Abstract
The aim of this study is to give a hands-on description of the successful monitoring procedure established for extended liver resections and liver transplantations in rats and to provide the typical range of data as obtained before and after a hepatobiliary surgical procedure (right median hepatic vein [RMHV] ligation) in healthy male Lewis rats. All manipulations were performed in anaesthetized (3% isoflurane in O2 1 L/min) healthy male Lewis rats (250–350 g) with an integrated multiple-channel intraoperative monitor (Powerlab® system) using a series of sensors for data acquisition. Vital parameters (body temperature, electrocardiogram, respiratory rate and heart rate), haemodynamic parameters (mean arterial blood pressure [MAP] and central venous pressure) and liver perfusion parameters (inferior hepatic venous pressure, portal vein pressure [PVP], blood flow of portal vein and inferior hepatic cava) were monitored. Catheters were placed in microsurgical technique after careful exposure guided by anatomical landmarks. Vascular incisions were closed with interrupted sutures. Complete instrumentation of animals was performed within 1 h. No specific complications occurred. Vital and haemodynamic parameters such as MAP (94 ± 16.2 mmHg) or portal pressure (9.6 ± 1.34 mmHg) were in the same range as known for humans (MAP = 100 mmHg, portal pressure = 5–10 mmHg), whereas parameters dependent on the size of the body or organ such as flow rates (portal blood flow = 16.2 ± 6 mL/min) were obviously different compared with those of humans (portal blood flow = 800 mL/min). In conclusion, the normal range for vital, haemodynamic and liver perfusion parameters was reported as reference values to allow quality control for future surgical hepatobiliary research projects. As the procedure can be easily learned, the extensive intraoperative monitoring can be used routinely.
One hundred years ago, Dr Harvey Cushing was the first to monitor blood pressure (BP) and heart rate in patients during and after operations. 1 In 1986, the American Society of Anesthesiologists concluded that oxygenation, ventilation, circulation and temperature should be monitored continually during a surgical procedure. 2 As a warning system, intraoperative monitoring helps physicians to identify problems at the earliest time point possible. 2
Instability of surgical manipulation or technical errors during the procedure may lead to undetected effects on the animal, subsequently influencing the results of a given scientific project. It is crucial to standardize all surgical procedures and to ensure that all procedures are performed similarly throughout an experiment. Intraoperative monitoring is an essential tool to detect any physiological abnormalities indicating either a technical procedure related problem or a previously unnoticed pathophysiological state of the subject undergoing the procedure. Modern intraoperative monitoring allows us not only to visualize physiological parameters but also to record them for later comparison between procedures. Exact monitoring of a given experimental surgical procedure performed by an experienced surgeon helps to establish a range of ‘normal values’ characteristic for the course of a given operation. These parameters can then be used as qualify benchmarks in the training of young fellows. Completion of training for a given procedure is based not upon reaching a survival rate but upon performing the procedure according to predefined intraoperative physiological parameters.
Nowadays, sensors and data acquisition systems that allow for an extensive intraoperative monitoring of rats are commercially available. In our research group, full and partial rat liver transplantations as well as liver resections, including 90% extended liver resections, are performed frequently. Intraoperative monitoring helped us to assess the effect of intervention and to demonstrate the reproducibility of surgical procedures.
The aim of this study is to give a hands-on description of the successful monitoring procedure established for experimental hepatobiliary surgery including extended liver resections and liver transplantations in rats. Furthermore, we provided the typical range of data as obtained before and after a hepatobiliary surgical procedure (right median hepatic vein [RMHV] ligation).
Materials and Methods
For the first part of the experiment, eight rats undergoing 70% partial hepatectomy were used to establish the procedure. Prior to using living animals, topographical anatomy for later vascular access was studied on rat cadavers. The description of the surgical details of the monitoring procedure is based on our initial experience. Animals were observed for potential complications related to the monitoring procedure during the postoperative period. Rats were euthanized 28 days after the intraoperative monitoring was performed. In the second part of the experiment, the range of normal values was determined based on data acquired from the additional six rats which were independent of the first part of experiment and received RMHV ligation.
Animals
Male inbred Lewis (Lewis/Han™Hsd) rats (weight range 250–350 g, Laboratory Animal Department, University Hospital Essen, Germany) were employed in this study. All rats were bred in the Laboratory Animal Department, University Hospital Essen. All rats were housed in groups of four in single-sex cages on sawdust bedding. Animals were kept under standard care conditions (humidity 45–70%, temperature 21 ± 1°C, 12 h light/dark cycle). Tap water and standard laboratory animal chow (Ssniff® complete feeds V1324, Ssniff Spezialdiaeten GmbH, Soest, Germany) were offered to the rats ad libitum. Housing and all procedures carried out were in accordance with German Animal Welfare Legislation. The animal protocol was reviewed and approved by the Landesamt für Natur, Umwelt und Verbraucherschutz Nordrhein Westfalen; Germany (§8 Tierschutzgesetz).
Surgical procedure
Surgical procedures were performed in a specific rodent operation unit, set up following the recommendations outlined by Hoogstraten-Miller. 3 The preparatory, anaesthesia induction and recovery areas were separated from the operation room by an airlock to avoid contamination with animal hair and bedding dust. All surgical procedures were performed in the separated, uncluttered operation room with minimal traffic flow.
Rats were placed in an anaesthesia induction chamber. Induction of anaesthesia was performed using an isoflurane vaporizer (Sigma Delta, UNO, The Netherlands). Isoflurane (Nicholas Piramal (I) Ltd, London, UK) concentration was 5% and oxygen flow 0.5 L/min. Both surgical sites, the abdomen and the right side of the neck, were shaved and disinfected. Anaesthetized and shaved animals were transferred into the ‘clean’ operation room. As we performed multiple rodent survival surgeries (minimum of 3 procedures in sequence), we followed the main principles of the current guidelines (e.g. Guidelines for rodent survival surgeries, 4 Aseptic technique for multiple rodent survival surgeries, 5 which are in accordance with the Principle of sterilization and disinfection 6 ). The instruments were wiped clean of blood and body fluids with a small amount of alcohol and disinfected between each surgical procedure with 72% isopropyl alcohol (Cutasept®, Bode Chemie, Hamburg, Germany). Isoflurane was used in a concentration of 2–3% and an oxygen flow of 0.5 L/min to maintain full anaesthesia during the surgical procedure.
The Powerlab® system (Powerlab® 16/30, ML-880, ADI Instruments, Castle Hill, NSW, Australia) as an integrated multiple-channel intraoperative monitor device was used for data acquisition. Data were transferred to a standard PC. Data acquisition, intraoperative data visualization and postoperative data analysis were performed using Chart 5 for Windows® (version 5.5.4, ADI Instruments).
A rectal temperature probe (MLT1403, ADI Instruments) was used to measure the core temperature of the rat (Figure 1a). The temperature sensor was connected to a T-type Pod signal conditioner (ML312, ADI Instruments). The signal conditioner was linked to the Powerlab® system. Before the measurement, the stainless steel shaft of the sensor was moistened with saline to facilitate the insertion. The body temperature was measured by gentle insertion of the stainless steel shaft of the temperature probe 5 cm into the rectum of the animal. Chart 5® displayed the stable read-out of body temperature 5–10 s after insertion of the thermometer.
Intraoperative vital monitoring on rat. (A) Placement of temperature sensor. (B) Placement of ECG sensor. (C) Placement of respiratory rate sensor. (D) Catheterization of right carotid artery for mean arterial pressure (MAP), (E) Catheterization of right jugular vein for central venous pressure (CVP)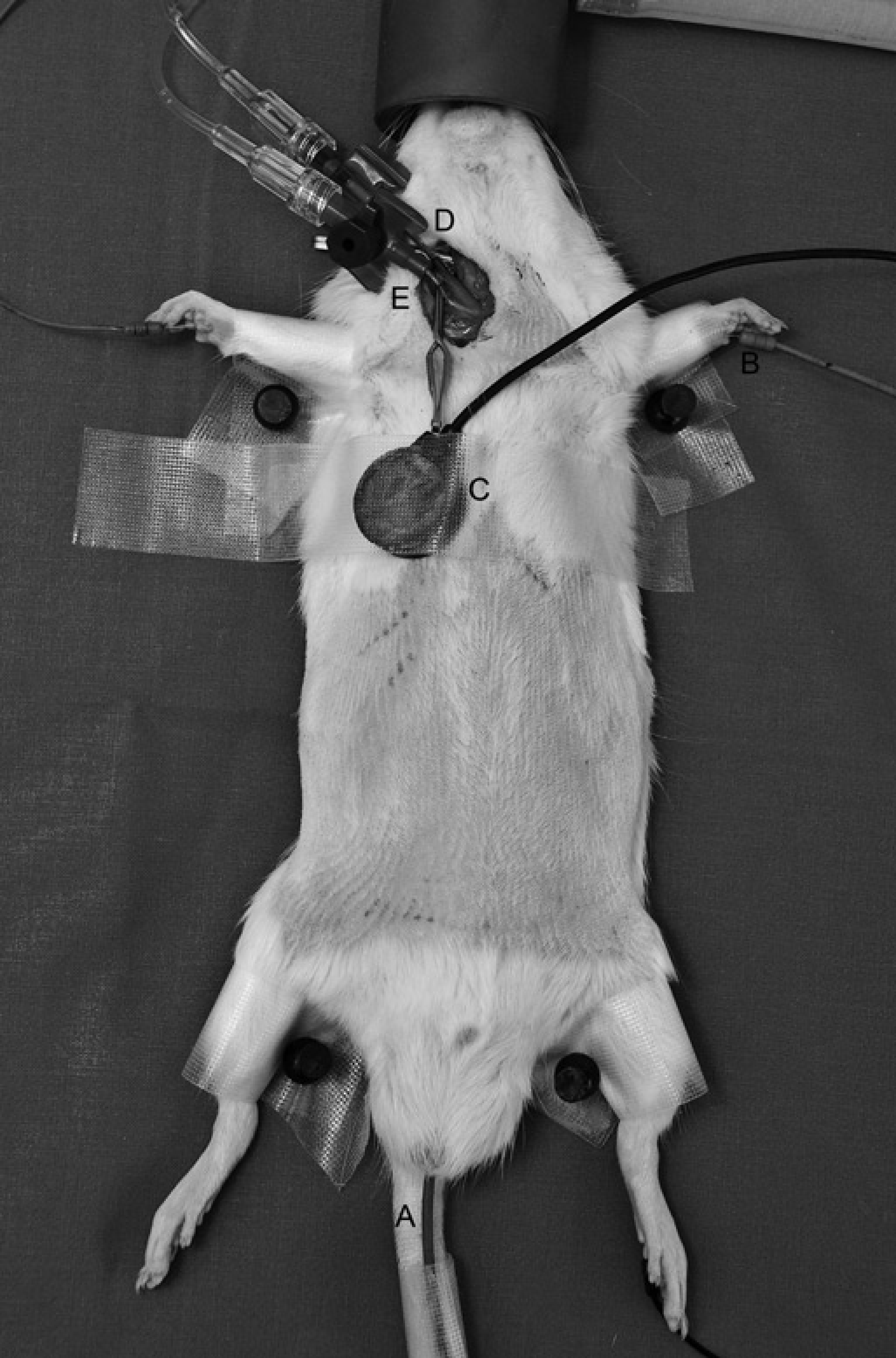
Electrocardiogram (ECG) was recorded using three stainless steel needle electrodes with different colors (MLA1204, ADI Instruments) (Figure 1b). The sensors were connected to the bio-amplifier Animal Bio Amp (ML136, ADI Instruments) via its corresponding input sockets. The amplifier was amplified and filtered the raw data before transfer-ring the signal to the Powerlab® system.
Heart rate was recorded on the basis of the ECG. Three needle electrodes were inserted subcutaneously into both fore-paws and the left hind-paw in accordance with the Lead-II placement 7 for ECG. Heart rate was computed by Chart 5® using the recorded ECG data. The scale of heart rate was precalibrated indicating a range of 0 to 500 beats per minute to filter signal noise from the ECG data.
Respiratory rate was recorded using a Pulse Transducer (MLT1010, ADI Instruments) linked directly to the 8-pin DIN Input on the Powerlab® system (Figure 1c). The pulse transducer was gently fixed on the chest of the anaesthetized rat positioned 1 cm right of mid-line by an adhesive tape.
Blood pressure was recorded invasively using intravascular saline filled catheters (Braun AG, Melsungen, Germany), which were connected to reusable BP Transducers (MLT0380/D, ADI Instruments). The transducers were connected to a QUAD Bridge amplifier (ML118, ADI Instruments), which was wired to the Powerlab® system. The diameter of the catheters and access routes were selected according to the target vessel.
To measure the mean arterial blood pressure (MAP), the right carotid artery was cannulated (Figure 1d). A virtual line was drawn from 1 cm caudal to the mandible to a point just cranial to the sternal manubrium. An incision of the same length was made 0.5 cm lateral and parallel to this virtual middle line. The skin was retracted to both sides as far as possible using a small retractor (Aesculap GmbH, Tuttlingen, Germany). The omohyoid muscle was pulled to the right to expose the carotid bed. The right carotid artery was carefully isolated from the vagal nerve using micro-forceps (Aesculap GmbH). Two vessel clamps (FST GmbH, Heidelberg, Germany) were placed on the distal part of the right carotid artery close to the mandible and the proximal part close to the clavicle. One 6–0 silk (Resorba, Nuremberg, Germany) loop with a preformed knot was placed around the carotid artery cranial to the proximal clamp. With micro-scissors (Aesculap GmbH) pointing towards the heart, an oblique cut was made on the ventral surface of the artery close to the distal clamp. A 22G catheter (Braun AG) was inserted, bevel up, into the artery. After removal of the proximal clamp the catheter was advanced. The preformed knot in the silk loop cranial to the proximal clamp was tightened gently to fix the catheter throughout the monitoring procedure (Figure 2a). The MAP could be monitored after briefly flushing the catheter with heparinized (Liquemin® N20000, Hoffmann-La Roche AG, Grenzach-Wyhlen, Germany) saline (5 IU/mL).
Catheterization of the right carotid artery and right jugular vein for measurement of blood pressure. Soft plastic catheter and plastic tube for invasive catheterization (A). The oblique cut on right carotid artery was closed using three to four interrupted sutures with 8–0 Prolene (B)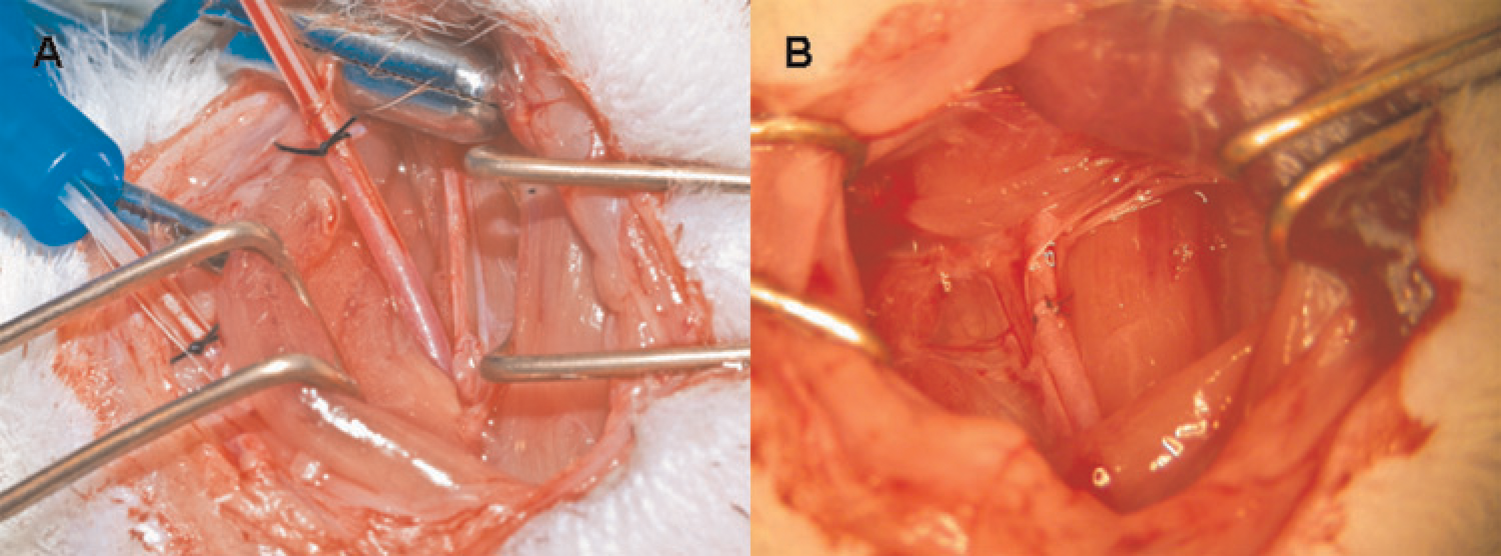
At the end of the surgical procedure, the proximal clamp was placed on the right carotid artery in the previous location. The catheter was withdrawn after opening and removing the 6–0 ligature. The oblique cut was closed using three to four interrupted 8–0 Prolene sutures (Resorba) (Figure 2b).
To measure the central venous pressure (CVP), the right jugular vein was used, employing a procedure similar to the one described for the carotid artery (Figure 1e). After implantation of the catheter into the right carotid artery, the sternocleidomastoid muscle was released and slipped into its anatomical location. The right jugular vein was carefully isolated from the lymph nodes and surrounding fat tissue. Two vessel clamps were placed on the distal and proximal part of the vein. A 22G catheter was inserted through the transversal incision on the ventral surface of the right jugular vein and then temporarily fixed with 6–0 ligation. After monitoring, the incision was closed with three to four interrupted 8–0 sutures. Four single sutures with 3–0 Nylon (Resorba) were used to close the skin incision after completing the monitoring procedure.
To measure the inferior hepatic venous pressure, the right iliolumbar vein was catheterized after median or transverse laparotomy. In rats, the right iliolumbar vein drains transversely into the inferior hepatic vena cava 2 cm below the right kidney. The intestine with the great omentum was removed from the abdominal cavity and placed to the left side of the abdomen. Gauzes were saturated with 0.9% saline prior to covering the abdominal organs. The right iliolumbar vein was exposed and cannulated with a 22G catheter in the same way as described for the other vessels. The tip of the catheter was advanced until it reached the lumen of the inferior hepatic vena cava (Figure 3a).
Catheterization of vessels and placement of blood flow sensors during the operation. (A) Catheterization of the right iliolumbar vein for measurement of inferior hepatic vena cava (IHVC) pressure. (B) Placement of blood flow probe on portal vein (PV). PV was placed between the body (ultrasonic transducer) and the hook (ultrasound reflector) of the probe. (C) Catheterization of the mesenteric vein for measurement of blood pressure of PV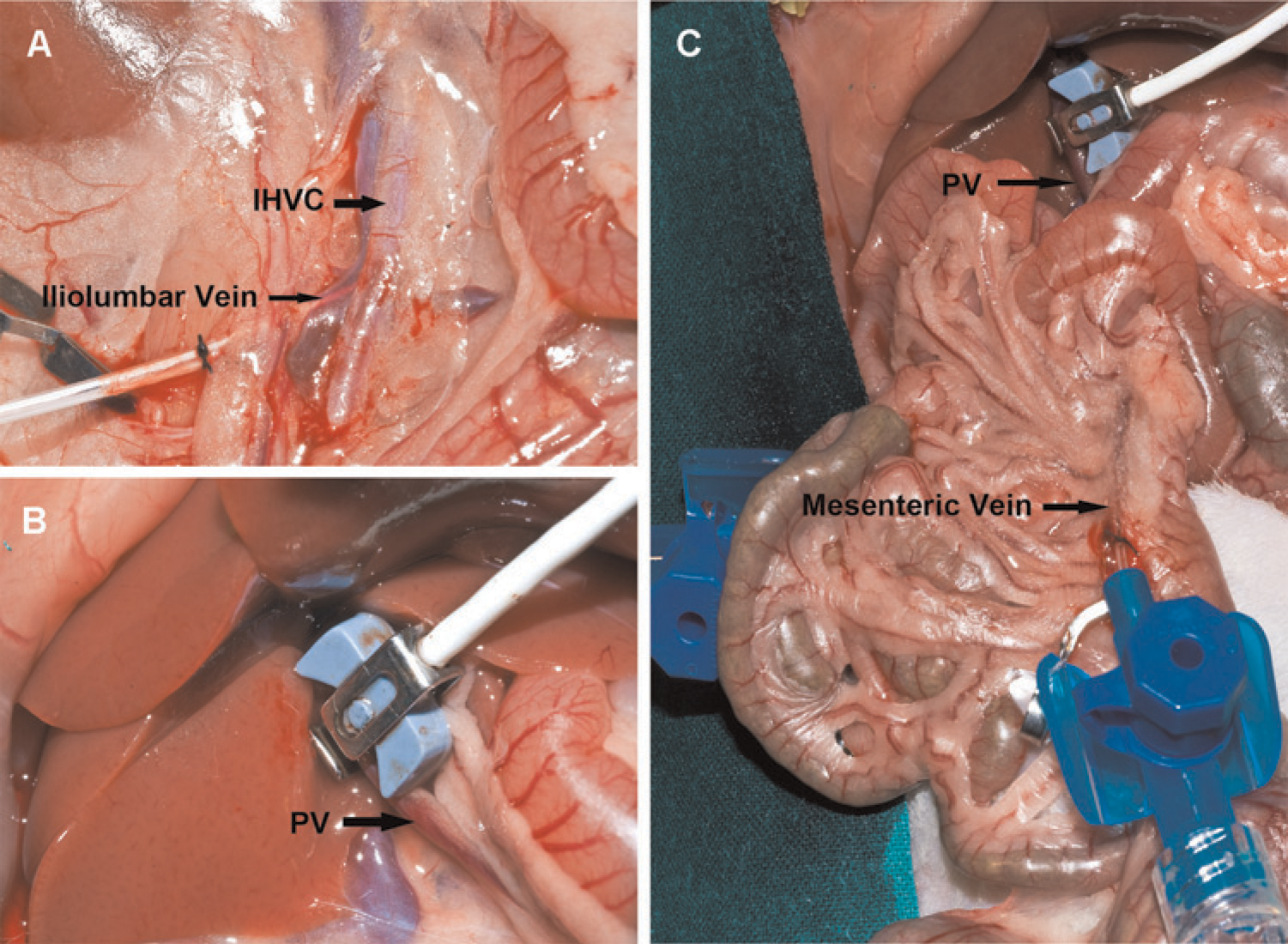
To measure the portal vein pressure (PVP), the superior mesenteric vein was isolated from surrounding fat tissue. Because of the limited length of the main stem of the mesenteric vein, three microvascular clamps were placed, one on the proximal point and the second on the distal point of the main stem. The third was placed on the small branch draining into the main stem of the mesenteric vein between the other two clamps. After performance of a small incision near the distal clamp, a 24G catheter (Braun AG) was inserted through the hole to measure the BP in the portal vein. One 6–0 silk suture was pre-set for intraoperative fixation of the catheter (Figure 3c). Four interrupted sutures with 8–0 Prolene were placed on the incision after withdrawal of the catheter.
Blood flow was recorded using a specific probe consisting of two ultrasonic transducers (body of the probe) and a fixed acoustic hook (reflector) (CM4, Transonic Systems Inc, Ithaca, NY, USA) was used. The probe was connected to a small animal research flowmeter (T106, Transonic Systems Inc) which was linked to the Powerlab® system. To measure portal blood flow and blood flow of the inferior hepatic vena cava, both vessels were freed from surrounding connective tissue. The ultrasonic probe was gently positioned around the inferior hepatic vena cava or portal vein (Figure 3b).
In the first part of the experiment, rats were subjected to 70% partial hepatectomy. Left lateral lobe and median lobe were resected using our previously described technique. 8 Attention was paid to place the piercing sutures exactly on the hepatic vessies after clamping the median lobe.
In the second part of our experiment, RMHV ligation was performed. After a transverse abdominal incision, the falciform ligament was dissected to mobilize the median liver lobe. The drainage point of the RMHV into the superior hepatic vena cava was exposed. A 1–2 mm incision vertically above the hepatic vein was made by a coagulator (battery-operated small vessel cauterizer, Fine Science Tools GmbH, Heidelberg, Germany) until the vessel surface was visible. The RMHV was then isolated from the liver mass by micro-forceps. The tips of the micro-forceps were inserted through the liver mass underneath the RMHV and the RMHV was ligated using Prolene 6–0 (Resorba).
Postoperative period
Postoperative analgesia was achieved by subcutaneous injection of buprenorphine (Temgesic®, Essex Pharma GmbH, Muenchen, Germany) at one dose of 0.01 mg/kg body weight immediately after surgery. Postoperatively, animals had free access to 20% glucose solution and rat chow ad libitum. During the observation time, clinical condition of rats was observed and judged by using a semiquantitative scoring system that was described previously. 9 Briefly, rats with normal activity, physiological position, no jaundice, and no signs of bleeding were regarded as healthy (±±±); animals showing a weaker activity, hunched back position and/or signs of jaundice or bleeding were regarded as weak (±±); and animals with no spontaneous activity and lying position and signs of jaundice or bleeding were regarded as severely ill (±). Body weight of all rats was monitored and recorded daily. Normalized body weight recovery rate was calculated (body weight after operation/original body weight x 100, expressed as %) and was used as the indicator of recovery after operation.
During the observation time, activity of the rats was monitored. Anatomical sites potentially affected by the monitoring procedure were carefully checked in respect to thrombosis, leakage or infections, throughout the observation period. For the first part of the experiment, eight rats were euthanized 28 days after the intraoperative monitoring. Animals were deeply anaesthetized by increasing the concentration of isoflurane to 5% and euthanized by exsanguination. For the second part of the experiment, the other six rats which were independent of the first part of experiment were euthanized by exsanguination immediately after collection of haemodynamic results.
Data analysis and statistics
Chart 5® software (ADI Instruments) was used to analyse the data acquired throughout the whole length of the intraoperative monitoring procedure. Raw signals captured for 0.5 s with time intervals of 0.5 s were analysed after manual selection. In total 60–270 data-points indicating 1–4.5 min were selected for each parameter before and after operation. Selected signals were converted automatically by Chart 5® and exported as a data table. All captured data were saved and then transferred into an Excel sheet (Version Excel 2003, Microsoft Corporation, Mountain View, CA, USA) or Sigma Plot (Version Sigma Plot v.9, Systat Software, Inc, Chicago, IL, USA) sheet for further analysis. Normal values of different parameters were expressed as mean ± SD. Statistical analysis used the one-way analysis of variance test. All statistical analyses were performed using Sigma Stat v.3.5 (Systat Software, Inc). P < 0.05 was denoted as statistically significant.
Results
Technical observations
Body temperature monitoring
Two problems interfering with the proper read-out of the temperature were pinpointed during the first part of the experiment. Firstly, the stainless steel shaft of the thermometer occasionally slipped out when stool in the rectum stimulated the defecating movement. Thus, before inserting the rectal probe into the anaesthetized rat the stool near the anal orifice should be cleaned out using micro-forceps. Secondly, applying too much force during insertion of the thermo-sensor may lead to laceration, bleeding and even perforation of the rectum and should be avoided.
ECG and heart rate monitoring
Pain was inflicted when the electrode needles of the ECG sensors were inserted into the paws of animals. Therefore, they should be placed only after the rat is fully anaesthetized.
Respiratory rate monitoring
Since the transducer is sensitive to movements, any movement of the sensor on the chest can interfere with the reading. Unintended manipulation of the sensor by the surgeon could be one reason for such movements. Lack of fixation of the sensor could lead to a lack of proper contact and influence the signal. In contrast, fixing the sensor too tight might impair the respiratory activity of the rat.
Blood pressure monitoring
Prior to cannulation of the targeted vessel, the catheter was filled with heparinized saline (5 IU/mL) and was connected to the transducer. The system was calibrated prior to the first measurement and once per month thereafter as recommended. The calibration procedure was performed by measuring the height of a water column in a plastic tube connected with the BP transducer. Plastic tube and BP sensor were placed in the same horizontal plane which was regarded as 0 cmH2O. Subsequently, the plastic tube was raised right above the BP transducer vertically to the table and the height of the tube was measured with a standard ruler. The corresponding voltage displayed on Chart 5® was recorded for two repeated measurements and subsequently converted to a pressure unit (mmHg) by Chart 5®.
Excessive traction on either the sternocleidomastoid or omohyoid muscle might cause injury to the muscle and lead postoperatively to limitations of neck movements. The anatomic position of the right carotid artery is relatively deep with respect to the skin incision. If fixation of catheter was not tight enough, the catheter could slip out causing severe bleeding, which would impair its reinsertion. Thus, the catheter should be secured in the artery by a suture immediately after insertion.
The BP value would be falsely low if the catheter could not be inserted into the portal vein. The vessel receiving the catheter should be selected so that the tip of the catheter can reach the drainage point of the mesenteric vein into the portal vein. Thus, the catheter should be inserted as deep as necessary to reach this drainage point. The vessel wall of the superior mesenteric vein is thin and fragile; manipulation and catheterization should therefore be performed very gently and carefully to prevent laceration of the vessel wall.
Air bubbles and thrombosis formation inside the tube would not only impair data acquisition but also threaten the life of the experimental animal. Single air bubbles would decrease the raw signal and lead to false readings. Therefore the BP transducers should be sealed with saline. Complete filling of the transducer cable system with saline can be reached by correct positioning during this procedure. The diaphragm of the transducer should be covered with saline solution. Furthermore, the dome of the transducer should be screwed tightly to the body of the transducer. Next, the connecting tube and the catheter are connected with the transducer via one port on the dome of the transducer. The catheter is flushed with a saline–heparin mixture (5 IU/mL) using a 20 mL syringe to remove all remaining air bubbles.
A height difference between the vessels of the rats and the pressure transducer placed on the operation table led to falsely high BP readings, increasing the true value of the BP by 1–3 mmHg. This effect was of utmost importance when monitoring the pressure in the low pressure venous system. Hence, the BP transducer should be placed at the same level as the catheter after insertion into the vein.
Blood flow monitoring
The probe had to be placed in de-aerated saline 4 h prior to use to adjust to the environmental conditions. Saline was introduced into the abdominal cavity near the probe in order to immerse both the vessel and the blood flow meter for better contact.
Insufficient contact between vessel and probe led to an attenuation of the ultrasound signal and subsequently to the loss of blood flow data. According to the manufacturer's recommendations the space between probe and vessel should be filled completely with saline, ultrasound gel or clotted blood to ensure appropriate ultrasonic coupling. In our hands saline was sufficient to obtain reliable measurements.
Another potential problem occurred when the size of sensor did not match the size of the targeted vessel. If the space for fixing the vessel between the sensor and the hook of the probe is too small, the vessel would be compressed leading to an artificial ‘stenosis’. Blood flow would be increased due to artificial narrowing of the vessel.
Recovery after intraoperative monitoring
Eight rats were used to establish the procedure and to assess the influence of monitoring on survival after a hepatobiliary standard procedure, the 70% partial hepatectomy. Complete instrumentation of animals was finally achieved within one hour and did not lead to specific intraoperative complications. Animals were observed for up to 8 h postoperatively. Animals appeared without pain after waking up fully within 1–2 h. Activity as well as food and water intake did not appear to be less than normal. Thus, the analgesia treatment was limited with one dose of Temgesic® injection. Animals lost about 10% of their original body weight within the first five postoperative days, as typically observed after minor/moderate operations. Body weight recovered thereafter following the same course (slope of body weight increase) as observed after this operation without performing full cardiovascular monitoring (Figure 4). No rat died subsequent to the placement of multiple sensors invasively and non-invasively. Catheterization did not cause any complication after operation. No limitation of movement in the neck or right legs related to the catheterization was observed during the postoperative observation period of 28 days. No thrombosis, anastomotic leakage, wound infection or vascular insufficiency was observed during the euthanizing procedure. However, in one incident the catheter slipped from the right carotid artery during a training operation and severe bleeding occurred. Bleeding stopped after the catheter was reinserted into the artery and properly fixed.
Normalized body weight recovery rate after 70% partial hepatectomy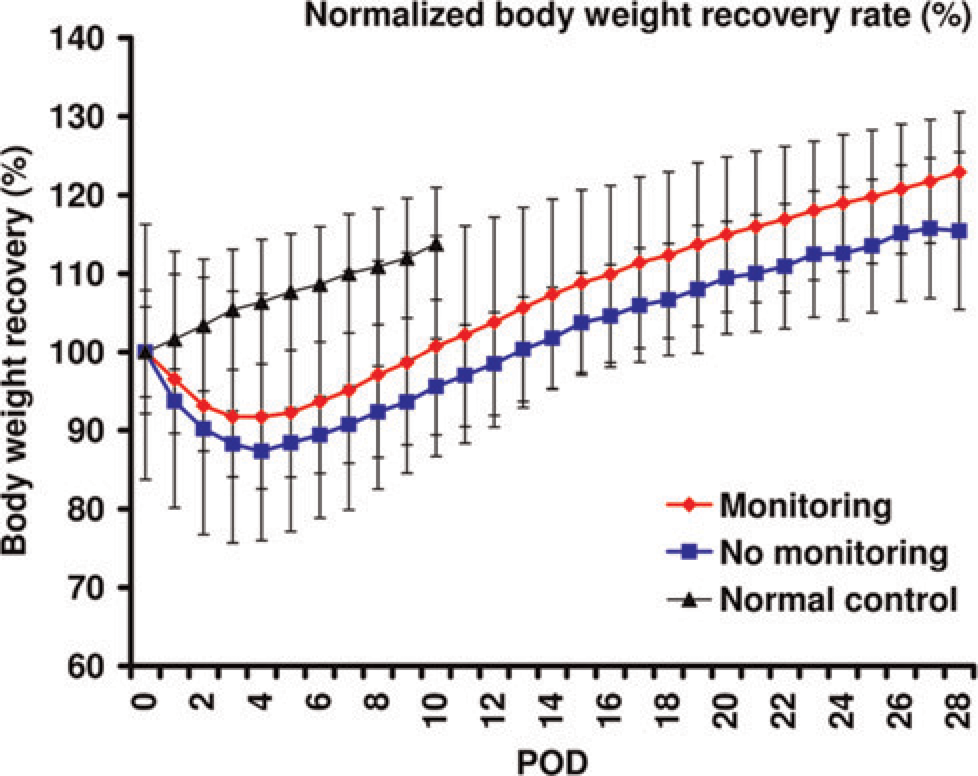
The typical range of vital and haemodynamic data as obtained before and after a hepatobiliary surgical procedure
Altogether six normal healthy Lewis rats were subjected to a complete intraoperative monitoring procedure before and after RMHV ligation. This hepatobiliary procedure takes about 45 min. We observed that respiratory rate and MAP were significantly lower after operation than before operation (Table 1; 31 ± 3 versus 42 ± 3 cycles/min and 67 ± 4.8 versus 94 ± 16.2 mmHg, P < 0.05). PVP was slightly increased after ligation compared with before ligation (12.1 ± 1.32 versus 9.6 ± 1.34 mmHg, P < 0.05). Other vital and haemodynamic parameters remained stable throughout the operative procedure of RMHV ligation. Vital and haemodynamic parameters such as MAP or portal pressure before operation were in the same range as known for humans (MAP = 100 mmHg, portal pressure = 5–10 mmHg). Parameters dependent on the size of the body or organ such as flow rates (portal blood flow = 16.2 ± 6 mL/min) obviously differed from the range observed clinically (portal blood flow = 889 ± 284 mL/min10).
Range of normal values of vital and haemodynamic parameters in rats (n = 6) obtained intraoperatively using 3% isoflurane/oxygen (1 L/min) anaesthesia
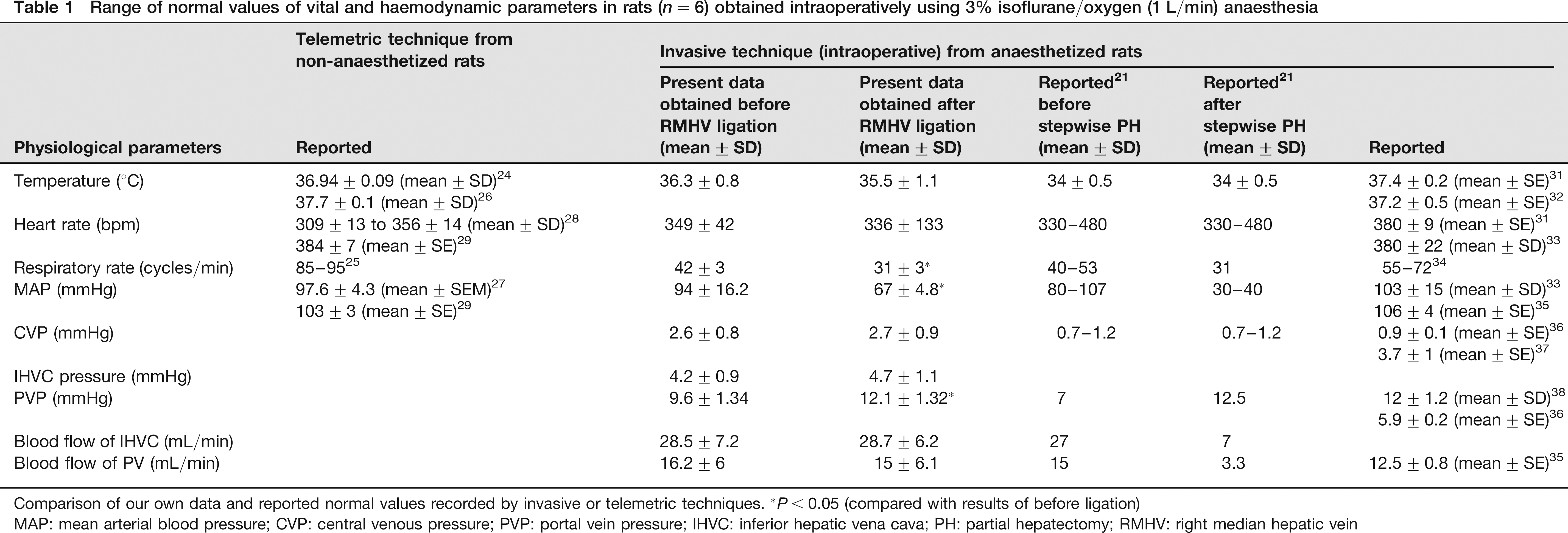
Comparison of our own data and reported normal values recorded by invasive or telemetric techniques. *P < 0.05 (compared with results of before ligation)
MAP: mean arterial blood pressure; CVP: central venous pressure; PVP: portal vein pressure; IHVC: inferior hepatic vena cava; PH: partial hepatectomy; RMHV: right median hepatic vein
The range of normal values (body temperature, HR, RF, MAP, CVP, diastolic venous pressure, PVP and blood flow of IVC and portal vein) in normal isoflurane-anaesthetized rats (n = 6) is shown in Table 1.
Discussion
It is one of the key aims of surgical research to assess the impact of a surgical procedure on a given set of parameters. In surgical research the procedures are often performed in rat models, especially in hepatobiliary and transplantation research. Operations such as extended liver resection and liver transplantation are extremely complex and technically challenging, multistep procedures. In a multistep procedure, the risk of a mistake or a problem looms in each and every step. Thus, high quality and reproducibility of complex surgical procedures in research quality control have to be ensured.
Surgical errors or complications however might influence the read-out parameters of interest. Thus results and conclusions based on read-outs attributed to the respective experimental settings might in reality just reflect procedural problems. Therefore it is of utmost importance to establish rigid quality control. In acute experiments involving hepatobiliary surgical procedures, tight control of cardiovascular parameter might be demanded, e.g. stable systemic circulation must be demonstrated if microcirculatory parameters are needed to answer an experimental question, as microcirculation is clearly affected by systemic circulation.
Measurement of vital parameters and extensive intraoperative monitoring are carried out routinely in clinical surgical practice during complex operations. Intraoperative monitoring helps to detect problems at an early stage and allows for a proper response. Even surgical problems such as extensive bleeding 11 can be underestimated by the surgeon in action. However, it may lead to detectable abnormalities such as hypotension, hypothermia, consumption of clotting factors and acidosis that put the patient at risk. During liver transplantation, haemorrhage can lead to reduced cardiac output and electrolyte imbalance 12 potentially associated with cardiac arrhythmia.13,14 After implantation of the liver graft, a decrease in MAP and heart rate can occur and might contribute to the so-called postreperfusion syndrome. 12 In brief, procedure-related problems such as blood loss may be associated with increased morbidity and mortality after liver resection and liver transplantation. 15 In the clinical situation, these problems must be detected as early as possible to enable fast counteractive treatment. In the experimental situation, these problems must also be detected as early as possible to decide whether the procedure should be excluded from analysis. From the scientific point of view it is a lot better to discover a procedure-related problem early and perform an additional procedure instead of having highly variable and inconclusive results.
Liver perfusion data (portal blood flow and portal pressure) can be extremely useful to demonstrate the stable standardized performance of a hepatobiliary vascular procedure such as the ligation of the RMHV. This is especially important if investigations of hepatic microcirculation are the focus of the scientific question. These values can also be useful for monitoring the surgical quality of more complex procedures such as a liver transplantation.
Quality of the intraoperative monitoring data is dependent on the precise placement of the sensors, irrespectively of whether they are placed in humans or in experimental animals. Incorrect manipulation of sensors during the operation can not only affect data acquisition and lead to falsely high or low results but also cause complications (Table 2).
Potential intra- and postoperative complications and solutions during intraoperative monitoring in rats
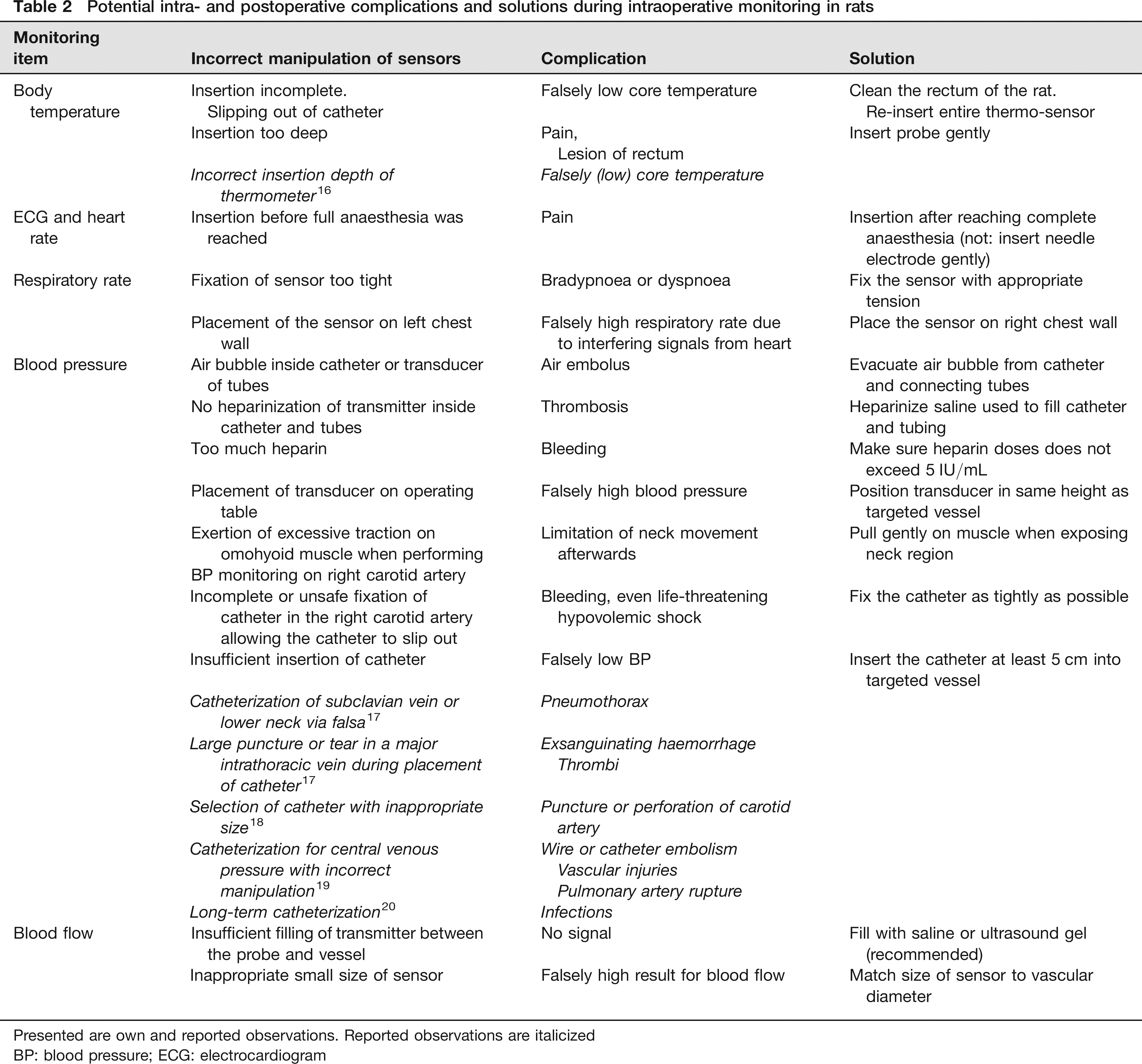
Presented are own and reported observations. Reported observations are italicized
BP: blood pressure; ECG: electrocardiogram
Normal body core temperature in rats has been reported to be highly dependent on the depth of thermometer inserted. Temperature was 32°C at 2 cm ab ano, 35.5°C at 4 cm and 37.5°C at 6 cm and the asymptote was reached after 7 cm, where the core temperature was 38°C. 16 Pneumothorax and damage to veins and arteries after central venous catheterization were reviewed. 17 Incidence of pneumothorax was higher in catheterization of subclavian vein than in internal jugular vein, and also higher in approach to lower neck than to higher neck. 17 Exsanguinating haemorrhage could be produced by large puncture or tear in a major intrathoracic vein. Accidental insertion of large CVP catheter even caused puncture or perforation of the carotid artery. 18 Data on cardiac complications such as wire or catheter embolism, vascular injuries and pulmonary artery rupture when performing the catheterization to measure CVP have been collected previously. 19 Vascular insufficiency was reported as a complication of arterial catheterization. 17 Catheter-related infection was also reported as a significant problem. Therefore, we used our clinically recommended skin disinfectant 72% isopropyl alcohol. Alternatively, Chlorhexidine gluconate was recommended for prevention of infections from the cannulation of sensors. 20 To prevent thrombosis subsequent to placement of catheters, it has been suggested that an anticoagulant (heparin) be administered intraoperatively through the corresponding catheters inserted in either the jugular vein or pulmonary artery, as also done by us. 17
As outlined above, the monitoring procedure itself is subject to technical errors and subsequent influences on the results. Therefore, it is advisable to practise the procedure and compare the results to the reported normal range. Once the expected normal range is observed in normal animals, the monitoring procedure can be used for surgical quality control purposes. Intraoperative quality control might be achieved by defining thresholds eliciting an automatic alarm during the surgical procedure.
This knowledge is of utmost importance in the experimental setting for the correct scientific analysis and interpretation of the experimental results. However, these data are hard to find and must be collected one by one from a variety of studies dedicated to different purposes. This experience motivated us to share our experience and present these results to the scientific community.
In previous studies, several intraoperative monitoring techniques have been used in rat experiments (Table 1), resulting in a similar range of normal values as we obtained in our present study. However, it was rather difficult to collect these data. We did not find a comprehensive description of the entire procedure and the normal values as required for complex monitoring in rat hepatobiliary surgery.
In the present study, a decrease in respiratory rate and MAP was observed after laparotomy. We found similar alterations after stepwise partial hepatectomy in previous experiments 21 (data shown in Table 1), but more pronounced, which might be related to the length of the procedure. However, PVP increased slightly, possibly due to the inflow resistance in the area of focal hepatic outflow obstruction after ligation of RMHV.
In the present study, we used invasive techniques for intraoperative monitoring. However, other ‘less or non-invasive’ monitoring techniques such as telemetric technique or tail cuff technique have been established for research. The telemetric technique has been widely accepted as useful for the acquisition of reliable physiological data from conscious laboratory animals. 22 By using telemetric technique, free movement of the animal and elimination of artefacts of anaesthesia, stress and potential infections could be achieved. 23 Normal vital parameters of rats such as body temperature,22–27 heart rate,22,23,25,27–29 respiratory rate 25 and MAP27–30 have been monitored by telemetric technique in different studies. However, the use of a telemetric monitoring system normally requires firstly implantation of the sensors and secondly waiting until complete recovery of the animals is achieved and only then performing the desired experiment. This approach is not practical for acute experiments with the key focus on obtaining intraoperative data from anaesthetized animals undergoing surgery. In our present study, we focused on realtime monitoring during the surgical procedures to ensure stable surgical quality. Furthermore, it is currently impossible to use telemetric techniques for monitoring blood flow and pressure of inferior hepatic vena cava and portal vein, parameters that were of great interest in our context of hepatobiliary surgery. In addition, no substantial differences between telemetrically and invasively recorded data were observed. Only respiratory rate recorded in conscious animals by telemetric technique 25 was different from the results we obtained in anaesthetized animals undergoing abdominal surgery (respiratory rate in conscious rats twice as high as in anaesthetized rats) (Table 1).
We established a complete intraoperative monitoring procedure in the rat. Using the Powerlab® system, a commercially available integrated multiple-channel monitoring system, safe and precise intraoperative monitoring can be performed routinely in any surgical experiments in rats. Normal physiological data of rat obtained from intraoperative monitoring provide quality control as well as an additional teaching and feedback tool, especially during the training phase.
Footnotes
Acknowledgements
This research was supported by the grant Klinische Forschergruppe 117-Optimierung der Leberlebendspende (Grant number: Da251/5–2 and 3, Project B2, KFO117). We thank Anne Gale of the German Heart Institute Berlin for editorial assistance.
