Abstract
Research in mammalian hair cell regeneration is hampered by a lack of in vivo model of adult mouse inner ear injury. In the present study we investigated the effects of a combination of a single dose of aminoglycoside followed by a loop diuretic in adult mice. The auditory brainstem response threshold shift, extent and defining characteristics of the cochlear lesion were assessed and verified at different time points post-treatment. Our data indicated that this drug combination caused the rapid and extensive death of outer hair cells (OHCs). OHC death presented throughout the cochlea that commenced in the basal turn by 24 h and progressed apically. In contrast, inner hair cell (IHC) loss was delayed and mild. Terminal deoxynucleotidyl transferase dUTP nick end labelling-positive nuclei demonstrated that the majority of OHCs died via an apoptotic pathway. Auditory threshold shifts of up to 90 dB SPL indicated a profound hearing loss. In addition, the endocochlear potential (EP) in the drug-treated animals displayed a significant decline at 12 h post-treatment followed by recovery by 48 h post-treatment. Despite this recovery, there was a significant and progressive decrease in strial vascularis thickness, which was predominantly due to atrophy of marginal cells. The present study reproduced an adult mouse model of aminoglycoside-induced hearing loss. The mechanism underlying the recovered EP in the model with extensive hair cell death is discussed.
The guineapig has been the major animal organism for hearing and deafness research for a long time. Guineapig anatomy permits surgical access to the cochlea more easily than other species and has well characterized cochlear anatomy and physiology. 1 The guineapig has also been proven to be a reliable hearing loss model because of the robust pathological response to noise and drug insults. Furthermore, the pattern of threshold shift and hair cell (HC) loss in guineapig models resembles the impairment in humans. Yet nowadays the availability of abundant antibodies, probes and genetic mutants has made the mouse a primary model for auditory study. A mouse model of ototoxic drug-induced sensorineural hearing loss will help to unravel the molecular mechanism of cochlear pathologies and may consequently lead to prevention and intervention strategy for hearing loss.
Unfortunately, adult mice have shown little drug-induced ototoxicity under conditions that would produce severe auditory damage in the guineapigs or chinchillas. Gentamycin, the most common aminoglycoside, has been widely used in experimental studies of ototoxicity. However the optimal dosage of gentamycin for significant inner ear damage has not been established in mice. Lower doses of the drug given successively induce little HC loss while single injections at higher doses may be lethal. Currently, repeated systemic administration of aminoglycoside is applied to achieve HC damage. 2,3 But the need for multiple injections makes this procedure difficult to follow, especially with a large number of animals. Topical ototoxicity is another approach to auditory injury in mouse by transtympanic injection of aminoglycosides and placement of aminoglycoside-soaked gelfoam in the round window niche. 4,5 However, topical ototoxicity protocols are cumbersome, surgery-involved, and lesions induced in this way are unsteady and typically limited in the basal region of the cochlea. Comparably, a combination of a single dose of aminoglycoside and a single dose of loop diuretic seems much simpler and it has been reported to induce rapid HC loss in humans and experimental animals. 6–8 Recently, a single injection of high-dose kanamycin followed by a single injection of the loop diuretic, such as bumetanide or furosemide, was found to induce rapid cochlear lesions, including extensive HC loss and severe stria vascularis (SV) atrophy. 9,10 However the relationship between severe sensorineural hearing impairment and the function of the organ of Corti and SV has rarely been described. We therefore investigated in detail the effects of this combination insult on auditory function and morphology in an adult mouse, focusing on the functional response of cochlea to the ototoxic injury.
Materials and methods
Mouse husbandry and handling
Three- to four-week-old CBA/J male mice were supplied by the Model Animal Research Center of Nanjing University (Nanjing, China). Mice were housed in cages in groups of six, with wood shaving litter, under controlled conditions: 12 h:12 h lighting cycle, room temperature of 20–22°C, relative humidity of 45–55% and 20–22 air changes per hour. Standard mouse food and water were provided ad libitum. All mice were housed in a quiet colony (<30 dB SPL) and allowed to acclimatize to the animal facility for at least one week before the first test was performed. All experimental protocols were approved by the Animal Research Committee, Graduate School of Medicine, Huazhong University of Science and Technology. Animal care was under the supervision of the Institute of Laboratory Animals, Graduate School of Medicine, Huazhong University of Science and Technology.
Experimental animal and deafening procedure
A total of 70 CBA/J mice were employed in the study. Kanamycin (Sigma, St Louis, MO, USA) was dissolved in phosphate-buffered saline (PBS). Eight animals not receiving ototoxic drug insult served as normal controls. All the rest of the animals were treated with the combined drug insult that consisted of a single subcutaneous injection of kanamycin (1 mg/g body weight) and a single intraperitoneal injection of furosemide (0.4 mg/g; Tianjin Pharmaceutical Group, Tianjin, China) 30–45 min later. At various time points after treatment, auditory function and endocochlear potential (EP) were assessed, and the cochleas were obtained from both ears for morphological examination. Figure 1 represents the schematic of the experimental design.
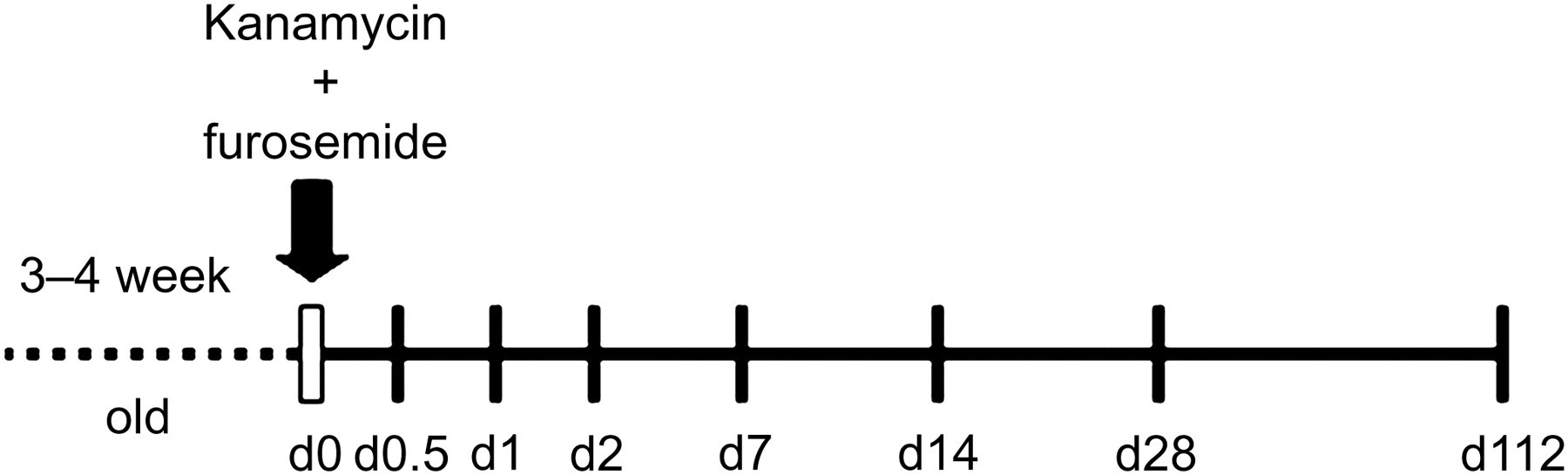
Time scheme for the protocol. Mice (3–4 weeks old) were injected with kanamycin and furosemide. The day of injection was defined as day 0 (d0). Mice were determined for auditory function and sacrificed for cochlear pathology on the indicated days. Control mice did not receive the drug insult and were analysed on d0. n = 8 in each time point
Evaluation of auditory function
Auditory threshold was determined by auditory brainstem response (ABR). Threshold was taken in control mice (untreated) and the drug-treated mice at 12 h (d0.5), 24 h (d1), two days (d2), seven days (d7), 14 days (d14), 28 days (d28) and 112 days (d112) after the co-administration. Mice were anaesthetized with an intraperitoneal injection of tribromoethanol (Shunqiang Company of Biology and Technology, Shanghai, China) at 300 mg/kg and kept warm with a heating pad. An active needle electrode was inserted subcutaneously at the vertex, in the midline of the scalp between the external auditory canals. A reference electrode was placed below the pinna of the left ear and a ground electrode was inserted contralaterally. Click and tone bursts of 2, 4 and 8 kHz (10 ms duration, 1 ms rise/fall time) were generated with the Nicolet Compass ABR System (Natus Medical, Inc, San Carlos, CA, USA). The average response to 1000 stimuli was obtained by reducing the sound intensity at 10 dB steps and finally at 5 dB intervals near the threshold. Threshold was defined as the lowest dB level of stimulus at which a positive waveform in the evoked response tracing was evident. All results of thresholds were verified at least twice.
Measurement of EP
Measurement of EP was performed following ABR evaluation when the animals remained anaesthetized. EP was measured in control mice (untreated) and the drug-treated mice at d0.5, d1, d2, d7, d14, d28 and d112. Body temperature was maintained at 37°C with heating pads and heat lamps. The animals were placed into a head holder. The auditory bulla was exposed via a ventral approach opening and the cochlea was visualized with a stereomicroscope. A small fenestra was made in the bony wall of the cochlea over the SV of the basal turn. A quartz-glass microelectrode was pulled to a tip diameter of 1–2 μm and filled with 150 mmol/L KCl. Then the microelectrode was placed just above the fenestra. An Ag–AgCl wire was immersed in the microelectrode and connected to a calibrated direct current amplifier. An Ag–AgCl indifferent electrode was placed in the neck musculature. Using a micromanipulator, the microelectrode was advanced into the scala media while the EP was recorded in realtime. The recording potential was zeroed when the microelectrode came into contact with fluid just laterally to the SV. The microelectrode was then advanced in a 50 μm step until a sudden positive voltage was measured at the point at which the electrode entered the scala media. Upon withdrawing the electrode from the scala media there was a rapid return of the measured potential to zero. After EP measurement, the animals were euthanized with an overdose of pentobarbiturate (Shunqiang Company of Biology and Technology) and the cochleas were obtained immediately for various morphological studies.
Propidium iodide and phalloidin staining
To investigate cochlear morphology with confocal microscopy, whole bodies of three mice in each group were fixed by cardiac perfusion with 4% paraformaldehyde after the animals were anaesthetized. Then cochleas were collected and placed in a fresh fixative solution for 12 h. The basilar membrane was dissected, rinsed in PBS and immersed in 0.25% Triton X-100 for 5 min and then placed in flourescein isothiocyanate (FITC)-conjugated phalloidin (Sigma; 1:200) in PBS for 30 min. After rinsing with PBS, the cochlear specimens were stained with propidium iodide (PI) (Sigma, 10 mg/mL) for 10 min, mounted on glass slides in 50% glycerol and coverslipped. Confocal microscopy was conducted using an Olympus V500 (Tokyo, Japan). FITC-conjugated phalloidin labels filamentous actin that is abundantly expressed in stereocilia. PI labels cell nuclei by binding to DNA.
Terminal deoxynucleotidyl transferase dUTP nick end labelling
The cochleas obtained directly at sacrifice were immediately fixed with 4% paraformaldehyde overnight at 4°C. The surface preparations of the organ of Corti were incubated in 0.5% Triton X-100 for 15 min at room temperature, and then washed twice with PBS. Subsequently, 100 μL of terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) reaction mixture (Roche Diagnostics Corporation, Indianapolis, IN, USA) containing dUTP-FITC and TdT was added on each surface preparation and incubated with the samples at 37°C in the dark for 60 min. The surface preparation was then washed and incubated with PI-staining solution (10 mg/mL PI in PBS) for 30 min at room temperature. Finally, the surface preparation was dissected into individual turn and mounted and examined with the laser confocal microscope.
Scanning electron microscopy
The cochleas were perfused with phosphate buffered (0.1 mmol/L, pH 7.4), 2.5% glutaraldehyde and immersed in the fixative for 2 h. The cochlea was rinsed with PBS, postfixed for 2 h with 1% phosphate-buffered OsO4, rinsed with PBS and dehydrated in a graded ethanol series up to 100%. Afterwards, the sample was critically point dried, mounted on aluminum stubs, painted with conductive graphite, sputter coated with gold and examined with a sanning electron microscope (Hitachi S-520; Hitachi, Tokyo, Japan).
Semi-thin section preparation and transmission electron microscopy
Cochlear tissue strips were immersed in the fixative overnight at 4°C and then immersed in 1.0% OsO4 for one hour at room temperature. After rinsing, the tissue strips were dehydrated in a graded series of ethanol solutions, stained en bloc with 2.0% uranyl acetate for 30 min and embedded in plastic. Sections approximately paralleled to the modiolus that included the whole height of the cochlea were taken at five to six successive levels to enable all turns of the cochlear spiral to be examined through the entire depth of the cochlea. At each level, an initial 1 μm thick semi-thin section was prepared and stained in toluidine blue for light microscopy. A series of thin sections (90 nm) were then cut and counterstained in uranyl acetate and lead citrate. The thin sections were examined and micrographs were taken with a transmission electron microscope (TEM) (Philips CM-120; Philips, Amsterdam, The Netherlands).
Assessment of SV
The thickness of SV was assessed with cochlear semi-thin sections. At each basal cochlear turn in each section in each cochlea, three images containing the entire thickness of the SV (from endolymphatic surface to interface with spiral ligament) were taken from the central portion of the strial width (along the axis of Reissner's membrane to spiral prominence). Using Photoshop v6, measurements of the thickness of the SV were obtained from each image.
Statistic analysis
Data of ABR, EP and SV thickness were presented as mean ± standard deviation. One-way analysis of variance with Tukey's correction was used for statistical analysis.
Results
In total, 64 out of 70 mice survived the long-term study. Body weights of the surviving animals continued to increase and were close to those of controls (data not shown). The mortality rate was near 9% and six animals died within 2 h after kanamycin injection, likely from acute renal failure (data not shown). This mortality rate was a little bit lower than that in previous report with the same protocols. 10
ABR thresholds
Figure 2 shows ABR thresholds determined in normal cochleas from control mice and in damaged cochleas from the mice treated with co-administraion of kanamycin and furosemide. This injection set resulted in a significant increase in ABR threshold shift beginning at 12 h at click and pure tone of 2, 4 and 8 kHz, and the increase continued during the first two days and then remained around 90 dB SPL thereafter.
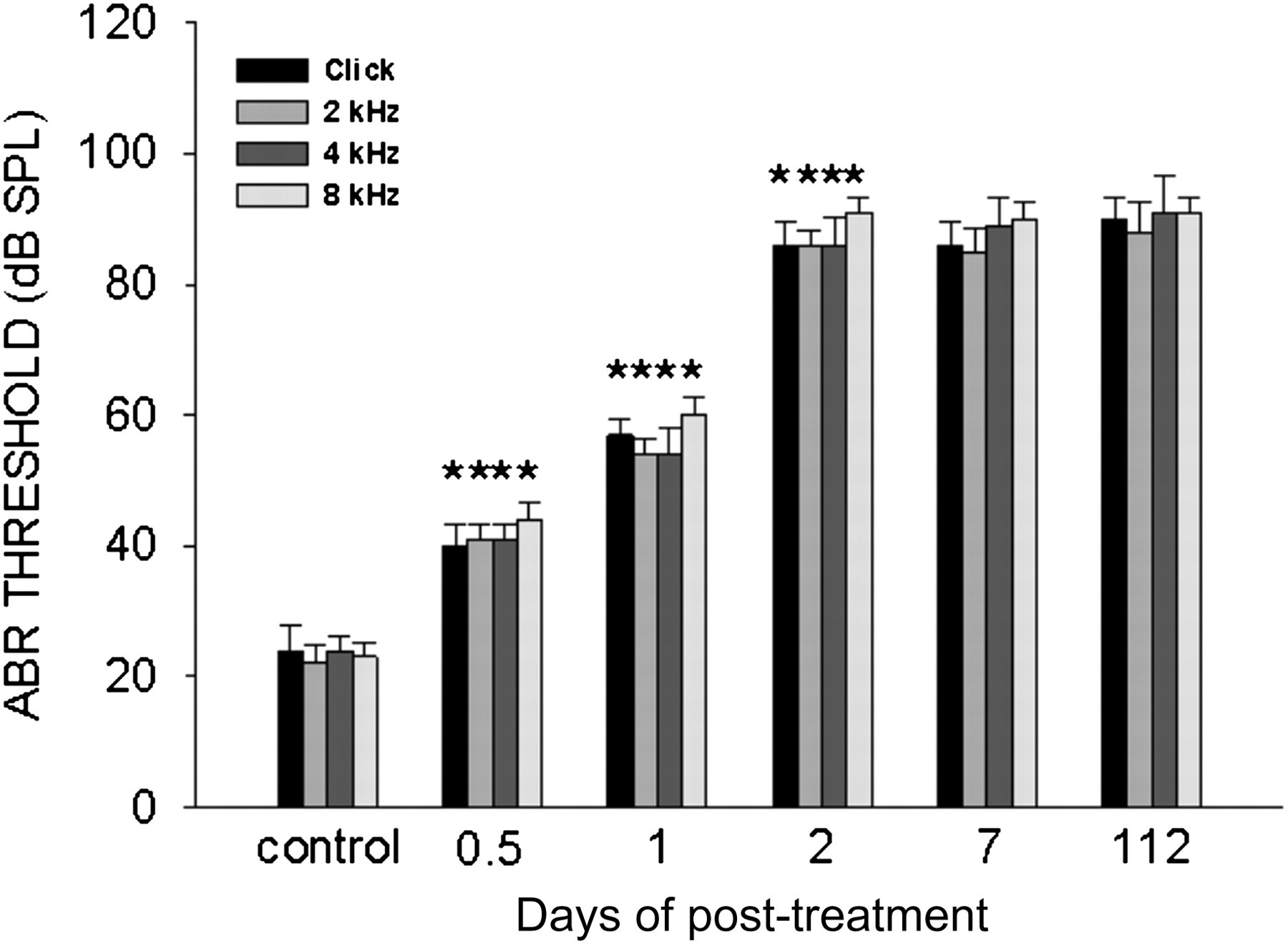
Change in threshold shift over time post-treatment. Threshold measured prior to the drug injection was defined as control. *P < 0.001 compared with the control. n = 8 at each time point
Deafness-related changes in the EP
Figure 3 illustrats EP recordings from four representative animals (Figures 3a–d) and summarized EP data from all animals (Figure 3e). Compared with untreated animals, EP in drug-treated animals significantly decreased at 12 and 24 h post-treatment. However since d2, a quick recovery of EP in treated animals was observed. The EP in these mice stayed at the levels similar to the one in controls throughout the rest of the 112-day period while the treated mice were deafened based on ABR threshold measurement.
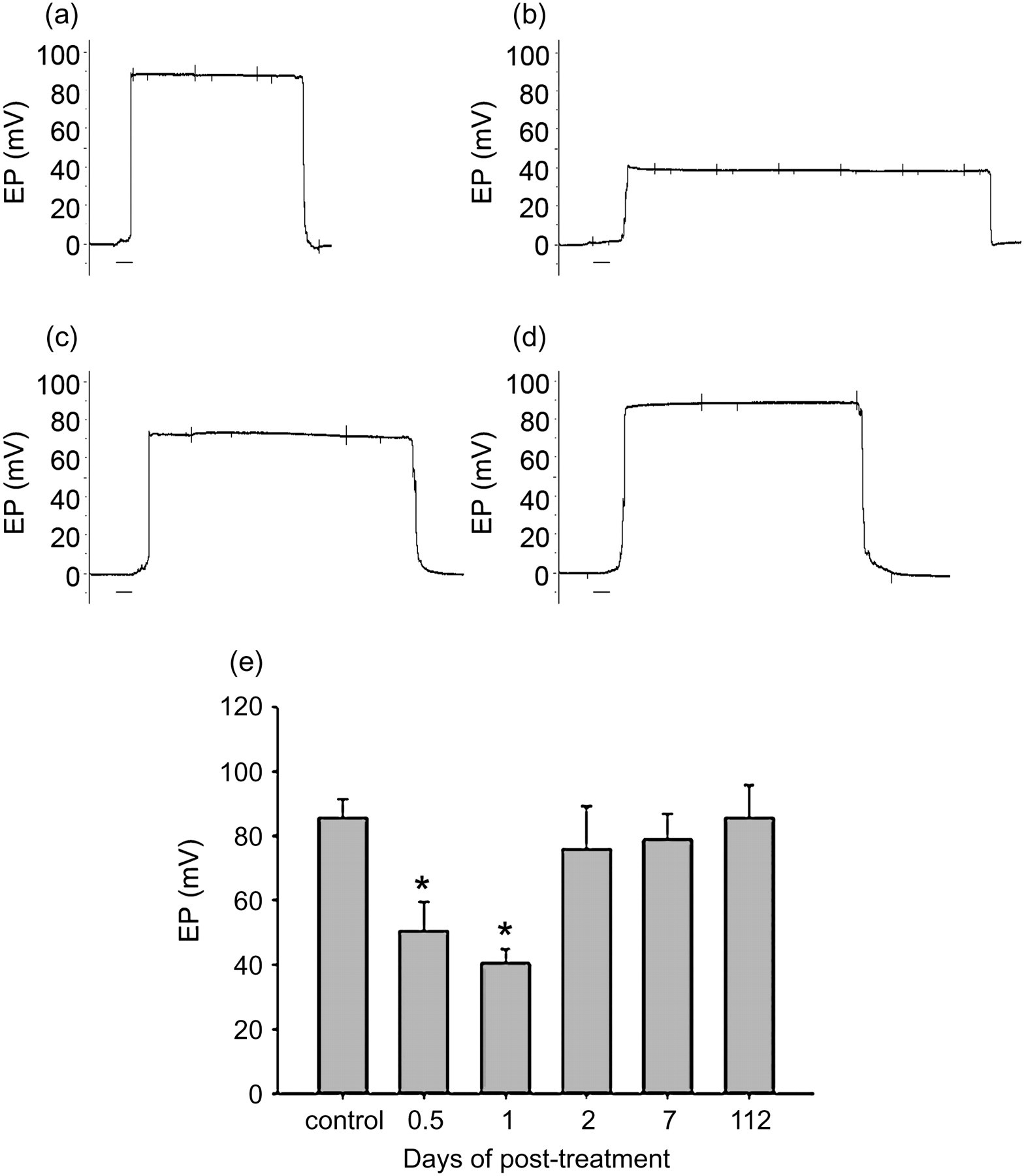
Change in endocochlear potential (EP) over time post-treatment. Control indicated EP detected from untreated mice. Panel a showed EP recordings from an untreated mouse. Panels b–d are EP recordings from three animals representing typical changes in EP after co-administration of drugs. (b) d1; (c) d2 and (d) d112. *P < 0.001 compared with the control; n = 8 at each time point. Scale bars: 5 s
Morphological studies
Our data of cochlear pathology were consistent with the functional deficits determined by the ABR measurements. Confocal microscopy of surface preparations of the organ of Corti displayed a well-defined outline of HCs both in basal turn and apical turn in untreated mice (Figures 4a and d). Shortly following the ototoxic co-administration, extensive HC lesions were observed. Outer hair cell (OHC) loss was first observed at the base of the cochlea at 12 h postinjection (Figure 4b). By 24 h complete loss of OHCs extended to nearly the entire basal turn (Figure 4c) and to the apex (Figures 4e and f). The most observed lesion at d2 was that all three rows of OHCs were destroyed throughout the length of the cochlea. In two treated animals, a few OHCs were seen in the extreme apex of the cochlea. The image data at d112 showed the presence of supporting cell scars in the OHC region. No spontaneous recovery or regeneration of OHCs was observed. In contrast, the pattern and extent of inner hair cell (IHC) loss was distinctly different from that of OHCs. Based on confocal microscopy, IHC loss was not detected until d7 and could be frequently observed at 14 or 28 days post-treatment. Generally IHC loss was much less severe than OHC loss, and some cochleas showed intact IHCs but absent OHCs at d7 and later (Figure 4h).
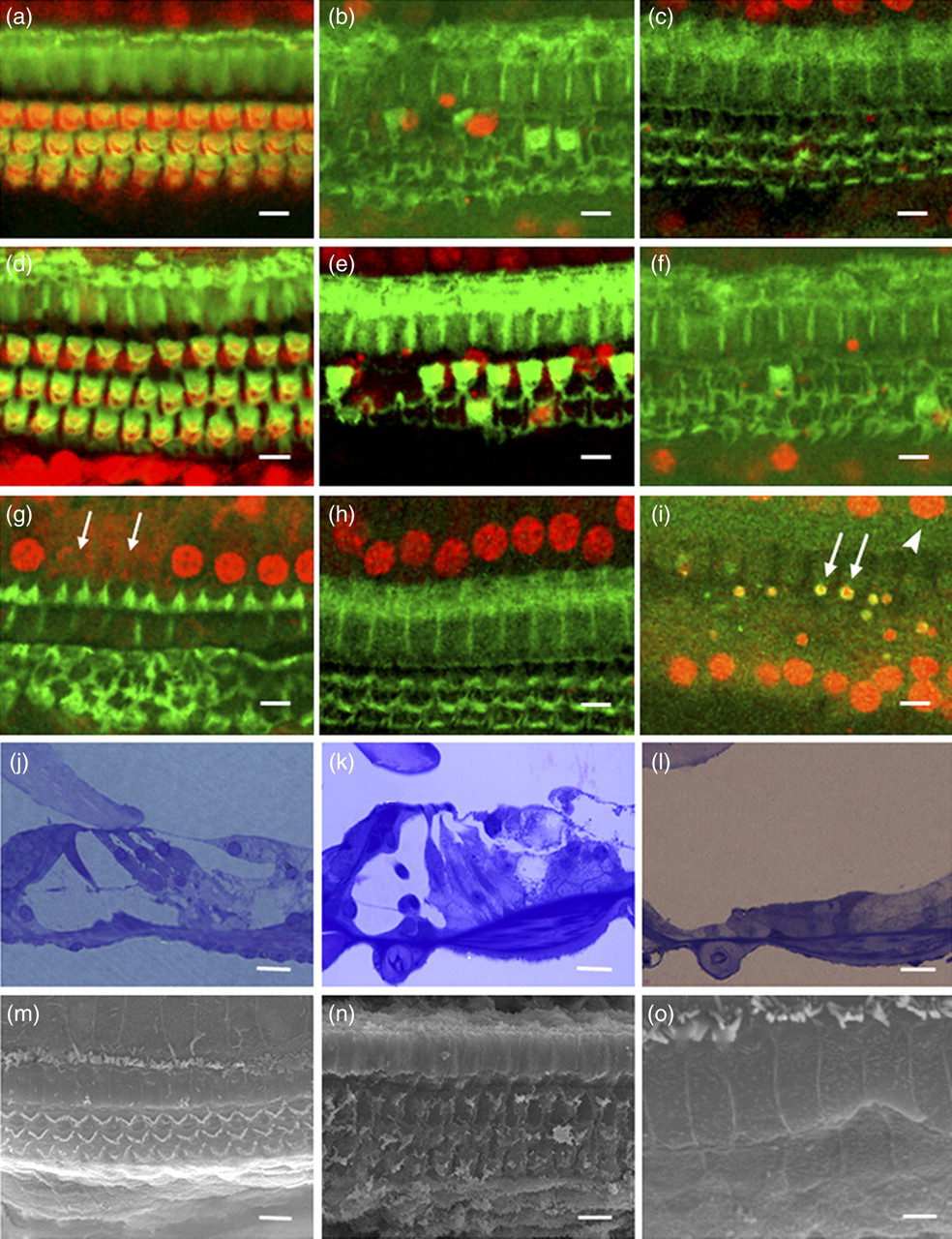
Progression of hair cell loss in the organ of Corti detected by confocal microscopy (a–i) and semi-thin section (j–l) and scanning electron microscopy microphotographs (m–o) of the organ of Corti. Flourescein isothiocyanate-phalloidin (green, in a–h) labelled actin assemblies at the reticular lamina and in hair cells. Nuclei were counterstained with propidium iodide (red, in a–i). (a) Basal turn from an untreated mouse; (b) basal turn at 12 h post-treatment; (c) basal turn at d1; (d) apical turn from an untreated mouse; (e) apical turn at 12 h; (f) apical turn at d2; (g) basal turn at d7. Arrows indicated absent IHCs. (h) Basal turn from one mouse at d28, showing undamaged inner hair cells (IHCs). (i) Terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) in the region of ongoing hair cell death in basal turn at d1 postinjury. Arrows indicated TUNEL-positive (green) nuclei of condensed outer hair cells (OHCs) and arrowhead pointed to an IHC. (j) Normal organ of Corti from an untreated mouse. (k) The organ of Corti in mice at d1, showing the loss of OHCs and remaining of the arch of Corti. (l) Collapsed organ of Corti in mice at d28. (m) Orderly three rows of OHCs and one row of IHCs in the basal turn in an untreated mouse. (n) OHC damage in the basal turn at d1. (o) OHCs were replaced by expansion of heads of the supporting cells in basal turn at d28. Scale bars: 10 μm (a–n), 4 μm (o). (For colour images, refer to the online version)
TUNEL labelling showed apoptosis located in the region of OHC loss at both d0.5 and d1. In all TUNEL-positive cells, the nuclei were pyknotic, with condensed chromatin (Figure 4i). There were no TUNEL-positive nuclei in the cochleas of untreated mice.
In agreement with the FITC-phalloidin and PI staining, semi-thin section and scanning electron microscopic (SEM) examination showed typical HC death over the post-treatment period. Light microscopy of control mice showed an intact structure of organ of Corti including one row of IHCs, three rows of OHCs and other supporting cells (Figure 4j). At 24 h post-treatment the OHCs disappeared although the arch of Corti remained (Figure 4k). By d28 the organ of Corti collapsed completely and supporting cells spread into the area where HC typically existed (Figure 4l). SEM of the organ of Corti in control mice showed three orderly rows of OHCs and one row of IHCs (Figure 4m). At 24 h after drug injection, extensive OHC death was detected and reticular lamina collapsed but remained intact (Figure 4n). By d28, all OHCs were replaced by expansion of heads of the supporting cells (Figure 4o). Contrastingly IHCs were present in all treated cochleas although a mild to moderate IHC loss was seen in some cochleas.
The impact of the drug insult on SV was determined with SEM. In untreated mice marginal cells (MCs) of the SV facing the endolymph were hexagonal-shaped and covered by numerous microvilli which form well-defined bands at cell–cell junctions (Figure 5a). At d2 MCs were swollen and plenty of microvilli were lost (Figure 5b). By d14 almost all microvilli disappeared and MCs presented a stone-like appearance (Figure 5c). Compared with the controls, TEM images showed a significant decrease in SV thickness in mice that lost HCs since d7 (Figures 5d and e). The major cause of decrease in SV thickness appeared to be a reduction in MC size (Figure 5e). Additionally the extent of strial atrophy was highly associated with the duration of deafness from d7 to d112 (Figure 6).
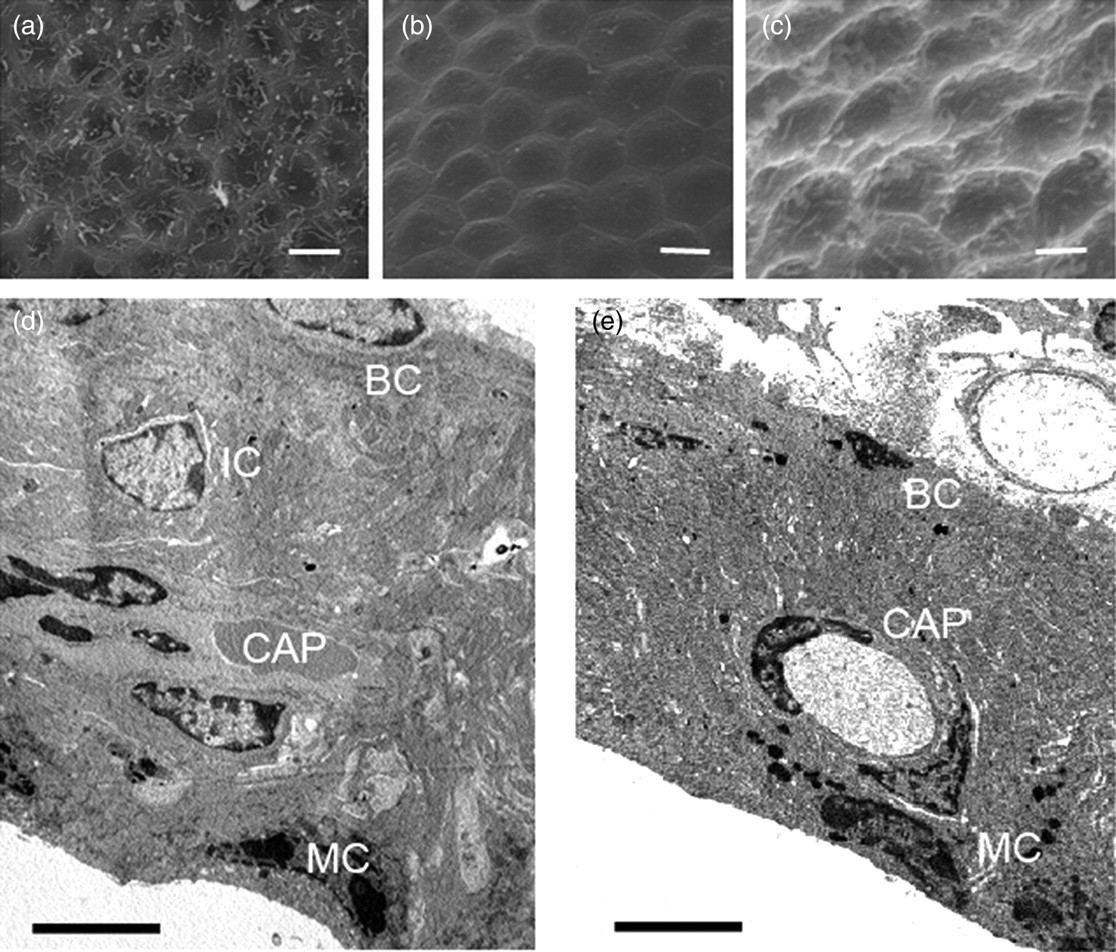
Scanning electron microscope (a–c) and transmission electron microscope (d and e) of the stria vascularis. (a and d) Normal stria vascularis from an untreated mouse. Marginal cells were hexagonally shaped and covered by numerous microvilli. (b) The stria vascularis at d2. Marginal cells were swollen and most of mircovilli were lost. (c) The stria vascularis at d14, showing stone-like appearance of marginal cells without microvillus coverage. (e) The stria vascularis in a mouse at d112, showing a significant decrease in strial thickness. BC: basal cell; CAP: capillary; IC: intermediate cell; MC: marginal cell. Scale bars: 10 μm (a and b), 5 μm (c–e)
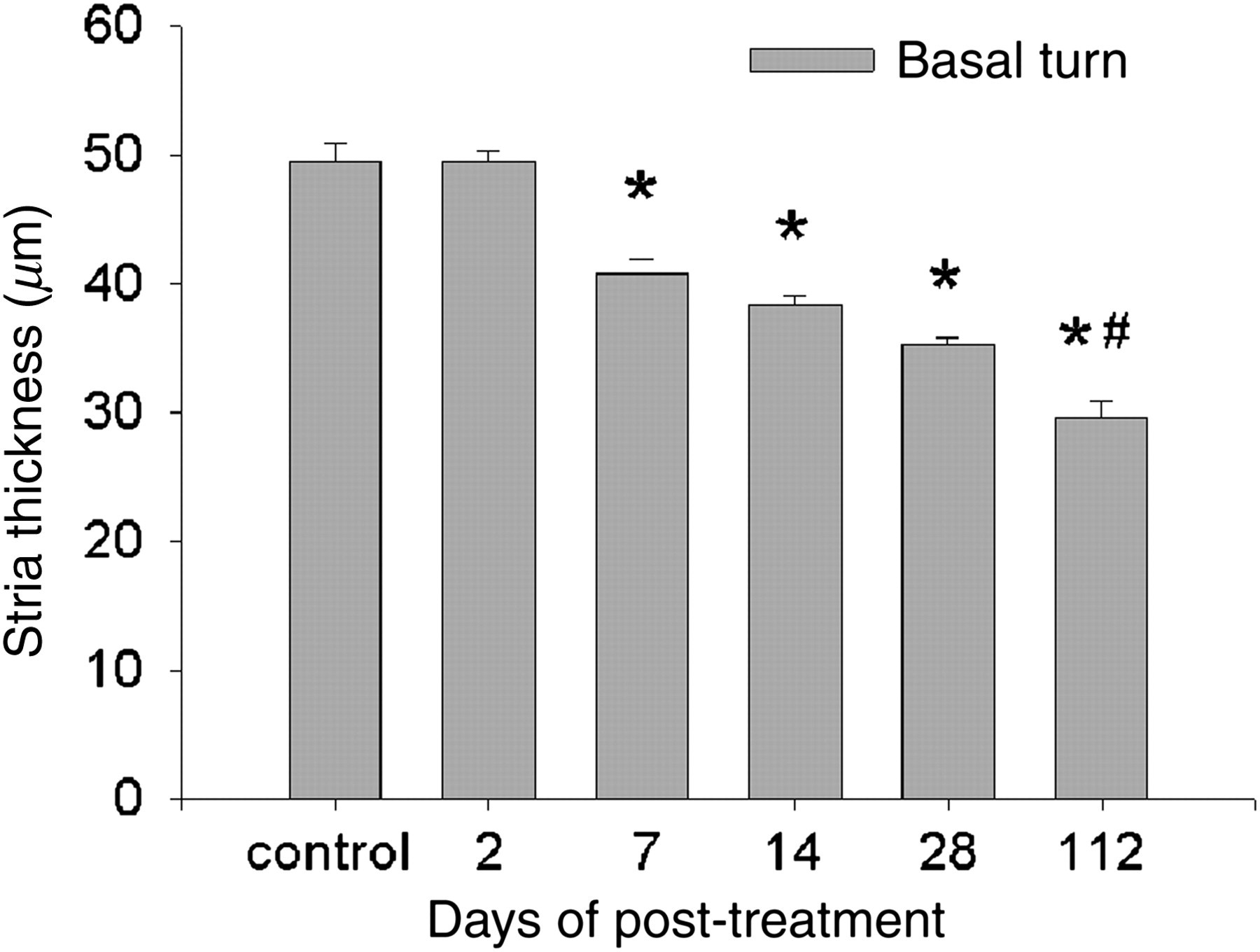
Change in strial thickness over time post-treatment. Control indicates data from untreated mice. *P < 0.001 compared with the control; # P < 0.001 compared with d28. n = 5 at each time point
Discussion
In the present study, we successfully reproduced an ototoxic model in adult mice with the protocol of kanamycin–furosemide co-administration. 10 Our functional and morphological data confirmed profound hearing loss and severe HC damage in the model animals. Additionally we detected for the first time EP alteration in response to the sensorineural hearing loss in mice and analysed the relation between functional and morphological changes in the cochlear lateral wall.
Compared with untreated animals, the drug-treated mice showed significant elevation of ABR threshold shifts responding to both broadband and frequency stimulus and reversible EP decline. Accompanying the functional impairment, there was extensive HC damage and remarkable change in the SV.
The damage of OHCs in adult mouse cochlea occurred as early as 12–48 h after the drug injection. The defined pattern of OHC death is identical to that occurring in other situations such as acquired hearing loss and genetic defect-related hearing loss. The presence of PI- and TUNEL-positive nuclei in the organ of Corti confirmed that OHCs were dying via programmed cell death, which resembled OHC death initiated by noise trauma. 11
In contrast, IHC loss was delayed and less extensive than OHC loss in our study and this agrees with previous reports in which the prolonged survival of IHCs after extensive OHC loss was also evident in mouse models of chronic kanamycin treatment-induced deafness and mice with certain genetic defect-related hearing loss. 12 Moreover, the phenomenon was observed when the animals were injured with either amikacin or gentamicin instead of kanamycin. The observations from us and other researchers suggest a significant difference in the sensitivity to injury between IHCs and OHCs in mice and IHCs may have inherently stronger resistance to damage than OHCs. With the models used in these studies, further in vivo study would help to elucidate the mechanism underlying different survival between IHCs and OHCs in the mature organ of Corti.
There is limitation of auditory function measurement in our study. While the frequency range of mice is 1–70 or 90 kHz, our frequency-ABR measurement was from 2 to 8 kHz, the range generated by vibration in the apical area of the basilar membrane. Our observation and published studies showed that the cochlear basilar membrane damage from ototoxicity starts in the basal area and progresses to the apical area. The damage is most severe at the basal area and less severe at the apical area. Therefore it is reasonable to assume that measurement of auditory threshold in higher frequencies would generate a larger shift.
It is notable that relatively mild IHC loss induced dramatic ABR threshold shift. At least three factors may account for the phenomenon. Firstly, it is generally assumed that OHCs act as a cochlear amplifier and modulate IHC function. It has been reported in an animal model that the sensitivity of auditory neuron reduces by at least 40 dB when OHCs are destroyed entirely but IHCs remain intact, 13 indicating OHCs and IHCs may interact with each other and are both involved in the primary signal processing of coding intensity of sound stimulus. Secondly, it is also possible that, although the EP recovers to normal levels, the ionic composition or potassium concentration in the endolymph remains altered and the mechanotransduction process is affected. Last but not least, ABR tests integrity of the auditory nerve and its ability to properly transmit a sound signal along the whole pathway to brainstem. It may be used to evaluate hearing indirectly, but is not the real subjective hearing threshold. Therefore, an ABR threshold shift induced by kanamycin–furosemide administration may only provide a general evaluation of hearing loss.
The ototoxic and nephrotoxic effects of aminoglycoside antibiotics have long been known in clinical practice. 14 Furosemide, bumetanide and other loop diuretics are also ototoxic clinically, but hearing loss induced by these drugs is mostly reversible. The predominant ototoxic effects of furosemide on cochlear function are believed to result from its interference with SV function. 15 Furosemide, and other loop diuretics such as bumetanide, causes an acute enlargement of the intrastrial space that is associated with a decline in EP. 16 Bumetanide inhibits the Na+-K+-2Cl− co-transporter, which is abundantly expressed in the basolateral membrane of the MC and is crucial for EP generation. 17 These diuretics are also well-known potentiators of aminoglycoside effects. The combined administration of aminoglycoside with loop diuretic leads to a rapid and profound hearing loss in humans and animals. 18 Taylor et al. 9 proposed that a loop diuretic induces an EP reduction from positive values at normal to negative and this could facilitate entry of the positively charged aminoglycosides into the endolymph, whereas aminoglycoside levels in perilymphatic fluids are not increased. Then aminoglycosides become trapped in the HCs where they accumulate and induce cell death. 19 The other viewpoint was that diuretic causes temporary disruption of blood flow in SV and consequent ischaemic–reperfusion injury, which leads to HC damage by free radicals and reactive oxygen species in the endolymph. 20 The mechanisms underlying the synergistic effects of aminoglycosides and loop diuretics on HCs remain to be fully elucidated.
Our study showed the kanamycin–furosemide treatment protocol caused not only extensive HC loss but also a conspicuous response both functionally and morphologically in the SV. Regardless of a significant decline at 12 h after deafening, the EP returned to normal levels soon in the animals with a long-term sensorineural hearing loss. The SV thickness decreased in the deafened mice and the reduction seemed proportional to the duration of deafness. The MC appeared to be the major target of the injury. The reduction in the volume of MCs was similar to the characteristics of strial pathology related to ageing, noise trauma and chronic aminoglycoside treatment. 21,22 The similarities of the SV response to damage paradigms suggest that there are common pathological events at the tissue and cellular levels, although the initiators of the injurious events differ from study to study. Hellier et al. reported that in guineapigs although strial volume was significantly reduced, the EP was maintained at near normal levels for long term after severe HC loss. 23 Likewise, the EP was not affected significantly even after several weeks of progressive strial atrophy that followed chronic, repeated aminoglycoside treatment, noise trauma and with ageing. 21,24 It may therefore be that the strial response may represent a damaging event possibly unrelated to ototoxic effects on the organ of Corti, but secondary to a reduced metabolic burden following the loss of the organ of Corti and the resultant reduction in K+ leakage from the endolymph. This seems to be supported by a recent study of noise-induced hearing loss. 21
While the EP was maintained at normal levels in long-term deafened animals, it was significantly reduced at 12–24 h following the deafening procedure. This is consistent with a previous study in which the EP recovered to normal levels in less than 72 h after the co-administration of an aminoglycoside with a loop diuretic. 25 These short-term changes in the EP presumably result from the effect of loop diuretic on the SV, although as discussed above, such changes may also be related to temporary K+ leakage associated with HC loss. Loop diuretics act at the level of the Na-K-Cl co-transporter in the SV, leading to a rapid fall in the EP followed by a rapid recovery when diuretics were eliminated in the body within about two days.
In conclusion, the present study successfully reproduced aminoglycoside-induced hearing impairment in mature mice. The co-administration of kanamycin and furosemide induced significant hearing damage and severe cochlear lesions in the treated animals. Furthermore our observation of changes in the SV suggested that SV damage plays a minor role in either temporary or permanent ototoxic hearing loss and atrophic SV is capable of maintaining a normal EP under conditions of severe loss of HCs.
Footnotes
ACKNOWLEDGEMENT
This work was supported by a grant from the National Natural Science Foundation of China (No. 30672307).
