Abstract
Hypothermia can be caused by anaesthesia and/or surgery and represents a daily challenge in the operating room. Experimental animal surgery settings typically use heating pads or warming blankets to maintain the rodent's body temperature during long-lasting experiments. Warming is crucial in small animal experiments because these animals quickly lose temperature due to their large body surface to body weight ratio. While establishing a left ventricular infarction model in rats, we inserted a rectal temperature probe. The heating pad's set point was 37°C. Although a dual set point control circuit should prevent overheating, we observed a maximum heating pad's surface temperature of 43°C between the animal's back and the surface of the heating pad. At the end of the experiments, which lasted up to 8 h, the animals showed severe haematuria and segmental kidney damage. We hypothesized that overheating of the heating pad and uneven distribution of temperature led to kidney damage. Therefore, the maximal temperature of commonly used heating pads must be tightly controlled to avoid overheating, which may cause kidney or tissue injury, may falsify the experimental data and could influence the study results.
Establishing a myocardial ischaemia reperfusion model in rats we observed bloody urine in our animals after 8 h of experiments. We retrospectively examined the impact of a thermal injury on the kidney, liver, muscle and myocardium of the animals.
The ability to regulate and maintain core body temperature is impaired by anaesthesia due to cessation of muscle movement, convection, conduction and evaporation. An increase in body temperature is known to increase heart rate (HR) and oxygen utilization. This becomes even more relevant when chronic experiments are planned and the animals wake up after the initial experimental procedure. Rodents are especially susceptible to heat loss due to their high surface to body weight ratio. Correct adjustment and measurement of body temperature is crucial for experimental settings with a focus on organ protection. Chien could positively correlate the myocardial infarct size to the animal's body temperature. In the range between 35 and 42°C temperature, differences of 1°C already change the resulting infarct size by about 10%. 1 The comparison of four technically different heating devices in a model of hepatic ischaemia–reperfusion injuries in mice has shown the different influence of these devices on the hepatic enzyme levels and thereby confounding the results. This becomes even more important when the animals are positioned on their belly and the heating pad has direct contact with the liver. 2 In rabbits it has been shown that the elevation of the body's core temperature up to 43°C lead to a proportional increase of the creatinine level indicating a progressive deterioration of the kidney function by the temperature rise. 3 Mild hyperthermia (38–39°C) will increase cerebral infarct size after mild (30 min) and moderate ischaemia (60 min) by 23% and 78%. 4 Therefore, the settings of the control circuits of used thermal heating devices and especially the temperature between the rodent's body and the heating pad has to be controlled as well as the correct measurement position has to be considered to avoid overheating.
Materials and methods
Animals
All of the experiments were performed in accordance with the German legislation governing animal studies and followed the
Male Sprague–Dawley rats weighing between 350 and 400 g were purchased from Charles River (Charles River, Sulzfeld, Germany). The animals were allowed to acclimatize to the local housing facilities for at least one week prior to the experiments. The animals were maintained as specific pathogen free according to the FELASA Guidelines.
5
The animals had free access to a standard diet (Ssniff® r/M-H Extrudat; Ssniff GmbH, Soest, Germany) and were provided with sterile, acidified water
We retrospectively compared three groups of animals in this study: the infarct group received an operatively applied left ventricular infarction (
Surgical technique
On the day of the operation, the animals received an intraperitoneal injection of 90 mg/kg thiopental (Altana Pharma, Konstanz, Germany). This final anaesthesia regimen with thiopental provided a good controllable depth of anaesthesia without pharmacological preconditioning of the myocardium. After the onset of anaesthesia, the animals were shaved and the skin was disinfected (Poly-Alcohol-Skindesinfectant; Antiseptica, Pullheim, Germany). The thiopental injection provided a deep anaesthesia level for oral intubation of the animals with a 14G cannula (Abbocath, Nr. G 713; Abbott, Wiesbaden, Germany) by sight. The animals were thereafter mechanically ventilated in a pressure-controlled mode with a closed-circuit anaesthesia machine (Physioflex®; Draeger, Luebeck, Germany) with a respiratory frequency of 45–50/min. A pressure gradient of 13 mmHg was corrected to an end-tidal CO2 concentration that was between 32 and 36 mmHg. The right jugular vein was catheterized with a 22G cannula (Leader Flex; Vygon, Germany, Ref No. 1212.04) using Seldinger's technique for continuous drug administration and blood withdrawal. Continuous blood pressure measurements were taken by placing a 2F conductance catheter (SPR-869; Millar Instruments, Houston, TX, USA) through the right carotid artery into the left ventricle. The data were collected and sampled using an acquisition and analysis system (Power Lab 8/30, LabChart 6 Pro v 6.11; ADInstruments, Colorado Springs, CO, USA). A continuous infusion of Ringers solution (10 mL/kg/h) was administered to compensate for perioperative fluid loss, and an ECG lead II was recorded throughout. No muscle relaxation was given for a better judgement of the depth of anaesthesia. A tail pinch test was used every 30 min to verify an adequate depth of anaesthesia. If the animal showed a limb flinch, trembling of the body or a rise in HR, then an additional intraperitoneal injection of thiopental (50 mg/kg) was given. After repetition of the thiopental injection any surgical stimulation was delayed for another 5 min.
The heart was exposed by a lateral thoracotomy in the fifth intercostal space, and the pericardium was widely opened. A 7–0 ligature (Prolene Visi-Black DA-3, Cat. No. 1854; Ethicon, Germany) was placed around the left coronary artery but was not tightened. After complete instrumentation, the animal was given 90 min to recover from the operative stress. The ligature around the left coronary artery was tightened for 60 min to induce myocardial ischaemia, which was verified by ST segment elevation in the echocardiogram. After 60 min, the coronary ligature was loosened, and the reperfusion was verified by re-colouring of the myocardium and by a flattening of the ST segment elevation. The sham-operated rats were subjected to the same surgical procedures, except that the 7–0 Prolene suture was not tightened; therefore, no myocardial ischaemia occurred in this group. The control group did not undergo any surgical procedures but did receive 90 mg/kg of thiopental intraperitoneally for anaesthesia. The hearts were withdrawn for an isolated organ perfusion study, whereas the blood and kidneys were immediately harvested.
Temperature maintenance
The rectal temperatures were monitored with a rectal temperature probe (RET-2, MLT1403; Physitemp, Clifton, NJ, USA) and were maintained at 37 ± 0.5°C with a feedback-controlled heating plate (TCAT-2 Controller, ML 295/R; Physitemp) throughout the entire experiment. The accuracy of temperature measurement by the rectal probe RET-2 is given with ±0.1°C and a thermal response time of 0.8 s. For the heating plate a temperature resolution of ±0.1°C and a measurement accuracy of ±0.5°C is stated. The rectal temperature was displayed on the front side of the control unit and recorded on the data sheet every 15 min. The heating plate controller uses a dual set point control circuit to prevent the heating element from overshooting the control point. The heating plate is made of black anodized aluminium and measures 216 × 111 mm. We determined the set point at the front panel of the control unit to 37°C. The actual temperature of the heating plate was not displayed. For measuring the oral temperature we used a T-type-Pod (ML 312; ADInstrument) with a T-type implantable thermocouple probe (IT-18, MLT1401; ADInstruments). The accuracy of temperature measurement is given with ±0.1°C and a thermal response time of 0.1 s. Intermittently the rectal probe was placed between the heating pad and the animal's back. The temperature of the heating pad itself was measured at the end of the experiments by placing the temperature probe (RET-2) directly onto the heating pad, isolated to convective temperature loss, by a gauze compress.
Blood and tissue sampling
Blood samples were collected after 120 min of reperfusion. The animals were killed by intravenous administration of 1 mL potassium chloride to induce diastolic cardiac arrest. Samples from the heart, kidneys and liver tissue were immediately snap-frozen in liquid nitrogen (−180°C) and stored at −80°C. Myoglobin, Troponin I, neutrophil gelatinase-associated lipocalin (NGAL) and liver fatty-acid binding protein (L-FABP) protein concentrations were quantified by enzyme-linked immunosorbance assays in the serum (ELISA Reader Biotek, ELx800) (Myoglobin: 2110–2-N; Rat Cardiac Troponin I: 2010–2-HS; H-FABP: 2310–2-HS; LifeDiagnostics, West Chester, PA, USA; NGAL: Kit 041, BioPorto Diagnostics, Gentofte, Denmark; TNFa: RTA00, R&D Systems, Minneapolis, MN, USA).
Pathological examination
The kidneys were inspected macroscopically, sagittally cut and fixed in 10% buffered formalin. The microscopic slices were stained with haematoxylin and eosin and then examined with a digital microscope (Leica 6000D; Solms, Germany).
Statistical analysis
The primary endpoints of the study were serum and tissue markers of kidney injury (NGAL), and the secondary endpoints were serum markers of myocardial and liver injury (TnT I, L-FABP) and inflammation (IL-6). Differences between the markers between the various animal groups were analysed with a one-way ANOVA with Scheffe's
Results
Repeated temperature measurements in the back of the animal with an additional temperature probe and the heating pad showed that the temperatures increased by up to 43°C, even though the measured rectal temperature never exceeded 38°C and the oral temperature did not exceed 37.5°C.
Postmortem macroscopic examination of the kidneys revealed an irregular, pale yellow-tan zone on the dorsal part beneath the capsule. The pale area had a normal consistency and extended into the kidney, wherein the margins were sharply delineated. The kidneys were further examined microscopically, and we saw that the zone of thermal injury was always sharply delineated, although the damaged area varied in size. Figure 1 shows the microscopic morphological changes in the kidney tissue after thermal injury. Haematoxylin and eosin staining showed a sharp delineation between intact and destroyed glomerula, whereas the tubules were widely destroyed. Additionally, a massive infiltration of leukocytes was observed in the sharply circumscribed area of the damaged kidneys. Up to two-thirds of the medullary cone of the kidneys was necrotic. The damaged area corresponded to the dorsal part of the kidneys, which faced the heating pad during the operation.
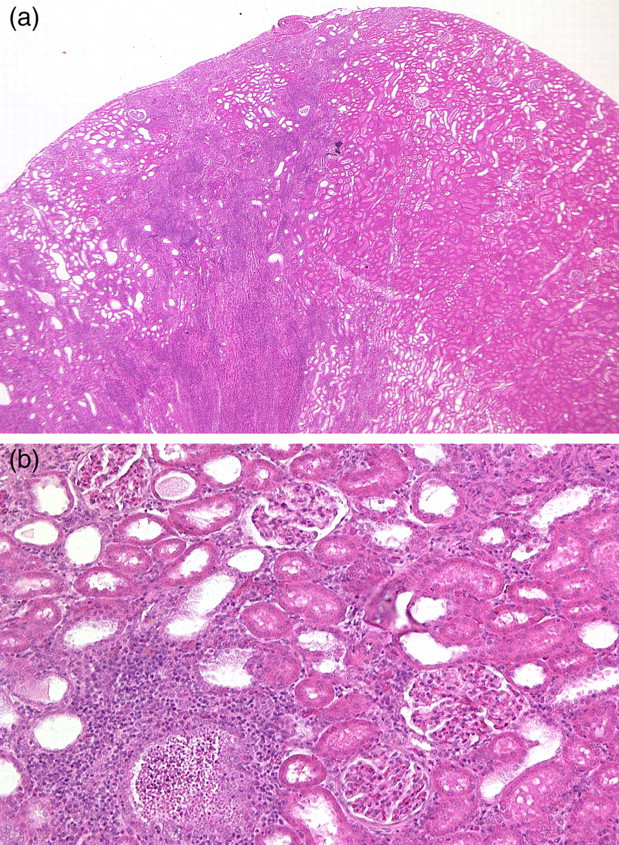
Haematoxylin–eosin staining of a representative thermally injured rat kidney (a). The area of necrosis extends from the subcapsular region and is sharply delineated from the normal parenchyma (b). The glomerula are still intact, although the tubuli are destroyed and a high number of neutrophil granulocytes are present
Thermal injury increased the NGAL concentration in both the serum and kidney homogenates in the operated animals (sham, infarct group) compared with rats that did not have an operation (negative control) (Figures 2a and b). The NGAL ratio of the kidney to serum concentration was decreased in animals that underwent an operation compared with the control animals (Figure 2c).
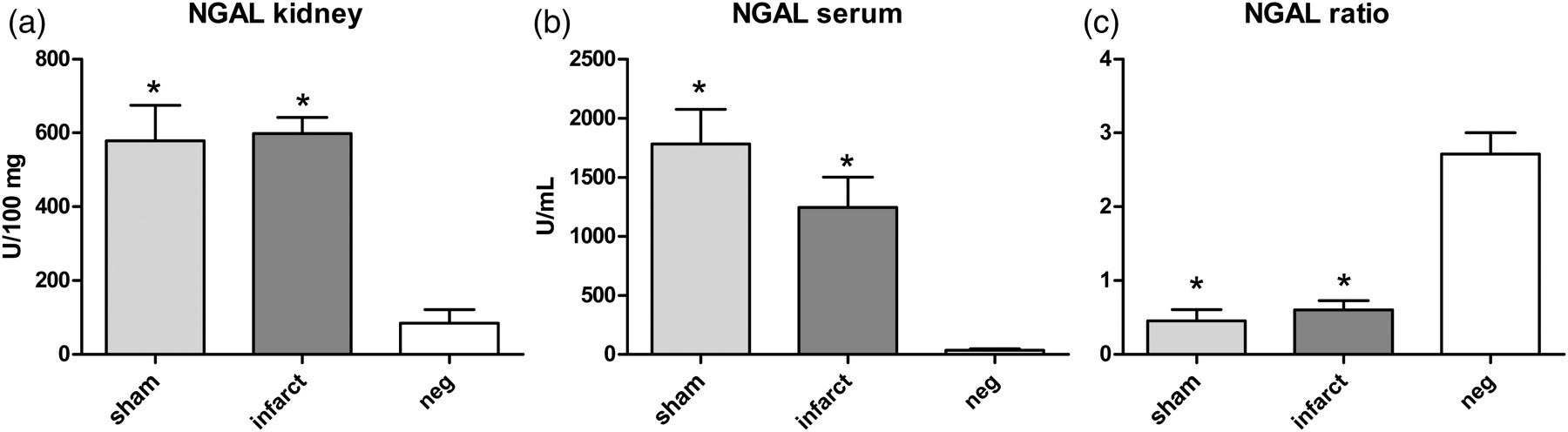
The neutrophil gelatinase-associated lipocalin (NGAL) concentration in the kidney homogenate (a) and serum (b). The ratio of the kidney to serum NGAL concentration (c). *
The troponin T concentration was significantly elevated in the infarct group compared with the sham and the negative control groups (Figure 3). The myoglobin values were not significantly different among the three groups and remained within a normal range (data not shown). Elevated L-FABP values were only present in the infarct group (Figure 3), which also had the highest IL-6 values. Although the IL-6 concentration was somewhat elevated in the sham group, this difference was not significant when compared with the negative control group (Figure 3). The animals with a myocardial infarction had slightly higher HRs during ischaemia (Figure 4) and significantly lower systemic blood pressure during myocardial ischaemia and reperfusion (Figure 4) than the sham-operated animals.
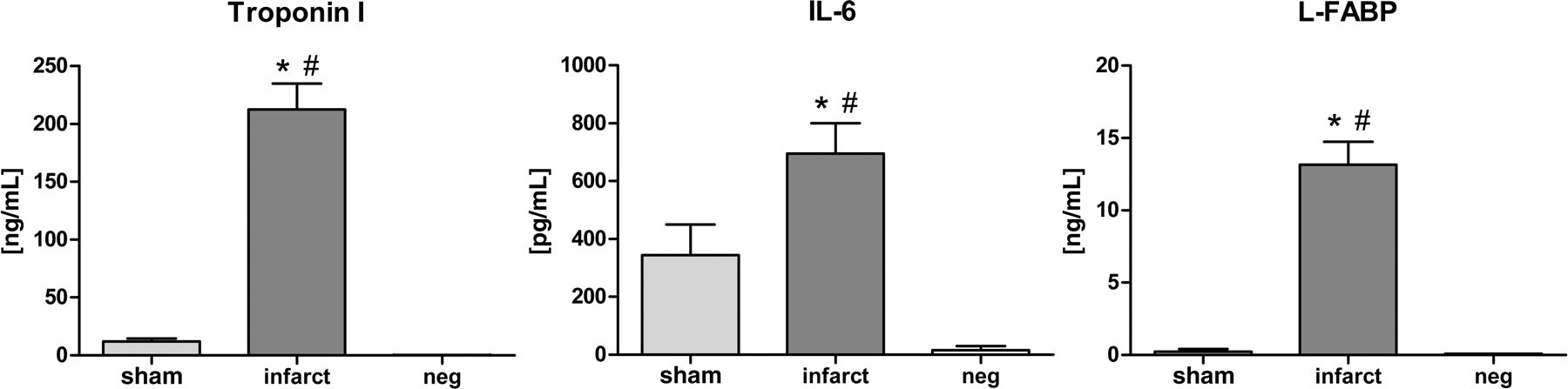
The troponin I concentrations as well as the IL-6 and liver fatty-acid binding protein (L-FABP) concentrations were significantly raised in the infarct group compared with the sham and the negative control group. *
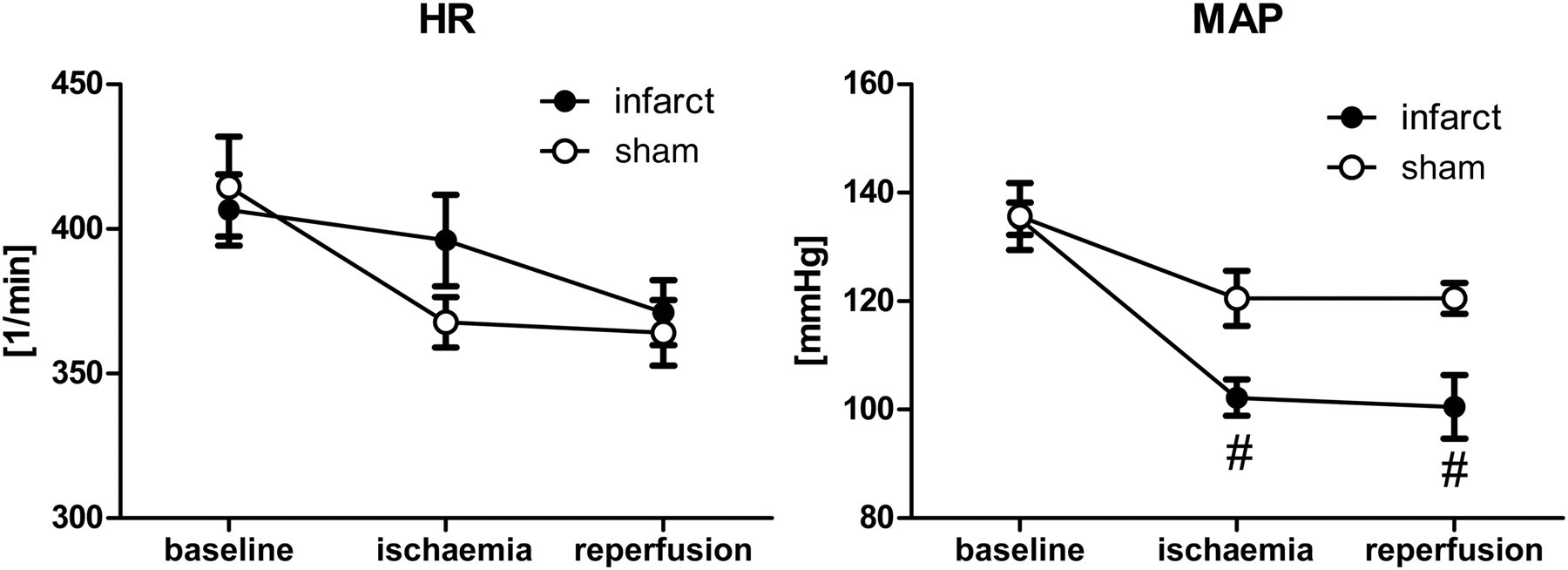
The heart rate (HR) did not differ significantly in the infarct and sham group. The mean arterial pressure (MAP) was significantly lower in the infarct group, although it was still sufficiently elevated for adequate organ perfusion. #
Discussion
The current study describes an unexpected, isolated renal injury that was induced by a thermal heating plate, despite correct use and on the first sight functioning temperature feedback. Elevated enzyme levels for kidney and inflammation markers would have been misinterpreted to be a consequence of the myocardial infarction. However, muscle and liver injury could not be related to the heating plate.
Comparably high NGAL values in both of the operated groups (infarct and sham) suggest that the heart is not a relevant source for NGAL. Elkon
Elevated IL-6 levels in the infarct and sham group show that the heart and kidneys are the source of this cytokine. IL-6, a proinflammatory cytokine, reaches a peak concentration in the serum by 4–8 h after renal ischaemia reperfusion injury. 9 IL-6 is a well-known general marker of inflammation, and the IL-6 concentration correlates with the myocardial or cerebral infarct volume. 10,11 However, in our model, elevated IL-6 levels could have been misinterpreted as the only origin of myocardial injury because IL-6 triggers structural and functional damage as well as aggravating left ventricular remodelling. 12 High IL-6 levels due to kidney injury would confound the results of myocardial ischaemia reperfusion studies.
As was expected, only the group with a myocardial infarction had an elevated concentration of a marker for liver injury (L-FABP) and a high troponin concentration, which was likely a consequence of heart congestion. We have previously observed that the slight increase in troponin concentration in the sham-operated group is a consequence of myocardial injury that is induced by the Prolene ligature.
The animals used in this study did not have any arteriosclerotic disease, were not diabetic and were not exposed to nephrotoxins, all of which could confound the results and hinder the identification of early biomarkers for ischaemic acute renal injury. Temperature control is crucial for ischaemia–reperfusion injury models. The extremely large body surface area to weight ratio of small animals renders them particularly prone to heat loss during surgery, which has to be compensated for with external heating devices. The rectal temperature measurements could be tampered by isolation of the probe by faeces, recurrent movement of the probe by the propulsive movement of the rectum or by the position of the animal, depending on the heating device used. The exact temperature of the heating plate is not measured and therefore not displayed on the temperature control unit. Only the set point temperature of the temperature control unit is displayed, which led to the overheating of the heating plate while the animal's temperature measured within the rectum is still below the set point temperature. The internal potentiometer of the control unit is not accessible for the user. Other test points should be considered that would more precisely reflect the animal's core temperature. Relevant temperature gradients between the oesophagus and rectum could be measured. 13 Thus, the place of temperature recording for the feedback control unit will have an additional impact on the real temperature in the organ. As a consequence of this, measurement of oesophageal temperature would be appropriate for studies on cerebral or spinal cord injury. Inhomogeneous distribution of temperature within the animal could be reduced by simply covering the animal with a blanket.
Despite their temperature feedback control algorithms, commercially available heating devices should be controlled for its maximal temperature on the surface and limited to 39°C. More stringent temperature control with adequate site of measurement would limit tissue injury and their effects on pathomechanisms and markers, which are the primary focus of animal studies.
Footnotes
Acknowledgements
Special thanks are extended to Renate Nadenau, Christian Bleilevens, Christian Beckers (Department of Anaesthesiology) and Thaddäus Stopinski (Institute of Laboratory Animal Sciences) for their help in the laboratory. We would like to thank the IZKF Biomat Laboratory for their histological work and technical support with the microscopy. Finally, we would like to thank Professor Dr F Eitner, (Department of Nephrology) for his expertise in renal histopathology in rodents. This study was supported by the German Research Foundation (DFG; fund number Ro 2000/10–1).
