Abstract
In the present study, we examined whether standard chow (SDS versus Purina 5001; both low fat, high carbohydrate) and reductions in hexokinase (HK) II (wild-type versus HKII+/− mice) affect (1) growth parameters, (2) HK levels in cardiac and skeletal muscle and (3) low-flow cardiac ischaemia–reperfusion (IR) injury. Total HK activity and HKI and HKII expressions were determined, and low-flow IR injury was examined in isolated hearts subjected to 40 min 5% low-flow ischaemia and 120 min reperfusion. Standard chow, but not HKII reductions, significantly affected body weight, heart weight and cardiac hypertrophy. Both standard chow and reduced HKII diminished total cardiac and skeletal muscle HK activity. For the heart, the Purina chow-induced decrease in total HK activity was through decreases in HKI expression, whereas for skeletal muscle post-translational mechanisms are suggested. Both standard chow and reduced HKII demonstrated a non-significant trend for affecting cardiac IR damage. However, the low-flow ischaemia model was associated with mild sublethal injury only (∼1% cell death). In conclusion, standard chow affects body weight, heart weight and HK activity and HKI expression in the heart, without altering HKII expression. This implicates standard chow as an important factor in genomic, physiological research models and demonstrates that large differences in fat or carbohydrates in the diet are not necessary to affect growth. In a cardiac low-flow IR model, resulting in only mild injury, standard chow or reduced HKII does not significantly affect IR damage.
To our knowledge, standard chow has received little attention in the field of animal experimentation. In contrast, much attention has been given to diets that differ largely in fat or carbohydrates, comparing e.g. low-fat with high-fat diets or low-carbohydrate with high-carbohydrate diets. However, often these diets not only differ in their fat or carbohydrate composition but also in many other components because diets are obtained from different suppliers. 1–4 Uncertainty then exists as to whether observed effects can be ascribed to alterations in the main constituent (fat or carbohydrate) or to differences in the background standard chow used. Obviously, information is needed concerning the effects that standard chow per se may have on growth and other physiological parameters. We examined this question using wild-type (WT) and HK+/− mice. The HK+/− mice were shown to have a 50% reduction in hexokinase (HK) II activity in adipose, skeletal and heart tissues, without an effect on HKI activity. 5 However, our own initial results with the HK+/− mice on HK activity did not show a significant difference between WT and HK+/− hearts. Interestingly, a recent study 1 demonstrated that difference in cardiac HKII content between WT and HK+/− hearts disappeared when animal nutrition was changed from low-fat to high-fat chow. However, the diets used also differed in background standard chow. It cannot be excluded that part of the observed changes in HK can be ascribed to differences in the standard chow used. Therefore, in the present study, we first of all examined the effects of two standard laboratory chows and reductions in HKII on body weight, heart weight and HK (activity and isoform expression) in heart and skeletal muscles in mice.
As a second aim, we investigated whether HK may confer cardioprotection against ischaemia–reperfusion (IR) injury. The possible role of HK in cell survival first became evident from cancer research, because tumour cells are able to survive due to enhanced HK activity and expression. 6 HK binds directly to the mitochondrial permeability transition pore, where it exerts its protective effect on cell death. 7–9 Very recently, it was described that besides mitochondrial binding glucose phosphorylation is also required for cellular protection. 10 We demonstrated HK translocation to the mitochondrion in cardiac tissue in reaction to cardioprotective interventions 11,12 and showed increased mitochondrial HK activity before and after ischaemia following ischaemic preconditioning. 13 Although these data in an intact heart do show a possible role for HK in cardioprotection against IR injury, it remains unknown as to whether HK is causally related to IR injury. 14 Therefore, in the present study, we hypothesized that cardiac HKII reduction, which is the most abundant HK isoform in the heart, results in increased IR damage in the intact heart. For this purpose, we applied a low-flow IR model in isolated hearts of heterozygous HKII (HK+/−) mice.
Material and methods
All procedures were in accordance with the Animal Ethical Committee of the University of Amsterdam and conform to NIH guidelines.
Animals and standard chow
C57BL/6J mice containing a partial deletion of the HKII gene (HK+/−), shown to have a reduction in HKII activity in adipose, heart and skeletal muscle tissues, but not in HKI activity, 5 were in-house bred under standard AMC housing conditions (12 h dark/12 h light cycle; water and food given ad libitum). Breeding couples were assigned to either (the local standard chow) Rat and Mouse Breeder and Grower Expanded standard chow CRM (SDS; Special Diet Services Ltd, Witham, UK) or Purina Laboratory Rodent Diet 5001 standard chow (Purina; PMI Nutrition International, St Louis, MO, USA). No large differences in the major caloric chow macronutrients were present between chows (fat 12.1% versus 9.1%; protein 28.0% versus 22.2%; carbohydrates 59.8% versus 68.7%, for Purina and SDS chow, respectively; Table 1). At 21 days, pups were weaned and fed the same standard chow as their parents. Genotypes were determined by standard polymerase chain reaction technique after DNA extraction from toe tissue and dissolving in standard TE buffer.
Composition of SDS and Purina standard chows
Experimental procedures
Four different groups (n = 10–13 per group; male and female) were studied: group (1) WT mice fed SDS chow (3 male, 6 female); group (2) HK+/− mice fed SDS chow (4 male, 5 female); group (3) WT mice fed Purina chow (5 male, 8 female); group (4) HK+/− mice fed Purina chow (4 male, 7 female). HK+/− and their WT littermates at 12–15 weeks of age were anaesthetized with s-ketamine (125 mg/kg) and medetomidine (0.2 mg/kg) by intraperitoneal injection. Heparin (15 IU) was also injected intraperitoneally. After performing tracheotomy and thoracotomy, the aorta was cannulated in situ and perfusion immediately started before excision of the heart from the chest. Each heart was Langendorff-perfused at 37°C with Krebs–Henseleit buffer containing (mmol/L): 118 NaCl, 4.7 KCl, 2.25 CaCl2, 1.2 MgSO4, 25 NaHCO3, 1.2 KH2PO4, and 11 glucose, 1 lactate, 0.5 glutamine and 0.1 pyruvate, gassed with 95% O2/5% CO2. Perfusion was by constant flow with an initial perfusion pressure of 80 mmHg. A cannula was inserted in the myocardial apex to drain Thebesian venous effluent from the left ventricular lumen. A balloon made of clingfilm (Clingorap PVC Clingfilm, Terinex, UK) was inserted into the left ventricle, connected to a pressure transducer and inflated with water to achieve an end-diastolic pressure (EDP) between 2 and 8 mmHg. After a stabilization period of 20 min, measurements started with a 20 min period of perfusion (baseline). Subsequently, 40 min of global low-flow ischaemia was performed with flow at 5% of baseline flow. Ischaemia was followed by 120 min reperfusion at baseline flow. Throughout the reperfusion period, venous effluent was collected at fixed times (5, 15, 30, 60 and 120 min) for lactate dehydrogenase (LDH) enzyme activity measurements. The time-integrated sum of activities from all time points resulted in the total release of LDH enzyme from the heart, as parameter for total necrosis occurrence during reperfusion.
Tissue homogenization and fractionation
To determine the effect of standard chow and HK genotype on basal HK activity and isoforms in heart and gastrocnemius medialis (GM) muscles, four additional groups (WT, SDS: 3 male, 4 female; HK+/−, SDS: 3 male, 2 female; WT, Purina: 3 male, 4 female; HK+/−, Purina: 3 male, 3 female) were examined (n = 5–7 per group). The heart and GM were excised from the anaesthetized animals and directly homogenized in 1.5 mL ice-cold isolation buffer containing (mmol/L): 250 sucrose, 20 HEPES buffer (pH 7.4), 10 KCl, 1.5 MgCl2, 1 EDTA, 0.1 phenylmethylsulfonyl fluoride, 5 µg/mL leupeptin, 5 µg/mL aprotinin and 1 µg/mL pepstatin A. Samples were directly stored at −80°C until further analysis.
Biochemical analysis – enzyme activity measurements
GM and heart homogenate were treated with 0.5% Triton X-100 and 0.9 mmol/L KCl to maximally solubilize HK, 15 followed by centrifugation (12,000 g; 1 min) to pellet crude undissolved remnants. All enzyme activities were spectrophotometrically determined in the resulting supernatant at 25°C. HK activity was measured using glucose-6-phosphate dehydrogenase, glucose, ATP, NAD and rotenone. GM and heart HK activities were normalized to protein content as determined by the Bradford method. 16,17 LDH, as index of necrosis, was measured using KH2PO4 buffer, NADH and pyruvate.
Immunoblotting
HKI and HKII protein levels were detected using the Western blot technique. Equal protein amounts of GM and heart (both 100 µg/lane) homogenate were loaded on a 10% sodium dodecycl sulphate-polyacrylamide gel, separated by reducing electrophoresis and transferred to a polyvinylidene fluoride membrane. Blots were blocked using a 5% bovine serum albumin solution and subsequently incubated overnight at 4°C, with either anti-HKI (1:40,000; Santa Cruz, CA, USA), anti-HKII (1:20,000; Chemicon, Hampshire, UK) or α-tubulin (α-TUB; 1:40,000; Sigma, St Louis, MO, USA). Following 2 h of incubation with horseradish peroxidase-conjugated secondary antibody and washing of blots using TBS-T solution, immunoreactive bands were visualized by chemiluminescence detection on X-ray film (Hyperfilm ECL, Amersham, Piscataway, NJ, USA) using enhanced chemiluminescence solution (Santa Cruz). For quantification a Kodak Image Station 440CF (Eastman Kodak Comp, Rochester, NY, USA) was used. Each sample was analysed in duplo. HKI and HKII band densities were expressed relative to the corresponding α-TUB band density. Validity of quantification was checked for each antibody by calibration curves (data not shown).
Statistics
Values are presented as mean ± SEM. Two-way analysis of variance (ANOVA) was used to analyse for main and interaction effects of standard chow and HKII reduction. Two-way ANOVA for repeated measurements was used for analysis of EDP measurements at different time points during ischaemia and reperfusion. Changes were considered statistically significant when P < 0.05.
Results
Effect of standard chow and HKII on body weight, heart weight and baseline cardiac physiological characteristics
Surprisingly, despite the marginal differences in the major caloric constituents between chows (Table 1), Purina chow-fed mice had significantly lower heart and body weights (12–15%) than SDS-fed animals (Table 2). In addition, Purina chow also significantly decreased heart weight relative to body weight, suggesting less cardiac hypertrophy. These standard chow effects were independent of HK genotype (P > 0.05 for interaction effect). Body and heart weights were not different between genotypes fed similar chows, indicating that HKII reduction does not affect growth parameters. Chow and HKII reduction were without effect on baseline cardiac functional parameters (Table 2).
Animal and haemodynamic characteristics of Langendorff experiments
BW: body weight; HW: heart wet weight; HW/BW: heart to body weight; LVEDP: left-ventricular end-diastolic pressure; DLVP: developed left-ventricular pressure; HR: heart rate; WT: wild type; HK: hexokinase
Data are presented as mean ± SEM, 10–13 animals per group
*P < 0.05 to SDS chow
Standard chow and HKII effects on total HK activity and isoforms in the heart
Standard chow significantly affected total cardiac HK activity, with Purina chow decreasing HK activity by −33% as compared with SDS chow (Figure 1a). Reductions in HKII also significantly decreased total cardiac HK activity (Figure 1a; −30%). No interaction between chow and HKII on HK activity was observed (P = 0.411). Standard chow also significantly decreased cardiac HKI expression, where Purina chow decreased HKI protein content by −46% (Figure 1b), whereas chow was without effect on cardiac HKII protein content (Figure 1c). The data therefore suggest that the decrease in cardiac HK activity with Purina chow can mainly be ascribed to diminished expression of the HKI isoform, and not the HKII isoform.
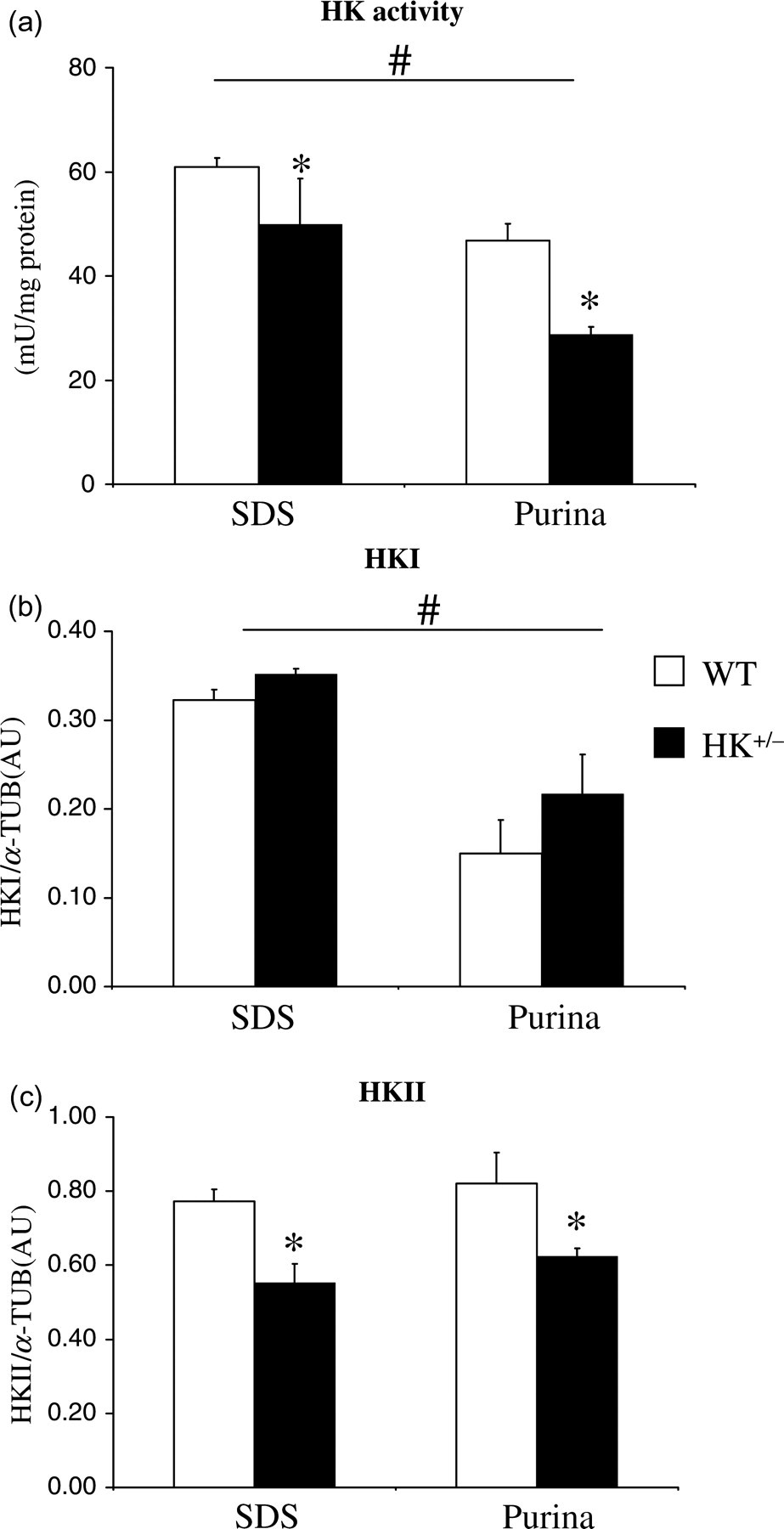
Standard chow and HKII effects on HK activity and isoform expression in the heart. Total HK activity is normalized to protein (a), and HKI (b) and HKII (c) protein content are measured by immunoblotting and normalized to α-tubulin (α-TUB) and shown as arbitrary units (AU). Values represent mean ± SEM, 5–7 animals per group. # P < 0.05 for standard chow effects; *P < 0.05 for HKII+/− effects within similar chow. WT: wild type; HK: hexokinase
For both chows, the deletion of the HKII allele did not result in an increased HKI expression (Figure 1b), disputing a possible compensatory HKI increase. However, there was a tendency for increased HKI protein content in HK+/− mice fed Purina chow. Finally, the results demonstrate that the decrease in HKII isoform in the HK+/− hearts was similar for both chows (−26%; Figure 1c).
Standard chow and HKII effects on HK activity and isoforms in skeletal muscle
To investigate whether chow effects on HK activity and isoforms in the heart extend to other insulin-sensitive tissue, the GM muscle was also examined. Standard chow also significantly affected HK activity in skeletal muscle (Figure 2a), although the effect was smaller as observed for the heart: Purina chow decreased HK activity by −25%. However, HKI protein content (Figure 2b) and HKII protein content (Figure 2c) were not affected by chow, suggesting that chow effects on total HK activity in skeletal muscle are mediated through a post-translational mechanism (and not through altered HKI expression as was observed for the heart). No interaction between chow and HKII on HK activity was observed (P = 0.262).
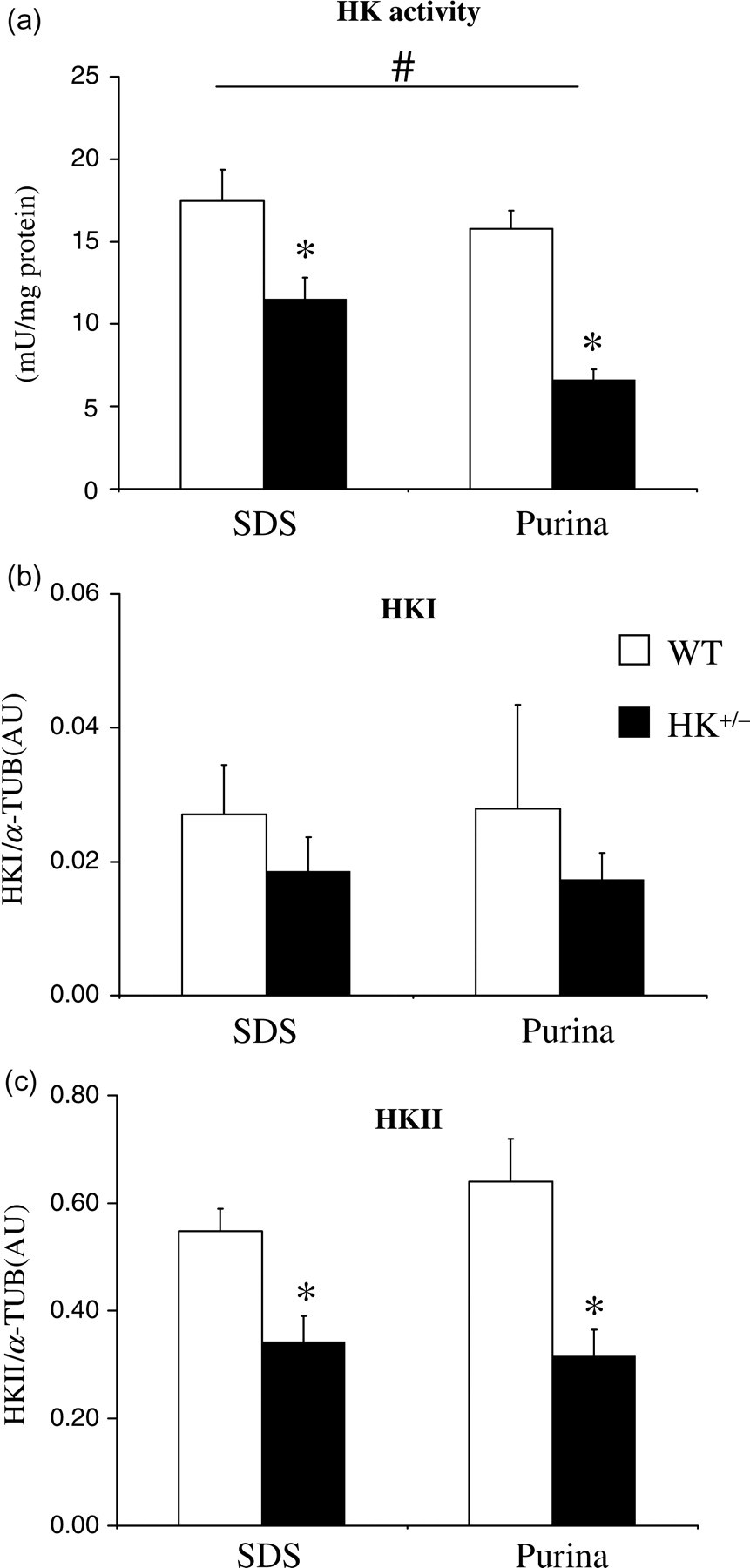
Standard chow and HKII effects on HK activity and isoform expression in skeletal muscle GM. Normalized HK activity (a), HKI protein content (b) and HKII protein content (c). Values represent mean ± SEM, 5–7 animals per group. # P < 0.05 for standard chow effects; *P < 0.05 for HKII+/− effects within similar chow. WT: wild type; HK: hexokinase
Also for the GM, reduced HKII resulted in decreased total HK activity for both chows in the GM (Figure 2a), with a decrease considerably larger as was observed for the heart (−58% versus −30%, for Purina, GM and Purina, heart, respectively). The larger decrease in total HK activity with deletion of one HKII allele in the GM is likely due to the fact that HKI is considerably less present in skeletal muscle than in the heart (Figure 2b versus Figure 1b). No compensatory increase in HKI was observed with partial deletion of the HKII gene in skeletal muscle (Figure 2b). The decrease in HKII protein content in the HK+/− skeletal muscle amounted to −46% (Figure 2c), coming close to the theoretically anticipated 50% decrease when knocking out one HKII allele. Overall, the data demonstrate that HK activity in the GM muscle is predominantly of the HKII origin and is affected by standard chow, probably through post-translational mechanisms. In contrast, HK activity in the heart is almost equally determined by HKI and HKII, with standard chow affecting total HK activity through alterations in the HKI isoform expression.
Effect of standard chow and HKII on cardiac IR damage
We first analysed standard chow and HKII effects on mechanical performance of the heart during and following low-flow IR (Figure 3). Both standard chow and reductions in HKII were without effect on the recovery of the rate–pressure product at the end of the reperfusion period (Figure 3a). The development of diastolic contracture is depicted in Figure 3b. Standard chow demonstrated a non-significant trend for increased diastolic contracture with SDS standard chow, both during ischaemia (P = 0.115) and during reperfusion (P = 0.062). Statistical analysis of area-under-the curve for the EDP parameter during reperfusion yielded a similar non-significant trend effect of chow (P = 0.076). The chow effect on diastolic pressure development following IR was independent of HKII genotype (P ≥ 0.90 for the interaction term). In addition, a non-significant trend was observed for increased left-ventricular end-diastolic pressure (LVEDP) in HK+/− hearts as compared with WT hearts (P = 0.288 during reperfusion). Although not significantly different, mean LVEDP for HK+/− hearts was 38% and 31% increased as compared with WT hearts, for SDS and Purina, respectively.
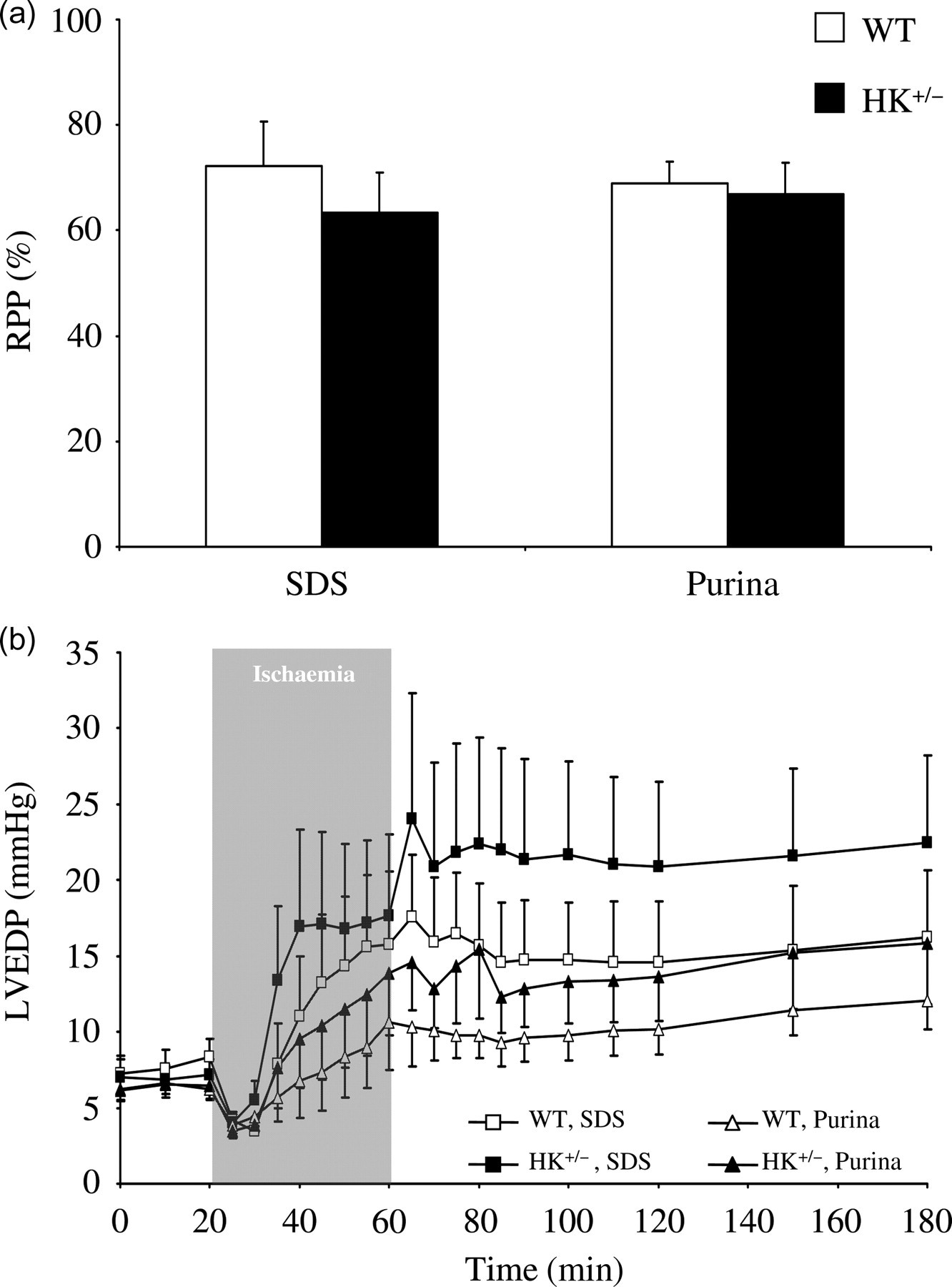
Standard chow and HKII effects on ischaemia–reperfusion (IR) functional parameters. (a) Recovery of rate–pressure product (RPP) at end IR protocol relative to baseline value; (b) Left-ventricular end-diastolic pressure (LVEDP) time course during protocol in heterozygous HKII knockout (HK+/−) and wild-type (WT) mice fed either SDS or Purina chow. Data are presented as mean ± SEM, 10–13 animals per group. HK: hexokinase
Concerning cellular damage caused by IR, we determined leakage of the enzyme LDH in effluent from the Langendorff-perfused heart. Interestingly, standard chow significantly affected basal cardiac LDH activity (P = 0.011), showing higher (+11%) LDH activity for mice fed Purina standard chow (Figure 4a). Reducing HKII was without effect on cardiac LDH activity. LDH release during reperfusion was therefore normalized to the amount of LDH present in heart at baseline for each specific standard chow (Figure 4b). First of all, the figure indicates that the 40 min 5% low-flow intervention resulted in a rather low incidence of cell death (approximately 1%). No overall effect of standard chow (P = 0.554) or HKII (P = 0.235) was observed on LDH release from the heart during reperfusion. Although there was a trend for HKII+/− hearts from both Purina-fed animals (+55%) and from SDS-fed animals (+28%) to have more LDH release as compared with WT hearts, this difference did not reach statistical significance (Figure 4b).
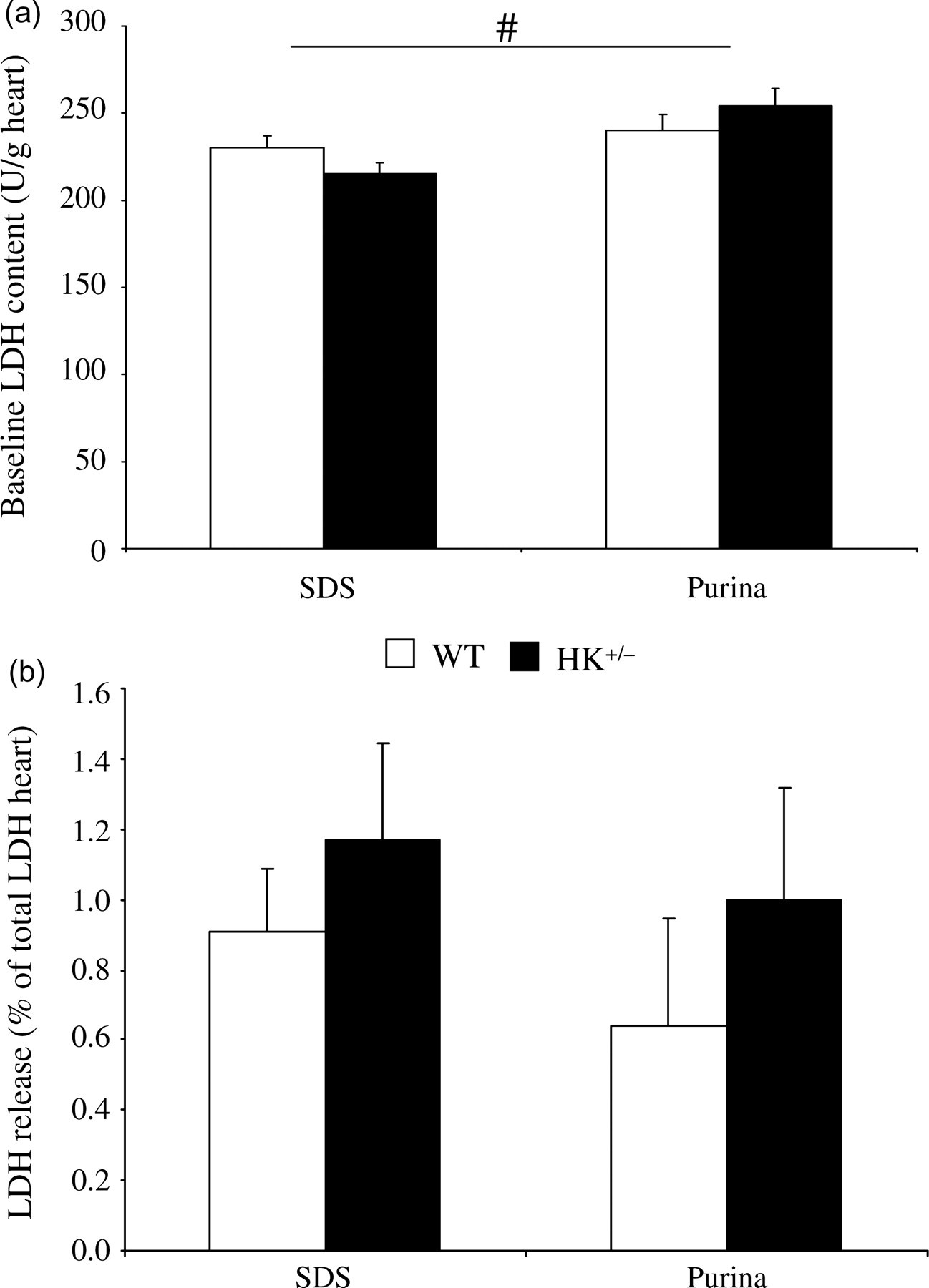
Standard chow and HKII effects on ischaemia–reperfusion (IR) cellular damage. (a) Standard chow and HKII effects on basal LDH activity (4 animals per group); (b) LDH activity cumulative for 120 min reperfusion period in heterozygous HKII knockout (HK+/−) and wild-type (WT) mouse hearts fed either SDS or Purina chow. 10–13 animals per group. Data are presented as mean ± SEM. # P < 0.05 for standard chow effects HK: hexokinase; LDH: lactate dehydrogenase
Discussion
In this study, our most important findings are: (1) standard chow affects body weight and heart weight, resulting in different degrees of cardiac hypertrophy, (2) standard chow significantly alters total cardiac and skeletal muscle HK activity, albeit through different mechanisms (altered HKI expression for heart and post-translational HK effects for skeletal muscle) and (3) in a low-flow cardiac IR model resulting in mild IR injury only, standard chow or a moderate decrease in HK activity does not significantly exacerbate IR damage.
It is traditionally thought that large changes in the major diet constituents (carbohydrates, fat and proteins) are needed to induce metabolic alterations in tissue and total body weight. The present study clearly demonstrates that such large differences in the major constituents are not necessary to observe these changes. Both chows used in the present study, being standard chows, are to be considered as normal to high-protein, high-carbohydrate, low-fat diets and use soy, maize, cereals and alfalfa as main ingredients. The decrease in body weight observed in the present study, when switching to the Purina chow with 13% less carbohydrates and 30% more fat and 21% more proteins as compared with the SDS chow, supports the suggestion that diets only moderately higher in fat and protein and lower in carbohydrates result in body weight reduction. 18–22 The directional changes in the major nutrients may also partly reconcile the changes observed in HK expression. The isoforms HKI and HKII are differently regulated. HKII is regulated by insulin 23,24 and is therefore most apparent in insulin-regulated/sensitive tissues, like heart and skeletal muscles. 25 HKI is not insulin regulated, is expressed ubiquitously throughout the body and is considered more as a housekeeping protein. Surprisingly, in the present study, standard chow affected cardiac HK activity through alterations in HKI and not HKII expression, whereas the majority of studies have demonstrated HKII to be most responsive to hormonal and metabolic perturbations. Although we were unable to find specific literature regarding dietary effects and cardiac HKI expression, there is evidence showing that increasing dietary fat and protein content at the cost of the carbohydrates content results in diminished HK activity, 26–28 as was found in our Purina-fed WT mice. Why this dietary effect on HKI was mitigated in the HK+/− mice is unknown, but is similar to the observations of Fueger et al. 1 showing that a high-fat diet only reduced HKII in WT but not in HK+/− mice. Interestingly, although Fueger et al. 1 ascribed many of their observed effects to the higher fat content, these effects may also possibly be ascribed to changes in their background standard chow, because their high-fat diet was Bio-Serve-based whereas the control diet was Purina-based. In addition, it is possible that the genomic reduction of the HKII gene from two to one allele impairs this dietary effect, because it was reported that HKI expression is regulated by the HKII gene. 29 Further studies will be necessary to examine cardiac HKI expression regulation in more detail. Also not much is known about probable post-translational modifications. Concerning nutritional regulation, it is known that HKII expression is inhibited by nutritional methyl donors like the vitamins folic acid, vitamin B12, choline and betain vegetable hormones. 30 Other nutritional regulators like phytoestrogens, originating from vegetables like soy, maize and alfalfa, 31 may also be of influence, because these are the main ingredients of both chows used in the present study. However, we found no interference of chow on HKII amount. Whether HKI might be affected by these nutritional components is not known and should therefore not be excluded as a possible regulator of expression. It should also be realized that both standard chows are so-called closed-formula chows, meaning that although dietary ingredients are known their concentration is not, because this varies from batch to batch, with availability of ingredients and season. 32 It has been reported that the Purina 5001 chow is rather high in phytoestrogens, 32 at least suggesting that there may be a role for phytoestrogen in the chow effects described in the present study.
In order to start to understand which component(s) in standard chow causes the effects described in the present study, subsequent studies are needed using a single standard chow to which known amounts of each suspected component are added and examined for effects on growth, biochemistry and pathophysiology of cardiac IR. Taking the observed standard chow effects on growth and cardiac HKI and HK activity into account, our data suggest that standard chow is an important, often neglected, 1–4 factor in genomic, physiological and metabolic research models.
Previous research demonstrated a protective role of HK against oxidative injury in cellular systems and a correlation between mitochondrial HK association and cardioprotection in the intact heart. 14 Mechanisms of protection may relate to increases in energetic flux through increased glucose phosphorylation (glycolysis) capacity with increases in HK and/or to mitochondrial protection through increased HK association with mitochondria and stabilization of the permeability transition pore. To examine both possible beneficial mechanisms, we exploited a low-flow ischaemia model. In contrast to no-flow ischaemia, where glycolysis is halted due to accumulation of metabolic end-products, low-flow ischaemia allows for continuous glycolysis-from-exogenous glucose, and thus glucose phosphorylation, during ischaemia. 33,34 It therefore allows evaluation of the effect of possible diminished glucose phosphorylation in the HK+/− hearts on the developing IR injury. In addition, low-flow ischaemia models more closely reflects the clinical condition of myocardial infarction, where residual flow is often present, 35 than a no-flow ischaemia model.
We observed no effect of reduced HKII on contractility during baseline or reperfusion. These results are commensurate with studies using over-expression of yeast cardiac HK in cardiac low-flow IR, 34 and suggest that the glucose phosphorylating capacity of the heart is not a critical determinant of active force production, and recovery thereof following IR. We did observe a non-significant trend towards increased irreversible damage with reductions in HKII as LDH release during reperfusion was 30–55% higher in the HK+/− heart. It is possible that the use of low-flow ischaemia and the consequent low incidence of cell death (∼1% in the present study), hampering statistically sensitive testing of HK effects on cell death. Such testing may await further experiments using this HK+/− heart genotype in settings of a more severe ischaemic injury model (i.e. no-flow ischaemia). From the present study it can be concluded that a moderate reduction in cardiac HK activity is without effect on cardiac IR injury originating from low-flow ischaemia.
Taken together, we have shown that standard chow modifies our genetic mouse model concerning cardiac HK, emphasizing standard chow as an important variable in cardiac genotype–phenotype characterization. Furthermore, this study demonstrates that in a mild, low-flow ischaemia model in isolated hearts, functional performance and IR functional recovery and necrotic damage were not affected by cellular HKII reduction.
