Abstract
In humans, cleft palate (CP) is one of the most common malformations. Although surgeons use palatoplasty to close CP defects in children, its consequences for subsequent facial growth have prompted investigations into other novel surgical alternatives. The animal models of CP used to evaluate new surgical treatments are frequently obtained by creating surgically induced clefts in adult dogs. This procedure has been ethically criticized due to its severity and questionable value as an animal model for human CP. Dogs born with a congenital CP would be much better for this purpose, provided they developed CP at a sufficient rate and could be fed. Up until now, feeding these pups carried the risk of aspiration pneumonia, while impeding normal suckling and chewing, and thus compromising orofacial growth. We developed a technique for feeding dog pups with CP from birth to the time of surgery using two old Spanish pointer dog pups bearing a complete CP. This dog strain develops CP in 15–20% of the offspring spontaneously. Custom-made feeding teats and palatal prostheses adapted to the pups' palates were made from thermoplastic plates. This feeding technique allowed lactation, eating and drinking in the pups with CP, with only sporadic rhinitis. To determine whether the use of this palatal prosthesis interferes with palatal growth, the palates of three littermate German shorthaired pointer pups without CP, either wearing or not wearing (controls) the prosthesis, were measured. The results showed that the permanent use of this prosthesis does not impede palatal growth in the pups.
In mammals, the palate is the structure that separates the nasal and oral cavities to allow simultaneous suckling and breathing during lactation. The palate develops from two anlages. The primary palate is derived from the junction of the two medial nasal processes, which extend from the frontonasal process of the head. This part of the palate gives rise to the anterior part of the hard palate, which supports the incisors. The rest of the hard palate and the soft palate develop from the secondary palate, comprising two bars of mesenchymal tissue of neural crest origin covered by ectoderm, the palatal shelves, which appear in the medial aspect of each maxillary process of the first branchial arch. The palatal shelves first grow downwards on both sides of the tongue and then elevate, approach each other and contact at the midline. Fusion of all these anlages completes the palatal development in most mammals, 1,2 with the only variation being a question of timing. For example, in humans this takes place between weeks six and 13, in mice between days 11 and 15, 1 while in dogs the secondary palate becomes fused between days 25 and 28. 3
The failure of any of the mechanisms described above leads to the appearance of a cleft palate (CP), a congenital defect in which the oral and nasal cavities communicate to a greater or lesser extent. 4 In humans, the frequency of CP ranges from 1:500 to 1:2500 live births. 5 In animals, CP has been described in a variety of species, 6 although its incidence is unknown. Any strain of dog may develop sporadic CP. Breeding studies show evidence of an inherited pattern with incomplete penetrance in the Shih-Tzu breed, as well as possibly in bulldogs, pointers and Swiss sheepdogs. 7 Its frequency ranges between 1.1 per 1000 in in-bred beagles 8 to 26.9% in a colony of Brittany spaniel–beagle crossbred dogs. 9 The old Spanish pointer dog, a Spanish dog strain, has an incidence of spontaneous CP in about 15–20% of its offspring. 10
To date, the only permanent treatment of CP is surgery, with surgeons selecting from different palatoplasty techniques. However, all of the current surgical methods have negative consequences for the growth of the maxilla. Palatoplasty impairs facial growth in children because the advance of the two mucoperiosteal flaps to the midline during surgery causes scars at the lateral margins of the palate, which impede the growth of the maxilla. 11 This leads to the non-aesthetic appearance of the face, which usually requires an average of four surgeries during infancy and adolescence, and expensive orthodontic treatment.
Alternative procedures to palatoplasty for CP repair are being investigated. Some have been done in humans or in vitro, 12,13 although the majority use large animal models. Most frequently, a surgical CP is created in adult dogs with a normal palate to produce a standard defect to be treated with the desired technique. 14–19 This procedure has been ethically criticized due to its severity and its questionable value as an animal model for human CP, 20,21 since it does not mimic the natural defect, which is congenital, nor the current palatal closure in humans, which is performed early in childhood on unoperated tissues. Less frequently, congenital CP is caused using mechanical or chemical means 22–24 or it is selected as an inherited trait, 9,25,26 although the difficulties in rearing these animals to the point of surgery complicate their use. Natural lactation is impossible and, if standard bottle-feeding is established, milk flows into the nasal passages, leading to sneezing, gagging, coughing and nasal discharge. Affected animals are at greater risk of developing aspiration pneumonia, which may result in death. The use of devices that fill the oral–nasal passageways has been reported, 27,28 but their permanent placement makes both removal and cleaning difficult, leading to nasal infections. The most commonly used option is feeding these animals through an orogastric tube. 25 This technique consists of the insertion of a feeding tube through the oesophagus into the stomach. The rubber-feeding catheter is connected to a syringe charged with an amount of milk determined after weighing the puppy. 29 However, there is still a risk of pneumonia from the inadvertent delivery of milk to the lungs instead of the stomach, and infection of the tube is frequent. 30 In addition, the tube does not encourage the suckling reflex of the neonate and the gradual delivery of milk to the stomach. 31 Correct suckling and chewing are essential for developing the proper equilibrium of muscular forces during orofacial growth, which is required for normal jaw morphology and dental occlusion. 32 Any research project planning to test new approaches for CP surgical repair should consider their consequences for orofacial growth.
Here, we report on a pilot project validating how to manage spontaneous CP (its natural form) by using individualized and custom-made feeding teats during lactation and a removable flexible palatal prosthesis after weaning. This method considerably reduces the risk of respiratory infections, allows normal suckling and eating, and does not interfere with the normal palatal growth of the puppies. It constitutes a refinement in how the animals are used and managed in experimental procedures for CP surgical research purposes.
Material and methods
Animals
The dog pups used in this study were a male and a female old Spanish pointer, and one male and two female German shorthaired pointers. All were obtained from the dog breeding and selecting centre in Olmeda de Extremo (Zoological Centre C19053125, Guadalajara, Spain). The research project presented here and the use of these dog pups and the old Spanish pointer strain for CP surgical research was ethically approved by the Animal Protection Area of the Community of Madrid (the body responsible for the use of animals for experimentation in Spain/Madrid – Ref. 04/262662.9/07), given that there are no large-sized animal models with congenital CP. Ethical approval was also obtained from the Ethical Committee of Animal Welfare of the Hospital Clínico San Carlos of Madrid (Ref. 08/19–18), where the dogs were housed, based on the approval from the Animal Protection Area of the Community of Madrid. These ethical approvals were in accordance with the stipulations of the Spanish Royal Decree 1201/2005 regarding the protection of animals used for experimentation and other scientific purposes, which is based on the EU Directive EC86/609 and its modification (Directive 2003/65/CE).
On arrival, the old Spanish pointer male was five days old, and presented a complete cleft of the primary and secondary palates and no other malformations (Figures 1a and b). The female of the same strain was one day old and had a complete cleft of the secondary palate (Figures 1c and d). The German shorthaired pointer pups were one week old and had normal palates. All of them were comfortably transported from the breeding centre to the Medical and Surgical Experimental Unit of the Hospital Clínico San Carlos under the supervision of two members of the research team.
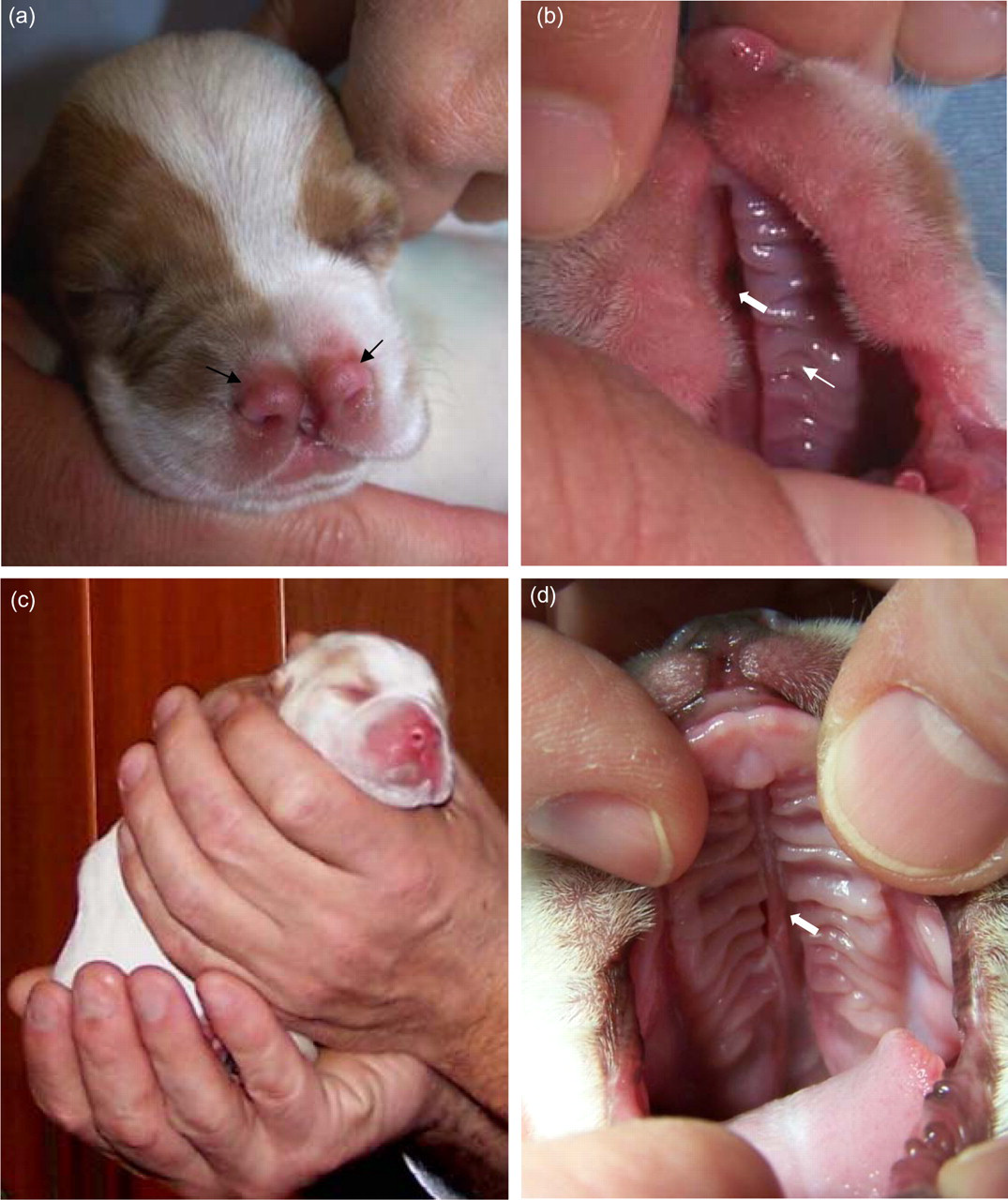
Cleft nose and palate in the old Spanish pointer dog pups. (a) Old Spanish pointer male dog aged five days. The arrows indicate the cleft nose. (b) Presence of complete cleft palate (thick arrow) in this dog's mouth. The thin arrow points to the palatal shelf. (c) Same strain female pup with normal nose aged one day. (d) Complete cleft of the secondary palate (arrow) of the pup is shown in (c)
All dog pups were housed in the Medical and Surgical Experimental Unit of the Hospital Clínico San Carlos, which is legally approved as an animal centre for research use (Ref. ES280790000088). This centre fulfils all the requirements of the Spanish Royal Decree 1201/2005 and the EU Directive EC86/609 mentioned above, and has two qualified veterinary personnel in charge, who were also members of this research team. The dogs in this study were closely supervised by the personnel in the Unit, where they were housed independently (9.7 m2 rooms), with a dry floor for resting, and humidity maintained at 45 ± 10%, 32–21°C, depending on the age of the pups, and 12 h light (300 lux)/dark cycles. Toys adequate for each age were provided. An assistant vet and an investigator from the research team took direct care of the dogs (no less than 7 h/day), including their hygiene, playing and daily walks. All routine vaccines for the puppies were administered (Parvigen®, Virbac, Carros, France; Duramune® Puppy DP + C, Fort Dodge Laboratories Ireland, Sligo, Ireland; Novibac® Puppy DP, Intervet International BV, Boxmeer, The Netherlands; Maxivac Hepta®, Girona, Spain). The intranasal vaccine for kennel cough was not given. Antiparasitic treatment (Vithaminthe, Virbac; Zipyran, Calier, Barcelona, Spain), microchips and other dog needs were administered. Since the fate of all of these dogs was adoption, socialization was carried out by exposing the dogs to different people in the Unit and in a nearby public garden, where they played daily. Socialization was especially important from day 21 onwards.
Humane endpoints were established based on four parameters related to each of the following items: (1) physical appearance (hair/coat); (2) posture; (3) temperature; (4) respiratory frequency; (5) spontaneous behaviour; and (6) handling response. Each parameter was scored from 0 (normal) to 3 (highly abnormal). Total scores were calculated by combining the subscores assigned after subjective evaluation of each parameter. Total scores were calculated and ranged from 0 to 18. Pups with a score of 0–5 were considered healthy, whereas pups with a score of 6–12 were considered as demonstrating clinical signs associated with morbidity and requiring the establishment of therapeutic measures. Scores of 13–18 were considered humane endpoints for euthanasia.
Feeding the pups with a CP during lactation
The male pup with a CP had been fed naturally for two days, after which he had become lethargic and dehydrated. Once he was brought to the Unit he was injected with 18 mg/kg body weight amoxicillin/clavulanic acid (Sinulox, Pfizer Inc, New York, NY, USA) and 10 mL of a 50% Ringer–50% glucosate solution to restore hydration. Antibiotic therapy was given prophylactically and continued for three days. The female was naturally fed for only 20 h, before starting artificial lactation.
A custom-made feeding teat that could be used with any standard feeding bottle was then manufactured (Figure 2). First, an impression of the pup's palate was taken by carefully introducing and maintaining a very thin piece of fast-setting silicone putty (Dentalite, Madrid, Spain) into the dog's mouth with the little finger, which did not cause distress to the pup. A plaster cast was then produced from this impression (Figure 2a). Using the teat of a commercial feeding bottle, a cast was made with silicone putty (Figure 2b). More silicone was added to the suckling end (Figure 2b) to adapt its palatal surface to the palatal surface of the plaster cast (Figure 2c). With the help of a thermo-vacuum-forming machine (Easy Vac, Ultradent Products Inc, South Jordan, UT, USA) (Figure 2d), feeding teats made of 1.5 mm thick thermoplastic plates (Mouthguard 060, Dentaflux, Madrid, Spain) were produced, which moulded the silicone cast (Figures 2e and f). Three small holes were made on their lingual surfaces for milk outflow (Figure 2g) and their palatal side was labelled (Figure 2f). The pups were fed with Babydog Milk (Royal Canin, Paris, France), which was prepared and administered according to the manufacturer's instructions. The pup's mouth was wiped gently with a sterile cloth each time the feeding teat was removed. Immediately after the meal, each nostril was cleaned by aspiration or with a plastic Pasteur pipette.
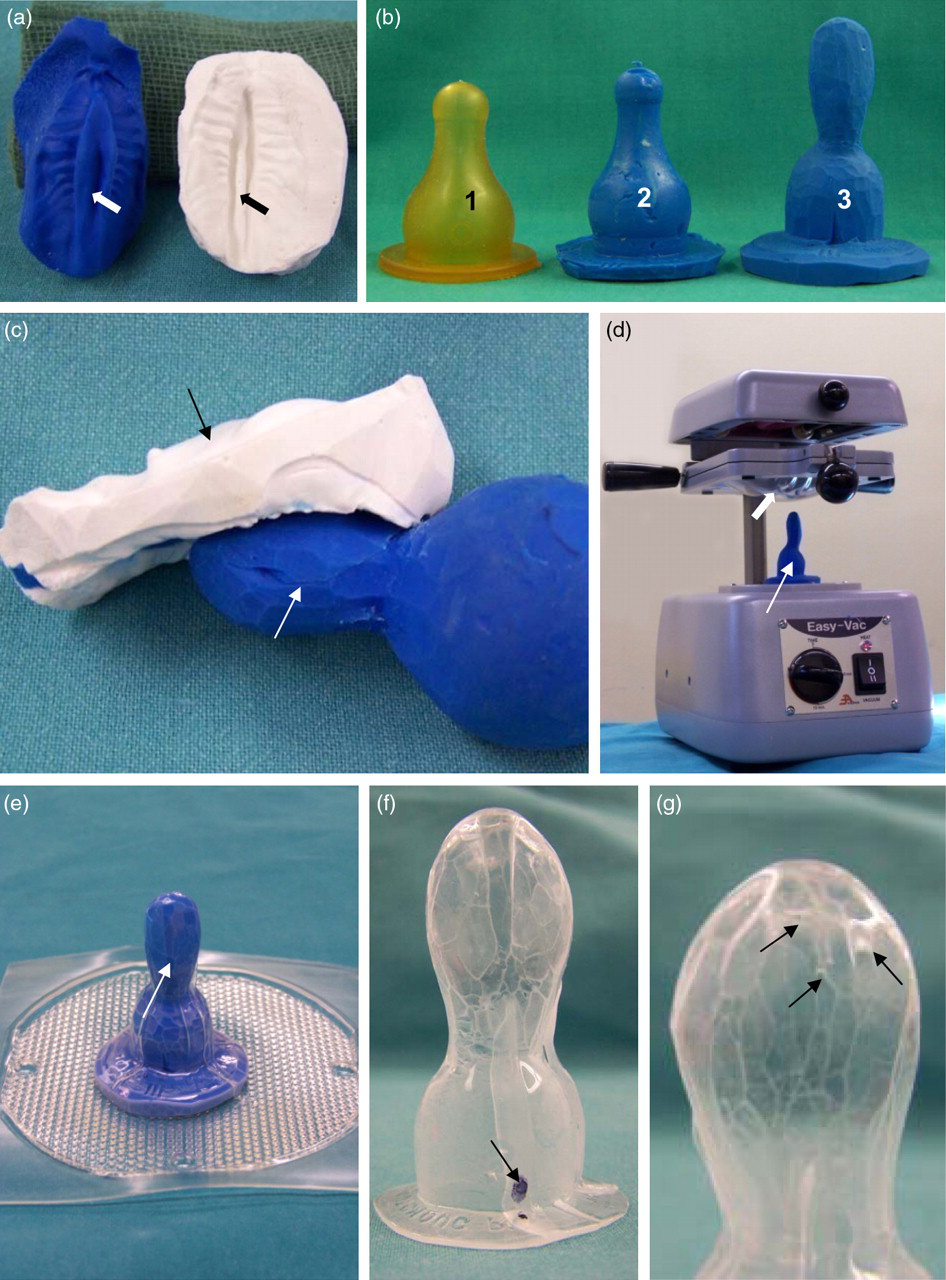
Production of the feeding teat. (a) Impression of the pup's palate in silicone putty (black) and plaster cast (white). The impression of the nasal septum (arrows) between the cleft palate edges is observed. (b, c) Steps to obtain the silicone mould of the feeding teat: a standard feeding teat (1) is filled with silicone putty (2); more silicone is added to the silicone mould end (3 and white arrow in c) to adapt to the plaster cast of the palate (black arrow in c). (d) Thermovacuum-forming machine with the silicone cast of the feeding teat (thin arrow) in place and the thermoplastic plate (thick arrow) ready for use. (e) Silicone cast of the feeding teat covered by the thermoplastic plate (arrow) as seen after the use of the thermovacuum-forming machine. (f, g) Palatal (f) and lingual (g) views of the feeding teat produced. Three small holes are observed in the lingual side (arrows in g) and the palatal side is labelled (arrow in f)
Feeding the pups with a CP after weaning
Weaning took place during week 5. Temporary incisors, canines and two premolars appeared in the mouth, and the CP was clearly visible (Figures 3a and b). At this time, a prosthesis that was adapted to the dog's palate and the alveolar ridge was made. First, under isoflurane induction anaesthesia (isoflurane 2–3% and oxygen 3 L/min) and with the help of a prefabricated tray made of a 1.5 mm soft thermoplastic plate (Dentaflux), an impression of the palate was taken with silicone putty (Dentalite; Figures 3c–e). Before the silicone in the tray was used, a small portion of it was placed in the cleft area and allowed to harden (Figure 3d). Both silicone portions were fused together afterwards (Figure 3e). The anaesthetic face mask was removed for only a few seconds to allow the processes of introducing and removing the silicone in the mouth. Next, a plaster cast was obtained from the silicone mould (Figure 3f). The palatal prosthesis was then made with the thermovacuum-forming machine from a 0.9 mm thick thermoplastic plate (Soft-Tray, Ultradent Products Inc), using the plaster cast as a mould. The prosthesis was extracted from the rest of the thermoplastic plate by cutting it away through a line that allowed it to cover most of the oral vestibulum rostrally and laterally, the alveolar ridge, the whole hard palate and the anterior part of the soft palate (Figure 3g). The manufactured prosthesis was flexible but consistent enough to prevent it from easily folding. Holes were then made in the prosthesis at the positions of the teeth (Figure 3h).
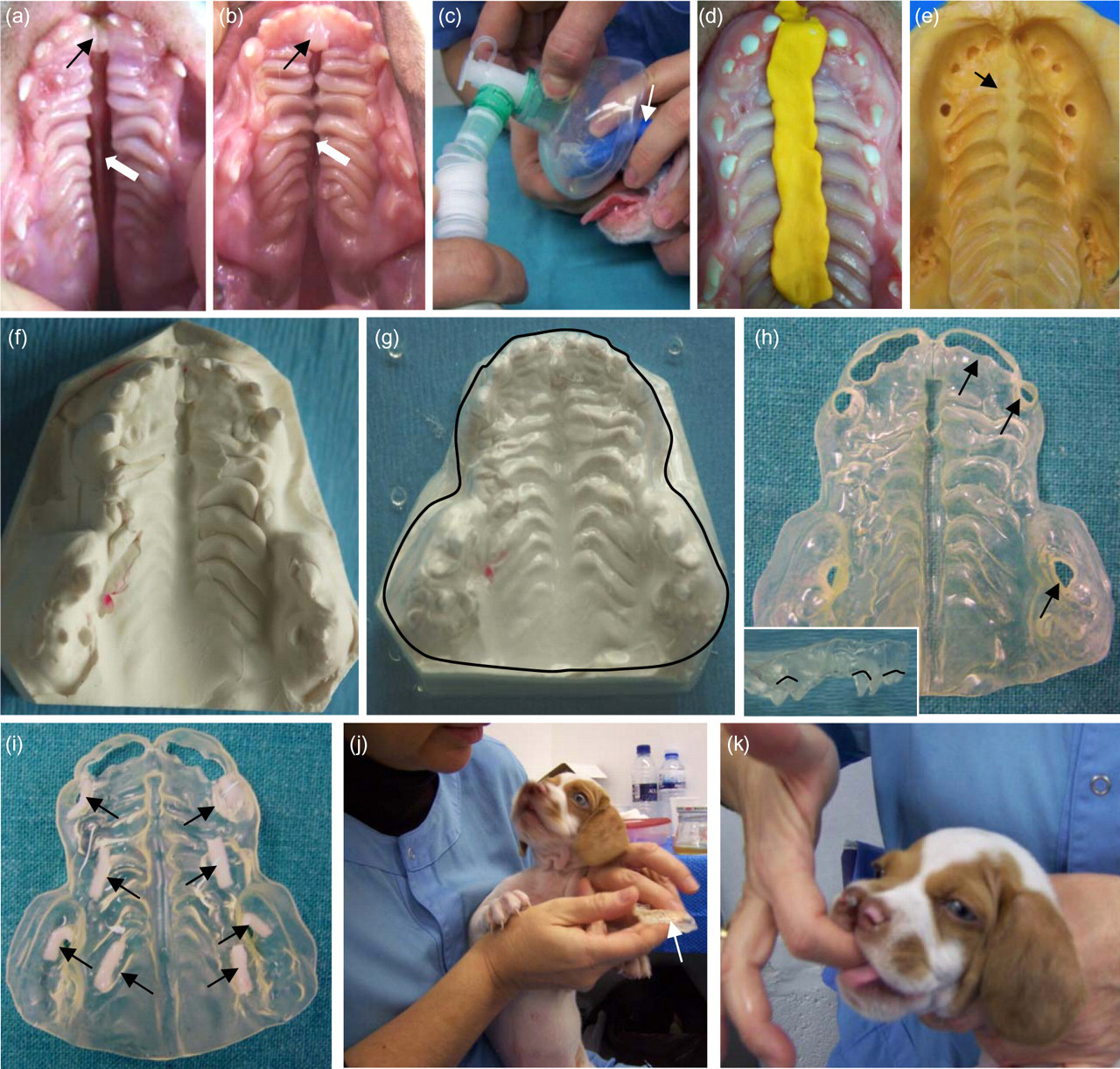
Production and placing of the palatal prosthesis. (a) Complete cleft of the primary (black arrow) and secondary (white arrow) palate in the old Spanish pointer male dog aged five weeks. (b) Complete cleft of the secondary palate (white arrow) with a normal primary palate (black arrow) in the female pup at the age of five weeks. (c–h) Preparation of the thermoplastic prosthesis: dog under isofluorane anaesthesia induction (c), while the impression of the palate is taken in silicone putty (arrow in c); sealing the cleft with a small portion of silicone (d) prior to taking the whole palatal impression; both silicone portions adhere perfectly (arrow in e) in the final palatal impression (e), from which the plaster cast is obtained (f); with the help of the thermovacuum-forming machine, the thermoplastic plate moulds to the plaster cast (g); the prosthesis is cut away along the black line (g); holes are then made (arrows in h and lines in the inset) at the sites where the teeth are present. (i–k) Placing of the palatal prosthesis in the pup's mouth: several wet Corega® Strips are placed on the palatal side of the prosthesis (arrows in i), which is immediately placed in the pup's mouth using the fingers (j, k)
Immediately prior to placing the prosthesis into the dog's mouth, eight pieces of Corega® Strips (GlaxoSmithKline, Brentford, Middlesex, UK), moistened with drops of water, were placed on its palatal surface (Figure 3i), which helped its adhesion to the dog's palate (Figures 3j and k). It remained in the dog's mouth during the daytime hours, and was easily removed at night to prevent any possible nasal infection caused by the permanent use of the prosthesis in these pups with CP. A new prosthesis was manufactured following the same procedure every other week.
From weeks 5 to 11, the food consisted of pieces about 1 cm thick, formed by 50% mashed feed (Initial Advance, Affinity Petcare SA, Barcelona, Spain), and 50% Savoury Chicken Puppy food (Science Plan, Hill's Pet Nutrition Ltd, Watford, UK). Water and food were offered four times a day. When the prosthesis was not in place, neither food nor water was administered. From week 11 onwards, the food was Royal Canin Maxi Junior feed, following a three-meal-a-day schedule.
Measurement of palatal growth in pups bearing the thermoplastic palatal prosthesis
The palatal growth of three German shorthaired pointer dogs with a normal palate, one of which (a female) was a control and two of which (a male and a female) wore a thermoplastic prosthesis 24 h a day from weeks 1 to 11, was compared. The prosthesis was made as described earlier. It was changed every other week according to new impressions of the palate, and was only removed for cleaning purposes every 12 h.
Transverse and longitudinal measurements were recorded on maxillary dental casts taken from the three pups weekly (Figure 4). Six longitudinal segments were defined along the mid-palatal suture, starting at the centre of the alveolar ridge and ending at the following points: (L1) posterior end of the inter-incisal papillae, (L2) posterior end of the second palatal rugae, posterior end of the (L3) fourth, (L4) fifth and (L5) eighth palatal rugae and (L6) posterior nasal spine. The six transverse dimensions recorded on each cast were: (T1) width of the anterior alveolar ridge measured between the distal sides of the lateral incisors, (T2) width between the two canines, (T3) width of the fourth palatal rugae, (T4) width between the first two premolars, (T5) width of the fifth palatal rugae and (T6) width of the eighth palatal rugae. All measurements were taken using an electronic digital calliper (Ceosa®, Madrid, Spain) with a precision of 0.01 mm. Although the sample only consisted of three dogs, longitudinal and transverse changes were compared using non-parametric tests. Non-parametric tests can be applied to small sample sizes, since they do not have sample size restrictions. Differences in measurements between the observation periods (Friedman's test for repeated measures with Dunn's multiple comparison test as a post hoc test) and between the two experimental and control pups (Kruskal–Wallis test with Dunn's multiple comparison test as a post hoc test) were assessed.
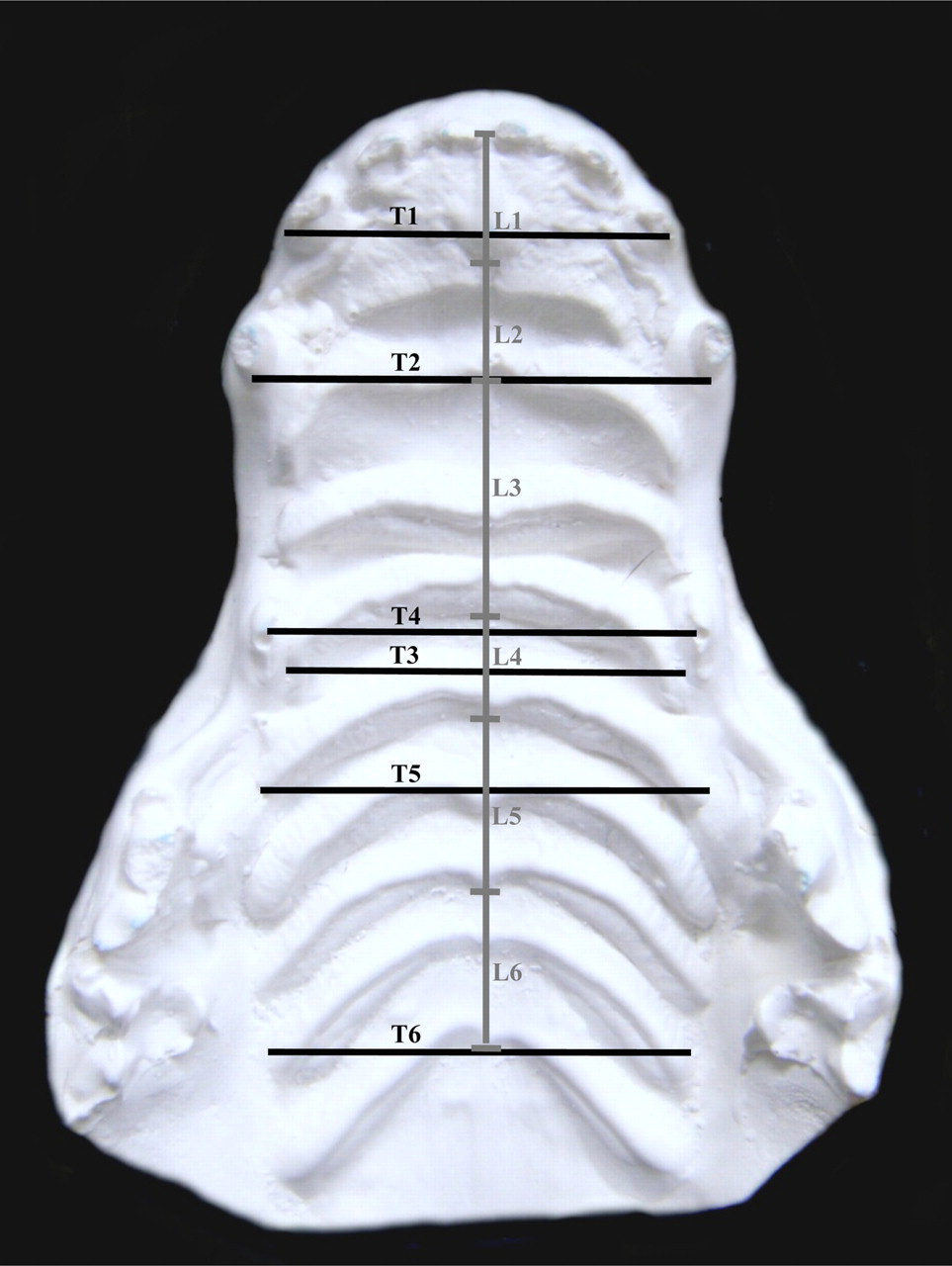
Measurement of palatal growth in the German shorthaired pointer pups' palatal plaster casts. Representative plaster cast of the palate of a German shorthaired pointer pup. The longitudinal measurements (L1–6) taken are shown in grey and correspond to six longitudinal segments defined along the mid-palatal suture. The transverse measurements (T1–6) are shown in black and represent the distance between pairs of teeth (T1, 2 and 4) or specific rugae (T3, 5 and 6)
Results
Clinical situation and development of the pups with a CP fed by the new technique
During the entire lactation period (5 weeks), three different sized teats were produced using the method described above to adapt to the growing palates (Figures 5a and b). These feeding teats were easily used with any standard feeding bottle (Figure 5c) and were accepted by the pups, which suckled like any bottle-fed animal (Figure 5d). In the pups on which this study was performed, some sneezing and nasal discharge was observed up to the third week of life, which required the use of antibiotics (18 mg/kg body weight amoxicillin/clavulanic acid, Sinulox, Pfizer Inc) for six days only in the male pup. This clinical situation was reflected in the lower body weight gain observed in the male compared with the female in week 2 (Table 1). Coughing was never observed and there were no signs of lung aspiration.
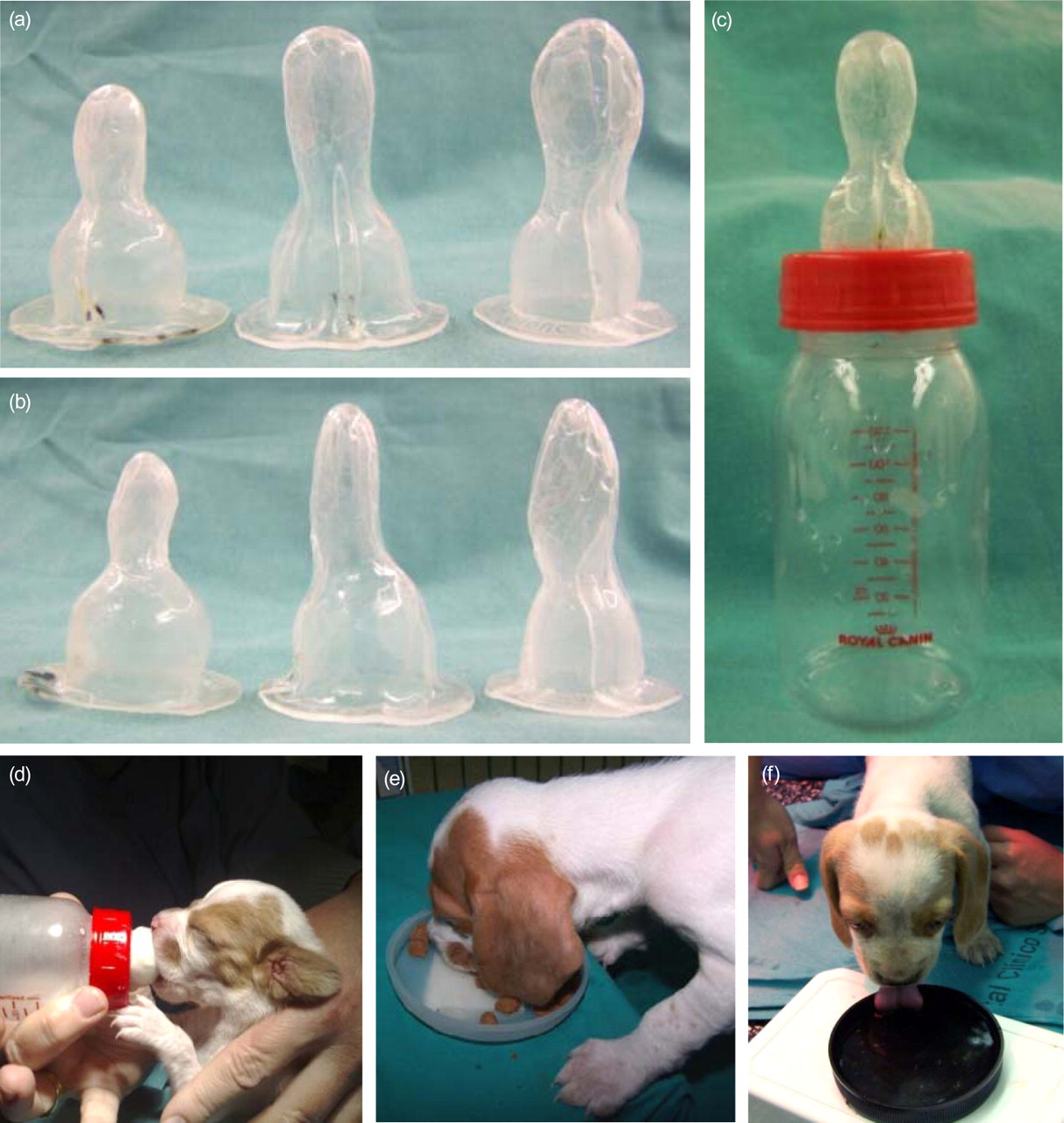
Feeding the pups with a cleft palate by the new technique. (a, b) Appearance of the three feeding teats manufactured during the lactation period observed from a palatal (a) and lateral (b) view. (c) The feeding teats were adapted to standard feeding bottles. (d) The use of the feeding teats allowed the pup's normal suckling behaviour. (e, f) The pups with a cleft palate ate food (e) and water (f) normally when wearing the thermoplastic palatal prosthesis
Body weight and weekly body weight gain of the two pups with a cleft palate fed by the new technique
*Lactation period
†Mixed nutrition
‡After weaning period
Weaning took place at week 5. The custom-made palatal prosthesis was initially placed in the dog's mouth between meals, but after only one day of adaptation, the pups were used to wearing it and did not show any signs of discomfort. The use of the palatal prosthesis allowed the pups to eat and drink as any dog without CP would (Figures 5e and f). From week 6 onwards their weight began to increase more rapidly (Table 1), while rhinitis and coughing, which are typical in CP animals, were never observed.
The material from which both the feeding teat and the palatal prosthesis were made did not cause halitosis or gum irritation, nor did it interfere with dental eruption. The adhesive Corega® strips were also tolerated by the dogs at all times. The dogs with CP in this study showed the normal behaviour of a healthy dog, including very good socialization skills in their relationship with other dogs and humans.
Palatal growth in pups using the palatal thermoplastic prosthesis
The longitudinal and transverse measurements taken from the plaster casts obtained from the palate of the three German shorthaired pointer dogs from weeks 1 to 11 are presented in Figure 6. Most of the anteroposterior growth of the palate took place in the posterior region (starting at L3, the fourth palatal rugae, increasing towards L6, the posterior nasal spine) with minor changes in the anterior region. Late transverse growth of the palate (weeks 9 to 11) was mostly observed in the central-posterior portion (T4, width between the first two premolars). No differences were found between the control pup and the two experimental pups at any time or between the observational periods.
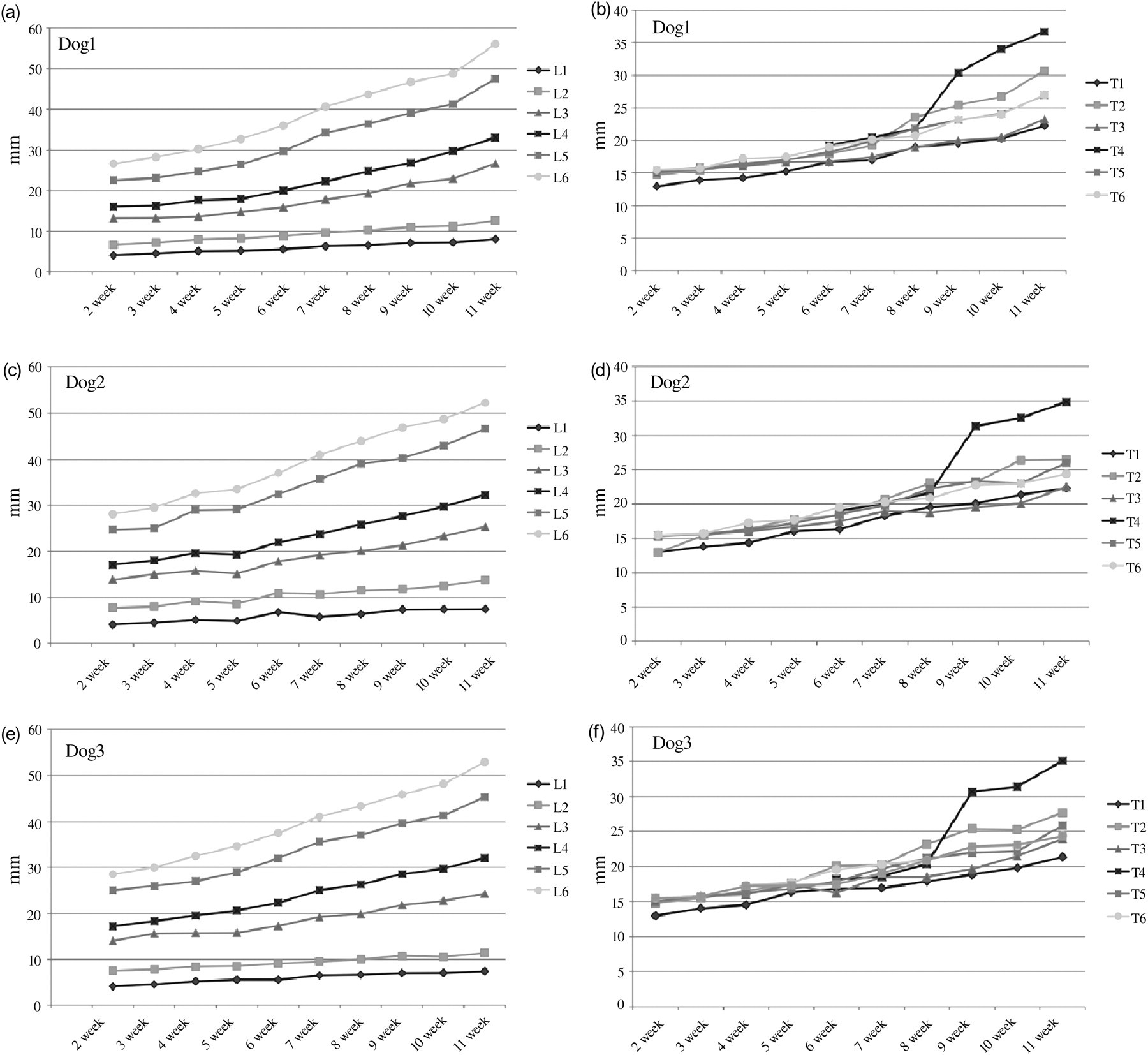
Palatal growth of the three German shorthaired pointer pups wearing and not wearing the palatal prosthesis. Longitudinal (a, c, e) and transverse (b, d, f) measurements of the palate of the two German shorthaired pointer pups wearing (dogs 1 and 2) and not wearing (dog 3) the thermoplastic palatal prosthesis. L1–6 and T1–6 correspond to the measurements taken in the plaster casts obtained from their palates in weeks 1 to 11. No differences were observed
Discussion
Congenital CP in animals is quite common. 6 Some strains of dogs develop CP 9,10,26 at such a rate that they could constitute good animal models for research on new surgical approaches. The old Spanish pointer dog with CP is such an example, since CP appears spontaneously in 15–20% of its offspring. 10 The work performed here represents the starting point for investigating, within the framework of two funded research projects (Osteology Foundation Ref. 09/012, and Spanish Ministry of Science and Innovation Ref. PS09/01762, ethically approved by the Experimental Animal Committee of the Universidad Complutense of Madrid, as it fulfils the deontological and ethical requirements of the Spanish Royal Decree 1201/2005 and EU Directive EC86/609), whether or not it is possible to repair CP with a minimally invasive technique that causes less complications for facial growth than palatoplasty. Following the hypothesis posed previously by this research team, 13 the experimental approach to be assayed will include the injection of BMP2 onto a hydrogel carrier at the edges of the pups' CPs with the aim of increasing their volume such that they reach the midline in one or two sessions. Once ossification is completed, refreshment of these edges will allow final palatal closure. To investigate the benefit of this new method for facial growth, craniofacial radiographs, axial tomography and magnetic resonance images taken from dogs of this strain with CP, treated by the new approach and by palatoplasty, and from dogs with a normal palate will be compared. We expect the absence of a lateral scar using the experimental method will benefit the dogs' facial growth. Regardless of the results obtained, the CP of all dogs in the study will be repaired prior to them being given up for adoption.
To date, animals with congenital CP have been fed using an orogastric tube, which requires specialized nursing training, 25 may have clinical complications such as aspiration pneumonia and tube infections, 30,31 and which compromises neuromuscular development in the orofacial area due to the lack of suckling. 32 Here, we present a method for feeding dogs bearing CP with a minimal risk of lung aspiration and respiratory tract infections and a lack of interference with normal suckling and chewing behaviours. This method was successfully tested in two dog pups with complete CP, a severe form of this disease, thus suggesting that it may be used in any kind of CP.
Although the special nipples to be placed in feeding bottles are available for children with CP, we have not found any described for animals. The individualized feeding teat presented here allows suckling as for any bottle-fed animal. It shows the great advantage of the particular adaptation to each pup's palate, closing the cleft during suckling. Another advantage is that the holes for the milk outflow are located on the lingual side. These two aspects reduce the entry of milk into the nasal cavity, which greatly minimizes the occurrence of respiratory infections.
To the best of our knowledge, this is the first time that a removable thermoplastic palatal prosthesis has been used to close a CP in animals, although individual trays to take palatal impressions in children with CP prior to producing a palatal prosthesis in acrylic have been made using this material. 33 The prosthesis used here was easily placed and removed from the dog's mouth; it was flexible and did not damage the oral mucosa. The pups of this study adapted to wearing this prosthesis very quickly, eating and drinking like pups with a normal palate. This is unusual in weaned animals with CP, as they need a longer time to learn how to masticate and swallow solid food, while developing abnormal chewing habits to prevent the entry of food into the nasal cavity through the cleft. 34 Importantly, respiratory infections, typical of CP animals, were absent with the use of the prosthesis, and the pups increased their body weight.
The material from which both the feeding teat and the palatal prosthesis were made is not toxic and is currently used in humans to whiten or fluoridate teeth. It has never been clinically tested in animals, but in these dogs we did not observe any apparent detrimental consequences of its use. The use of the adhesive Corega® strips (indicated for human prosthesis denture adhesion) did not have any adverse effects. A remarkable advantage of the use of these thermoplastic plates to close the CP is that they do not interfere with palatal growth. Analysis of the measurements taken from the palate of the three German shorthaired pointer pups both wearing and not wearing the palatal prosthesis indicated that its use allowed normal growth of the maxillae. A likely explanation for this fact could be the lack of rigidity of the material used. When an original plaster cast mould was compared with the prosthesis obtained from that mould after two weeks of use, an expansion of the material was observed in all cases, thus indicating that it had accommodated itself to the palatal growth. This is a great advantage compared with the material usually used in the palatal prosthesis for closing human CP, i.e. acrylic resin, 35 which is a rigid material and impossible to stretch. In children with CP, some soft materials may be positioned prior to dental eruption, thereby lining the inner part of the acrylic prosthesis, which is removed as the maxilla grows. 36 However, in dogs, maxillary growth and dental eruption occur very quickly, and the use of a rigid prosthesis could compromise both.
Although this study was performed on dogs bearing spontaneous CP, any animal with a congenital CP could be fed using this technique. None of the equipment or materials needed to obtain the palatal impressions, feeding teats, and palatal prosthesis was expensive and would probably be affordable for many veterinarian clinics. Therefore, this method could be conducted by veterinary surgeons in a clinical situation for the benefit of the animal. Its use would make possible the utilization of animals with a high incidence of spontaneous CP as animal models of this disease for the design and testing of new surgical approaches, avoiding the need for other feeding techniques that may lead to severe clinical complications. The possibility of rearing these animals will hopefully prevent the use of animal models in which the CP defect is created surgically in adulthood, 14–19 making CP surgical experimentation more ethical and adequate for the objective of improving CP treatment in children.
Footnotes
ACKNOWLEDGEMENTS
We are very grateful to C Contera and M Contera for their generous gift of the pups in this study; Dr P Torres, from the Hospital Clínico de San Carlos, of Madrid, Dr J Murillo, Mrs D Carrasco, SJT, CAG, Royal Canin and GlaxoSmithKline for their support, the Proof-Reading-Service (UK) for the English revision of this manuscript and the Editorial Board of Laboratory Animals and the anonymous referees for their valuable comments. This work was supported by grants from the Spanish Ministry of Health (PI06/0184) and University Complutense-Comunidad Autónoma de Madrid to the Complutense Research Group 920202 in 2007.
