Abstract
The aim of this study was to report the cardiorespiratory events observed during coronary artery occlusion and reperfusion in a minimally invasive closed chest myocardial occlusion–reperfusion model in rhesus monkeys. We hypothesized that a minimally invasive technique may lead to fewer cardiac arrhythmias and complications. Eight male rhesus macaques 10–15 kg and 10–15 years old were sedated with ketamine (2 mg/kg), midazolam (1.3 mg/kg), atropine (0.01 mg/kg) and buprenorphine 0.02 mg/kg intramuscularly. Etomidate 1–2 mg/kg was injected intravenously to allow tracheal intubation. Anaesthesia was maintained with isoflurane. Pulse oximetry, electrocardiogram (ECG), heart rate, mean arterial blood pressure (MAP), inspired isoflurane fractions (FIISO) and core temperature were recorded every 10 min. The coronary artery occlusion was induced by a balloon-tipped catheter advanced via the femoral artery into the left anterior descending artery and inflated to completely occlude the vessel for 20–50 min (IT) before reperfusion. Sequences of elevated ST segment, QRS complex prolongation, ventricular premature complexes and ventricular fibrillation were observed with a lower incidence than previously described in the literature. IT was (min: 17; max: 50) min long. FIISO was lower than the minimal alveolar concentration in these species. Hypotension (MAP < 70 mmHg) and hypothermia (T°C < 36°C) were observed in all macaques. This minimally invasive closed chest model was successful in providing better cardiorespiratory physiological parameters than reported in previous models. The benefit (achieving ischaemia) versus risk (lethal arrhythmia) of the duration of the coronary occlusion should be considered.
Cardiovascular diseases are the world's largest killers, claiming 17.1 million lives a year (World Health Organization). Animal models of ischaemic myocardial disease are now focused on myocardial reperfusion after a short period of ischaemia (induced by coronary artery occlusion) in order to mimic acute coronary condition (ACS), which affects millions of patients worldwide. 1,2 The size of the infarct area correlates with the severity of the heart failure and mortality. 3 This model is used to study the cardioprotective effects of drugs on the myocardial infarct size or necrotic area after reperfusion. 4,5
During the occlusion, myocardial ischaemia can lead to severe arrhythmia 6,7 and has been observed up to 25 h after experimental coronary ligation. 8
It is thought that because of the high cDNA sequence identity (97%) between man and non-human primates, the macaque may constitute a better model of the human heart than the hearts of other more commonly used laboratory species. 9 For example, primate hearts appear to infarct more slowly than those of other species. Between non-human primates some differences in the vascularization of the heart may be observed. For example, collaterals are less frequent and smaller in size in rhesus monkeys than in baboons. 10,11 However, such data are limited in monkeys due to ethical reasons, the expense of the animals and the need to house them in specialized facilities. 10 Cardiovascular changes during experimental myocardial infarction have been previously studied in unanaesthetized monkeys several days after a coronary ligature was placed by means of a thoracotomy under general anaesthesia. Opening the chest and pericardium, however, is painful and caused a high complication and mortality rate. 8
We hypothesized that a less invasive technique might lead to fewer cardiac arrhythmias and complications. To test this idea we developed a minimally invasive closed chest myocardial occlusion–reperfusion model in rhesus monkeys monitored by contrast-enhanced ultrasound imaging during long-term anaesthesia with isoflurane. 12 The lessons learned during this study will hopefully contribute to the goal of developing new therapies to treat ACS in man.
This present paper reports the cardiorespiratory events observed during the procedure.
Materials and methods
This prospective study was approved by the ethics committee of the National Veterinary School of Lyon on 28 April 2009, number 0925.
The study was performed in compliance with standard operating and quality procedures following published guidelines (OECD Principles on Good Laboratory Practice [as revised in 1997] and Council Directive of 24 November 1986 on the approximation of laws, regulations and administrative provisions of the Member States regarding the protection of animals used for experimental and other scientific purposes [86/609/EEC]).
Animals
Eight purpose bred adult male rhesus macaques (Macaca mulatta) 10–15 kg, 10–15 years old, were anaesthetized to allow transient occlusion of a coronary artery by balloon catheter followed by reperfusion. The series of experiments were designed so as to reduce the number of animals. Adult large-size animals were chosen in order to facilitate the introduction of the catheter into the coronary artery. Smaller animals with correspondingly smaller hearts had proven difficult to catheterize previously and were therefore excluded.
Macaques were imported from an European approved primate centre and entered into the testing facility two months before the beginning of the study.
Animals were housed individually in stainless-steel cages in order to prevent fights as these mature males had not previously been housed together. All the cages were placed together and no other species were housed in the same room. Ventilation was greater than 10 air changes per hour, with no air recirculation. Lighting was 12 h light/12 h dark photoperiod, and room temperature was 22 ± 3°C. Animal room and cage cleaning was performed on a daily basis. SDS Primate Diet (back numbers 6758, lapsing 27 August 2009) was provided daily in amounts appropriate for the size and age of the animals. Non-familiar food items were also occasionally given to the animals as part of the Testing Facility Environmental Enrichment Program. Tap water was available ad libitum to each animal via an automatic watering device.
Different games were also offered to the animals. The study animals were acclimatized to their designated housing for at least 28 days prior to the first day of the experiment. Complete examination, under sedation, conducted by a veterinarian, included abdominal palpation and observations of the condition of integument, respiratory and cardiovascular systems.
Animals were starved for 8 h for food and for 2 h for water. Five macaques were randomly selected for euthanization at the end of the procedure by an intravenous injection of pentobarbital 4 g (Doléthal ND; Vétoquinol, Lure, France). The heart, kidneys, liver, lung, spleen and adrenal glands were collected for the purpose of model development. 12 The three other macaques were allowed to recover.
Methods
The eight monkeys were premedicated with ketamine 2 mg/kg (Imalgene, Merial, Villeurbanne, France) and midazolam (1.3 mg/kg) (Midazolam Aguettant, Aguettant, Lyon, France) mixed in the same syringe and injected intramuscularly in the animal facilities. Stress during injection was limited as much as possible.
Atropine (0.01 mg/kg) (Atropine Aguettant, Aguettant) was injected subcutaneously after recumbency. Buprenorphine 0.02 mg/kg (Vetergesic, Sogeval, Laval, France) was administered intramuscularly.
Macaques were wrapped up in a blanket and transferred to the theater. An intravenous catheter was introduced in the left cephalic vein and lactated Ringer's solution was infused (5–10 mL/kg/h intravenously). Etomidate 1–2 mg/kg (Etomidate Lipuro, B-Braun, Boulogne Billancourt, France) was injected intravenously slowly to effect if necessary to allow tracheal intubation. An endotracheal tube was introduced into the trachea. Isoflurane in oxygen 100% or in oxygen (25%) and air (75%) was administered by an anaesthetic machine and a small animal circle breathing system.
Ventilation was maintained spontaneously. Body temperature was maintained with a heating pad (circulating hot water).
Pulse oximetry (SpO2) (probe on the finger), electrocardiogram (ECG), heart rate (HR), mean arterial blood pressure (MAP) by oscillometry (with the cuff positioned over the brachial artery), invasive blood pressure (catheter placed in the femoral artery), respiratory rate (fR), end tidal carbon dioxide (PE
At the end of the procedure, arterial blood samples were analysed for pH, partial pressures of oxygen (PaO2), carbon dioxide (PaCO2), and haemoglobin oxygen saturation (SaO2) with a blood-gas analyser (Opti CCA; AVL, Schaffhausen, Switzerland). Anaesthesia time (AT) was also recorded.
Acute myocardial infarction model
The right inguinal region was shaved and surgically prepared and drapped. A sterile 5F introducer sheath was placed percutaneously in the femoral artery over a guide wire. Then a 5F guiding catheter (launcher, Medtronic, Tolochenaz, Switzerland) was advanced under fluoroscopic guidance and placed in the left main coronary artery. Baseline angiograms were performed with different angles of incidence to detail the left anterior descending artery (LAD) anatomy. Angiograms were performed using either an OEC Fluorostar™ 7900 digital mobile C-arm fluoroscopy system (GE Healthcare, Little Chalfont, UK) or a BV PULSERA digital mobile C-arm fluoroscopy system (Philips medical system, Eindhoven, The Netherlands), using pulsed acquisition (30 pulses/s) with a cardiovascular pack. Isosorbide dinitrate 0.5 mg (Risordan®, Sanofi Aventis, Paris, France) was injected intracoronary. A 0.014 inch wire (Balance Middle Weight, Abbott, Rungis, France) was positioned in the distal part of the LAD and finally a balloon-tipped catheter (Maverik 2 TM, Boston Scientific, Nanterre, France) was advanced into the LAD and was inflated to completely occlude the vessel (8–12 bars) just after the first diagonal branch. The diameter of the balloon was chosen according to the angiogram to reach a 1.1 balloon-artery ratio. Aspirin 200 mg (Vetalgine®, CEVA, Libourne, France) and 500 UI of heparin were administered intravenously just before occlusion. Angiograms were performed to control the localization of the balloon, the inflation, the LAD occlusion and the reperfusion after deflation using 5–10 mL injections of Ioméprol (Iomeron®, Bracco Suisse, Plan-les-Ouates, Switzerland). The LAD was totally occluded for 20–50 min (IT) then the balloon was deflated. An angiogram was performed to confirm total coronary reperfusion, and the catheter was removed afterwards.
The ischaemic period was followed by at least 3 h of reperfusion with the animals continuously maintained under anaesthesia.
Myocardial perfusion status was assessed before and during occlusion and immediately after reflow using ultrasound contrast agent BR38 (BR38, Bracco Research, Geneva, Switzerland).
This ultrasound examination confirmed a complete occlusion of the LAD when the balloon was inflated without persistent perfusion defects during reperfusion. 12
Resuscitation protocol
When ventricular fibrillation (VF) occurred during the procedure, anaesthetic drug administration was stopped, oxygen was given at 100%, external defibrillation was performed by incremental currents 2; 3; 4 J/kg. The number of VF periods was recorded. Other adverse events (arrhythmias) were also recorded.
Postoperative care
The three macaques received buprenorphine (20 µg/kg) every 12 h during three days and ketoprofen (2 mg/kg) (Ketofen 10%, Merial) during five days. Clinical examinations were performed in the morning and late afternoon. The macaques were weighed the week after the procedure. No adverse effect was experienced either in the immediate or longer term (at least 2 years) postoperative recovery period. The ethics committee was involved in the follow-up during the postoperative period.
Results
Arrhythmias
Anaesthesia time (AT), coronary artery occlusion time (IT), pH, partial pressures of oxygen (PaO2) and carbon dioxide (PaCO2), haemoglobin oxygen saturation (SaO2) with a blood-gas analyser (Opti CCA; AVL, Schaffhausen, Switzerland) recorded just before recovery or euthanasia (after 330–460 min of anaesthesia) in eight macaques undergoing coronary artery occlusion and myocardial reperfusion under anaesthesia with isoflurane
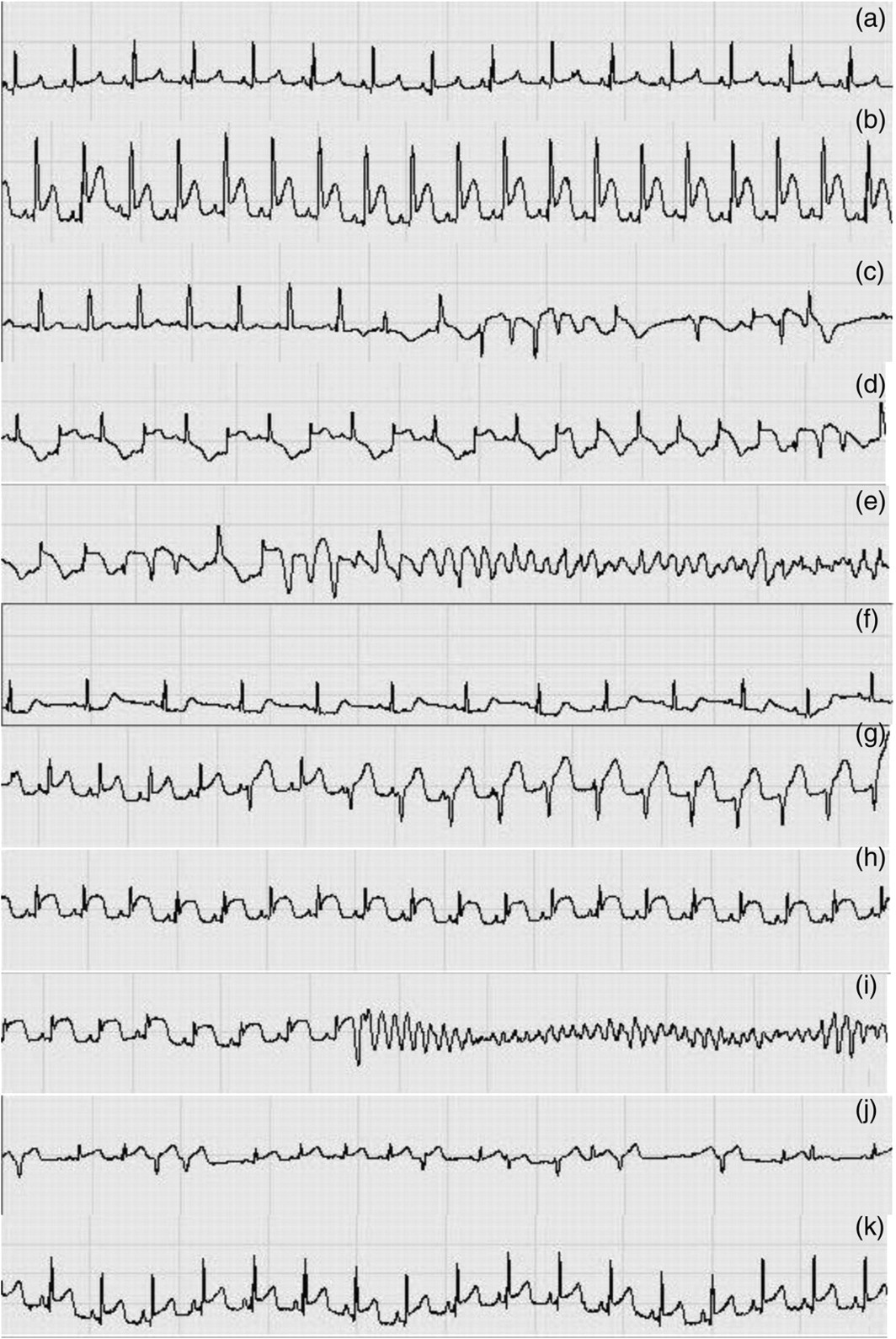
Electrocardiogram (ECG) recording during VI176 anaesthesia (time from induction). This figure is representative of the modifications observed in the other macaques’ ECG records. a (1 h 10 min): Normal ECG, before beginning of surgery. b–f: Introduction of the guide wire and placement of the guiding catheter in the LAD (1 h 10 min–2 h 40 min). b (2 h 18 min): ST segment elevation, QRS complex prolongation. c (2 h 20 min): QRS complex prolongation and VEC. d (2 h 21 min): PVC. e (2 h 22 min): VF. f (2 h 22 min): normal rhythm early after defibrillation. g–i (2 h 45 min–3 h 25 min): LAD occlusion. g (2 h 46 min): PVC. h (3 h 12 min): elevated ST. i (3 h 25 min): VF. j (3 h 28 min): PVC during early reperfusion. k (3 h 48 min): normal ECG. LAD: left anterior descending artery; VEC: ventricular ectopic complexes; PVC: premature ventricular complexes; VF: ventricular fibrillation
Cardiorespiratory parameters
As shown in Figure 2, during the LAD occlusion HR remained constant in all the macaques. The blood pressure dropped during the first 160 minutes after the induction of anaesthesia, independent of the time of the occlusion, with the mean arterial pressure value being below 70 mmHg. When the surgery was completed, during reperfusion, inspired fraction of isoflurane was decreased and anaesthesia depth was lightened, which was concurrent with an increase in blood pressure (Figure 3). Indeed, the mean inspired fraction of isoflurane required to maintain anaesthesia (0.9 ± 0.3%) was lower than the minimal alveolar concentration defined for this species (1.46%). Also shown in Figure 3 too, TC decreased from 36°C to as low as 32°C (for some macaques) with time despite the use of a circulating water heating pad until surgery was completed. Body temperatures were stabilized during reperfusion when macaques were wrapped in thick blankets and when additional active warming was initiated with a heater below the operating table.
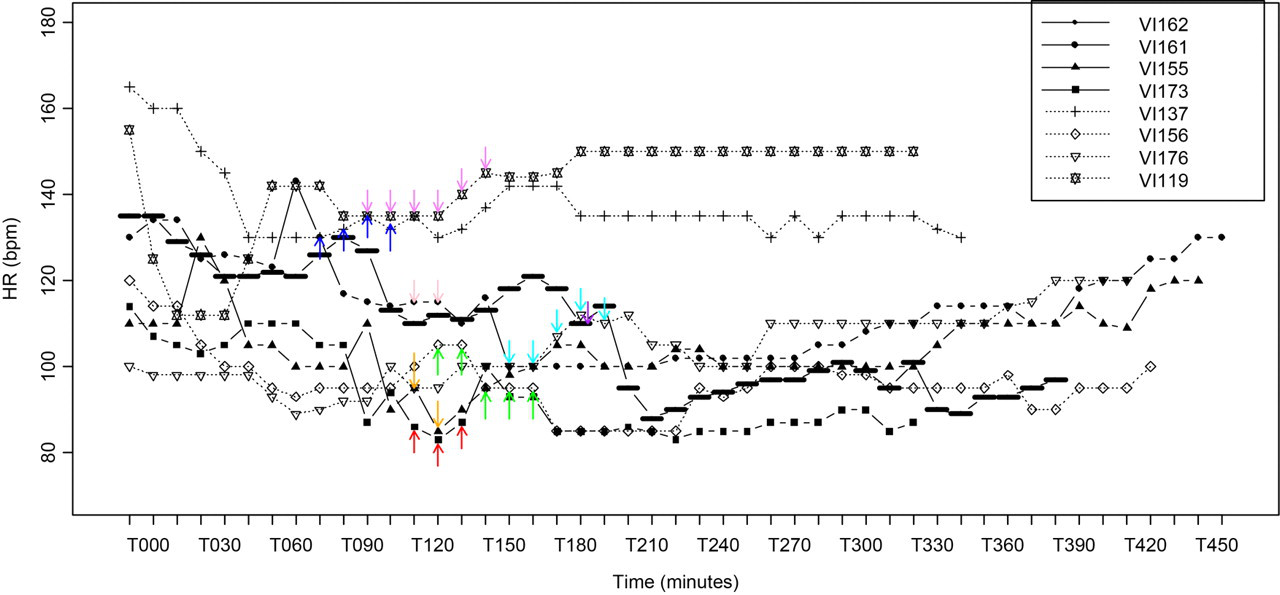
Heart rate (HR) in beats per minutes (bpm) recorded every 10 min in eight macaques (VI) undergoing coronary artery occlusion (arrows) and myocardial reperfusion under anaesthesia with isoflurane (VI173-155-161-162-176-156-119-137). Time is in minutes. A balloon-tipped catheter was introduced in the femoral artery, was advanced into the left anterior descending artery (LAD) and was inflated to completely occlude the vessel just after the first diagonal branch. Occlusion time (arrows) was 17–50 min. Then the balloon was deflated and reperfusion allowed until the end of anaesthesia (330–460 min)
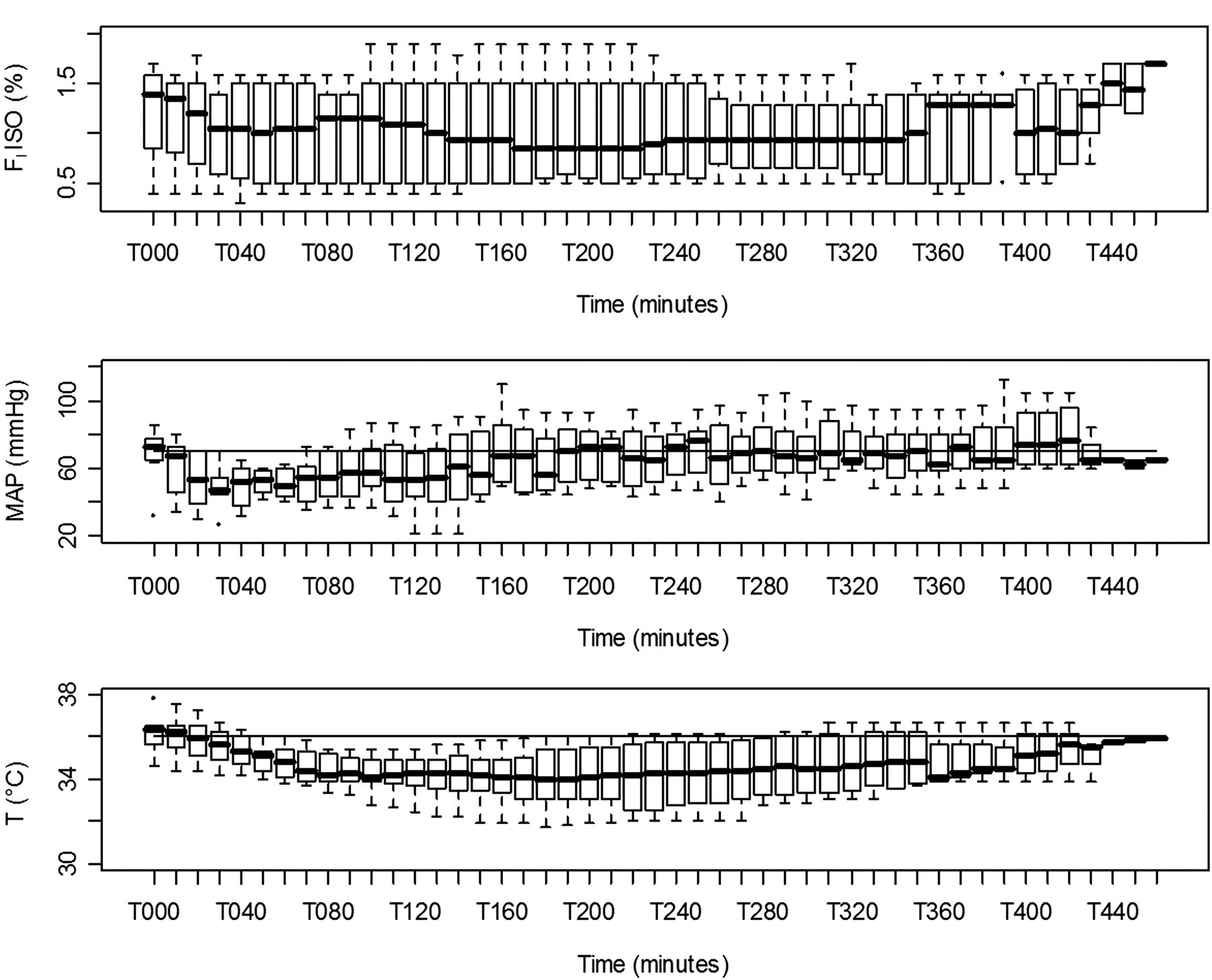
Inspired fraction of isoflurane (FIISO), mean arterial blood pressure (MAP), core temperature (T°C) recorded every 10 min in eight macaques undergoing coronary artery occlusion and myocardial reperfusion under anaesthesia with isoflurane. Time is in minutes. A balloon-tipped catheter was introduced in the femoral artery, was advanced into the left anterior descending artery (LAD) and was inflated to completely occlude the vessel just after the first diagonal branch. Occlusion time was 17–50 min. Then the balloon was deflated and reperfusion allowed until the end of anaesthesia (330–460 min). The box plots represent the first and third quartiles (Q1 and Q3 respectively) and the median. The lower dispersion bar represents, in the distribution, the value which is immediately superior to the adjacent value (Q1−1.5 [Q3–Q1]). The higher dispersion bar represents, in the distribution, the value which is immediately inferior to the adjacent value (Q1 + 1.5 [Q3–Q1])
Blood gases
As shown in Table 1 at the end of the procedure, acidaemia was observed with low arterial blood pH values (7.25 ± 0.15). This could have resulted from respiratory acidosis as arterial partial pressure in carbon dioxide was quite elevated (49 ± 8 mmHg). This hypercapnia may have resulted from hypoventilation because the macaques breathed spontaneously. Arterial partial pressure in oxygen was good (PaO2 was 291 ± 106 mmHg). Some macaques breathed 100% O2 whereas others breathed a mixture of oxygen (25%) and air which could explain this high standard deviation. Haemoglobin was almost fully saturated (99 ± 0.4%).
Discussion
Only two macaques out of eight exhibited VF during ischaemia. In comparison, in a canine model of myocardial infarction, 72% of the dogs presented with VF following transient ischaemia. 13 The onset of VF following coronary occlusion is a direct consequence of spatial chemical inhomogeneity (increased extracellular potassium and decreased ATP) that leads to an increase in the resting potential in the affected cells. 14 For the two macaques that presented with VF, this occurred at the end of 40 and 50 min occlusion time (the longest time that was applied during this study).
VF seemed to be associated with the duration of the LAD occlusion as it was observed in the macaques with the highest IT. Indeed, infarct size and perhaps severity of cardiac arrhythmias depend on the duration of ischaemia. 15,16 Yang et al. 10 reported VF in a macaque after 12 min of ischaemia while in three other macaques VF occurred during reperfusion. VEC were also observed. Hill et al. 8 reported on the outcome following experimental myocardial infarction with no reflow in unanaesthetized macaques. Of the 31 macaques involved in the study, five died postoperatively before ligation, 11 died within 6.5 h from myocardial infarction (cardiogenic shock) and only 15 survived more than 12 h. ST segment elevation and premature ventricular beats were observed immediately after the occlusion. Right bundle branch blocks were observed within one hour after infarction, but VF was the most serious and frequent arrhythmia recorded (18 animals). Ventricular flutter occurred in one macaque 25 h after coronary ligation. 8
In contrast with the above studies, in which frequent and severe arrhythmias were reported, within our minimally invasive closed chest myocardial occlusion–reperfusion model, an overall good cardiostability was observed during the procedure. This could have also resulted from the strain of macaque used (for example fatal arrhythmias in baboons during ischaemia reperfusion seem more common 17 than in cynomolgus macaque 10 ), from cardioprotective properties of the anaesthetic agents 18 and/or from the short duration of the coronary occlusion and/or from the lack of myocardial injury.
Nevertheless, arrhythmias and waveform changes (VF, VEC, ST segment elevation, QRS complex prolongation) were observed during the introduction of the catheter in the heart and coronary vessels and during the LAD occlusion. One of the macaques presented with a period of VF during the coronary artery catheterization. In other macaques VEC were also observed during the catheterization of the heart. Consistent with our results, arrhythmias during heart catheterization (pacemaker placement, coronary angioplasty, etc.) have been frequently reported by others. 19
Ventricular arrhythmias have been described as a sign of ischaemia achievement. ST segment elevation and QRS complex prolongation seem to be correlated to the degree of myocardial ischaemia/infarction. 20 Ischaemic tissues are characterized by low excitability, longer refractory period, reduction in action potential duration, slower conduction velocity and decreased frequency of ventricular tachycardia/VF. 21
In the present study, VF was successfully treated by external defibrillation with a current of 2 J/kg independent of the duration of the occlusion. This ease of VF treatment was similarly reported by Hill et al. 8 and Yang et al. 10 In a porcine model the defibrillation threshold was lower during coronary occlusion in comparison with non-ischaemic conditions. Nevertheless the energy required to defibrillate did not correlate with the duration of coronary occlusion. 22
No major changes in HR occurred during the procedure even during the LAD occlusion. Although the ECG was monitored continuously, the 10 min between recorded datapoints may have been too long and could have masked HR changes due to arrhythmias.
Hypotension was observed in most macaques and was unresponsive to colloids. Positively inotropic and pressor drugs were not allowed. The hypotension may have resulted from vasodilation induced by isoflurane. Isoflurane-induced hypotension has been exploited in experimental studies in rhesus macaques. 23 Other published studies in macaques did not report hypotension during anaesthesia under isoflurane, with mean MAP values of 75 and 77 mmHg at 1 and 0.8 MAC, respectively. 24,25 However, those procedures were short and the mean value may have masked some individual result. Indeed the lowest MAP value recorded in one study was 42 mmHg. 25
Marked hypotension during long-term anaesthesia with sevoflurane was reported in rhesus macaques by Corbach-Söhle et al. 26 Interestingly, in this study, MAP was not correlated to end tidal sevoflurane concentration, perhaps due to the limited range of concentrations used. In humans, isoflurane decreases MAP by 20%. 27 In Yang et al.'s study 10 anaesthesia was achieved with ketamine for sedation and sodium pentobarbital for induction and maintenance (bolus) and no changes were reported in haemodynamic variables (MAP and HR) during occlusion and reperfusion in comparison to baseline values.
In our model and for minimal invasive procedures, macaques can be immobilized with a much lower FIISO than the minimal alveolar concentration of isoflurane in oxygen given for the species (MAC = 1.46 ± 0.06% in rhesus macaques 25 ).
Previous studies indicate that the hypothermia experienced by the study animals may have enhanced the vasodilation induced by isoflurane. 28,29 Moreover, anaesthesia with isoflurane has been associated with greater hypothermia than ketamine–midazolam or propofol. 30 Finally, there is evidence that etomidate may have contributed to the hypotension as well. 31
Macaques were positioned in dorsal recumbency lying on a circulating hot water mattress. It was not possible to wrap them up until occlusion was completed, and hypothermia was observed in all of them. Immediately after reflow active re-warming was started.
This minimally invasive closed chest model was successful in providing better cardiorespiratory parameters than previous models during 20–50 min of myocardial ischaemia followed by reflow. Marked isoflurane-related hypotension was recorded in all macaques during anaesthesia. In order to improve cardiovascular function during minimally or non-invasive experimental studies, a much lower FIISO than the reported minimal alveolar concentration in macaques should be used.
In this myocardial ischaemia–reperfusion model, the benefit (achieving ischaemia) versus risk (lethal arrhythmia) should be considered when deciding what duration of coronary occlusion to use. VF was the most severe arrhythmia observed and was easily treated by external defibrillation.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to acknowledge Anais Michon and Alexandre Helbert for their technical assistance and Mathew Von Wronski for the English review.
