Abstract
Cage change is one of the unavoidable routines in laboratory rodent care. However, cage change disrupts the rodents’ olfactory environment and can evoke stress reactions. In this study, the short-term cardiovascular responses to three different cage change procedures were compared with telemetric monitoring. These procedures were: placing the rats into a new, clean cage (NEW), transferring the old cage lid into the clean cage (LID) and transferring an enrichment object into the clean cage (ENR) with the animals. Seven outbred rats (four Hsd:Sprague-Dawley and three HsdBrlHan:WIST) were instrumented with telemetric transmitters. The reactions were recorded during the 24 h following the cage change procedures. All cage change procedures (and also simple handling) caused elevated heart rate and mean arterial pressure levels for up to 5 h after the procedure, with the largest effect seen during the first hour. The reactions observed after cage change were significantly (P < 0.05) greater than those observed after simple handling. The reactions after NEW were significantly higher than the reactions after ENR or LID, though the results were dependent on the stock. In Wistar rats the LID procedure resulted in smaller reactions than ENR. In Sprague-Dawley rats, the differences between ENR and LID were not so clear, but the transfer of scent-marked material into the new cage decreased the reactions compared with the NEW procedure also in this stock. Based on these results, using the old cage lid on the new cage could reduce the disturbance of cage change in rats.
The way routine procedures are conducted in the laboratory can have a great impact on both animal welfare and quality of research results. One important and recurring part of the maintenance of laboratory rodents is the cage change. Most commonly, the rat cages are changed once or twice a week by removing the animals from their old home cage and placing them into a new, clean cage with new, clean bedding, nesting material and often also with new or clean enrichment objects. This practice may guarantee a high level of hygiene, but it does disrupt the rodents’ olfactory environment. Cage change has been shown to be a stressor for both rats and mice, evoking an elevation in blood pressure, heart rate (HR) and locomotor activity and affecting the animals’ behaviour. 1–6 The cage change procedure exposes the animals to a variety of stressors, such as handling and noise, 7 often during the daytime, when the animals’ reactions to disturbances are more pronounced than during their active period. 8 In mice, the specific effect of disruption of olfactory cues during cage change has been studied much more thoroughly than in rats. 9
In rats, olfaction is an important sense. 10–12 Problems with aggression are rare in group-housed rats and have not been reported to be a problem after cage change procedures. 1,10 However, rats do react to cage change by exploring their surroundings. 13,14 This might not be completely benign, since prolonging the interval between cage changes has been shown to have positive effects on animal wellbeing. 15
The aim of this study was to compare the short-term cardiovascular responses of rats to three different ways of cage changing: placing the rats into a completely new, clean environment, transferring the old cage lid with the rats to the clean cage and transferring an enrichment object (aspen tunnel) to the clean cage. These cage change procedures differed in the way that olfactory cues were transferred from the old home cage to the new cage. The response of the rats to these procedures was assessed by telemetric monitoring of HR and blood pressure with the goal being to determine the least disturbing way to change cages and also to establish the duration of the possible reactions. We hypothesized that moving the old enrichment to the clean cage would result in a less intense cardiovascular response than moving the rats to a completely new environment.
Materials and methods
The study protocol was reviewed and approved by the Animal Care and Use Committee of the University of Kuopio, Finland. The study was conducted in the Laboratory Animal Centre (LAC), University of Oulu, Finland and in the National Laboratory Animal Center (NLAC), University of Kuopio, Finland.
Animals and housing
A total of 21 male barrier-bred rats were used in this study. The study population consisted of 12 Hsd:Sprague-Dawley (Sprague-Dawley) rats from LAC (Oulu, Finland) and nine HsdBrlHan:WIST (Wistar) from NLAC (Kuopio, Finland). The barriers were free of the pathogens listed in the FELASA recommendations for health monitoring. 16 The rats were housed in solid bottom Makrolon® polycarbonate cages (55 × 35 × 20 cm) with a wire-grid lid (Bayer AG, Leverkusen, Germany). Ambient temperature was kept at 20 ± 2°C and relative air humidity at 50 ± 10%. Room illumination followed a 12/12 h cycle, with lights on at 07:00 h. Food (R36, Lantmännen Ab, Södertälje, Sweden) and tap water in polycarbonate bottles were available ad libitum. Aspen chips (3 L) were used as bedding (Tapvei Oy, Kaavi, Finland) and rectangular aspen tubes (20 × 10.5 × 10.5 cm, wall thickness 1.5 cm) as enrichment objects (Tapvei Oy). Cages, bedding, aspen tubes and water bottles were renewed once a week.
Study design
The rats were housed in groups of three. From each cage, one randomly chosen rat was implanted with a telemetry transmitter (TA11PA-C40; Data Sciences International, St Paul, MN, USA) at the age of 11–12 weeks (altogether 7 rats). The details of anaesthesia, analgesia, surgery and collection of the telemetric data have been described in a previous publication. 17 The animals were allowed to recuperate for approximately 10 days before the study began.
The experiment used a crossover design. Each of the implanted animals was subjected to the four different methods of cage change. The order of the cage change procedures was randomized. Between each treatment, the rats were allowed a washout period of two weeks.
Cage change procedures
OLD – the rat was lifted from its home cage, and immediately placed back into the same old cage with old bedding, food, water and enrichment object.
NEW – the rat was lifted from its home cage and placed into a new, clean cage with new, clean bedding, cage lid, food, water and a new enrichment object.
ENR – the rat was lifted from its home cage and placed into a new, clean cage with new, clean bedding, cage lid, food and water. The old enrichment object (aspen tunnel) was placed into the new cage.
LID – the rat was lifted from its home cage and placed into a new, clean cage with new, clean bedding, food, water and a new enrichment object. The old cage lid was moved onto the new cage.
During each cage change procedure, all rats in the cage were gently lifted by the base of the tail. The first rat taken from the cage was always the implanted rat. All rats were handled at a constant speed (3 s/rat). The cage change procedures were always performed at 14:00 h. Twenty-four hours after the procedure, the old enrichment object or the old cage lid was replaced with a clean version in order to make the cages equally clean during the rest of the washout period.
Data processing and statistical analyses
The telemetric data were recorded for 10 s every 5 min. The mean arterial pressure (MAP) and the HR recordings were collected. The 5 min means of MAP and HR values were then used in the statistical analysis. In Wistar rats, the activity (counts/min) was also recorded.
The different cage change procedures were compared with each other using linear mixed models for repeated measures. Fixed effects in the model were time, treatment and stock (stock was not used as a fixed effect when the two stocks were analysed separately). The individual was included as a random effect in the model to account for possible dependencies within the data. The effect of stock on the means of MAP and HR was also analysed as were the possible interactions between stock and treatment. All data were analysed with both stocks together in the analysis as well as separately for both stocks. In addition, the data from Wistar rats were analysed using all of the data and using only the time points where activity was close to zero (<1 count/min). In the statistical analysis, the 24 h following the cage change treatments was divided into four time periods: 0–1 h after the cage change, 1–5 h (i.e. the rest of the light phase), 5–17 h (i.e. the following dark phase) and 7–24 h (i.e. beginning of the next light phase up to 24 h of follow-up). The results are presented as means and their standard deviations (SD) for each time period.
The statistical software package used to process and analyse the data was SPSS 14.0 for Windows (SPSS Inc, Chicago, IL, USA). The graphs were created with SigmaPlot 11.0 software (Systat Software Inc, San Jose, CA, USA).
This manuscript was prepared according to the ARRIVE guidelines 18 and GSPC list. 19
Results
Effect of stock
The HR levels of Wistar and Sprague-Dawley rats did not differ significantly (P < 0.05) at any of the time periods. However, during the 5–17 and 17–24 h periods, the interactions between treatment and stock were statistically significant in the HR analyses. In MAP, the stocks differed significantly during all of the time periods except the 0–1 h period, with Sprague-Dawley rats showing higher MAP levels. In MAP, the interactions between treatment and stock were also statistically significant during all of the time periods except for the 0–1 h period. Due to these differences between the stocks and the interactions between the stocks and treatments, the results are reported separately for Wistar and Sprague-Dawley rats.
HRs in Wistar rats
During the first hour, the mean HRs ranged from 408 to 423 beats per minute (bpm). The differences between the cage change protocols were not statistically significant. During the 1–5 h time period, the means were between 342 and 351 bpm, without any significant differences between the groups. During the dark period (5–17 h) the HR was 378 (SD 57) bpm in OLD, 397 (54) bpm in NEW, 398 (54) bpm in ENR and 388 (54) bpm in LID. The HR of OLD was significantly lower than that of NEW, ENR and LID. Of the three actual cage change protocols, LID showed significantly lower values than NEW and ENR. Even though some statistically significant differences were found during the next day (17–24 h), the absolute differences were small. The means ranged from 337 to 344 bpm (Figure 1, Table 1).
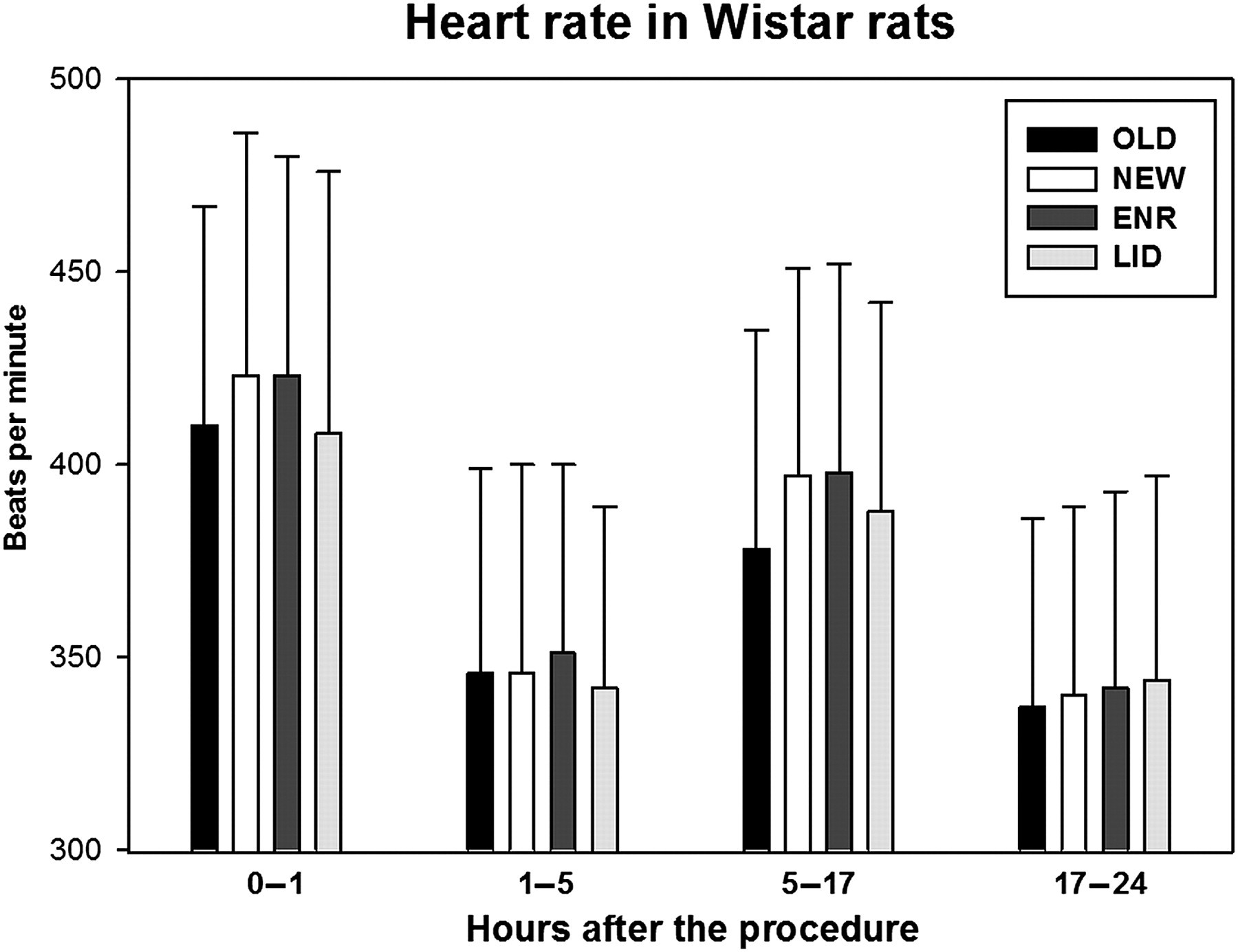
Heart rate in Wistar rats during the first 24 h following the different cage change procedures. The dark period is situated in the time window 5–17 h. The error bars represent the standard deviations of the data. ENR: clean cage, old enrichment; LID: clean cage, old lid; NEW: clean cage; OLD: same cage
Values of the heart rates (HR) and mean arterial pressures (MAP) at different times of the experiment
The four treatments are compared with each other by presenting the P values (P). When the difference is statistically significant, the relative difference of the means (%) is also presented
bpm: beats per minute; ENR: clean cage, old enrichment; LID: clean cage, old lid; NEW: clean cage; NS: not significant; OLD: same cage; SD: standard deviation
HRs in Sprague-Dawley rats
During the first hour, the mean HRs ranged from 386 to 399 bpm. The differences between the cage change protocols were not statistically significant. During the 1–5 h time period, the mean HR was 352 (45) bpm in OLD, 361 (42) bpm in NEW, 358 (46) in ENR and 359 (52) bpm in LID. OLD was significantly lower than NEW and LID. During the dark period (5–17 h) and the next day (17–24 h), the absolute differences were very small (means 388–393 and 314–321 bpm, respectively) (Figure 2, Table 1).
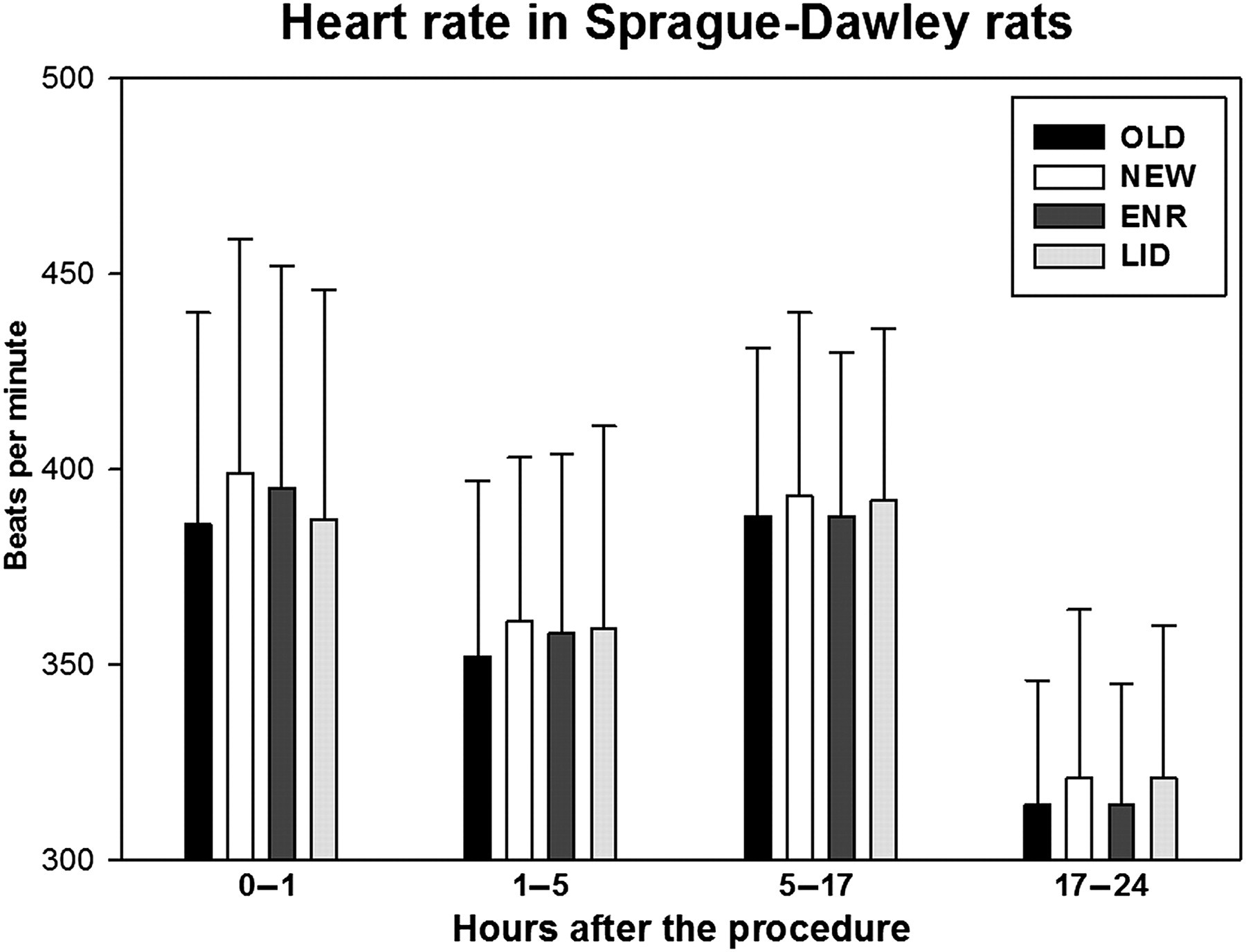
Heart rate in Sprague-Dawley rats during the first 24 h following the different cage change procedures. The dark period is situated in the time window 5–17 h. The error bars represent the standard deviations of the data. ENR: clean cage, old enrichment; LID: clean cage, old lid; NEW: clean cage; OLD: same cage
MAP in Wistar rats
During the first hour after the cage change procedures, the mean MAP was 107 (7) mmHg after the OLD procedure, 110 (9) mmHg after the NEW, 110 (8) mmHg after ENR and 106 (10) mmHg after LID. OLD and LID were significantly lower than NEW and LID was also significantly lower than ENR. During the 1–5 h time period after the cage change procedures, the differences were very small (mean 98–100 mmHg), but LID was significantly lower than any of the other groups. During the dark period (5–17 h), the means ranged from 104 to 107 bpm with OLD and LID significantly lower than NEW and ENR. During the next day (17–24 h), the means had declined to 99–103 bpm, with ENR significantly higher than the corresponding values in the other groups (Figure 3, Table 1).
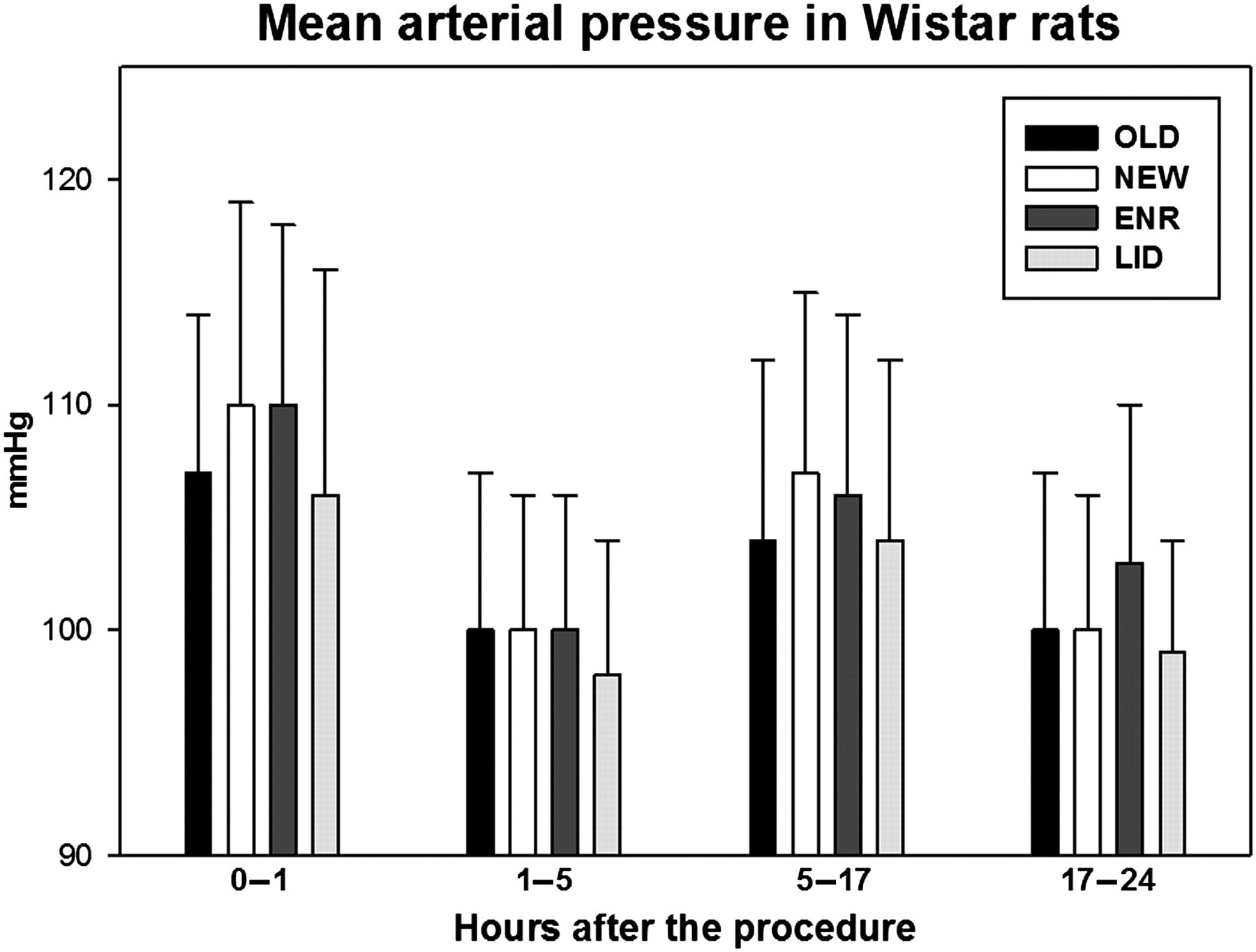
Mean arterial pressure in Wistar rats during the first 24 h following the different cage change procedures. The dark period is situated in the time window 5–17 h. The error bars represent the standard deviations of the data. ENR: clean cage, old enrichment; LID: clean cage, old lid; NEW: clean cage; OLD: same cage
MAP in Sprague-Dawley rats
During the first hour after the cage change procedures, the mean MAP was 112 (SD 9) mmHg after the OLD procedure, 116 (8) mmHg after the NEW, 115 (8) mmHg after ENR and 111 (11) mmHg after LID. OLD and LID were significantly lower than NEW and ENR (P < 0.05). During the remainder of the light period (1–5 h after the cage change) the mean MAP was 115 (8) mmHg after the OLD procedure, 17 (8) mmHg after the NEW, 106 (8) mmHg after ENR and 105 (9) mmHg after LID. OLD, ENR and LID were all significantly lower than NEW. During the dark period (5–17 h), the absolute differences were negligible (means 111–112 mmHg). The next day the differences were no longer statistically significant (Figure 4, Table 1).
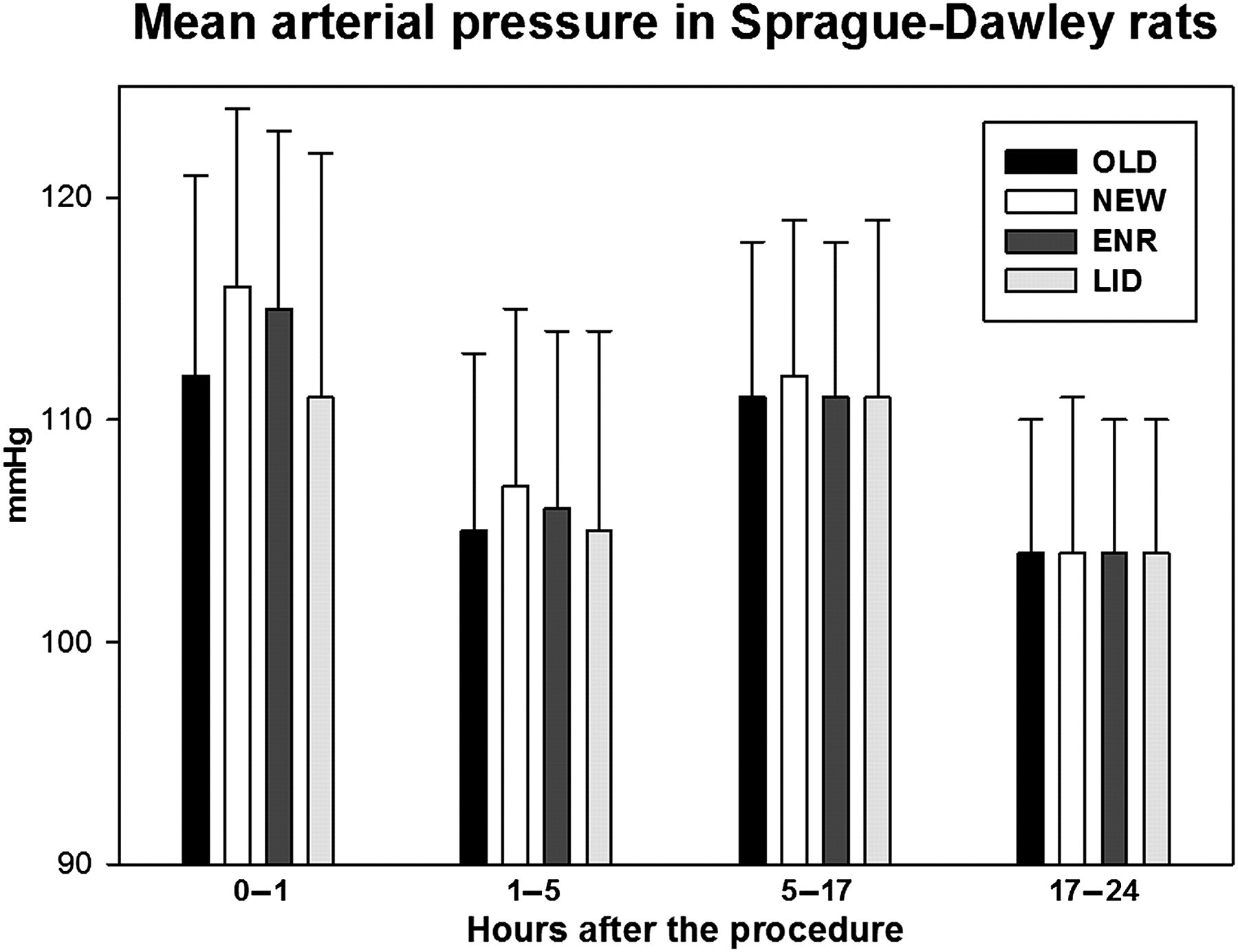
Mean arterial pressure in Sprague-Dawley rats during the first 24 h following the different cage change procedures. The dark period is situated in the time window 5–17 h. The error bars represent the standard deviations of the data. ENR: clean cage, old enrichment; LID: clean cage, old lid; NEW: clean cage; OLD: same cage
Activity in Wistar rats
The activity was not directly analysed with the linear mixed model due to its skewed distribution (over 50% of the recordings were very close to zero). The HR and MAP in Wistar rats were analysed twice, first using all recordings and then using only the time points where activity was <1 count/min. HR and MAP from the inactive time points were lower than the means from all data, but there were no differences in the relative differences between the cage change protocols.
Discussion
Rodents have a well-developed sense of smell and they communicate by scent-marking. 10,11,20,21 Cage change has been shown repeatedly to be a stressor for rats, resulting in elevated HR and blood pressure. 2–4,8,14 In mice, the possibility of reducing the stress caused by cage change by transferring some of the scent-marks from the old cage to the new cage has been investigated. Old nesting material reduced aggression after cage change, whereas transferring other scent-marks, such as bedding, seemed to intensify aggression. 9 It is recommended that odour cues be left for rodents during the cage cleaning process. 22
In rats, aggression is much more rare than in mice and is not considered a problem after cage change. 1,10 The possible benefits of transferring scent-marks during cage change in rats have not yet been demonstrated. On the contrary, in 2001 Duke et al. 2 reported that transferring a small amount of soiled bedding to the new cage did not decrease the cardiovascular stress responses to cage change. Furthermore, Burn and Mason (2008) conducted a preference test in rats between a soiled, familiar cage and a clean, unscented cage. The rats displayed no differences in their preferences for these two environments. 23 These results might indicate that scent-marks are not very important during cage change in rats.
In this study, we compared three different cage change procedures in rats to determine the least disturbing method for the animals. The procedures chosen were placing the rats into a completely new and clean cage (NEW), placing the rats into a clean cage with their old aspen tunnel from the previous cage (ENR) and placing the animals into an otherwise clean cage with their old cage lid from the previous cage (LID). As a control, the animals were picked up and placed directly into their own, old cage without changing anything (OLD).
The effects of the different cage change procedures were determined by telemetric monitoring of HR and MAP. Telemetric monitoring 24–26 is commonly used to assess the stress responses of animals to different procedures and housing conditions. 4,27–29 The elevation of blood pressure and HR is a fundamental component of the stress reaction. 30–33
The results of this study revealed that generally the reactions after NEW were significantly higher than the reactions after ENR or LID, where scent-marked material had been transferred to an otherwise novel environment. In Wistar rats, the LID procedure resulted in smaller reactions than ENR. In Sprague-Dawley rats, the difference between ENR and LID was not so clear, but the transfer of scent-marked material into the new cage decreased the reactions compared with the NEW procedure also in this stock.
The two rat stocks, Wistar and Sprague-Dawley, also differed significantly in terms of their MAP values, with Wistar rats showing lower values throughout the experiment. There were no statistically significant differences between the HRs of the stocks.
The high levels of MAP and HR observed during the first hour after the procedures are likely to be a result of handling in the OLD group and the combination of handling and novelty in NEW, ENR and LID groups. The effects of handling are reported to be limited to one hour, 17,27,34,35 which was also confirmed in our previous study comparing handling techniques in the same rats as used in this study. 17
HR and MAP follow a prominent diurnal rhythm, with low MAP and HR during the light period, and higher values during the dark period in undisturbed rats. 36,37 During the 1–5 h period after the procedures, HR and MAP decreased in all cage change groups to levels lower than those observed during the night time (5–17 h). During the night time, the OLD group still showed significantly lower values than the NEW and ENR groups. During the next day, the levels of HR and MAP were lower than during the previous day (1–5 h). This indicates that during the 1–5 h period after the procedure, the levels of HR and MAP in all groups were still higher than a normal daytime baseline. The duration of the effects thus exceeded the effects of mere handling, indicating that cage change may be a more intense stressor for rats than mere handling. Kemppinen et al. 4 reported that cage change also resulted in larger reactions than gavage, further emphasizing the significance of the cage change procedure in rat husbandry.
Due to technical limitations, we were only able to monitor activity in Wistar rats. The data from Wistar rats were analysed first using all observations and then restricted to only the observations where the activity count was very close to zero (animals inactive). The relative order, differences and statistical significance of the comparisons between the cage change protocols remained unchanged. Values of HR and MAP calculated from the inactive observations were consistently and significantly (P < 0.001) lower than those detected from observations during activity. The fact that HR and MAP were always lower during inactivity may indicate that the elevations observed in HR and MAP during the first 5 h were related to locomotion and exploration 1,13 rather than a pathological stress reaction. 29,33,38,39 On the other hand, increased locomotion is also considered a reaction to novelty stress. 40 However, it is difficult to judge whether this reaction to novelty is in fact detrimental to the animals.
It should be noted that telemetric monitoring yields a vast quantity of data and therefore the analysis may produce statistically significant differences even when the absolute differences are minor. Attempts to resolve this dilemma have been made by using a 6% threshold as guideline for deciding whether an observed effect is truly significant. There is no conclusive evidence to suggest that this would be the right threshold in all situations, but after Krohn et al.'s publications in 2003, 29,41 the 6% limit has been used by several authors. In this study, none of the absolute differences between the cage change procedures exceed the 6% limit (Table 1). Kemppinen et al. 4 described another method of estimating the biological relevance of observed differences in telemetric data. These workers compared the magnitude of the difference between treatments to the magnitude of the difference between the night and day averages in the same variable. They argued that if the difference is smaller than the night–day difference, its significance in terms of animal welfare is likely to be minor. In this study, none of the differences observed between the reactions to different cage change procedures exceeded that threshold.
In conclusion, cage change (and also simple handling without any concomitant change of the cage) elevated HR and MAP levels for up to 5 h after the procedure, with the largest effect seen during the first hour. The reactions observed after cage change were significantly greater than those observed after simple handling. The timing of cage change (and handling) should be adjusted to the research protocols; cage change during the same day may alter or confound the results obtained from the animals. The cage change procedure had statistically significant, albeit small in absolute numbers, effects on HR and MAP. In Wistar rats, transferring scent-marks from the old cage via the cage lid resulted in smaller reactions than the other methods. In Sprague-Dawley rats, the differences between the cage change methods were not as consistent as observed in Wistar rats, but it seemed that transferring scent-marks from the old cage via environmental enrichment or the cage lid resulted in smaller reactions than placing the animals into a completely new cage. Based on these results, using the old cage lid on the new cage could be a way to reduce the disturbance of cage change in rats. Another possibility to minimize the disturbance caused by cage change might be to decrease the frequency of cage change.
Footnotes
ACKNOWLEDGEMENTS
We thank Dr Marja Luodonpää, technicians Tuula Mäkinen and Veikko Lähteenmäki (University of Oulu, Finland) and Dr Hanna Leskinen and technician Heikki Pekonen (University of Eastern Finland, Finland) for their help with the procedures and data collection.
