Abstract
In the conventional shuttle box, animals are trained to avoid electric foot-shocks. As a consequence of these stress-inducing foot-shocks the animals become anxious and are difficult to train. The aim of the present study was to avoid the stress-inducing foot-shocks and to develop a fast and reliable conditioned avoidance behaviour task for guineapigs. We examined whether narrowband noises at four different sound levels above hearing threshold could be used as conditioned stimulus (CS). The unconditioned stimulus (UCS) was a stream of air, which was used instead of the conventionally used electric foot-shocks. The animals were initially trained with a CS of 78 dB sound pressure level (SPL). In this initial training, guineapigs learned to detect a narrowband noise of 78 dB SPL. Interestingly, during the first additional training session in which three other sound levels were applied, guineapigs did not immediately generalize the learned response at 78 dB SPL to lower sound levels of 58 and 68 dB SPL. However, in this session a noise level of 88 dB SPL led immediately to a high level of responses. The response latency decreased with increasing sound level, from ~7 s at 58 dB SPL to ~3 s at 88 dB SPL. The escape latency during the UCS was ~0.6 s. The present results demonstrate that after reducing the level of stress guineapigs can acquire a response in only a few sessions and furthermore, although the guineapigs were less anxious, training at sound levels of 78 and 88 dB SPL was influenced by an aversive reaction by the guineapig. The results indicate that this aversive reaction of the guineapig is crucial for the training.
Behavioural training of animals involves many confounding factors. Training guineapigs is even more complicated than training other rodents because guineapigs demonstrate a different behavioural pattern compared with rats and mice. When they are exposed to a novel environment, their behaviour is based more on precautionary rather than on exploratory behaviour,1,2 and a negative reinforcement may lead to stress affecting the behaviour.3,4 For this reason most of the experimental set-ups developed for rodents are not suitable for guineapigs. However, for some experiments guineapigs are preferred. As examples, from a practical point of view, in auditory research guineapigs are favourable above rats and mice because of the easily implantable cochlea. 5 From a toxicokinetic point of view guineapigs appeared to be a better model than the rat because, like in humans, their blood contains small amounts of carboxylesterases, which act as scavengers for Cholinesterase inhibitors. 6 In contrast, the blood of rats contains large amounts of carboxylesterases.
Although guineapigs react erratically and tend to freeze in the conventional shuttle box when foot-shocks are applied as the unconditioned stimulus (UCS), 7 several investigators succeeded in training guineapigs by conventional methods, such as the Morris water maze,8–10 an operant conditioning chamber 11 and the conventional foot-shock-motivated shuttle box.12,13 In the conventional shuttle box, animals learn on guidance of a conditioned stimulus (CS) to avoid an unpleasant and stressful foot-shock, the UCS. The advantage of the shuttle box is that animals learn rapidly to avoid the UCS. The disadvantage is that the shuttle box may induce stress.
The best method to avoid stress in a shuttle box is to use a positive instead of a negative reinforcement. A commonly used positive reinforcement is a food reward. Pilot experiments in our laboratory, using food as a positive reinforcement, were time consuming and not successful, possibly due to lack of motivational aspects. 14 For these reasons we decided to use a modified shuttle-box paradigm for guineapigs. 7 The foot-shock guided shuttle box, which is currently also used for mice, 15 was originally developed to test rats. 16 In the modified shuttle box for guineapigs a strong stream of air, imitating a natural outside atmospheric condition like a gust of wind, was used as UCS instead of foot-shocks to minimize stress-inducing effects. This type of stimulus was used because anxious guineapigs are difficult to train as stress may interfere with the behavioural outcome. Previously, the modified shuttle box only examined performance at a single sound level of the CS. The aim of the present study was to investigate whether modulation of the sound level of the CS increased the potential of the modified shuttle box as a psychoacoustic test for guineapigs.
Materials and Methods
Animals and experimental design
Fifteen albino female guineapigs (strain: Dunkin Hartley; body weight: 250–350 g) were purchased from Harlan Laboratories (Horst, The Netherlands) and socially housed, three or four together, in Makrolon cages (80 cm x 60 cm x 25 cm) in the animal care facility of the Rudolf Magnus Institute of Neuroscience. All animals had free access to food and water and were kept under standard laboratory conditions. Lights were on between 07:00 and 19:00 h. Temperature and humidity were kept constant at 21°C and 60%, respectively. Animals were acclimatized to laboratory conditions for a week. During this week the animals were handled daily by transporting them to the sound-attenuated chamber in which the shuttle-box tests were performed. All experimental procedures were approved by the University's Committee on Animal Research (DEC-UMC # 03.04.036).
The behavioural testing procedure was adapted from TNO (Rijswijk, The Netherlands). 7 The shuttle box consisted of two equal compartments (23 x 23 x 23 cm), connected by a passage through which the animal could cross from one compartment to the other. An infrared beam on each side of the passage detected entries into each compartment. The guineapigs had to learn to avoid an unpleasant stream of air (UCS: 6250 cm 3 /s, duration 20 s, air tube diameter 1 cm) by moving into the other compartment within 15 s after the CS (narrowband noise of 58, 68, 78 or 88 dB sound pressure level [SPL] passing through a band-pass filter [slope –6 dB per octave] with a centre frequency of 10 kHz) had been turned on. A shuttle response (defined as the presence of the guineapig in the other compartment) terminated the CS or UCS and initiated an inter-trial interval. The inter-trial interval was 20 s (± period randomly varied between 0 and 10 s). Responding to the CS was classed as a conditioned avoidance response (CAR) and responding to the UCS as an escape response. When the animal remained in the compartment during the UCS presentation (20 s) this was classed as a ‘no response’. A shuttle response during the inter-trial interval was classed as an inter-trial response. The CAR is expressed in percentage: CAR% = number of CARs x 100/number of CS presentations. During the first two sessions, animals received 10 trials of 78 dB SPL. On training sessions 3–5, 20 trials at 78 dB SPL were presented. From session 6 to 10, 20 trials were presented with random sound levels of 58, 68, 78 or 88 dB SPL (5 trials per SPL). Stimulus presentation and data collection were controlled with custom-written software and a personal computer. During the training sessions the performance of the guineapigs was continuously monitored. Animals were trained in 10 daily training sessions (sessions 1–10) starting on Wednesday. Animals were not trained during the weekends. All experimental manipulations were carried out in the morning.
Statistics
Statistical analyses were performed using SPSS® for Windows (version 15.0.1). To identify differences between sessions and sound levels, repeated measures analysis of variance (rm ANOVA) were used. To test differences between the numbers of animals responding at different trials during the same session the McNemar test (CochranQ test) was used. To test differences in response latencies of CARs paired t-tests (with Bonferroni correction) were used.
Results
Shuttle responses
The guineapigs rarely shuttled from one compartment to the other compartment between the trials (inter-trial responses).
On average they had two inter-trial responses during a session, which always occurred immediately after the animals entered the shuttle box. The number of trials scored as ‘no response’ was zero, which means that the animals always responded with an escape to the other compartment when they were exposed to the UCS. The mean latency for the escape response elicited with the UCS was 0.63 ± 0.26 s.
Conditioned avoidance responses
The mean percentages of CARs during the first five training sessions are presented in Figure 1. During these first five sessions the sound level of the CS was 78 dB SPL. Repeated measures ANOVA revealed a significant effect of session (rm ANOVA; F = 13.7, df = 4, P < 0.001). These data demonstrate that the animals had a higher percentage of CARs during the fourth training session than during the first training session. Interestingly, the percentage of CARs seemed higher during the first session than in the second session. To investigate the avoidance behaviour within a single session (especially within the first session), the number of animals demonstrating a CAR is presented per trial in Figure 2. McNemar tests showed that the number of animals demonstrating a CAR during the first trial was significantly higher than the number of animals demonstrating a CAR during trials later on in the session (P< 0.05). This effect was present in sessions 1–4 (Figure 2). In the fifth session the number of animals demonstrating a CAR is rather uniformly distributed across trials. The most interesting feature of these results is that during the first trial of the first session, 14 of the 15 animals shuttled to the other compartment during exposure to the CS. Another important feature is that the number of animals that demonstrated a CAR increased in the later trials during session 4.
Mean percent (± SEM) conditioned avoidance responses (CARs) of guineapigs (n = 15) for a sound level of 75 dB sound pressure level (SPL), during the first five training sessions of 10 (sessions 1 and 2) or 20 (sessions 3–5) trials each. Repeated-measures analysis of variance revealed that CARs at a sound level of 78 dB SPL increased during sessions 1-5 (F = 13.7, df = 4, P < 0.001) Number of animals responding correctly in single trials presented during the first five training sessions. During the first two sessions, guineapigs (n = 15) were exposed to 10 trials. During sessions 3–5 the guineapigs were exposed to 20 trials. The sound level of the conditioned stimulus during these first five sessions was 78 dB SPL *P<0.05, **P < 0.01. * and ** indicate that a statistical different number of guineapigs demonstrated a conditioned avoidance response during that trial as compared with the first trial of that session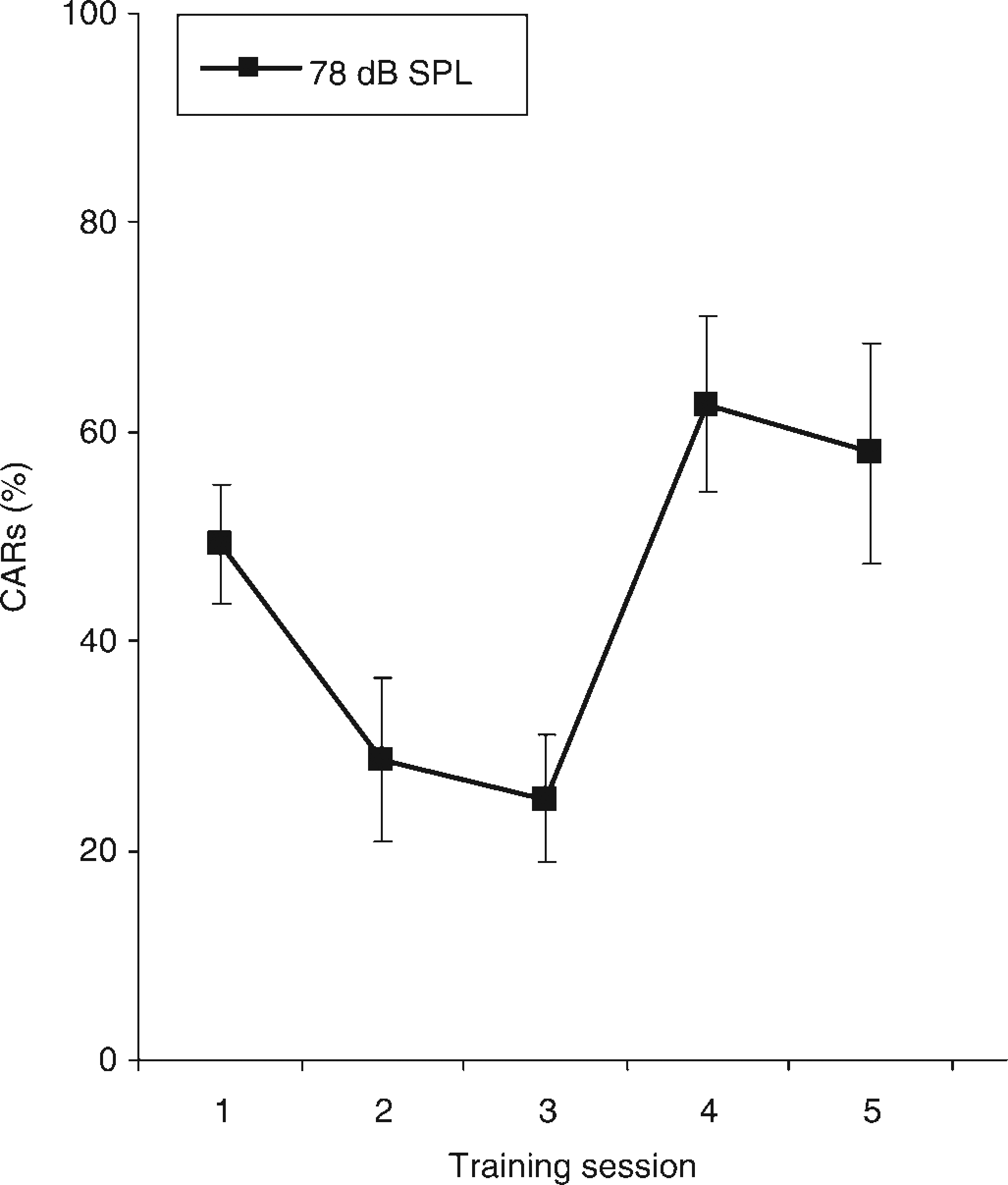
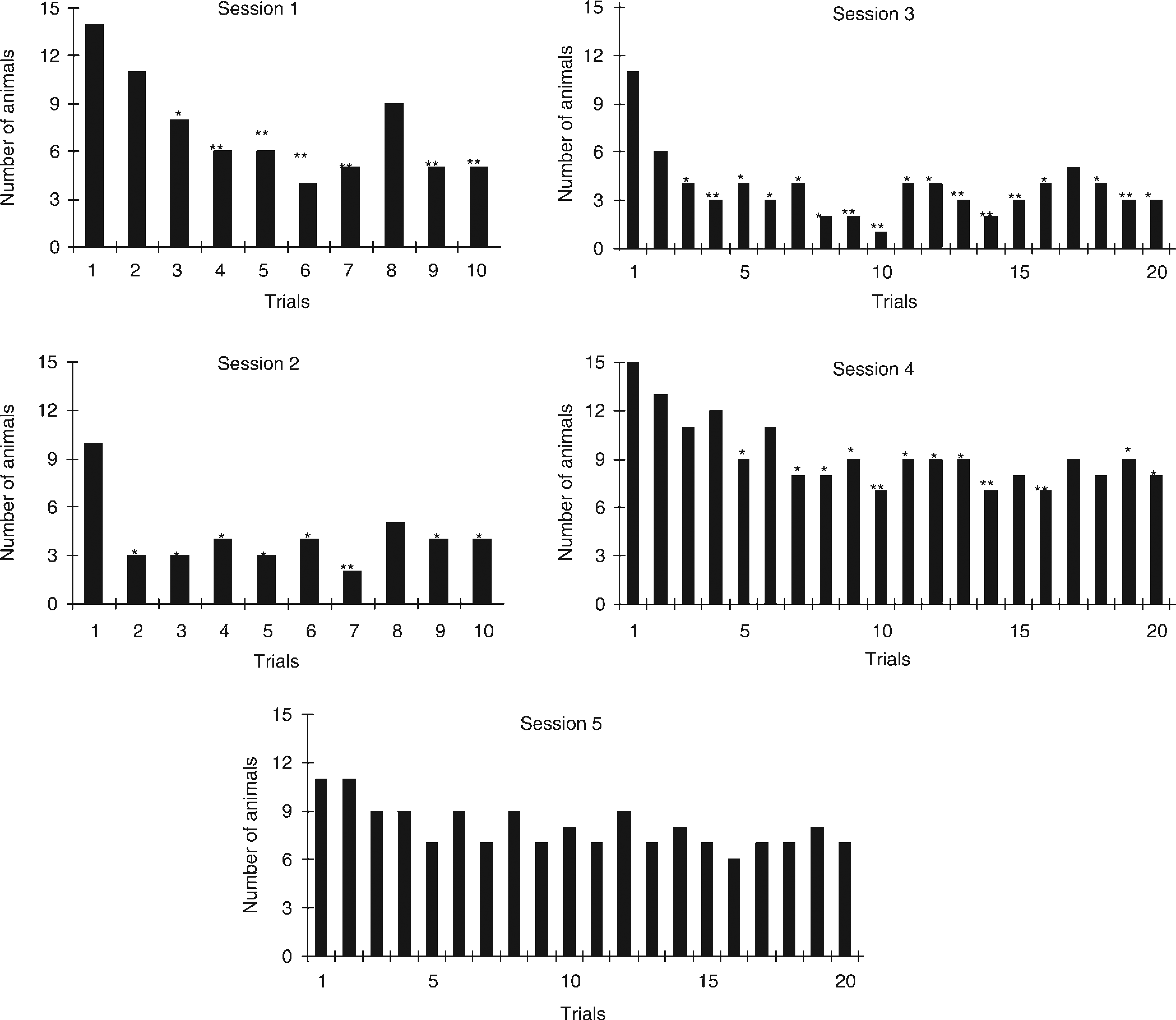
Figure 3 shows the percentage of CARs during training sessions 6–10. In these sessions the CS was presented randomly at different sound levels. Analysis of the data with rm ANOVA revealed significant effects of sound level (F = 79.43, df 3, 36, P < 0.001) and session (F = 3.92, df = 4, 48, P < 0.01). The effect of sound level was already prominent during session 6. The percentage of CARs at 88 dB SPL was close to 100% while the percentage of CARs at 58 dB was ~10%.
Mean percent (± SEM) conditioned avoidance responses (CARs) of guineapigs (n = 15), for all four sound levels, during training sessions 6–10. Two-way repeated measures analysis of variance revealed an effect of sound level (F = 79.4, df = 3, 36, P < 0.001) and session (F = 3.9, df = 4, 48, P < 0.01)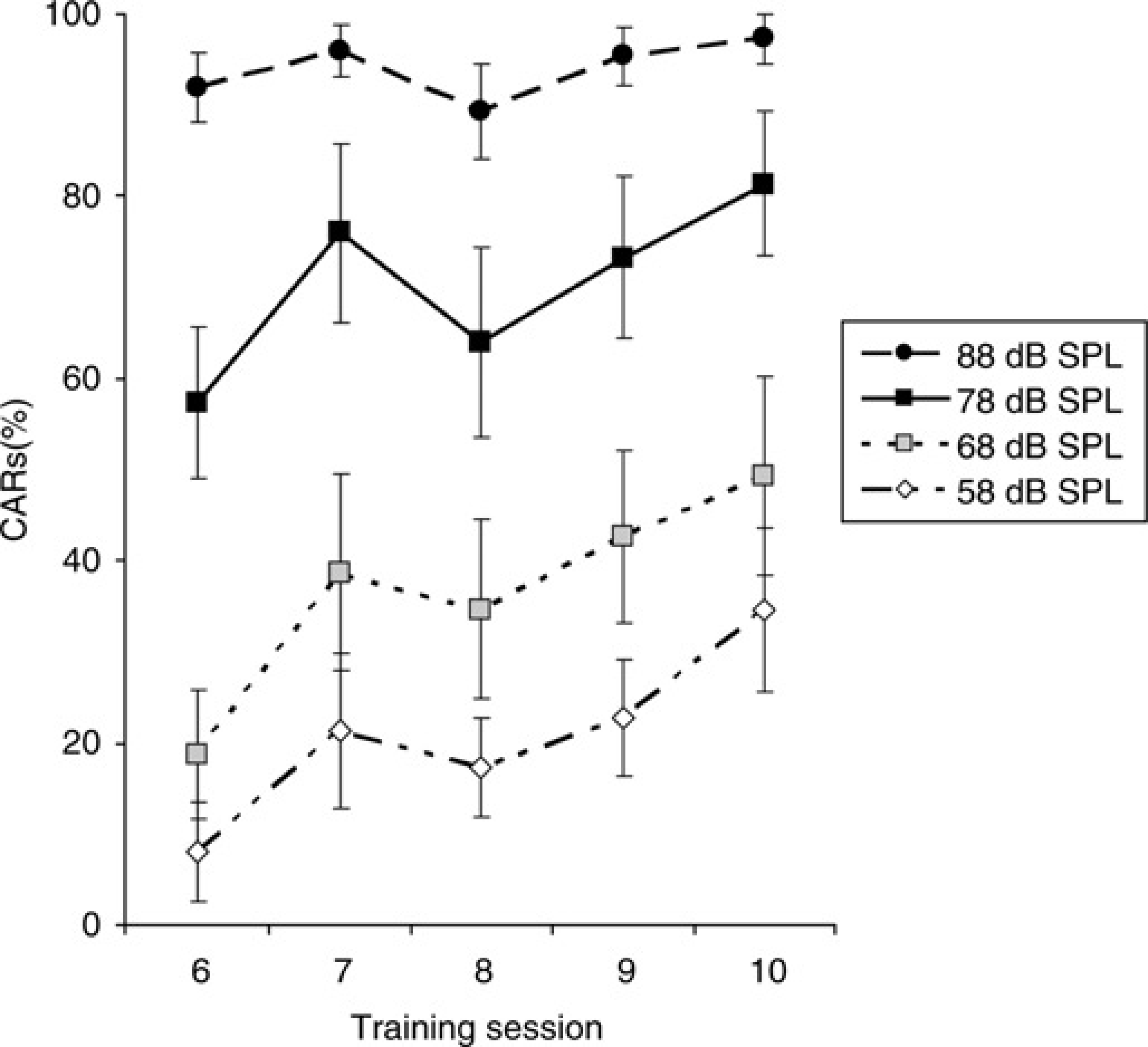
Latencies
Figure 4 shows the mean avoidance latency for each sound level. Avoidance latency equalled the time between the onset of the CS and the avoidance response. Paired t-tests showed statistically significant differences between the latencies at the four different sound levels. Latencies of CARs at 58 dB SPL were greater than the latencies for CARs at 68 dB SPL (paired t-test: P < 0.001), latencies of CARs at 68 dB SPL were greater than the latencies for CARs at 78 dB SPL (paired t-test: P < 0.01) and latencies of CARs at 78 dB SPL were longer compared with latencies for CARs at 88 dB SPL (paired t-test: P < 0.001). Latencies compared with latencies other than the latency of the next sound level were all statistically significant (paired t-test: P < 0.001).
Mean (± SEM) avoidance latencies (in seconds) averaged across all 15 guineapigs for the four different sound levels. Statistically significant differences with the next sound level are indicated. **P < 0.01, ***P < 0.001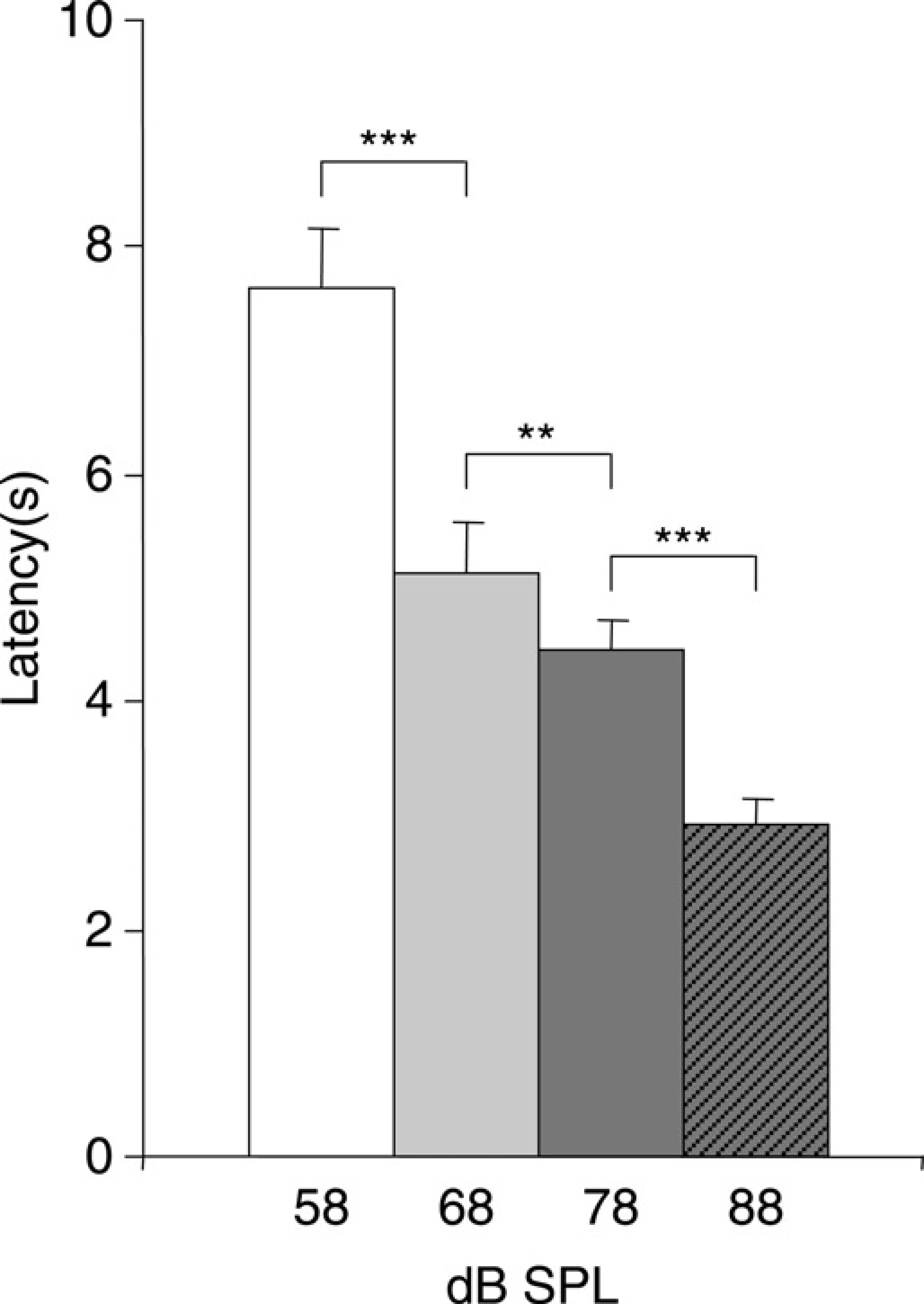
Discussion
The present study demonstrates that guineapigs learn easily, within five sessions of roughly 10 min, to avoid an unpleasant stream of air when the sound level of the CS was 78 dB SPL. During training session 6 they were tested for the first time at sound levels of 58, 68 and 88 dB SPL. Guineapigs did not immediately generalize the acquired CAR at 78 dB SPL to the sound levels of 58 and 68 dB SPL. They needed several training sessions to score 35% and 49% at 58 and 68 dB SPL, respectively. In contrast, it was found that during session 6 the CAR at 88 dB SPL was close to 100%.
The high level of CARs (49%) on the first training session with a sound level of 78 dB SPL (Figure 1), even before the animals experienced the relationship between the CS and the UCS, (i.e. associative learning 17 ), implies that this sound level initially evoked an aversive reaction. In other studies, in which the sound level of the CS was lower (~65 dB SPL), the performance was <20% during the first training session.7,13 The suggestion that a sound level of 78 dB SPL initially evoked an aversive reaction is in line with the observation that 14 of the 15 guineapigs shuttled to the other compartment during the first trial in the first session (Figure 2). In this trial they were exposed to the CS of 78 dB SPL for the first time. Analyses of the individual trials at sessions 2 and 3 also revealed that more animals shuttled to the other compartment during the first trial, as compared with trials later on in these sessions. In session 5 this outcome disappeared (i.e. the animals learned to respond to the CS of 78 dB SPL).
As mentioned before, when the animals were exposed for the first time to sound levels of 88 dB SPL (during session 6), the CAR was close to 100% (Figure 3). It might be that guineapigs demonstrate an aversive reaction when they are exposed to a CS of 88 dB SPL instead of generalizing the acquired avoidance response at 78 dB SPL to higher sound levels. This would mean that when the sound level of the CS is higher than 78 dB SPL, the guineapigs would not learn to avoid the UCS because they respond directly by escaping to the other compartment after exposure to the CS. That a sound level of 88 dB SPL evoked an aversive reaction is supported by several studies that reported that this sound level approximated sound levels that were used in acoustic startle paradigms performed in mice and rats.18–20 On the other hand, the response latency at 88 dB SPL was about 3 s, which is slow for an aversive reaction and five times slower than the escape response elicited by the UCS.
It is important to note that the UCS used in the present study produced noise. This noise was produced by pressured air leaving the tube. The sound level of this noise was about 90 dB SPL. This sound level equalled the sound level of the CS of 88 dB SPL. The exposure to this behaviourally relevant sound level during the escape responses probably contributed to the high level of CARs at 88 dB SPL during session 6.
In conclusion, the sound level of 78 dB SPL initially evoked an escape reaction but the presented data indicate that guineapigs apparently habituated to this sound level during the first five training sessions. The repetitive exposure to a sound level of ~90 dB SPL produced by the UCS might have altered the sensitivity of the guineapig and probably affected the training at other sound levels during later sessions. Within only a few sessions guineapigs acquired a CAR, and if appropriate consideration is given to the high sensitivity of guineapigs 5 the presented model is easy to use in a psychoacoustic test. This model is less stressful for the guineapigs and therefore presents a more ethical task for behavioural testing in guineapigs.
Footnotes
Acknowledgements
The authors would like to thank FWJ Albers, who sadly died on 30 October 2007, for his support of this study. René van de Vosse and Rolf Struikmans are acknowledged for developing the recording software and Elisa Bravo Rebolledo for her excellent technical assistance. We would like to thank Professor Dr VM Wiegant, Dr SFL Klis and Dr H Versnel for the helpful discussions concerning the study design. This study was supported by the Heinsius-Houbolt Foundation.
