Abstract
Thyroid status was assessed in adult female rhesus monkey breeders at the California National Primate Research Center at the beginning of the breeding season. The 95% confidence intervals for thyrotropin (TSH), thyroxine (T4) and triiodothyronine (T3) (n = 66–80) were similar to those previously reported in smaller samples of macaque monkeys. Based on human criteria, 10 of 80 monkeys (12%) were hypothyroid (TSH > 2.0 µIU/mL). Because hypothyroxinaemia can be a risk factor in pregnancy, T4 status was compared with past breeding history, breeding outcome for that season and general health records in a subset of 42 breeders. Age, weight and parity did not differ between monkeys in the lowest T4 quartile as compared with those in the upper three quartiles. However, T4 concentrations were significantly associated with the number of missed menstrual cycles during the previous breeding season. In additional work, three healthy lactating rhesus monkeys were given three different doses of environmental contaminant and thyroid iodine uptake inhibitor, ammonium perchlorate (0.006, 0.34, 12.8 mg/kg/day, respectively) in food for two weeks. Thyroid status variables (TSH, T4, T3, thyroid radioactive iodine uptake) were then measured. In the monkey receiving the highest perchlorate dose, iodine uptake was suppressed relative to baseline. The study shows the availability of tools to study thyroid status in rhesus monkeys, the variability of thyroid status in the breeder colony and the potential ability of environmental factors to influence thyroid status.
Thyroid hormones are key regulators of metabolism and development. 1 Thyroid disorders are known to disrupt fertility and reproduction in women 2 and environmental contaminants that are seen as ‘endocrine disruptors’ are thought to affect reproduction through their action on thyroid hormone systems. 2,3 However, little is known about links between thyroid, reproductive function and endocrine disrupting chemicals in non-human primates. Development of thyroid status immunoassays using an automated system has made it possible to survey thyroid status in a group of female rhesus monkey breeders. In addition to deriving population values for thyrotropin (TSH), thyroxine (T4) and triiodothyronine (T3), the relationship between thyroid indicators and reproductive history and performance in colony records was examined. Finally, the response of thyroid to the environmental contaminant perchlorate was studied in a small group of lactating females. Perchlorate was at one time used as a pharmaceutical to treat hyperthyroidism, 4 and is now a widespread environmental contaminant due to its use in rocket fuel and fertilizer. 5 The presence of perchlorate in ground water, soil and vegetation 6–9 may be a significant public health risk for sensitive subpopulations. It is uncertain what levels of perchlorate intake will affect health, particularly in ‘at risk’ populations (e.g. fetuses, neonates, children under the age of 12, females of reproductive age, and those with low iodine intake and perhaps genetically susceptible populations). Perchlorate acts via competitive inhibition of the sodium-iodide symporter, and also impairs thyroid hormone synthesis. 10 These effects have not been studied in non-human primates. Our study is the first preliminary study to help define the chronic toxic effects of perchlorate ingestion in a controlled feeding study using monkeys, which are considered the best animal model for human thyroid function assessment. 11–14
Materials and methods
Animals and animal care
The rhesus monkeys (Macaca mulatta) used in this study were born and raised at the California National Primate Research Center (CNPRC). They were part of the indoor breeding colony of rhesus females. They were pair-housed in rooms containing 30–40 animals under a 12 h light:12 h dark cycle with a temperature range of 21–25°C. Breeders sampled for this study came from one of three indoor areas where they had been housed in cages for 546 ± 47 (mean ± SEM) days, except for one area where the duration of housing was 24 ± 5 days. Monkeys for the perchlorate exposure portion of the study were housed for a full 15 days individually with their nursing offspring in metabolic cages. Monkeys were fed commercial diet (LabDiet #5038; Purina-Mills, Inc, St Louis, MO, USA) available ad libitum and had continuous free access to water. All monkeys were observed each morning for abnormal health signs. All procedures were performed according to a protocol approved by the Institutional Animal Care and Use Committee (IACUC) in compliance with National Institutes of Health guidelines.
Study design
Three related studies were conducted; screening of a group of indoor breeders for thyroid status; comparison of thyroid status to colony records on health and reproduction in a subsample of breeders, and administration of perchlorate and measurement of thyroid status in three lactating breeders.
Thyroid status of breeders
Blood samples were obtained in the fall of two successive years (2006–2007) prior to the onset of the indoor breeding programme. Available females were those not restricted by project status.
Comparison of thyroxine status with reproductive parameters
A subpopulation of breeders from screening was selected based on sampling conducted in the same year, availability of complete colony record information and housing across several different indoor colony rooms. They were divided into hypothyroxinaemic (lowest quartile based on the screening population, ≤3.5 µg/dL) and normal (>3.5 µg/dL).
Perchlorate effect on thyroid status
Three monkeys were selected for perchlorate administration based on normal thyroid hormone status prior to pregnancy during the screening of the colony, availability, similarity in age, weight, and parity, and location in the same cage room. They were nursing infants 116 ± 2 (mean ± SD) days of age at the initiation of dosing. Perchlorate was administered during lactation so that presence in milk and transmission to nursing infant could be evaluated. Ammonium perchlorate (99.8%, Sigma Aldrich Inc, Saint Louis, MO, USA), which contains about 85% perchlorate, was given orally each day in a favorite monkey food such as half an apricot or prune. We prepared three different concentrations of ammonium perchlorate solution. Each day one-third of the daily dose, which was 25 µL, was drawn into a pipette and put into a favorite monkey food. Treated fruits were kept in a refrigerator until use. The perchlorate-treated monkey food was given to the subject three times a day (06:30, 11:30, 16:30 h). Doses were based on the recorded body weight at the beginning of the study. The consumption of treated food was recorded. Perchlorate was administered over a 14-day period (days 1–14). On days 0, 8 and 15, thyroid hormones (serum T4, T3 and TSH) were assessed. On days 0 and 15, radioactive iodide uptake (RAIU) was assessed. Additionally, serum was sampled one week after completion of exposure in both dam and infant for perchlorate assay. Milk samples were obtained at the time of RAIU for perchlorate analysis.
Sample collection
Two millilitres of blood samples for analysis of thyroid hormone parameters (T3, T4 and TSH) were obtained from the cephalic vein of unanaesthetized, unrestrained colony breeders in their home cages. To obtain the sample, the cage door was opened just enough to allow the monkey to present its arm. If this was not done spontaneously the sliding rear cage wall was used to reposition the monkey to the front part of the cage. If necessary, an aluminium rod was used to position the arm. Blood samples were allowed to clot and were centrifuged at 1600
Maintenance diet, food items used for perchlorate administration and drinking water were analysed for perchlorate and iodide content. Samples were collected daily from days 8 to 15 of perchlorate administration from the home cages of perchlorate-treated monkeys and pooled for analysis.
Data from colony records
Historical data on each female breeder were obtained from the computerized database at CNPRC (‘Vitals’). This included general background and health-related variables (age, weight, number of ‘diarrhoea’ and ‘thin weak’ reports from the daily health check), breeding history variables (total conceptions, number of live births, number of fetal deaths/stillbirths/neonatal deaths, menstrual cycles previous year) and breeding season outcome variables (pregnant/not pregnant, number of matings, gestation length).
Perchlorate exposure
Ammonium perchlorate (99.8% purity, 85% perchlorate, Sigma Aldrich Inc) was diluted in distilled water and injected by pipette at a 25 µL volume into a preferred food item (apricot, prune) that was hand fed to the monkey. One-third of the daily dose (0.006, 0.34 or 12.75 mg/kg/day) was given to the subject three times a day (06:30, 11:30 16:30 h). The doses were based on the No Observed Effect Level (NOEL) of 0.007 mg/kg/day used as the point of departure for development of the USEPA health-based drinking water standard for perchlorate. 5
RAIU measurement
Monkeys were lightly sedated with ketamine hydrochloride (5 mg/kg, intramuscularly) and medetomidine (30 µg/kg, intramuscularly). Atipamezole (0.15 mg/kg, intramuscularly) was given as a reversal agent at the conclusion of the procedure after an approximate 6 h fasting for each animal (an overnight fast) according to the schedule. The solutions containing 100 µCi Na 123I for dams were administered by gastric tube. Accumulation of radiolabelled iodide was determined as previously described 15 at 6 and 24 h following radioiodine ingestion.
Thyroid hormone analysis
The following tests were performed: serum T4, T3 and TSH. These serum hormones were measured using competitive chemiluminescent assays developed for this study. The TSH assay was a two-site sandwich chemiluminescent immunoassay using direct chemiluminometric technology. All assays utilized the Siemens/Bayer ACS-180 platform (Bayer Healthcare, Tarrytown, NY, USA) and reagent kits for measurement of human TSH (Product #672221), T3 (Product #672206) and T4 (Product #672201). All samples were analysed in duplicate. Each batch included multiple samples from the same animals and from each group. Inter- and intra-assay coefficients of variance were as follows: T4 – 3.2% and 4.4%; T3 – 2.5% and 5.8%; and TSH – 3.6% and 4.3%, respectively. All analyses were conducted by the Endocrine Core Laboratory at the CNPRC.
Perchlorate and iodide analysis
Perchlorate and iodide were analysed by triple-stage quadrupole liquid chromatography-mass spectrometry (LC-MS/MS) using an Applied Biosystems 4000 Qtrap LC-MS/MS system 16 at UC Davis and confirmation analyses were performed at CDC by triple-stage quadrupole ion chromatography-mass spectrometry (IC-MS/MS) using an Applied Biosystems 4000 IC-MS/MS system. 17,18 Samples such as urine and food were diluted in water and filtered prior to analysis. Serum and milk samples were prepared by mixing a 4:1 v:v of acetonitrile to milk or serum and centrifuging to denature and remove proteins. The resulting supernatant was dried and reconstituted in water prior to analysis. An isotopically-labelled internal standard (18O-HClO4) was added to each sample extract prior to analysis to provide accurate quantification of native perchlorate. The LC-MS/MS was equipped with a Phenomenex RP-Polar HPLC column, which provides adequate separation of perchlorate from matrix components. Two separate MS/MS transitions for perchlorate using precursor ions of 35ClO4 − and 37ClO4 − were monitored to provide qualitative identification. Quantitation was performed using the ratios of the chromatographic peak area from the native perchlorate versus the internal standard as compared with those determined by analysis of 5-point calibration curves analysed before and after each set of samples. All anions were found at levels above the method detection limits, and no contamination problems were observed. Inter- and intra-assay precision was excellent during the analysis of the study samples, with coefficients of variation of less than 5% for quality control materials.
Background ingestion was low and appropriate for an experimental perchlorate dosing study.
Data analysis
Comparisons of thyroid status variables and reproductive variables were conducted by analysis of variance (JMP, SAS Institute, Cary, NC, USA). Regression analyses were also conducted using the absolute values of the TSH and T4 assays. Inadequate power was identified for some variables for which the incidence in the sampled population was very low (e.g. neonatal deaths, 7%).
Results
Thyroid status of breeders
Thyroid status evaluations of the rhesus monkey breeders are shown in Table 1. The mean values of TSH and T4 in the sample as a whole are given. Additionally, subpopulations identified as hypothyroid and hypothyroxinaemic are identified as a percent of the population. Identification of hypothyroidism in humans relies on elevation of TSH in response to inadequate thyroid hormone production. Recommendations for TSH threshold for hypothyroidism in humans have declined in recent years from 5 to 3.0 to 2.5 or 2.0 mIU/mL based on more adequate characterization of normative populations. 19–21 Using 2.0 mIU/mL as the most conservative value for elevated TSH, 12% of the monkey population could be identified as hypothyroid.
Thyroid status of female rhesus monkey breeders prior to the breeding season
T3: triiodothyronine; T4: thyroxine; TSH: thyrotropin
Hypothyroxinaemia is a distinct diagnosis based on low T4 levels. While T4 levels may not be low enough to trigger TSH production, they could prove inadequate to supply the fetus during the early in utero period before fetal thyroid hormone synthesis begins. 22,23 Based on available data, macaques have generally lower serum T4 than humans. Recently published data from NHANES give median values in 6182 adult women (disease-free, not pregnant) of 8.59 µg/dL (95% reference limits = 4.96–12.81). 24 The median T4 for our population was 4.19 µg/dL with a range of 2.44–8.37 µg/dL. Studies in rhesus macaques using smaller samples have also identified mean values between 4 and 6 µg/dL. 25,26 Thus human population values are not appropriate for identifying hypothyroxinaemia in rhesus macaque monkeys and hypothyroxinaemia was arbitrarily defined at the cut-off point for the lowest quartile of values in the population of monkeys sampled in this study (<3.5 µg/dL).
Comparison of T4 with reproductive parameters
Thyroid status endpoints (TSH, T4) were compared with background variables and reproductive performance. No relationship was found between hypothyroid rating and age, weight, parity or health (thin, weak or diarrhoea reports) of the breeders. However, all of the monkeys in the hypothyroid group (n = 5) came from the same wing of the animal facility. Animals were housed in one of three wings. The wing containing the hypothyroid group had been recently remodelled and the animals had been caged there less than a month in comparison of caging durations of about 18 months in the other two areas. This difference in incidence between wings was significant by χ 2 (P = 0.01) and Fisher's exact test (P = 0.01). However, a potential causal factor could not be identified. This group included three monkeys who were both hypothyroxinaemic and hypothyroid. Given the small number of hypothyroid monkeys it was not possible to statistically compare their reproductive performance to euthyroid breeders. However, no pattern of deficit was seen from examination of the data.
Given that a clinical criterion for hypothyroxinaemia could not be extrapolated from human literature, we designated monkeys in the lowest quartile of T4 distribution as ‘hypothyroxinaemic’. The relationship of thyroid status to demographic variables was analysed using information from colony records for a subset of the monkeys (n = 46, see Table 2).
Comparison of rhesus breeder females categorized as hypothyroxinaemic prior to initiation of the breeding season with breeders categorized with normal thyroid status and matched for year of sampling and cage location
These breeders were selected for comparison from the larger sample described in Table 1
T3: triiodothyronine; T4: thyroxine; TSH: thyrotropin
†Mean ± SEM
*P = 0.01
**P < 0.0001
No significant effects of low T4 were identified for the breeding history variables or the breeding outcome variables, some of which are shown in Table 2. Correlation between T4 and background and reproductive indices (Table 3) revealed a significant correlation with missed menstrual cycles during the breeding season.
Correlations between T4, menstrual cycles in the breeding season and dam background characteristics
T4: thyroxine
*P < 0.05
Perchlorate effect on thyroid status
The perchlorate and iodide content of water, regular food (monkey chow) and food items offered to animals as enrichment (prunes and dried apricots) are presented in Table 4. Analysis of dam blood, milk and urine and infant blood confirmed that perchlorate administration resulted in detectable concentrations of the chemical in serum of lactating dams, breast milk and serum of nursing infants (Table 5). RAIU measurements performed at 6 and 24 h after the last dose were consistent with the expected inhibition of iodide uptake by the thyroid in the middle and high dose animals compared with their baseline values (Figure 1). However, statistical confirmation of this anticipated outcome cannot be achieved without a full experiment. TSH was not apparently elevated (Figure 1). Serum T3 and T4 were apparently unchanged in the perchlorate-treated monkeys (data not shown).
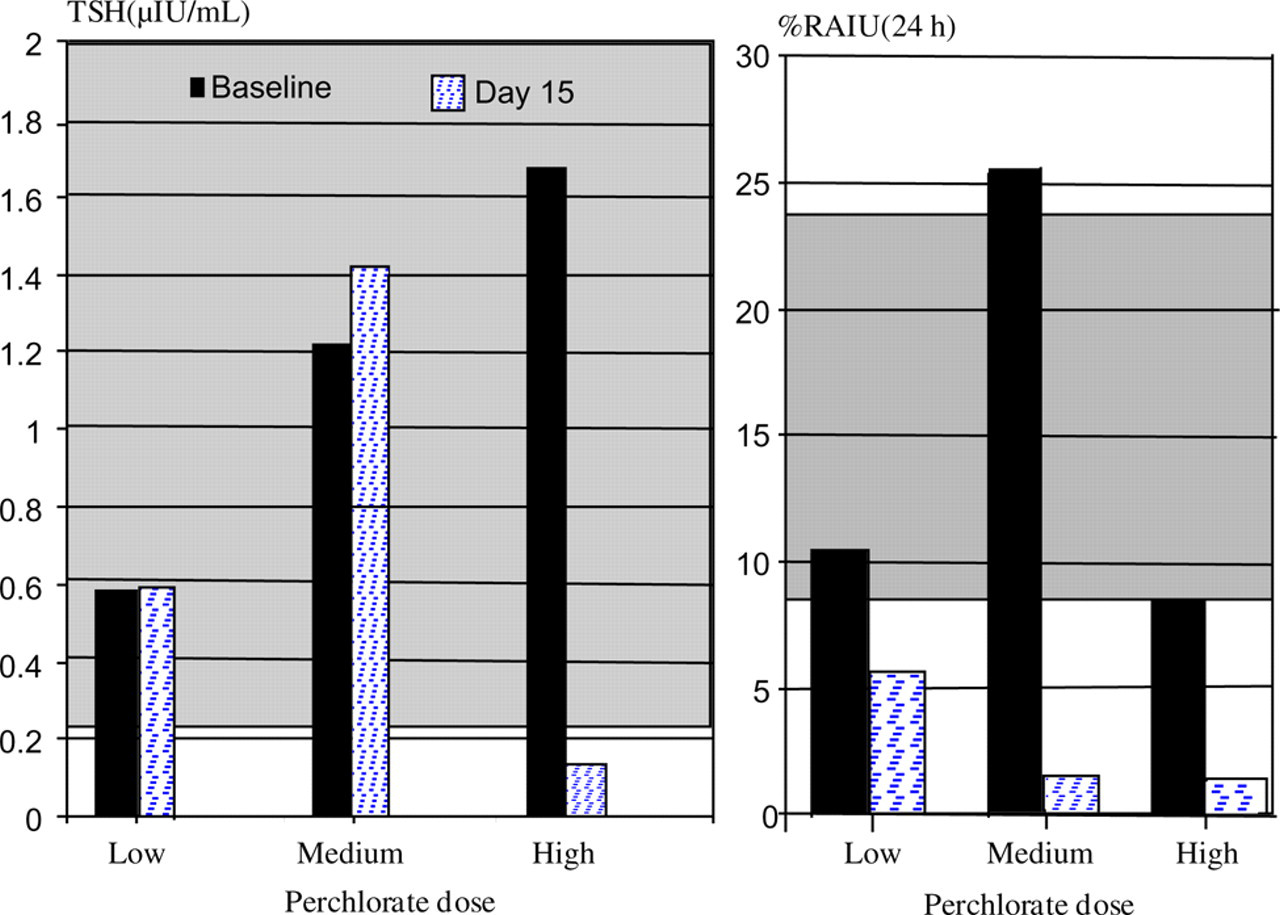
Radioactive iodine uptake, and of individual female rhesus monkeys before and after exposure to three different daily doses of perchlorate for 14 days (shaded area is the 95% confidence interval for colony breeders as described above). TSH: thyrotropin; RAIU: radioactive iodide uptake
Analytes in food and water samples (reported per kg dry weight)
ND = not detected
Perchlorate concentrations in maternal and infant samples
The maternal samples were obtained the day after completion of the 14-day dosing period. The infant sample was obtained one week after completion of the 14-day dosing period. NM = not measured
*Serum and urine perchlorate baseline levels were under the detection limits; milk perchlorate baseline levels were detected between 1.1 and 2.6 ppb
Discussion
Physiological regulation of thyroid function is a widely conserved feature of mammalian species. Some unique aspects have been reported in great apes 27,28 but existing data indicate a general similarity among non-human primates and humans. Existing data concerning macaque monkeys include studies on thyroid function in pregnancy and infancy of rhesus macaques 25,29,30 as well as in adult cynomolgus macque males. 31–33 Both rhesus and cynomolgus macaques have served as human biomedical models for testing therapeutics that affect the thyroid 34–37 and rhesus for effects of diet on thyroid function. 26,38 However, population-based studies in non-human primates are rare.
For rhesus monkey breeders studied here, the serum concentrations of TSH were similar to those reported in humans but the T4 levels were lower. Monkeys in the lowest quartile of T4 distribution had no apparent deficit in their reproductive ability. Neonates are not routinely examined at birth and birth weights are not routinely recorded in the breeding colony, so no information was available on neonatal status. Potential effects on growth and brain function of the infant postnatally would be valuable to ascertain as these are linked to gestational hypothyroxinaemia in women. 39 However, controlled experiments would be necessary.
In laboratory non-human primate facilities, adequate nutrition as determined by the National Research Council is provided and incidence of infectious disease is monitored and controlled. Sources of variability in thyroid status are likely to be due to genetic or environmental factors as in healthy human populations. Genetic origins have not been examined in this study. An indication of environmental influences was the finding that the small number of breeders found to be hypothyroid was housed in the same area of the colony.
Environmental contaminants can also affect thyroid function. In the pilot work reported here, thyroidal iodine uptake was apparently suppressed by oral exposure to perchlorate, as has been seen in human studies. The administered perchlorate was detected in urine, serum and breast milk of lactating rhesus. With the two-week exposure there was no apparent influence on thyroid status measures except for the apparent drop in TSH with high dose. Decreased TSH concentration is the opposite of what is expected when iodide uptake is blocked or in nutritional iodine deficiency. Human studies that evaluated thyroid status after exposure to known amounts of potassium perchlorate in drinking water ranging from 0.007 to ∼13 mg/kg/day 19,40–45 also found that TSH levels did not increase, and thyroid hormones did not decrease, although iodine uptake was suppressed. However, serum TSH concentrations decreased in some groups in the Brabant's study. 40 Uncontrolled confounding variables such as age, gender, variable diets and dosing regimen decreased the sensitivity of these studies. Identifying thyroid disease clinically can be challenging. The effect of perchlorate exposure on thyroid hormone homeostasis during short periods of time might be related to abnormal hypothalamus function leading to decreases in serum TSH concentration. 46
In summary, rhesus monkey colonies show heterogeneity in thyroid status similar to healthy human populations and provide a possible model for studying the genetic and environmental origins of this variability.
Footnotes
ACKNOWLEDGEMENTS
We would like to thank Sarah Davis and Toni Traill for their help handling the primates, Lisa Laughlin for performing the thyroid status analyses, Casey Hogrefe for assembling colony record data and Mike Filigenzi for perchlorate analysis. This study was funded by Western Institute for Food Safety and Security (WIFSS) and RR00169 (California National Primate Research Center).
