Abstract
The present study investigated the postoperative plasma concentrations of corticosterone and buprenorphine in male Wistar and Sprague-Dawley rats, treated with buprenorphine administered either through subcutaneous (SC) injection or through voluntary ingestion (VI). The animals were treated with buprenorphine for pre-emptive analgesia prior to surgical placement of a jugular catheter, followed by automated blood sampling during 96 h. Buprenorphine was administered on a regular basis throughout the experiment, and blood was collected on selected time points. Body weight was measured before and 96 h after surgery. It was found that the two rat stocks responded in a similar manner to both buprenorphine treatments, with the exception of body weight change in Wistar rats, in which body weight was reduced after SC treatment. The plasma concentration of corticosterone was significantly higher in the SC-treated animals than in the VI-treated animals during the first 18 h of the study, while plasma buprenorphine concentration was at least as high and more even over time after VI treatment. The present study shows that buprenorphine administration through VI is suitable for both Wistar and Sprague-Dawley rats, with lower stress response and higher plasma concentrations of buprenorphine than after the traditional SC route of administration.
Developing adequate methods for pain and stress alleviation in laboratory animals is important for humane reasons and because pain and stress may affect the outcome of an experiment, due to larger variation between animals and within the individual animal. 1 Common experimental procedures associated with pain and stress include anaesthesia combined with invasive surgery and postoperative recovery. Surgical stress triggers the release of corticosteroids from the adrenal cortex. In rats, corticosterone is the most biologically relevant corticosteroid. 2 During an allostatic response to environmental stressors, corticosterone has many beneficial effects on circulatory functions and mobilization of energy for relevant organ systems. 3 However, if high levels of corticosteroids persist in the circulation for a prolonged period of time, adverse effects such as altered physiological and endocrinological functions, impaired immune system, impaired reproductive ability and pathological lesions are found. 4–6 Adequate perioperative treatment, including pre-emptive analgesia, has been found to reduce postoperative stress in rats. 7,8 The partial μ-opioid receptor agonist buprenorphine is the most common compound used for perioperative analgesia in laboratory rodents. 9 It is known to possess analgesic properties in most analgesiometric tests and to benefit postoperative recovery. 10 A popular route of administration is by subcutaneous (SC) injection. Recently, however, increased interest has emerged in non-invasive methods for administration. By mixing buprenorphine in flavoured jelly or sticky nut paste, the animals are allowed to voluntarily ingest the mixture without human interference. This type of administration has been found to improve postoperative recovery. 11,12
Corticosterone is generally considered as a relevant marker for acute stress, and several studies have demonstrated the usefulness of quantifying plasma corticosterone or faecal excretion of corticosterone and corticosterone metabolites for the assessment of surgical and postoperative stress. 12–17 Recently, we investigated the postoperative effects of pre-emptive buprenorphine on the corticoid system, both after SC injection and after voluntary ingestion (VI), and found that buprenorphine after VI administration was able to reduce the corticosterone levels in the circulation during 18 h after surgery in Sprague-Dawley rats. 12 In addition, the oral administration resulted in beneficial effects on postoperative body weight gain, equal to the effects after SC administration.
The aim of the present investigation was to study the concentrations of corticosterone and buprenorphine in the circulation of Sprague-Dawley and Wistar rats subjected to jugular vein catheterization, treated with perioperative buprenorphine administered by SC injection or by VI. It was hypothesized that the concentrations in both stocks of rats would be similar, and that the corticosterone response would be lower and the concentration of buprenorphine in blood would be higher after oral administration through VI than after traditional SC administration. It was found that postoperative buprenorphine treatment through VI of the drug mixed in Nutella® in Wistar rats has similar effects on blood corticosterone levels and body weight change as in Sprague-Dawley rats, with lower corticosterone levels than after the SC administration, and that the oral treatment results in at least as high buprenorphine plasma concentrations as the SC administration.
Materials and methods
The animal experiments performed in this study were approved by the Animal Ethics Committee in Uppsala, Sweden.
Animals
Eighteen male Wistar (Sca:WISTAR) rats and 18 male Sprague-Dawley (Sca:SD) rats at the age of 10–12 weeks, obtained from Scanbur, Sollentuna, Sweden, were used in the study. Females were not included in this study to avoid oestrus cycle hormonal fluctuations as confounding variables. The average body weight of the rats at the beginning of the experiment was 335 ± 22 g (mean ± standard deviation).
Study design
The animals were divided into three groups, with 12 animals in each, six out of each stock: one control group that did not undergo surgery or any treatment, used for monitoring normal body weight gain; one experimental group that underwent surgery and received buprenorphine through SC injection and one experimental group that underwent surgery and received buprenorphine through VI of the drug mixed in Nutella®. No vehicle control was included, since a recent study has demonstrated that ingestion of Nutella® alone affected neither corticosterone levels nor body weight in rats. 15 Group sizes were determined using a power analysis as described in the literature, 18,19 based on data from previous investigation where corticosterone levels in blood have been investigated. 12,17 Treatments were administered according to a randomized block design sorted by stock.
Husbandry during experiment
Before the experiments, the rats were housed in Makrolon type IV cages (2–3 rats per cage) for at least one week in animal rooms with standard animal house conditions: diurnal rhythm was regulated with a 12 h light:12 h dark cycle with lights on from 06.00 to 18.00 h; temperature was kept at 19–21°C; relative humidity was 30–60%; air was changed approximately 15 times per hour; and cages were cleaned twice a week. Aspen chips (Scanbur B&K, Sollentuna, Sweden) were used as bedding material. The animals had free access to food pellets (R36, Lantmännen, Stockholm, Sweden) and tap water at all times. Food pellets were placed on the bedding to improve accessibility after surgery. Cages were randomly placed on the racks in the animal room, and cages were enriched with wooden tunnels for the animals to hide in and paper sheets were used as nesting material. Two days before surgery, the rats were transferred to single housing in Makrolon type III cages and moved to a designated laboratory with similar environmental and housing conditions, where the experiments were conducted. After the transfer, all cages were placed at the same bench in the laboratory, close to the blood sampling equipment, and the animals were handled daily to habituate them to the experimenter. All animals were handled by the same person, to minimize the risk of bias of experimental data due to the change of personnel.
Preoperative and surgical procedures
Two days before surgery (Day −2), 2 g/kg body weight (bw) Nutella® hazelnut and chocolate cream (Ferrero, Pino Torinese, Italy) was placed in the cages of the rats in the oral treatment group to habituate them to the flavour. Body weight was measured on Day −2 and Day 0 to ensure that all animals included in the study gained weight as expected before subjected to surgery.
Prior to surgery, rats were treated for pre-emptive analgesia with buprenorphine (Temgesic®, Schering-Plough Europe, Brussels, Belgium), either 0.05 mg/kg bw administered SC (1.7 mL/kg bw; 30 min before surgery), or 0.4 mg/kg bw dissolved in Nutella® (2 g/kg bw; 1 h before surgery) for VI administration. The orally-treated animals consumed all of the buprenorphine-mix within 30 min in each case. The mixture was prepared immediately prior to administration, and no more than two rats were investigated at the same time. Thereby, it was not necessary to store the mixture, which could have inflicted degradation of the buprenorphine. Analgesic doses were based on those recommended in the literature 11,20 and on our previous investigations where VI administration has been used. 12,16,17
The rats were placed in an induction chamber and anaesthesia was induced with 5% isoflurane delivered in pure oxygen. Once the paw withdrawal reflex was absent, the rats were shaved at the incision sites and attached to a Simtec anaesthetic mask for spontaneous respiration. Isoflurane was maintained at approximately 2.5–3% to ensure adequate anaesthesia, and rectal body temperature was maintained at 37–38°C, using a heated pad. The shaved parts were washed with iodine (Jodopax vet®, Pharmaxin AB, Helsingborg, Sweden). An incision was made in the skin of the neck and the external jugular vein was catheterized with a vein catheter (DiLab, Lund, Sweden) filled with heparinized saline. The catheter was placed with the tip close to the entrance of the right atrium of the heart, secured in the vein by two sutures and led subcutaneously through a DiLab Dacron button attached by sutures in the muscle and skin in the nape of the neck. The catheter was led further through a metal spring and connected to an AccuSampler® (DiLab) for automated blood sampling. The procedure, from induction to recovery, was completed within 45–60 min. The rats were observed regularly during the recovery phase. To avoid bias in corticosterone levels by disturbing the animals during the experiment, the observations took place only immediately after blood withdrawal and in connection to drug administration. None of the animals exhibited any overt abnormal behaviour related to pain or distress that would have required a premature termination of the experiment.
Blood sampling and postoperative treatment
All surgeries were completed before noon and blood sampling started between 11:00 and 12:00 h, starting immediately after the rats regained consciousness. Thereafter, blood was collected after 2, 6, 10, 14 and 18 h, followed by a sample collected at 12:00 and 00:00 h until 96 h after surgery. The rationale for treating the animals for such a long period of time, despite the relatively minor surgical procedure, was to monitor the return to basal circadian rhythmicity of corticosterone, similar to a previous study, 12 and investigate possible stock-related differences. In total, 13 samples of 150 μL each were collected from the rats, corresponding to a blood withdrawal of approximately 0.5% of the animals' body weight during the whole experiment. The AccuSampler® compensated the loss of body fluid by injecting saline in volumes similar to the withdrawn blood volume.
Besides the presurgical buprenorphine treatments described above, animals in the experimental groups were treated according to the following: rats in the SC group were given an additional dose 8 h after surgery at approximately 08:00 h, followed by an injection every 12 h until the end of the experiment, 96 h after surgery. Rats in the VI group were treated with buprenorphine in Nutella® in the morning every 24 h.
The postoperative body weight was recorded at the end of the experiment (96 h postoperative) in all three groups. The experiments were terminated by an infusion of 100 mg/kg bw pentobarbital (Apoteket, Uppsala, Sweden) into the blood circulation via the catheter, except for animals in the control group that were used in unrelated investigations.
Corticosterone quantification
The blood samples were collected in tubes and stored overnight at 4°C, after which they were centrifuged to remove blood cells and obtain plasma. Plasma was stored at −20°C until analysis. The plasma concentration of corticosterone was quantified with enzyme-linked immunosorbent assay (ELISA), using a commercial ELISA kit for corticosterone (Correlate-EIA, Assay-Designs Inc, Ann Arbor, MI, USA) according to the manufacturer's manual. The kit has been verified to have a cross-reactivity equivalent to 28.6% against deoxycorticosterone, 1.7% against progesterone, 0.13% against testosterone, 0.28% against tetrahydrocorticosterone, 0.18% against aldosterone and less than 0.05% against cortisol, pregnenolone, beta-oestradiol, cortisone and 11-dehydrocorticosterone acetate.
Buprenorphine quantification
Plasma was analysed in duplicate using the Buprenorphine One-step ELISA (International Diagnotic Systems Corp, St Joseph, MI, USA) in accordance with the manufacturer's instructions. For improved accuracy, the provided standards were supplemented with additional dilutions to yield a five-point standard curve consisting of concentrations 0, 0.5, 1, 1.5 and 3 ng/mL. All known cross-reactivities are reported by the manufacturer at <0.06%, with the exception of norbuprenorphine, cross-reacting at 1.1%. No analytical sensitivity is given by the manufacturer, but the analysis kit has previously been validated for samples with corticosterone concentrations as low as 0.2 ng/mL. 21 The absorbencies were recorded at 450 nm (reference wavelength, 650 nm) using a Thermo Multiskan Ex microplate reader (Thermo Fisher Scientific Inc, Slangerup, Denmark).
Statistical procedures
Body weight change, corticosterone levels and buprenorphine concentrations were analysed using a two-factor (stock and treatment) analysis of variance (ANOVA) with Bonferroni's post-test, using GraphPad Prism 5.0. Body weight values were normalized and indexed based on values Day 0, to facilitate comparison in body weight change. Numeric values were used for corticosterone and buprenorphine concentrations. Since no stock-related differences in corticosterone levels or buprenorphine were observed, the stock data were pooled, and the pooled data were further analysed with SPSS 15.0, using a general linear model (GLM) with repeated measures for determination of differences between treatments during the first 18 h of the study, supplemented with t-tests for each individual time point during the same period. P values <0.05 were considered significant.
Results
There were no significant differences between the initial body weights Day –2 in any of the groups (WIST control 338 ± 11 g [mean ± SEM]; WIST SC 333 ± 8 g; WIST VI 343 ± 13 g; SPRD control 331 ± 6 g; SPRD SC 339 ± 11; SPRD VI 330 ± 6), and all animals gained body weight between Day −2 and Day 0 as expected. Therefore, the body weight values were normalized to percent difference from Day 0, to facilitate comparison of the effects of surgery and treatment. The effects of surgery and the two perioperative buprenorphine treatments on body weight changes are shown in Figure 1. A two-factor ANOVA showed that both treatment (P < 0.001) and stock (P < 0.05) had significant impact on the postoperative body weight change. Bonferroni's post-test showed that both SC and VI treatments caused a decreased body weight gain compared with control in both stocks (WIST VI P < 0.01; WIST SC, SPRD SC and SPRD VI P < 0.001), while the SC treatment was different from the VI treatment only in Wistar rats (P < 0.01).
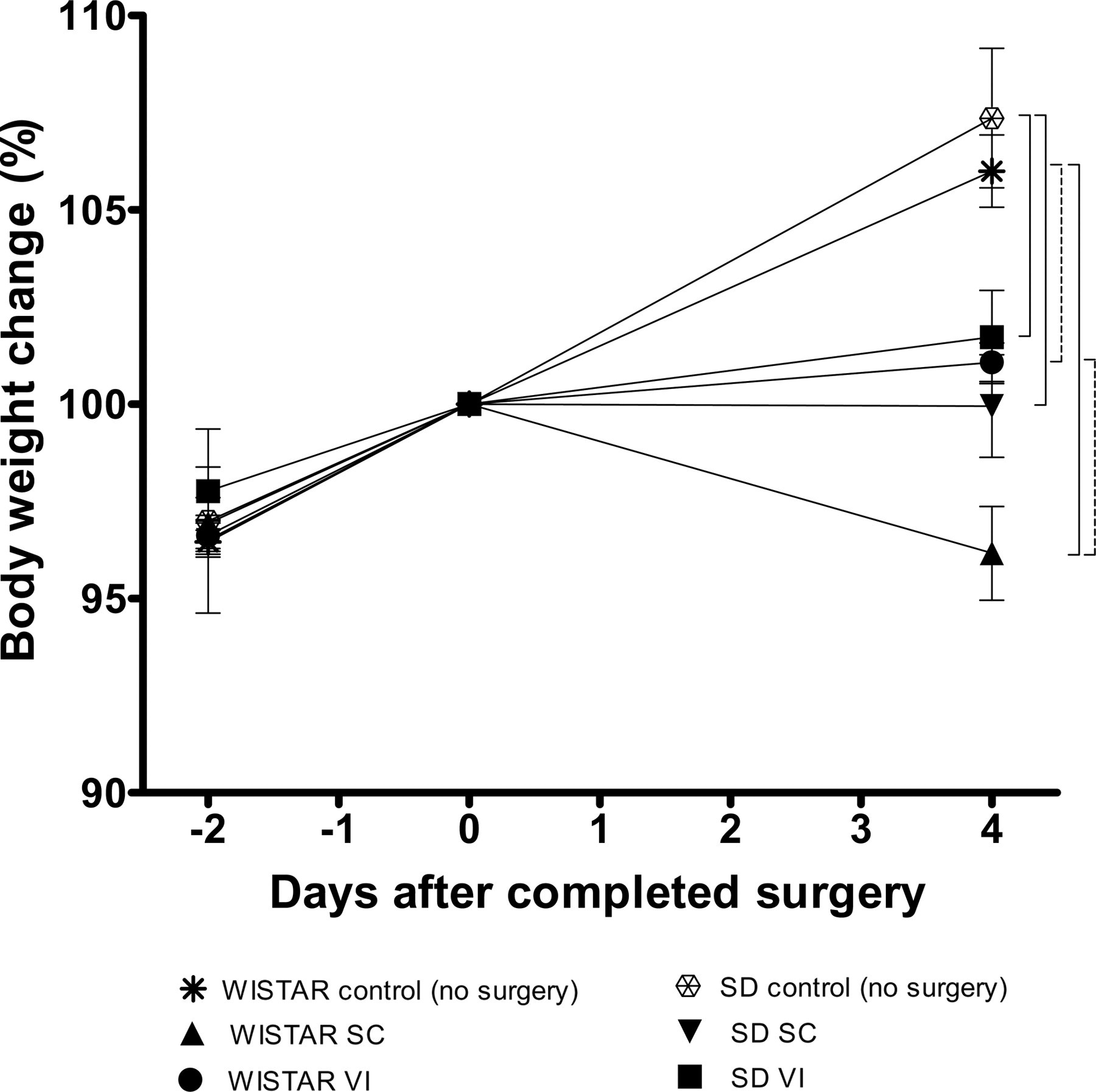
Percentage change in body weight from two days before until four days after surgery, using values at Day 0 as index. Hatched brackets represent statistical significance between groups at P < 0.01 and continuous brackets P < 0.001, as determined with the two-factor ANOVA with Bonferroni's post-test. The error bars display SEM. ANOVA: analysis of variance; SEM: standard error of the mean; SC: subcutaneous; VI: voluntary ingestion; SD: Sprague-Dawley
The effects of surgery and the two perioperative buprenorphine treatments on plasma corticosterone levels in Wistar and Sprague-Dawley rats are shown in Figure 2. A two-factor ANOVA with Bonferroni's post-test showed significant differences between treatments at time points 2 h (P < 0.05) and 10 h (P < 0.01), with higher corticosterone levels in animals subjected to the SC treatment. No statistical differences could be observed between the two stocks at any time point. Therefore, data from both stocks were pooled and further analysed with respect to the effect of treatment only, focusing on the first 18 h period (Figure 3). A GLM analysis with repeated measures showed that the effects of the two treatments were different during 18 h after surgery (P < 0.01). In addition, the supplementary t-test showed that there were differences between treatments at time points 2 h (P < 0.05) and 10 h (P < 0.01).
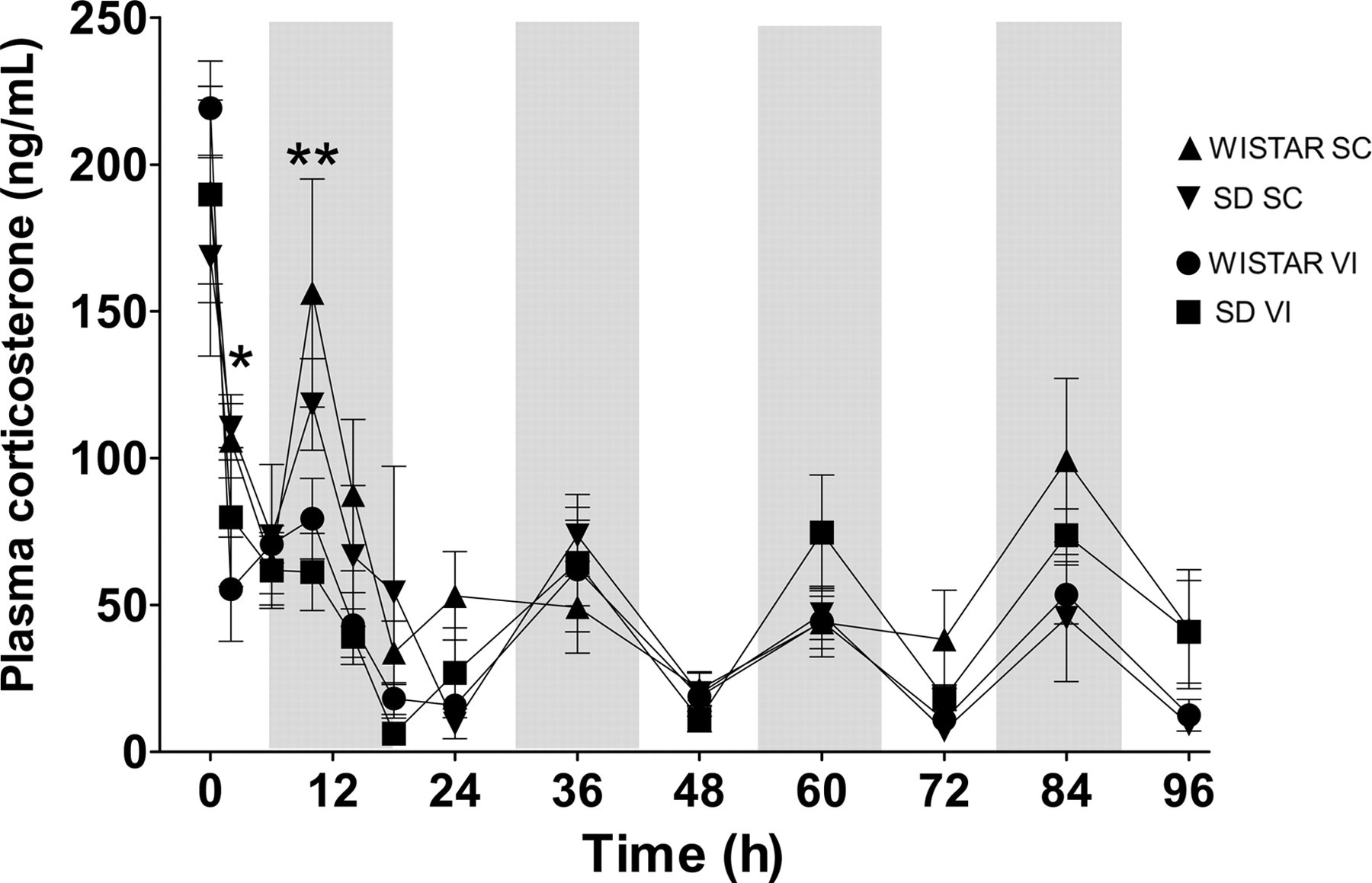
Plasma corticosterone levels in both SC and VI groups during the whole 96 h period, divided by rat stock. *Represents significant difference between the two treatments at P < 0.05 and ** at P < 0.01, as determined with the two-factor ANOVA with Bonferroni's post-test. The shadowed boxes indicate dark periods. The error bars display SEM. ANOVA: analysis of variance; SEM: standard error of the mean; SC: subcutaneous; VI: voluntary ingestion; SD: Sprague-Dawley
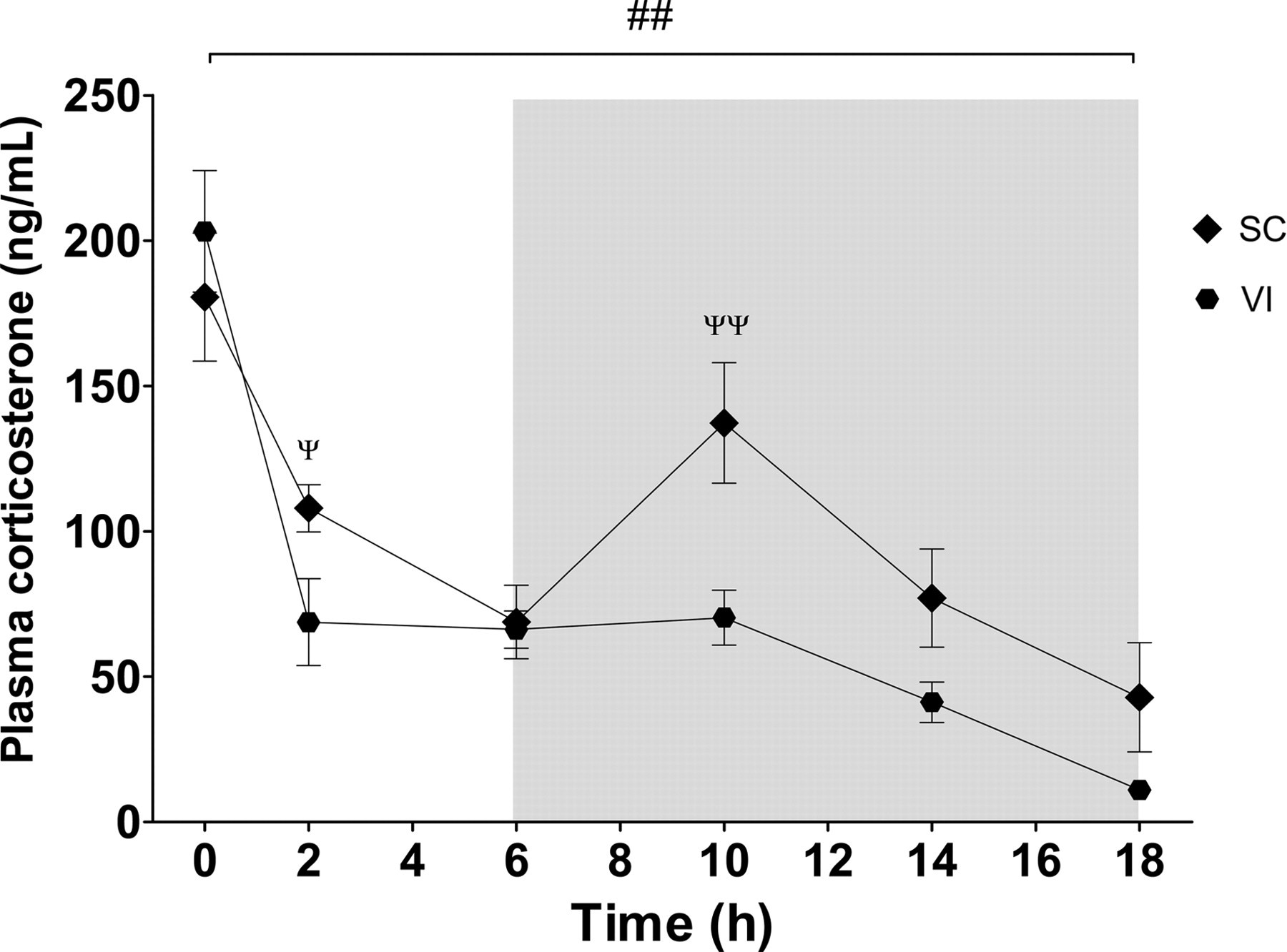
Plasma corticosterone levels in both SC and VI groups during the first 18 h period, the two rat stocks pooled. The bracket with ## represents difference between the two treatments during the entire period (P < 0.01), as determined with GLM with repeated measures. Ψ and ΨΨ represent differences between treatments at the indicated time points, P < 0.05 and P < 0.01, respectively, as determined by t-test. The shadowed box indicates the dark period. The error bars display SEM. SEM: standard error of the mean; SC: subcutaneous; VI: voluntary ingestion; GLM: general linear model
The buprenorphine concentrations in plasma during 18 h after surgery for each stock and treatment are shown in Figure 4. The buprenorphine concentrations after SC treatment were high immediately after completed surgery, but decreased within 2 h and were close to 0 after 6 h. At time point 10 h, 2 h after the second injection, the concentrations were high again, followed by a similar decline, although the concentrations apparently did not reach as low a level as after the first injection. A two-factor ANOVA with Bonferroni's post-test showed that there was no stock-related difference between the two stocks at any time point. Similar to the corticosterone data, the stocks were pooled and data analysed with a t-test for each individual time point. As shown in Figure 5, it was found that the buprenorphine concentrations were different at time points 2 h (P < 0.01), 6 h (P < 0.001) and 10 h (P < 0.01).
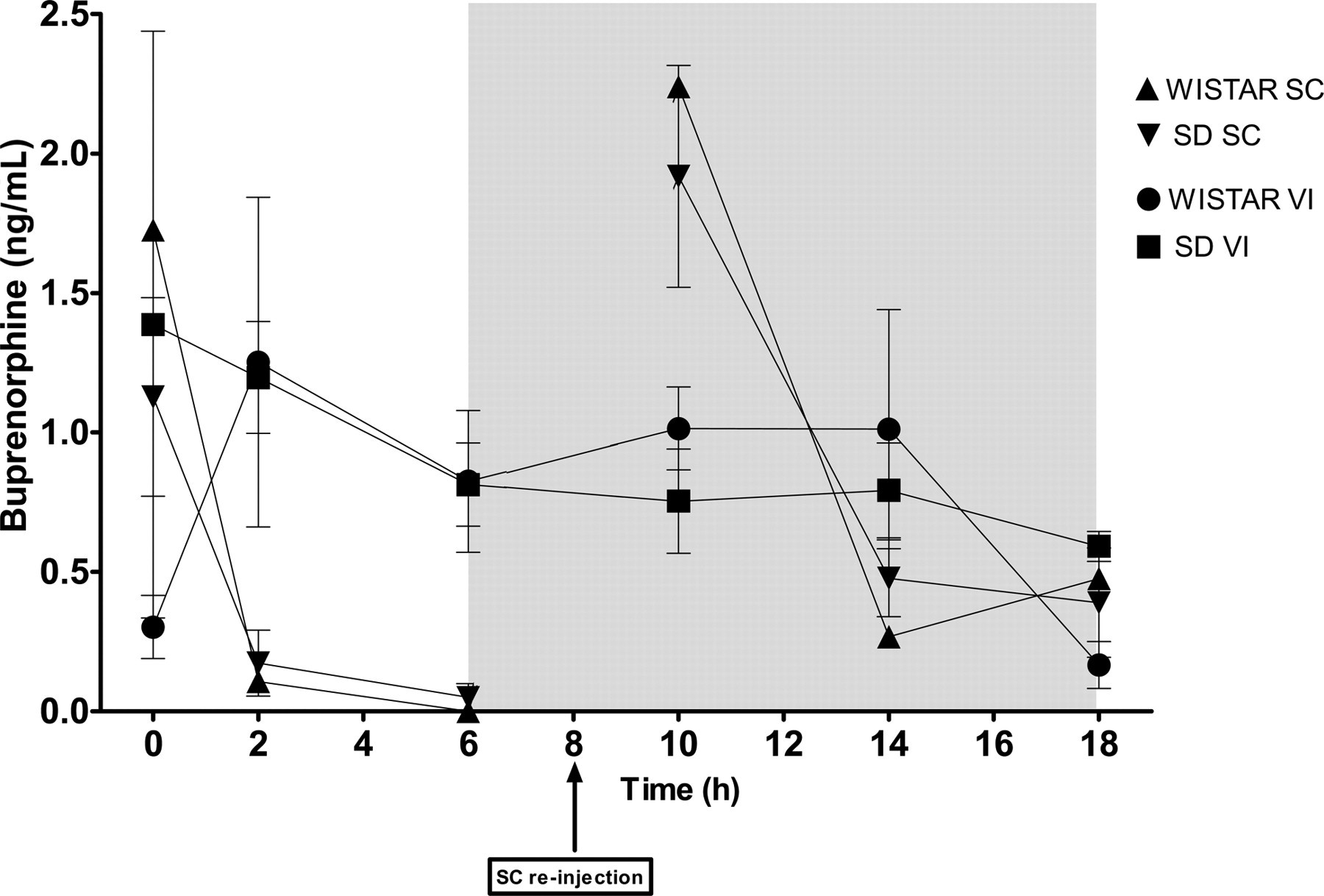
Concentration of buprenorphine in plasma in both SC and VI groups during the first 18 h after surgery, divided by rat stock. The shadowed box indicates the dark period. The error bars display SEM. SEM: standard error of the mean; SC: subcutaneous; VI: voluntary ingestion; SD: Sprague-Dawley
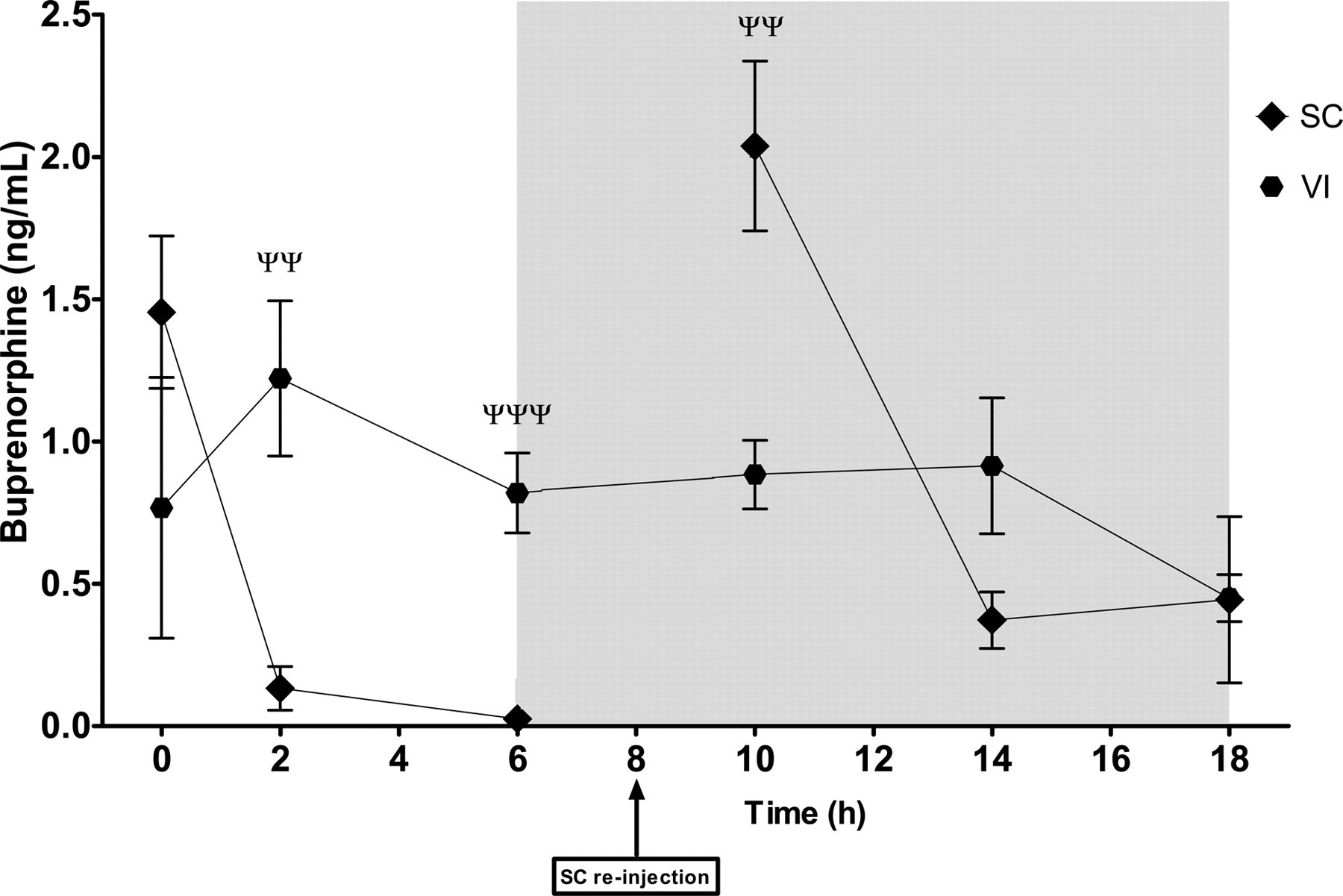
Concentration of buprenorphine in plasma in both SC and VI groups during the first 18 h after surgery, the two rat stocks pooled. ΨΨ and ΨΨΨ represent differences between treatments at the indicated time points, P < 0.01 and P < 0.001, respectively, as determined by t-test. The shadowed box indicates the dark period. The error bars display SEM. SEM: standard error of the mean; SC: subcutaneous; VI: voluntary ingestion; SD: Sprague-Dawley
Discussion
Investigating stress response in laboratory animals may be difficult, since handling and experimental procedures or other disturbances in the environment may increase experimental variation and reduce the likelihood of obtaining reliable corticosterone levels. The automated blood sampling technique used in the present investigation has previously been investigated in several studies, and appears to be a sensitive non-invasive method causing minimal disturbance to the baseline levels of corticosterone. 17 The transfer of the animals to the laboratory two days before surgery was done to allow the animals to acclimatize to the laboratory. There is a potential risk that this could inflict additional stress to the animals. However, this is not likely. A recent study demonstrated that group-housed animals transferred to another location and then separated to single housing did not exhibit any significant stress response after two days that could be measured by the quantification of corticosterone excreted in faeces. 22
The present study demonstrated that the method for oral buprenorphine administration that we previously found to be effective in Sprague-Dawley rats, 12 results in a similar corticosterone profile and body weight change in Wistar rats. Both stocks are among the most commonly used laboratory rats, due to their high vigour and reproductive ability and their docility. The Wistar rat was developed already in 1906 at the Wistar Institute, and was the first rat stock developed as a laboratory rat. 23 The Sprague-Dawley rat was established around 1925, with the Wistar rat as origin on the maternal side. 23 Despite the relationship, there are a number of differing features between the two stocks. For instance, the Wistar rat has been found to respond more strongly to predator odour than the Sprague-Dawley rat, 24 while the opposite response has been reported regarding the response to an alarm substance from the same stock during immersion in water. 25 Stock differences have also been reported in response to and inhibition of acoustic startle response, 26,27 and in the effects of novelty on locomotor behaviour and of morphine on dopamine release from the nucleus accumbens. 28 In addition, marked differences in several endocrine parameters including corticosterone levels, and in growth rate, have been described between the two stocks. 29 Due to these differences in behavioural and physiological properties, it could not be excluded that the two stocks would respond differently in a postoperative situation. In the present study, stock-related differences were only observed in postoperative body weight change after the SC treatment. The precise mechanisms responsible for this difference are not known, but may be related to the differences in endocrine functions regulating growth rate mentioned above. The body weight gain was reduced after surgery in both stocks in the present study, compared with non-operated animals. However, this reduction is much smaller compared with what has been previously found in Sprague-Dawley rats, subjected to identical experimentation without buprenorphine treatment. In these animals, the body weight was significantly decreased compared with preoperative values. 12 Any effect on body weight gain after VI treatment is unlikely due to additional weight added by the ingestion of Nutella® itself, since the Nutella® mix provided was only approximately 0.7 g per rat daily. Thus, if the body weight would be affected by the ingestion of Nutella®, this is probably due to other nutritional factors than the weight of the vehicle.
The reduction in postoperative corticosterone levels observed in both stocks after VI compared with SC injection, confirms our previous findings that the VI treatment is effective in reducing the surgical stress response. 12,15 It is not fully understood whether the reduction of corticosterone observed is due to a direct effect on the HPA axis by buprenorphine, or due to an indirect effect as a result of reduced nociceptive signalling. There are indications that the latter may be the most likely. It has previously been demonstrated that SC injection of buprenorphine in non-operated rats does not affect corticosterone levels. 30 In addition, no reduction of the corticosterone levels were observed after the second SC injection in the present study, which one could expect if buprenorphine per se would have a significant effect on the HPA axis. Finally, it has been reported that different degrees of surgical invasiveness result in different levels of reduction of corticosterone during surgery. 31 The impact of Nutella® as a vehicle on the corticosterone response is not fully known. However, in a recent investigation, no differences in postoperative plasma corticosterone levels were found in rats that were not treated with any analgesia, with either no vehicle or Nutella® as a vehicle. 15 Therefore, it is not likely that Nutella® would be involved in the more pronounced corticosterone-reducing effect observed after VI treatment. However, possible effects on other physiological systems related to nutritional factors or caloric intake after Nutella® ingestion remain unknown. Although it is unlikely that such effects would be adverse, it is worth considering for future investigations.
The use of oral administration of buprenorphine has been questioned, due to the first-pass liver metabolism of the drug. In humans, oral administration is therefore not recommended. 32 In addition, a few studies in rats have found that the oral doses required for obtaining sufficient analgesia in rats are more than 10-fold higher than the dose used in the present study. 33,34 The differences between studies may be explained by the different parameters studied. The studies that claimed that higher doses were necessary assessed analgesic effect using analgesiometry instead of clinical parameters such as body weight gain, food and water intake or biological markers such as corticosterone, and the latter may be more relevant for assessing the success of postoperative recovery. 10 In addition, the data in the present study show that the buprenorhine plasma concentration after VI of 0.4 mg/kg was equal to or even higher than after SC injection, as determined by the t-tests. In addition, the buprenorphine concentrations at different time points were similar to each other after VI administration, compared with the very fluctuating concentration observed after SC administration. An even blood concentration over time is preferred when administering drugs. 35 After SC injection, buprenorphine was rapidly eliminated and reached low blood concentrations within 2 h. After the second injection, the concentration was again high followed by a rapid decline. The second decline seems smaller than the first, with apparently higher concentrations after the decline. However, the increase in buprenorphine concentration after the second injection seems not to be sufficient to decrease the levels of circulating corticosterone, since these levels were significantly higher after 10 h in the SC group. Hence, it seems as if an even and high buprenorphine concentration over time is important to achieve the desired reduction in corticosterone levels. However, it remains unknown what precise plasma concentrations and what time fraction is needed to attain to these concentrations that is necessary for buprenorphine to be efficient. The fact that oral buprenorphine undergoes first-pass liver metabolism, which could decrease the bioavailability of the drug, seems not to be a problem when using the dose of 0.4 mg/kg. Most likely, the hepatic metabolism of buprenorphine is saturated on the first pass when using a sufficiently high dose. This effect, resulting in higher bioavailability, has been described for other substances such as salicylamide 36 and ethanol. 37 In addition, the absorption of buprenorphine from the gastrointestinal tract is likely to be slow, which causes a slower elimination of the oral bolus, and thereby prolongs the duration of high serum concentrations. Altogether, the present data and previous finding support that the method of using VI for oral administration of buprenorphine are suitable for improving the postoperative recovery of rats, at least after the surgical procedures described in the present study.
In conclusion, the present study demonstrates that postoperative buprenorphine treatment through VI of the drug mixed in Nutella® in Wistar rats has similar effects on blood corticosterone levels and body weight change as in Sprague-Dawley rats, and that this treatment results in at least as high buprenorphine plasma concentrations as does the SC administration. Therefore, it is confirmed that the recently developed method for oral administration is a relevant method for perioperative analgesia, applicable in both Wistar and Sprague-Dawley rats.
Footnotes
Acknowledgments
The present investigation was supported by generous grants from the Swedish Research Council and from the Swedish Board of Agriculture. DiLab kindly donated two AccuSamplers. The authors wish to thank Trine Marie Nielsen for her technical assistance.
