a great challenge within current acute and intensive care. 1 The focus of therapy in these patients with elevated intracranial pressure (ICP) is maintaining cerebral perfusion and thus the concomitant oxygen supply to the brain. 2
Cerebral perfusion pressure (CPP) is defined as the difference between mean arterial pressure (MAP) and ICP: CPP = MAP − ICP. Consistent maintenance of CPP > 60 mmHg is generally accepted as one of the major therapeutic goals in adults with TBI with increasing ICP. 2 Cerebral blood flow and oxygen delivery are directly linked to CPP when cerebral autoregulation is compromised. Animal models may prove helpful in developing strategies to help reduce the negative effects of elevated ICP on cerebral metabolism in the future. 3 We report on a new method that is simple, effective and inexpensive for the reproducible, step-by-step reduction in cerebral perfusion via increasing ICP in a pig model in a controlled setting.
This laboratory technique was carried out on healthy female domestic pigs (Sus scrofa ferus domestica; age: 4 months, weight: 34–36 kg). The animals were cared for in accordance with the national standards of laboratory animal welfare in Germany. Pigs were housed in the ‘Central Animal Care Facility, Institute for Experimental Surgery’ (University of Rostock, Rostock, Germany) and had a one-week acclimatization period after arriving at their new animal facility. Animals were fed on a standard diet. Only water was accessible ad libitum in the 12 h before experiments. Experimental procedures were approved by the Local Ethics Committee on Animal Research at the University of Rostock (Germany). Ten acutely instrumented pigs were studied while under deep terminal general anaesthesia.
The animals were premedicated with flunitrazepam 0.2 mg/kg body weight (Rohypnol®; Hoffmann-La Roche AG, Grenzach-Wyhlen, Germany) and ketamine 10 mg/kg (Ketanest®; Parke-Davis/Pfizer, Freiburg, Germany) via intramuscular injection. General anaesthesia was induced via an auricular vein with fentanyl 6 µg/kg (Fentanyl® Janssen; Janssen-Cilag, Neuss, Germany), flunitrazepam 0.1 mg/kg and rocuronium 1.2–1.5 mg/kg (Esmeron®; Organon Laboratories Limited [Merck Sharp & Dohme Ireland Limited], Dublin, Ireland), and intubation was performed with a 7.0 mm cuffed tube. Anaesthesia was maintained with a continuous intravenous infusion of propofol 4–6 mg/kg/h (Propofol-®Lipuro; B Braun, Melsungen, Germany), fentanyl 3–4 µg/kg/h and rocuronium 2.2–2.7 mg/kg/h. The lungs were mechanically ventilated in a pressure-controlled mode using a Servo 900 ventilator (Siemens, Erlangen, Germany) with a positive end-expiratory pressure of 5 cmH2O and an FiO2 of 0.40. Respiratory rate and tidal volume were adjusted to maintain normocarbia throughout the experiment. After induction of anaesthesia, an arterial line was placed in the femoral artery to measure MAP. Normovolaemia was maintained by infusing a full electrolyte solution (Jonosteril®; Fresenius, Bad Homburg, Germany) at a rate of 10–15 mL/kg/h.
Vital signs (heart rate, body temperature, systolic, diastolic and mean pressures) were measured. All animals were monitored continually to assess adequate level of anaesthesia. The pedal withdrawal reflex, palpebral reflex and the tone of jaw were evaluated using standard protocol every 15 min during anaesthesia.
At the end of the experiments, all animals were euthanized under deep anaesthesia with a potassium chloride overdose, according to the German laws for animal studies. After induction of anaesthesia, the pigs were placed in a prone position so that a balloon catheter could be inserted into the left lateral ventricle and an ICP sensor could be placed in the parenchyma of the right cerebral hemisphere. A straight, 3 cm long incision was made in each pig's forehead approximately 1 cm over the eye and 1 cm lateral to the midline. A 5 mm burr hole was made and then widened with a punch to a diameter of about 1 cm. Any bone haemorrhage was sealed with bone wax.
The dura mater was opened with a small X-shaped incision. After thermocoagulation of the dura edges and the brain surface, a balloon catheter (Equalizer Balloon Catheter [EQL/33/7/2/65; Boston Scientific Corp, Cork, Ireland]) was introduced into the lateral ventricle. Correct placement was verified by collecting cerebrospinal fluid through the open catheter tip. A further, though not necessary, option to verify correct positioning is a contrast-enhanced X-ray of the ventricle system. The burr hole was then closed with an absorbable gelatin sponge and the catheter was fixed to the skin with sutures.
An ICP sensor catheter was placed in the parenchyma in the right hemisphere via a right-sided frontal burr hole (Codman MicroSensor ICP Transducer [LCN 171978-001/M; Codman, Wokingham, UK]) and then connected with a Codman ICP Express™ Monitor (Codman, UK). The placement of the burr holes in relation to each other is demonstrated in Figure 1a. Figure 1b shows the experimental set-up after insertion of the catheters and suturing of the skin.
(a) Placement of the bifrontal paramedian burr holes. The intraparenchymal intracranial pressure catheter is placed on the right side. (b) Experimental set-up after insertion of the catheters and suturing of the skin. A syringe is connected to the balloon catheter for filling at intervals during the experiment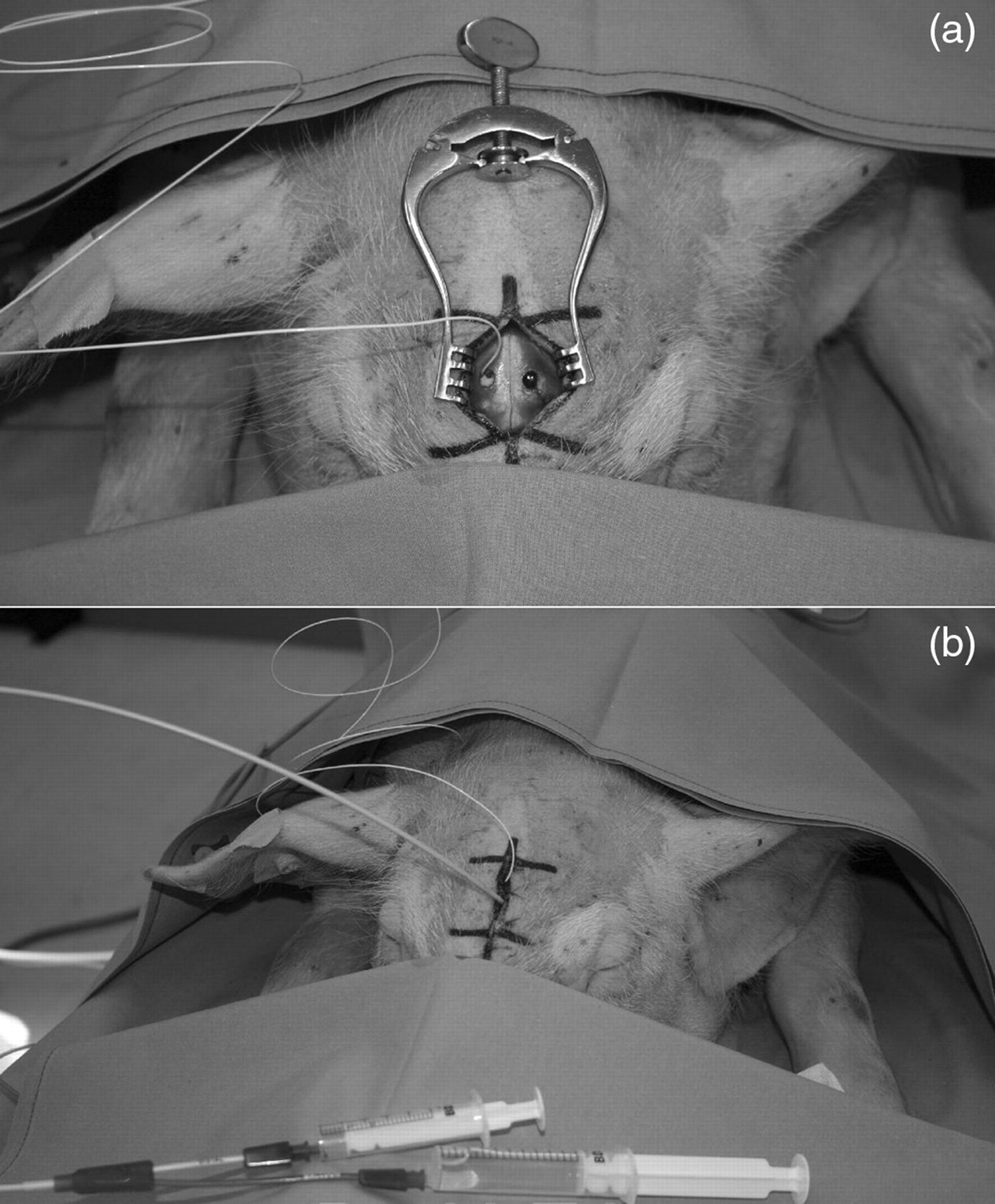
During the experiment, it was possible to inflate and deflate the balloon in order to vary the ICP according to the experimental setting.
The length of surgical procedure was 20 min to a maximum of 30 min, but greater expertise shortened the duration for instrumentation over time. There were no acute complications or losses of animals related to the procedure during the study. The step-by-step inflation of the intraventricular balloon allows one to achieve the desired ICP up to 46 mmHg and maintain it at this level. ICP values recorded across all animals ranged from a median of 2 (1–2) mmHg to 43 (29–45) mmHg. A selected experimental protocol is demonstrated in Figure 2.
Typical data recording of intracranial pressure (ICP) changes from baseline during the experiment. ICP was manually increased step-by-step by inflating the intraventricular balloon after one, 24 and 68 min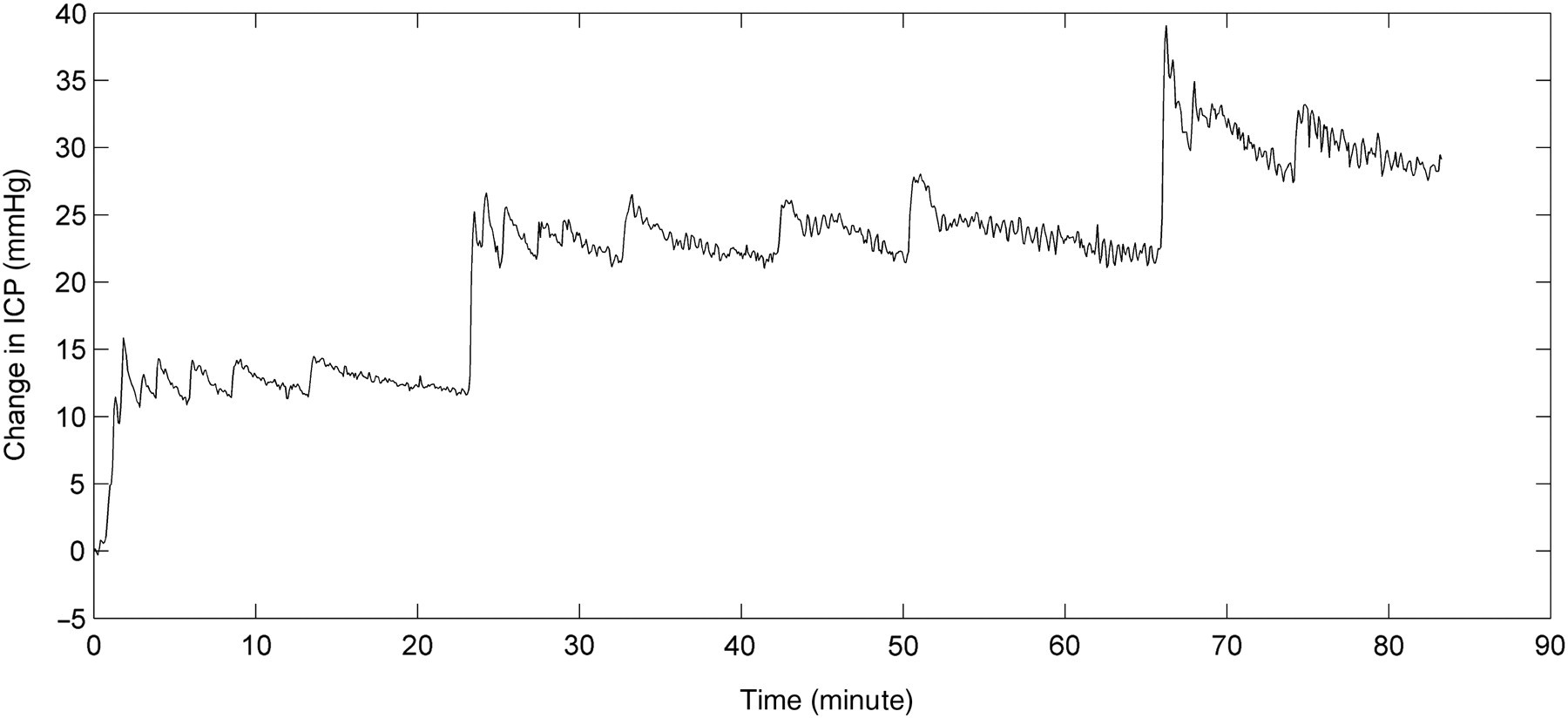
This study has provided a detailed description of an accurate surgical technique for placing a balloon catheter into the lateral ventricle in pigs for increasing and decreasing ICP in an experimental setting for the very first time. This technique allows for the variable adjustment of ICP without major brain damage.
Although experimental studies throughout the literature have accomplished diffuse ICP increase via ventricular infusion of artificial cerebrospinal fluid, 4 the inflation of an epidural balloon is a widely used method for the controlled elevation of ICP in animal models. 5–7 This method allows for reproducible though one-sided and mainly only a regional increase in ICP as an external approach. In contrast, the authors see that the advantage associated with the described technique is that it produces a uniform distribution of pressure originating from the centre of the brain, just as it can be found in vivo in generalized cerebral oedema after TBI.
The domestic pig was chosen because of the striking similarities in its anatomy and physiology compared with the human brain. 8 The study protocol described with a continuous intravenous propofol–fentanyl anaesthesia and controlled ventilation have a minor to moderate effect on ICP, CPP, arteriovenous oxygen difference (AVDO2) 9 and the cardiovascular system overall as well as body temperature and other physiological parameters. Furthermore, the dosages described induced deep surgical anaesthesia of long duration with a smooth induction. The authors can recommend this method of anaesthesia for non-survival experiments measuring ICP and CPP in an experimental setting as it produces sufficient cardiovascular stability for 8–12 h.
We found the described surgical technique to be a safe and easy way to elevate ICP in a pig model. Our previous experiences with the laboratory technique presented in this paper suggest that the procedure is useful and reliable and may be applied in numerous experimental settings.
