Abstract
In order to develop tissue engineering applications for oral and maxillofacial surgery, the surgical anatomy of the miniature pig's face was investigated in three miniature pig cadavers and three anaesthetized miniature pigs using identical procedures that were previously described for humans. A preauricular incision with a retromandibular and a submandibular extension was initially made through the facial skin and subcutaneous tissues. The underlying tissues were then carefully dissected in order to progressively expose the platysma muscle, the superficial layer of deep cervical fascia, the marginal mandibular branch and buccal branch of the facial nerve, the mental nerve and the mandibular skeleton. The marginal mandibular branch of the facial nerve has an upper and lower division. Stimulation of the facial nerve and its branches showed that the upper division of the marginal mandibular branch innervates muscles and tissues in the upper lip and nose region, and the lower division innervates muscles and tissues in the lower lip region. The gross anatomy of the maxillofacial region in the pigs was found to be similar to that of humans. Although the distributions of the marginal mandibular branch of the facial nerve and the mental nerve are different from that of humans, we concluded that miniature pigs are a suitable experimental model for the preclinical development of tissue engineering applications in oral and maxillofacial surgery.
Keywords
Animal models offer an unprecedented opportunity to preclinically evaluate the efficacy and safety of newly developed human therapies. 1 The development of tissue transplantation methods in animal-based tissue engineering research requires the same surgical methods for tissue transplantation in humans. Although a few reports on the anatomy of porcine face and facial skeleton have been published, 1,2 no descriptions of the surgical and sectional anatomy of the porcine face and facial skeleton have yet been published. Without such descriptions, it is therefore difficult to initiate a new research project in the field of experimental oral and maxillofacial surgery using pigs. In order to develop tissue engineering applications in oral and maxillofacial surgery, the surgical anatomy of the porcine face was investigated using identical procedures that were previously described for humans 3 in miniature pig cadavers and anaesthetized miniature pigs.
Materials and methods
All animal care and handling procedures were performed in accordance with the ‘Principles of Laboratory Animal Care’ of the Tokyo Women's Medical University Animal Experimentation Committee and the Japanese ‘Act on Welfare and Management of Animals’ (Act No. 105, Japan, 1973).
Surgical anatomy of the face in miniature pig cadavers
The cadavers of three healthy 6–10-month-old male Clawn miniature pigs, which weighed between 13 and 20 kg, were used for this study. The miniature pigs were bred in the Japan Farm Clawn Institute, and were vaccinated prophylactically against and veterinary checked for any zoonoses prior to their use. The three pigs were housed and cared for in individual cages (W: 95 cm × L: 95 cm × H: 120 cm) (Natsume Seisakusho, Tokyo, Japan) whose flooring was made of acrylic resin-coated wire netting. The room in which they were housed was kept at 22–24°C and 45–55% relative humidity on a 12 h light (08:00–20:00 h):dark cycle. The pigs were fed daily with 300–400 g of pig chow (Miniature swine diet M-16, CLEA Japan, Tokyo, Japan), had free access to fresh water and their cages were cleaned daily every morning. Prior to their use in this study, the three pigs were used in a cardiology research project, and were killed at the end of this project by an intravenous injection of potassium chloride under deep sevoflurane-induced anaesthesia. After confirmation of death, the cadavers were frozen at −15°C until required for this investigation.
When required, the cadavers were removed from the freezer, and kept overnight at room temperature. The surgical procedure for approaching the mandible was identical to that described by Ellis and Zide for maxillofacial surgery.
3
Specifically, the incision line is a preauricular incision with a retromandibular and a submandibular extension. In human maxillofacial surgery, this incision is called the modified Blair incision, and made for the removal of parotid tumours.
3
Briefly, the incision line, which starts in the preauricular region approximately 1.5 cm posterior to the manibular ramus on palpation in the retromandibular region and extends in a loose S shape 1.5 cm below the mandibular body on palpation in the submandibular region, was marked on the skin (Figure 1A). After making the incision through the skin and subcutaneous tissue, the skin flap was then dissected and elevated using scissors and a scalpel. The mandible was then excised using the identical method that was previously described by Sasaki
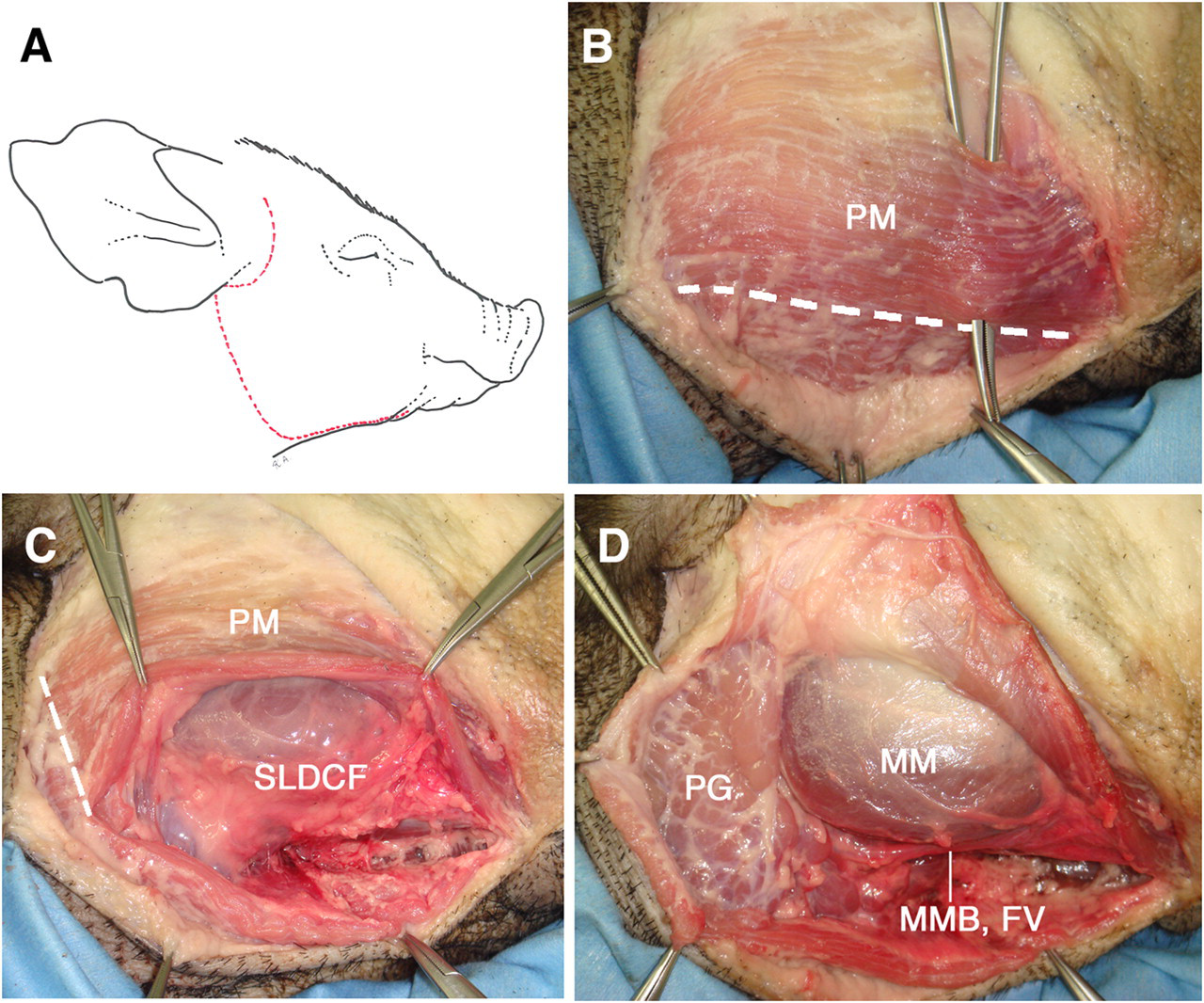
(A) The red dotted line describes the preauricular incision with a retromandibular and a submandibular extension on the miniature pig's face. (B) The platysma muscle (PM) was exposed by undermining of the skin and subcutaneous tissues. (C) The superficial layer of deep cervical fascia (SLDCF) was exposed by dissecting the PM along the dotted line. (D) After dissection of the SLDCF, the parotid gland (PG), the masseter muscle (MM), the marginal mandibular branch of the facial nerve (MMB) and the facial blood vessels (FV) can be observed in the SLDCF
Determination of the facial muscles and tissues that were innervated by the marginal mandibular branch and buccal branch of the facial nerve
In order to establish which facial muscles and tissues were innervated by the marginal mandibular branch and the buccal branch of the facial nerve, the two branches of the facial nerve were isolated and then stimulated using a nerve stimulator (Vari-stim® III, Medtronic, Jacksonville, FL, USA) in three anaesthetized Clawn miniature pigs. Their housing and husbandry were identical to that described above. Briefly, each pig was first premedicated by a combined intramuscular injection of medetomidine (80 µg/kg), ketamine (5 mg/kg) and butorphanol (0.2 mg/kg), and then anaesthetized by an intravenous injection of 1% propofol (induction: 20–30 mg), and maintained by repeated intravenous injections of 10 mg propofol that were given every 10 min. A retromandibular incision with a submandibular extension was then made on the swine face in order to expose the buccal and marginal mandibular branches of the facial nerve. Each exposed nerve was then electrically stimulated separately at 0.5–2 mA using the nerve stimulator. The facial muscles and tissues that were innervated by each nerve were determined by observing which muscles contracted during the stimulation. After this study, the pigs were used in a research project of facial nerve regeneration.
Results
Surgical anatomy of the miniature swine face
The platysma muscle was exposed by undermining the skin and subcutaneous tissue. The platysma muscle runs parallel to the mandible and covers the orbital rim (Figure 1B). The platysma muscle was then dissected in order to expose the superficial layer of deep cervical fascia (SLDCF) (Figure 1C).
The marginal mandibular branch of the facial nerve, and the facial artery and vein run in the SLDCF (Figure 1D). The marginal mandibular branch of the facial nerve runs along the ventral edge of the mandibular ramus between the medial pterygoid muscle and the submandibular gland, and parallel to the facial artery and vein on the medial margin of the masseter muscle (Figures 2A and B). The marginal mandibular branch of the facial nerve has an upper and lower division (Figure 3A). Nerve stimulation showed that the marginal mandibular branch innervates muscles and tissues in the upper and lower lip, and nose. Specifically, the upper division of the marginal mandibular branch innervates muscles and tissues in the upper lip and nose region, and the lower division innervates muscles and tissues in the lower lip region (Figures 3A and 4). The buccal branch of the facial nerve runs parallel to the zygoma approximately 0.5–1 cm below the zygomatic arch on the masseter muscle (Figures 2A and B). Nerve stimulation showed that the buccal branch also innervates muscles and tissues in the upper lip and nose region (Figures 3B and 4).
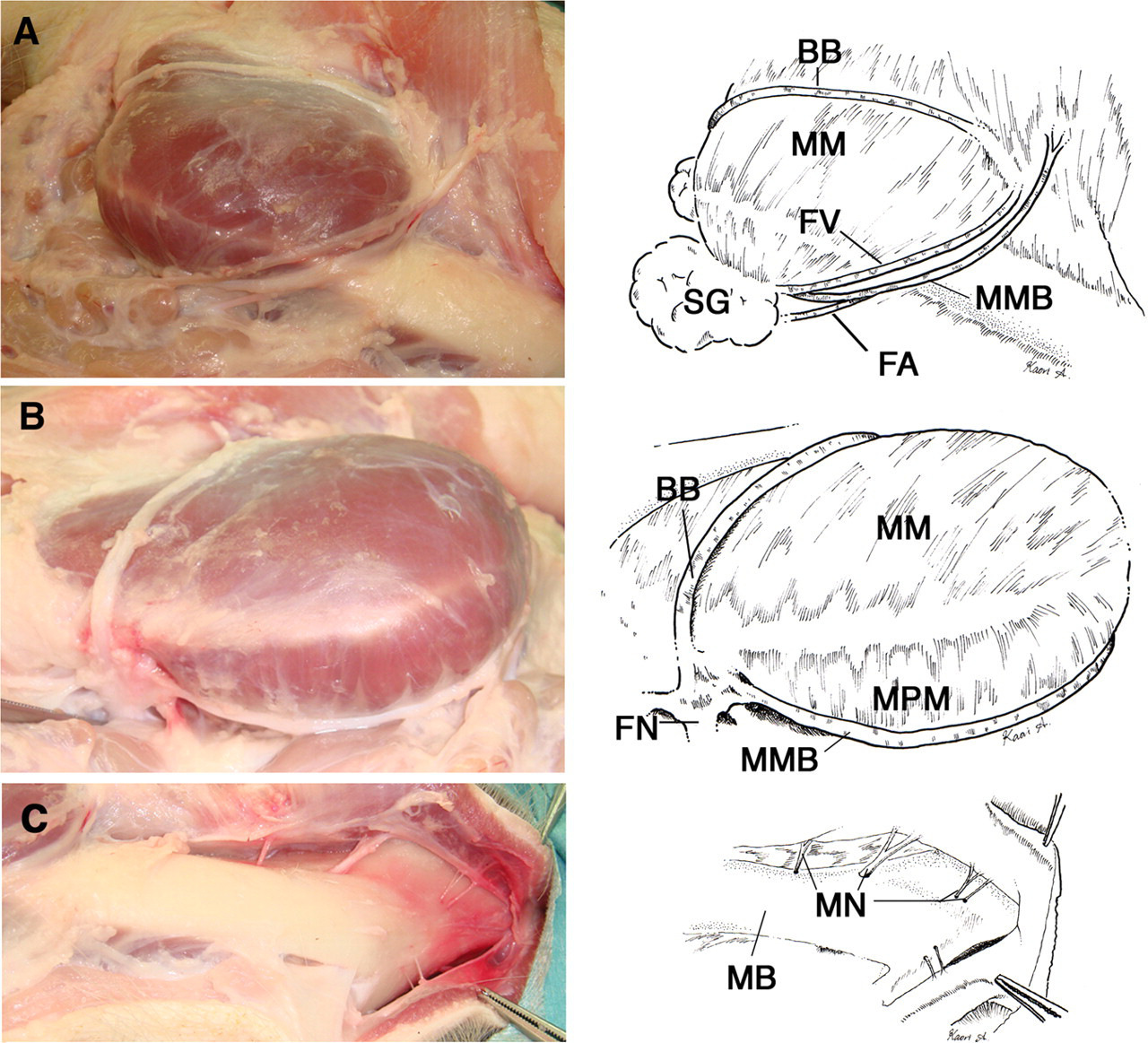
(A) Facial nerve (FN) and vessels of the miniature pig. The marginal mandibular branch (MMB) of the FN, and the facial artery (FA) and vein (FV) run along the medial margin of the masseter muscle (MM). The buccal branch (BB) of the FN runs along the superficial surface of the MM. SG: submandibular gland. (B) Retromandibular view. The MMB of the FN runs along the medial pterygoid muscle (MPM). The BB runs on the MM. The trunk of the FN divides into the MMB and BB branches. (C) The exposed mandibular bone (MB) of the miniature pig. The mental nerves (MN) pass through the mental foramina to innervate the lower lip region
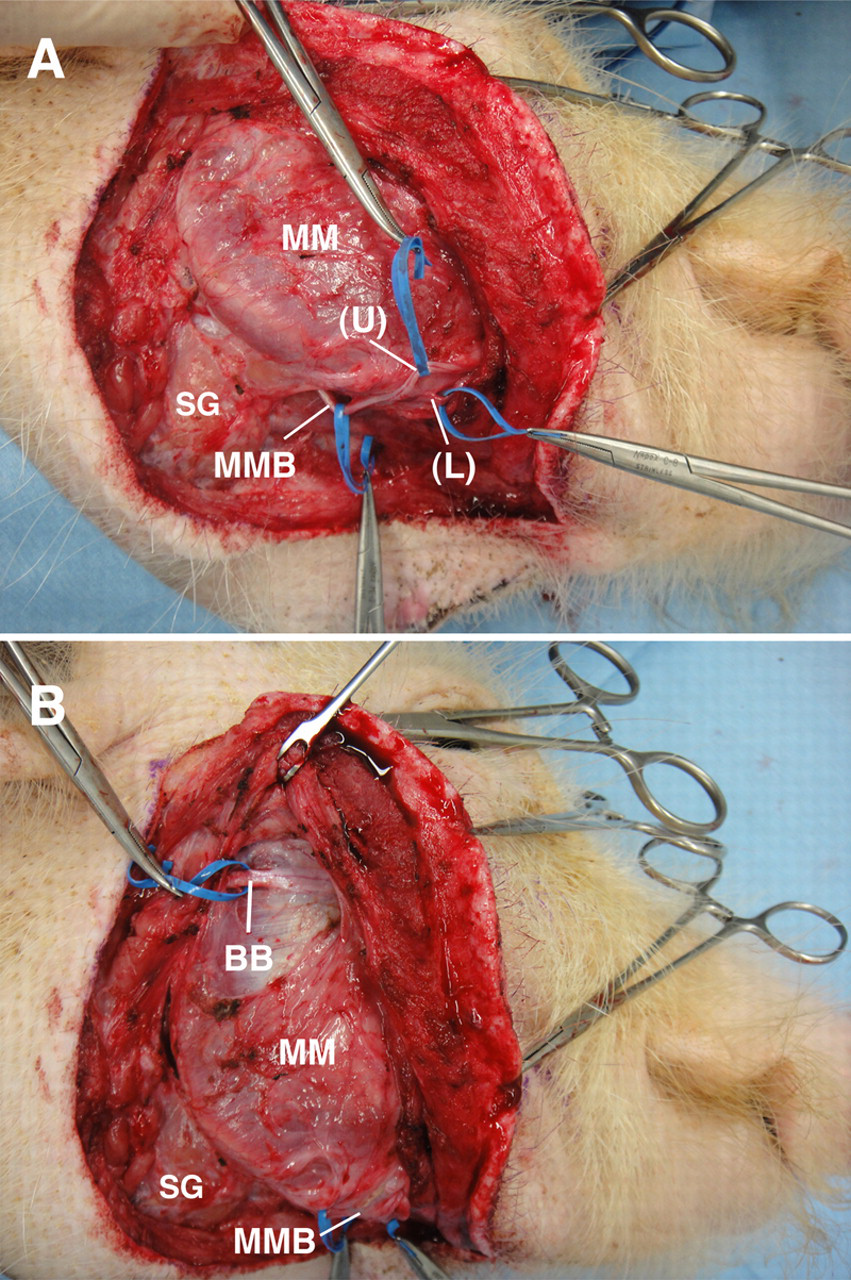
Facial anatomy of a 19 kg anaesthetized 13-month-old male Clawn miniature pig. (A) The marginal mandibular branch (MMB) of the facial nerve has an upper (U) and lower (L) division. The MMB and its upper and the lower divisions were retracted using blue vessel loops. Nerve stimulation showed that the mandibular branch innervates muscles and tissues in the upper and lower lip, and nose. The upper division of MMB innervates muscles and tissues in the upper lip and nose region, and the lower division innervates muscles and tissues in the lower lip region. (B) The buccal branch (BB) of the facial nerve runs parallel to the zygoma approximately 0.5 cm below the zygomatic arch on the masseter muscle (MM). The BB was retracted with a blue vessel loop. Nerve stimulation showed that the BB innervates muscles and tissues in the upper lip and nose region. SG: submandibular gland
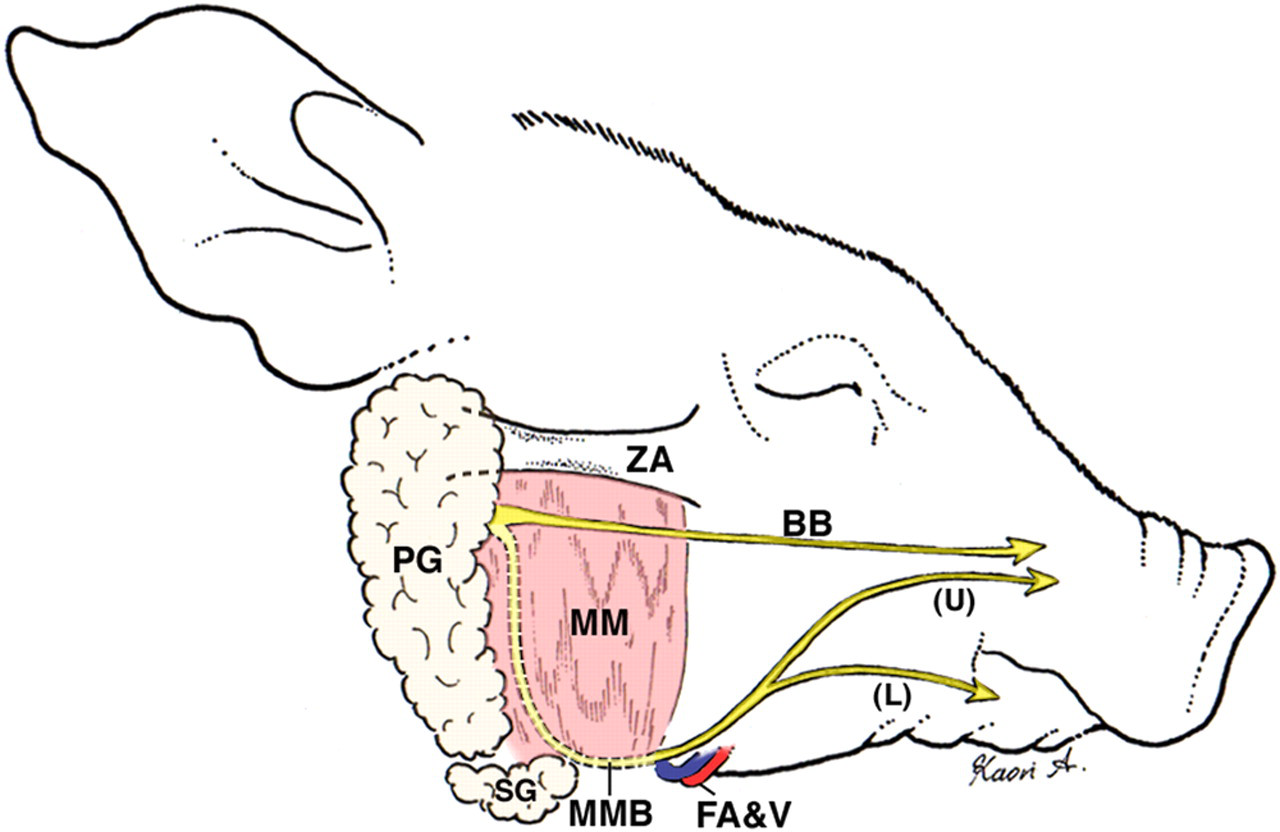
Schematic diagram of the surgical anatomy of the miniature pig's face, and the distribution of the buccal and marginal mandibular branches of the facial nerve. PG: parotid gland; SG: submandibular gland; MM: masseter muscle; ZA: zygomatic arch; BB: buccal branch of the facial nerve; MMB: marginal mandibular branch of the facial nerve; (U): upper division of the MMB; (L): lower division of the MMB; FA&V: facial artery and vein
In order to expose the mental neurovascular bundle, the periosteum of the mandible at the site between the medial pterygoid and the masseter muscle was incised along with the lower margin of the mandible (Figure 2A), and then lifted and removed using an elevatorium. The mandibular bone was then exposed by elevating the body toward the mental region in order to expose the mental neurovascular bundle. The periosteum around the mental nerve was cut using scissors in order to expose the mental nerve. The pig has four mental foramina in the lateral mental region because the mental nerve comprises four distinct nerves (Figure 2C).
Mandibular anatomy of the miniature pig
The mandibular dental arch is long and narrow, and the condyle has only one head and no condylar process. The mandibular foramen is located in the central part between the inferior and posterior ramus on the occlusal plane. An impacted molar tooth was observed distal to the erupted molar teeth (Figure 5).
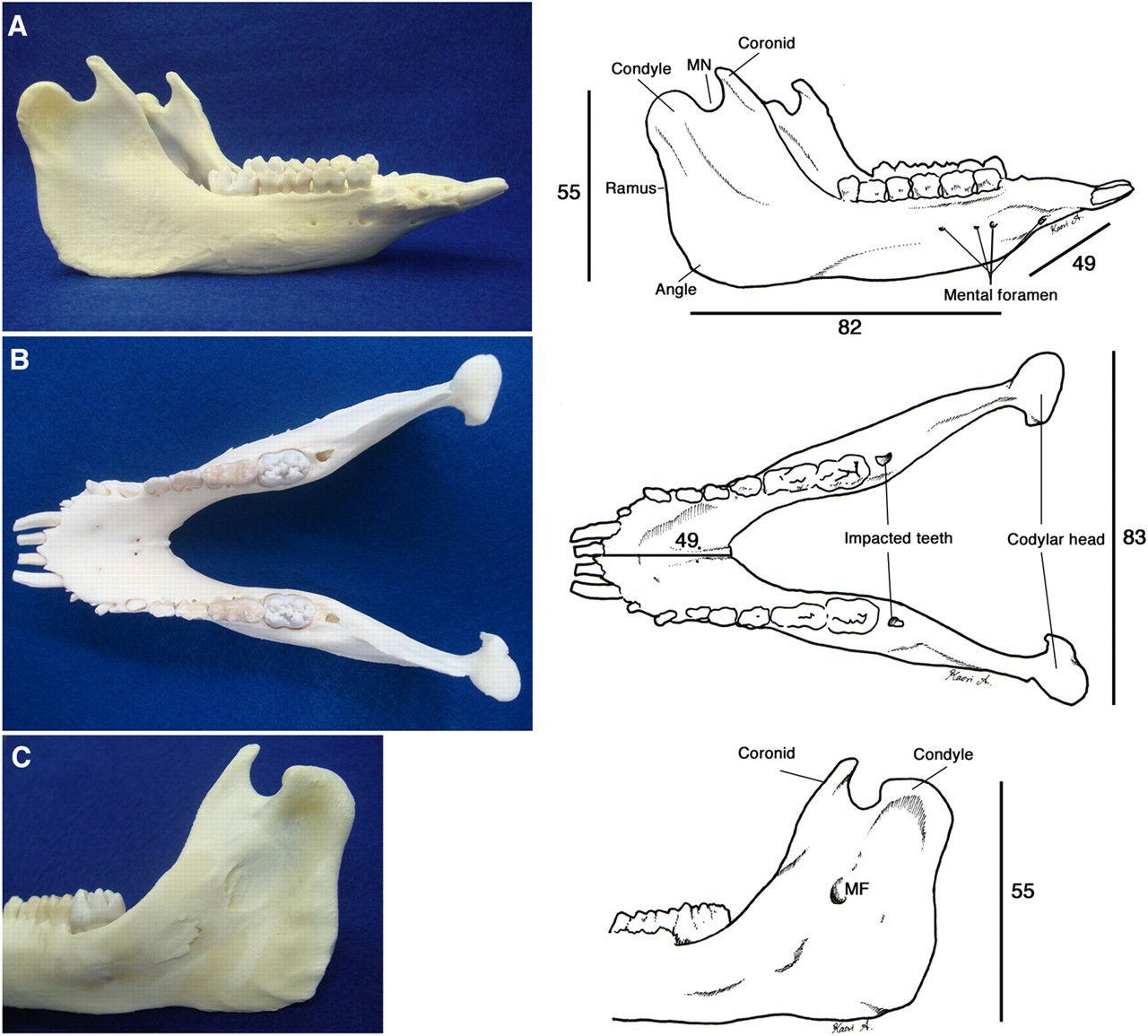
The mandibular bone of a 13 kg six-month-old male Clawn miniature pig. (A) lateral view. MN: mandibular notch. (B) axial view. Impacted molars are observed. (C) The inferior surface of the mandibular ramus. The mandibular foramen (MF) is located on the central part between the inferior and posterior ramus on the occlusal plane extension. Scale bars are in mm
Discussion
In order to develop tissue engineering applications for oral and maxillofacial surgery, the surgical anatomy of the miniature pig's face was investigated in miniature pig cadavers and anaesthetized miniature pigs using identical procedures that were previously described for humans. 3 Compared with the human platysma muscle, which is thin and runs vertical to the mandible and covers the mandibular margin, 3 the pig platysma muscle is more extensive and thicker, and runs parallel to the mandible and covers the orbital rim. The SLDCF in the pig is similar to that of humans in that it is located below the platysma muscle and that the marginal mandibular branch of the facial nerve and the facial artery and vein run through it.
We found that the marginal mandibular branch of the facial nerve has an upper and lower division in miniature pigs. In humans, the marginal mandibular branch innervates only the lower lip region. 3 In rats, the marginal mandibular branch also has upper and lower branches that innervate the upper and lower lips, respectively. 5 The facial nerve of the guineapig has six main peripheral branches: anterior auricular, cervical, inferior labial (IL), posterior auricular, superior labial (SL) and zygomatico-orbital. The IL connects to the SL in the maxillary region, and innervates the upper and lower lip region. 2
In humans, a single mental foramen is present on the lateral surface of the mandible and inferior to the interproximal region of the first and second mandibular premolar teeth, and accessory mental foramina are rarely found. 6 The pig has four mental foramina in the lateral mental region because the mental nerve comprises four distinct nerves.
The mandibular dental arch of the pig is longer and narrower than that of humans, and the condyle has only one head and no condylar process. Similar to what is found in humans, the mandibular foramen is located in the central part between the inferior and posterior ramus on the occlusal plane. We found an impacted molar tooth distal to the erupted molar teeth, and this could be likened to the third molar (wisdom tooth) in humans. It is also reported that miniature pigs have both deciduous and permanent dentitions, and the initiation of tooth development and eruption in miniature pigs is very similar to that in humans. 1
To summarize, the gross anatomy of the maxillofacial region in the pigs was found to be similar to that of humans. Although the distributions of the marginal mandibular branch of the facial nerve and the mental nerve are different from that of humans, we concluded that miniature pigs are a suitable experimental model for the preclinical development of tissue engineering applications in oral and maxillofacial surgery.
Footnotes
Acknowledgements
The authors thank Mr Tsutomu Kojima for helping prepare the cadavers, and Dr Kaori Agawa (Tokyo Metropolitan Police Hospital) for drawing the illustrations of the surgical anatomy of the miniature pig's face. The authors would also like to thank Dr Arieh Bomzon, Consulwrite (
