Abstract
Preference tests have often been performed for collecting information about animals' acceptance of environmental refinement objects. In numerous published studies animals were individually tested during preference experiments, as it is difficult to observe group-housed animals with an automatic system. Thus, videotaping is still the most favoured method for observing preferences of socially-housed animals. To reduce the observation workload and to be able to carry out preference testing of socially-housed animals, an automatic recording system (DoubleCage) was developed for determining the location of group-housed animals in a preference test set-up. This system is able to distinguish the transition of individual animals between two cages and to record up to 16 animals at the same time (four animals per cage). The present study evaluated the reliability of the DoubleCage system. The data recorded by the DoubleCage program and the data obtained by human observation were compared. The measurements of the DoubleCage system and manual observation of the videotapes are comparable and significantly correlated (P < 0.0001) with good agreement. Using the DoubleCage system enables precise and reliable recording of the preferences of group-housed animals and a considerable reduction of animal observation time.
Preference tests (choice tests), as well as consumer demand tests (strength of preference), have been used in many studies in different fields. Laboratory animal scientists often perform preference tests for evaluating specific environmental aspects. During tests animals have free access to the examined resources and are allowed to express their preferences (based upon location). Currently, preference tests and consumer demand tests are often applied in order to evaluate environmental refinement designs. These methods are frequently performed for understanding animals' preferences between two (or more) objects or housing conditions. 1–6 For observing the location of animals video analysis is the most applied method, which is extremely time-consuming. Besides human observation, possible techniques for automated registration, such as infrared recorder, tracking system based on video, ultrasonic or radio frequency, have been considered for detecting the location or movement of animals. 7–11 Automatic systems have been developed using light detectors (photoelectric cells, photo beam) to observe the movement of mice during preference tests (e.g. Blom et al. 12 , Fitchett et al. 13 ) or consumer demand tests (e.g. Sherwin 2 , Warburton and Nicol 14 ). Other possibilities such as electric balances 15 or micro-switches 16 have also been applied for registering the positions and transition of animals. However, none of these studies were able to detect individual animals when a social group was tested.
As group housing is recommended for social species such as mice, preference tests using group-housed animals are therefore considered as a necessary framework for further studies. However to perform preference tests using group-housed animals it would be necessary to analyse videotapes repeatedly for each individual animal in the same cage. This process is extremely time-consuming, especially when a long-term preference test is implemented, and has great limitations such as inefficiency and slow progress.
To overcome such limitations and be able to perform preference tests with socially-housed animals, an automatic system for detecting the location of each individual animal would be very helpful. For this purpose automated techniques, such as transponder (implantable radio frequency microchips) or video-tracking systems, have been reviewed. Although both systems are able to detect the location of animals, the transponder can identify individual animals more easily (e.g. animals need to be dyed for identification on a video-tracking system) and register more animals at the same time. It was therefore decided to focus on an automatic system based on the transponder system.
The system (DoubleCage) was designed in cooperation with the University of Zurich to meet the requirements of our institute. The system should be able to continuously monitor/record the location of animals and providing continual, real-time data access; even quick movements should potentially be detected.
The experimental set-up, the DoubleCage (Figure 1), consisted of two Makrolon® cages (32.5 × 16.5 × 14 cm) connected by a Perspex tube (ø 3 cm, 30 cm long). Microchip antennas (sensors) were installed at both ends of the connecting tunnel to register the transition and position of mice individually. Animals were able to move freely between the two cages, the movements of an individual mouse between the two cages being registered automatically (real-time-data, in seconds) by microchip sensors and transferred to the computer. As there were two antennas, the crossing direction could also be detected automatically. To avoid possible data misreporting the experiment was set up in such a way that the transition of an individual animal was only counted when detected by both antennas. This system could register up to 16 animals (four per cage) at the same time. Temperature and light intensity were also recorded during the experiment.

DoubleCage structure (microchip-sensors are installed in the black rings)
To ensure reliability and accuracy, the present study validated the data recorded by the DoubleCage system, comparing these with the results of a videotape analysis (manual). Two inbred strains, BALB/cOlaHsd and C57BL/6JOlaHsd female mice obtained from Harlan Winkelmann (Borchen, Germany), were chosen for the present study. These animals were used for the FELASA training course either before or after the experiment. Each animal received a subcutaneous microchip (transponder, Euro ID Identifikationssysteme) under ketamine and xylazine anaesthesia (90 mg/kg body weight for ketamine 10%®, Weilerswist, Germany and 6 mg/kg body weight for xylazine 2%®, WDT, Garbsen, Germany). The animals were also dyed (Poly Palette No. 909, Schwarzkopf, Henkel, Germany) for distinguishing each individual during the manual observation (video analysis).
All animals were kept at a room temperature of 22 ± 1°C and 55 ± 10% relative humidity, with a 12/12 h light/dark cycle and with a light intensity of 150 ± 10 lux (measured 100 cm above floor level). Tap water in drinking bottles and a pelleted diet (Altromin No. 1324, Altromin GmbH, Lage Germany) were given ad libitum. As bedding softwood shavings were provided for each cage (Altromin type Grade 5, Altromin GmbH), cage and bedding were changed once a week.
After arrival all mice were randomly allotted to experimental units (in stable groups) and transferred to the DoubleCage two weeks before the experiment started. During the experiment mice in the DoubleCage were registered by computer and also videotaped using a time-lapse recorder (s/w CCD camera Panasonic, WV-330). Infrared light was provided when animals were videotaped for 24 h in the dark period.
In total 44 female mice (at 8–9 weeks of age) were used for a total of four separate sets of recordings in the present study (no mouse was re-used). The experiment started with 30 observation hours during the light period (four mice were randomly observed every second day for two weeks), followed by two times 12 h of continuous observation during the light period (8 and 16 mice respectively). Finally, 16 additional mice were continuously observed for 24 h (light and dark period). After transferring the data to the computer, the dwelling time for cages 1 and 2 during the entire experiment was calculated.
Manual video analysis was performed after the experiments. Data were analysed using Prism (5.0b, GraphPad Software Inc. 2009) software, comparing the data automatically registered by DoubleCage and those of the videotapes. For analysing the correlation between both sets of data, the data from all experiments were pooled. Due to the failure of microchip implantation, the data from five mice were incomplete. Thus, these data were not included for statistical purposes (total n = 39).
Even though there were some misreported transitions (0.93%), in most of the cases the program was able to correct the data automatically after the mice had moved again to another cage. The final misreported crossing was 0.26%. No statistical difference could be detected between the data collected with the DoubleCage program and video analysis. A significant correlation between the measurements of DoubleCage and videotapes was found for the crossing frequency (R 2 = 1.000, P < 0.0001, Figure 2) and for the dwelling time (P < 0.0001 for the dwelling time in both cages, R 2 = 0.997 and 0.999, Figure 2). A good agreement was found for the crossing frequency and the dwelling in both cages (Bland-Altman analysis, Figure 2). The differences obtained by the two methods are not significantly different (one sample t-test, hypothetical value = 0.0, P > 0.5 for the crossing frequency and the dwelling in both cages). However, it should be mentioned that two extreme cases were found for the 12 h test during the light phase. After the misreporting both mice did not move again until the end of recording; therefore, the program was not able to correct the mistake automatically. This led to a slightly reduced correlation between the visual and automatic observations. Such errors can be avoided/reduced if the active period of the animals (dark phase) is included in the analysis.
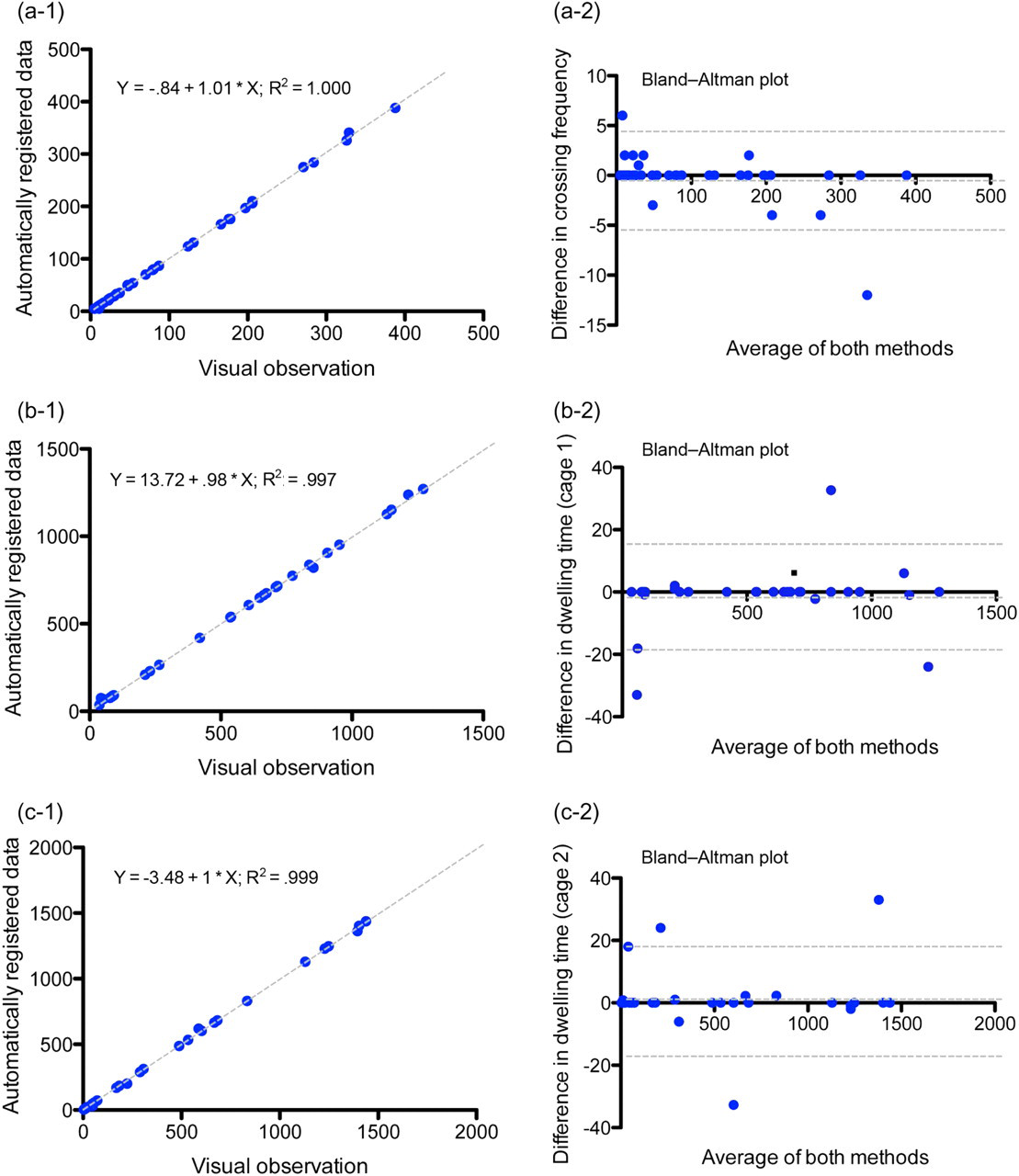
The correlation and Bland-Altman plot of crossing frequency (a) and dwelling time (b: cage 1, c: cage 2, n = 39)
According to the presented results the DoubleCage system can correctly determine the locations of each individual of group-housed animals during preference test and provide reliable results.
