Abstract
In this study, we have investigated the short- and long-term impact of toe clipping, a commonly used method for marking and simultaneously taking biopsies of pups, which is controversially discussed because of its potentially negative impact on animals. Furthermore, we have analysed animalwelfare aspects such as health, behaviour, development, stress and detrimental effects in young animals and in adults after toe clipping at postnatal days 3 (P3) and 7 (P7). Our findings indicate that for both P3 and P7 pups amputations at the second phalange of one toe of each paw do not have any negative effects on growth and physical development and that the clipped pups do not suffer from rejection by their mother. Our data indicate that even though at both ages no abnormalities have been detected in histology, clipping at P7 is the preferable age for an adequate marking mostly because of the small size of the toes at P3. This was also confirmed by grip tests at the age of 12 weeks where P3 animals had lower grip strength than control animals, whereas P7 pups did not show any impairment. Hotplate tests indicated that toe clipping performed at P3 and P7 did not cause hyperalgesia at the amputation stump. Serum corticosterone analysis directly performed on P7 pups after clipping indicated that major stress was provoked mainly through the handling and not because of the clipping itself. Taken together, these data lead to the conclusion that toe clipping is from a morphological, physiological and welfare point of view an acceptable method for marking and genotyping newborn mice.
Proper animal identification is one of the most critical steps when working with laboratory animals because it is the only connection between the animal and all its data: genotype, samples and research results. Toe clipping is regularly practised worldwide, but it remains a highly controversial animal identification method. It allows the permanent identification of newborn mice and simultaneously delivers sufficient amounts of tissue for genotyping – thus having a clear advantage over other commonly used marking methods that are subjectively considered as less stressful for the animals1,2 like tattooing, or microchip implantation and do not provide any biopsies. Usually, animals are genotyped at the age of weaning by tail biopsy and ear clipping, but for experimental procedures, which involve work with newborn mice, very little alternatives are available for the simultaneous marking and biopsy sampling. In these cases, a non-permanent marking method is often combined with a tail biopsy.
Despite the controversy surrounding it,1–4 toe clipping presents multiple advantages. It is fast, easy to apply, does not require anaesthesia or analgesia and is cheap. Moreover, it is easy to learn and perform, and offers a high numbering possibility depending on which scheme is used. Although toe clipping is a legally accepted methodology, a lot of concerns have been raised because of the assumed pain for the newborns and because of possible detrimental long-term effects, which may be encountered like alterations of the grasping ability or hyperalgesia. As a consequence, in many standard operating procedures all over the world1–3 it is mentioned that toe clipping must be used only if no other method is feasible and must be performed only on neonates. Nevertheless, because of its numerous advantages it is easy to assume that this methodology could also be applied as a standard procedure for animal marking. Since no explicit study on the short- and long-term impacts of toe clipping on the animals’ wellbeing is present, it is necessary to clearly analyse the physiological and behavioural changes in clipped mice in order to finally determine whether this methodology is acceptable or if its practice should be reduced to a minimum.
The research to date has focused basically on the presence and degree of bone regrowth after digit tip amputation in newborn mice.5–11 These studies indicated that the regeneration response is level-specific within the terminal phalangeal bone and the regrown digit tips look morphologically normal but shorter than control digits. Neufeld and Zao 8 showed that only amputation of the proximal levels of the first phalangeal bone prevents regrowth of the amputated digit.
To date, only two studies investigating toe clipping as an identification method in the mouse are known. Kumar 12 argued that toe clipping can be performed without anaesthesia on 14-day-old pups (P14) and induces minimal discomfort. His conclusions were based on the observation that the pups started to suckle again within a few minutes. Vachon 10 focused only on the detrimental long-term side-effects related to bone growth and tissue reorganization, and concluded that toe amputation (performed at P14) is an acceptable procedure to numerically identify mice. Despite the fact that both these works describe toe clipping as a valid procedure, far too little attention has been paid to animal welfare, and no studies have been published for analysing the eventual presence and intensity of pain and stress produced by toe clipping in the first week after birth and its possible long-term effects.
An objective determination of pain and stress in mice, especially if only moderate, is very difficult. In general, mice show few signs of distress. In animal experiments, especially if a person is present in the room as an observer, the mouse will hide all signs of pain.13 - 15 The monitoring of low to moderate pain by solely observing the animals is therefore very difficult. Classical methods, like measurement of blood corticosterone levels, imply the handling of the animals, which produces stress and therefore can influence the results obtained, unless the animal is immediately sacrificed. Since pups undergo a period of reduced responsiveness of the pituitary-adrenal axis in early postnatal development, which is characterized by very low basal corticosterone levels and an inability of many stressors to elicit a corticosterone response,16 - 18 it is very difficult to measure corticosterone changes in the first postnatal days.
Recently, new methodologies were developed for the objective assessment of pain and stress in animals. One of them is telemetry, which allows the simultaneous measurement of physiological parameters such as heart rate, core body temperature or blood pressure. This technology has been successfully used for the assessment of post-laparotomy pain 19 and for the detection of the impact of different tissue biopsies for the genotyping of laboratory mice. 20 However, due to the size of the transmitter, this technology cannot be used in newborn mice.
In the present work, we analysed the physiological and behavioural impact of toe clipping on mice at postnatal days 3 (P3) and 7 (P7). We have performed corticosterone measurements, analysed physiological parameter changes as well as the behaviour of the mothers towards the clipped pups. We have further assessed the long-term effects of clipping by performing behavioural and histological studies with adult animals, which had been clipped at P3 and P7.
Materials and Methods
Animals and housing conditions
Male and female B6D2F1 mice were obtained from RCC (Research and Consulting Company, Biotechnology and Animal Breeding Division, Füllinsdorf, Switzerland) at the age of 6-8 weeks for breeding. The mice were free of all viral, bacterial and parasitic pathogens listed in the Federation of European Laboratory Animal Associations recommendations. 21 Animals were kept in type III plastic cages (425×266×150 mm, floor area 820 cm2) with autoclaved dust-free wooden bedding (80-90 g/cage) (Schill AG, Mutenz, Switzerland) and autoclaved paper tissues (2/cage) and a paper house as nesting material. They were fed a pelleted mouse diet (Kliba No. 3431, Provimi Kliba, Kaiseraugst, Switzerland) ad libitum and had unrestricted access to sterilized drinking water. The light/dark cycle in the room consisted of 12/12 h with artificial light (40 lux in the cage) from 07:00 to 19:00 h. The temperature was 21 ± 1°C, with a relative humidity of 50 ± 5% and with 15 complete changes of filtered air per hour (HEPA H 14 filter, Vokes-Air, Uster, Switzerland); the air pressure was controlled at 50 Pa. The studies were approved by the Cantonal Veterinary Office (Zurich, Switzerland). Housing and experimental procedures were in accordance with the Swiss animal protection law and conformed to the European Convention for the protection of vertebrate animals used for experimental and other scientific purposes (Council of Europe no. 123, Strasbourg 1985).
The animals used for breeding had a two-week adaptation time after the arrival at the animal facility. Oestrus was induced by the Whitten effect. The animals were mated at a ratio of one male to one female per cage. On the first day the cage was partitioned, so that the male could not reach the female, but there was olfactory contact. On the third day, the partition was removed and the female was checked for a vaginal plug in the morning of the fourth day, with each female kept singularly in a cage when it was positive.
Toe clipping
Partial amputations in the second phalange of the toes were done in neonatal pups of different ages. We performed the experiments with two groups: group 1 (P3, P7; n = 120) was used for the analysis of short/long-term behavioural and physiological parameters, group 2 (P7, n = 150) was used for the determination of serum corticosterone and therefore sacrificed shortly after clipping.
For each group, 50% of each litter was used as control animals, which underwent the same handling and restraint, and had scissor blades closed close to each paw, to mimic the clipping.
For the clipping procedure, the whole litter (group 1) or single animals (group 2) were taken out of the nest and put into an extra cage laid out with paper tissues. Every pup was hand-held over a paper pad and, with gentle pressure, restrained. The second phalange of a toe was cut, in total three toes, one per paw, chosen randomly at the two forepaws (except of the pollex) and one specifically selected at one of the hind paws according to a 1-10 numbering system. The toes were stored either in 4% formalin for morphological analysis or on ice for genotyping.
Very sharp microsurgery scissors (Vannas Spring Scissors, 4 mmblades, straight, from FST, Heidelberg, Germany) were used. Between each animal, the instrumentswere wiped with alcohol. No anaesthesia or analgesia was used. No specific method was used for homeostasis because no substantial bleeding occurred. After the complete litter (group 1), or one animal (group 2) had been processed, the animals were put back to the mother in the home cage nest.
Developmental analysis/short-term behavioural observations
The animals of group 1 and their mothers were observed immediately, 1, 3, 5 and 12 h after clipping and then daily for three weeks with the help of score sheets specifically designed for this purpose, partially based on previous works.22,23 The weight of all pups was determined daily for three weeks, during this procedure the litter was separated from the mother (remaining within audible range) for maximally 10 min.
Serum collection and corticosterone analysis
The P7 pups were euthanized between 12:00 and 14:00 h by decapitation 7 min (an appropriate time point for the measurement of corticosterone was determined in a preliminary experiment, data not shown) after clipping or handling only (non-clipped [NC] control group) and trunk blood was collected. The blood of five animals at a time (same sex) was pooled to ensure an adequate amount of serum, with a total of 25 animals for each of the four conditions (clipped, NC, male and female). The blood was allowed to clot at 4°C for one hour, then centrifuged for 6 min at 8000 rpm and the serum was stored at -20°C for later enzyme immunoassay (EIA). Serum corticosterone levels were assayed using a competitive EIA kit with a sensitivity of 0.17 ng/mL (Corticosterone HS EIA, Immunodiagnostic Systems, Frankfurt am Main, Germany) according to the manufacturer's instructions.
Baseline values
Corticosterone basal values for unhandled P7 pups were also determined. Each animal was killed by decapitation within 10 s after withdrawal from the nest. The blood of five animals at a time (same sex) was pooled, with a total of 25 animals for each sex. Serum corticosterone values were determined as described above.
Behavioural tests for assessment of long-term effects on forepaw grip strength and hyperalgesia
Both tests were performed with group 1 mice at the age of 12 weeks on two consecutive days. Each animal was taken out of the home cage with the experimenter being blind to group membership. For the hotplate test, we additionally used a control group (n = 20) of naïve mice that had not experienced any neonatal handling.
(A) Grip test
The grip test was performed as previously described.24,25 The grip strength meter was positioned horizontally and the mousewas held by the tail and lowered towardsthe apparatus. It was allowed to grasp (forelimbs only) the smooth, metal, triangular pull bar connected to a tractive force indicator (3 N, AWYCO AG, Olten, Switzerland) and was then pulled backwards horizontally. The force applied to the bar at the moment the grasp was released was recorded as peak tension (N). The test was repeated five consecutive times and the five values were recorded for each animal. Mice were not trained prior to the testing and each mouse was tested once.
(B) Hotplate test26,27 for detection of hyperalgesia
Each animal was grasped by the tail, removed from the home cage and dropped froma height of approximately 10 cmabove the hotplate (delimited by a plastic cylinder), and heated to a constant temperature of 52 ± 1°C. The experimenter activated a timer when the mouse came in contact with the surface of the plate and stopped the measurement as soon as a response (jumping, fore- or hind pawlicking or shaking) wasmade and the latency to respond was recorded. The animal was then removed immediately. A cut-off time of 30 s was chosen. Each animal was tested once.
Morphological analysis
For morphological analysis of the amputation stumps of adult mice (group 1) the following staining techniques were used: haematoxylin/eosin paraffin staining28,29 and Alizarin Red S/Alcian blue 8 GS (Division Chroma, Münster, Germany) whole mount skeletal staining. 30
Statistical analysis
All data are presented as mean±standard deviation (SD). Statistical analysis was carried out to validate the results of the grip test, the hotplate test and the corticosterone analysis (SPSS 16.0, SPSS Inc, Chicago, IL, USA). Unpaired two-tailed t-tests were applied to compare values obtained for the different groups in the behavioural tests. A Bonferroni correction was used; for the grip test P values ≤0.0125 were considered significant, whereas for the hotplate test P values ≤0.0062 were considered significant. Corticosterone data were logarithmically transformed and an analysis of variance with factors gender and group followed by Bonferroni post hoc tests were performed.
Results
Behaviour and physiological parameters
The immediate reaction to the clipping procedure was very similar for P3 and P7 pups. During the clipping, 48.2% of P3 pups (n = 85) and 39.4% of P7 (n = 137) pups showed no visible reaction at all, the others showed sporadic paw withdrawal, mostly at one or both hind paws (Figure 1a). No substantial bleeding occurred; blood stains were barely visible on white paper pads.
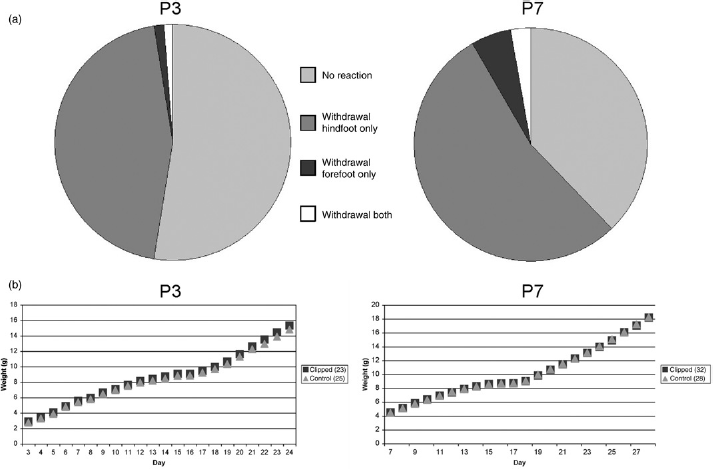
(a) Paw withdrawal as a frequent reaction to toe clipping. A total of 85 (P3) and 137 (P7) animals were tested. (b) Average weight development over three weeks of clipped pups compared with control animals
After the animals were put back into the nest, the mother immediately started grooming them without making any difference between clipped pups and control animals. Neither cannibalism by the mother, nor automutilation, inflammation or other abnormalities occurred. The mother kept all the pups inside the nest and milk spots were always visible through the furless skin. Righting of all pups worked correctly. Physiological development of fur, teeth, ears, eyes and walking was positive for all pups. The weight development over three weeks looked the same for the clipped and the control animals (Figure 1b).
Corticosterone analysis
Serum corticosterone values (Figure 2) were 25% higher in male than in female pups (P = 0.002). A negligible and nonsignificant increase of 3% (95% confidence interval (CI) -16% to +27%) could be seen in clipped compared with NC animals. Both clipped and NC pups had considerably (by factors 3.3 and 3.2 fold; P < 0.0001) increased corticosterone values compared with the non-handled (NH) animals (basal values, mean±SD: F5.33 ± 0.92 ng/mL, C4.07 ± 0.62 ng/mL).
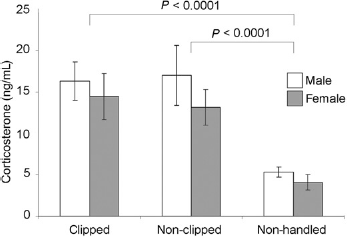
Serum corticosterone levels of P7 pups after clipping/handling compared with basal values of non-handled pups. Error bars show means±SD
Behavioural tests
The grip test (Figure 3a) revealed that animals clipped (C) at P3 had significantly lower paw strength than those clipped at P7 (P = 0.011). (Mean±SD: P3C 0.78 ± 0.23 N; P7C 0.98 ± 0.30 N.)
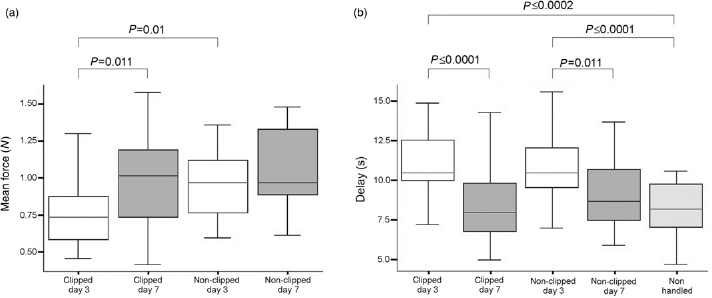
(a) Grip test results for the experimental groups P3 (n = 60) and P7 (n = 60) with clipped and non-clipped (control) animals, respectively; after Bonferroni correction, P values ≤0.0125 are significant. (b) Hotplate test results including an additional group of non-handled mice (n = 20) to control for neonatal handling or mother deprivation effects; after Bonferroni correction, P values ≤0.0062 are considered significant
In contrast, the NC groups of each age showed no significant differences (95% CI -0.24 N to 0.03 N; mean±SD: P3NC 0.99 ± 0.23 N; P7NC 1.06 ± 0.26 N). Comparing the clipped to the NC mice of each age, P is 0.01 for P3 animals, but for P7 mice there is no significant difference (95% CI -0.23 N to 0.06 N).
The hotplate test (Figure 3b) showed significantly (P ≤ 0.01) higher delay times for clipped (C) P3 animals compared with both clipped/NC P7 mice and NH mice (mean±SD: P3C 10.92 ± 2.44 s; P3NC 10.76 ± 2.16 s; P7C 8.35 ± 1.04 s; P7NC9.10 ± 3.21 s;NH8.17 ± 1.77 s). Nosignificant difference was found between clipped and NCP7 animals (95%CI -1.88 to 0.36 s).
Histological analysis of the amputation stumps 12 weeks after clipping
The second-phalanx amputations showed no sign of bone regeneration or bone hypertrophy and the bone stumps were histologically well-defined at their proximal ends (Figure 4).
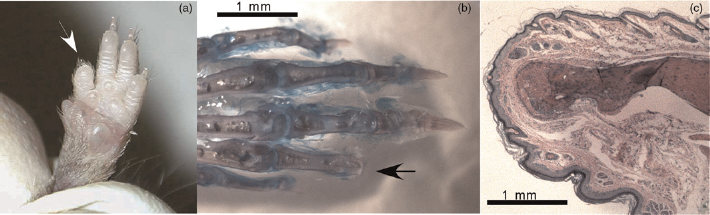
(a) Forepaw of a 12-week-old mouse by which a toe had been clipped at P7 (white arrow). (b) Alizarin Red S/Alcian Blue whole mount skeletal staining of a paw from a 12-week-old mouse that was clipped at P7 (black arrow). (c) Haematoxylin/eosin-stained paraffin section of the tip of an amputation stump. A well-developed dermis and epidermis reorganization covers the external surface of the stump and the medullar cavity of the phalangeal bone is closed
Discussion
In this study, we investigated the short- and long-term impact of toe clipping on animal welfare aspects such as health, behaviour, development, stress and detrimental effects in adulthood. Our findings demonstrate that for both P3 and P7 pups toe clipping does not have any negative effects on growth and physical development, and that clipped pups do not suffer from rejection by their mother. We could also confirm that the tissue of a single toe is sufficient for genotyping, even for P3 pups (data not shown). Our results have shown that the pups should be older than three days for adequate marking. On day 3, the pup's toes are still partially grown together. It is therefore difficult for the experimenter to cut them precisely. Furthermore, no matter how carefully the animals had been clipped at P3, a lot of them had very short stumps, which significantly decreased the grip strength of these animals in adulthood, as shown in Figure 3. We nevertheless think that this might not be considered a serious impairment for a laboratory mouse. This observation highlights the importance of choosing biopsy and marking procedures in accordance with the kind of experiments planned. For breeding of transgenic lines, for example, it is conceivable that any marking and biopsy method can be used, but if for example behavioural tests, where toes and tail play an important role (like beam walk, Morris water maze or grip tests), have to be performed, it would be important to decide if for example toe clipping would be a better alternative to tail biopsy or vice versa. Both methods have their advantages and disadvantages: in the case of toe clipping the grip strength of an animal can for example be impaired and in the case of a tail biopsy one can assume that the balancing or swimming capacity of a mouse could be affected. A recent study evaluating tail biopsy collection in laboratory mice 31 used a scoring system to qualitatively assess abnormal behaviour after the stimulus of tail biopsy and showed that, although the length of biopsy did not have a significant effect on behavioural responses, with increasing age more animals responded to the stimulus of conscious tail biopsy and the responses were also prolonged. The long-term effects of a tail biopsy on animal welfare have not been thoroughly investigated, although the removal of a large section of the tail has been demonstrated to cause thermal and mechanical hyperalgesia in the remaining part of the tail. 32 More experiments are needed to clearly assess for example the long-term effects of tail biopsy on balance and climbing capacity of a mouse.
In some cases, we observed that too short pieces of toewere cut and this resulted in more or less complete bone regrowth. In this case, the clipped toeswere hardly distinguishable from the NC ones, making the individual identification of the animal impossible. These difficulties did not occur for P7 pups; we would therefore recommend carrying out the clipping procedure at this age. In accordance, the results of the grip test (Figure 3) supported our previous observations, indicating that P7 is the preferable age for toe clipping, becausewe did not see significant differences in grip strength between clipped animals and controls for this age, in contrast to clipped P3 animals, whose grip strength was significantly lower compared with both clipped P7 and NC P3 mice. One reason for this could be that P3 pups often had very short amputation stumps, as mentioned previously.
Paw withdrawal, an immediate response to clipping, was observed for a relatively high number of pups of both ages (Figure 1a). We hypothesize that this can be due to a specific characteristic of prenatal pain development. It is known that neonatal rats, mice and even infants show enhanced and exaggerated basal pain behaviours compared with adults, associated with lower nociceptive thresholds and greater display of non-specific behavioural responses.33- 35 Hence, we assume that this kind of behaviour cannot be regarded as representative of the real pain intensity, mostly because only about half of the pups behaved that way.
We had to consider a specificity of the newborn organism, when we measured the level of corticosterone subsequent to clipping as an indicator for stress and pain. It was described that neonatal mice, just like rats, undergo a so-called stress hypo-responsive period due to the postnatal development of the hypothalamic-pituitary-adrenal axis. This period lasts from right after birth until P12. During this time, basal corticosterone levels are low and an exposure to moderate stress does not enhance them.16- 18 This observation was also confirmed by our preliminary experiments with P3 animals (data not shown). For animals clipped at P7 we found a significant increase (more than threefold) of serum corticosterone for the clipped pups as well as for the controls compared with the basal values of NH animals. We nevertheless found no significant difference between the clipped pups and the NC but handled controls (Figure 2). These findings indicate that first and foremost, the pups are stressed due to the handling and not because of the clipping itself. It is therefore clear that even short and inoffensive handling represents a stressor for the newborn mice and that the toe amputation is not the critical factor. A similar reaction had already been observed during the analysis of the impact of different biopsy methods used for genotyping. 20 We strongly believe that at this early age corticosterone measurement is an adequate indicator of stress and pain. It would be of interest to analyse vocalization of the pups during toe clipping; nevertheless, these data should be interpreted very carefully to prevent misinterpretations.
In order to determine whether the time point of clipping has an influence on the development of hyperalgesia, we performed a hotplate test. Interestingly, P3 pups, whether clipped or handled only, showed significantly higher delay times in the hotplate test compared with P7 pups and unhandled control animals (Figure 3b). It has been shown that chronic postnatal stressful handling procedures result in a hyposensitivity to thermal nociceptive stimuli by lowering the threshold for painful stimuli.36,37 This phenomenon could be the reason why we observed this increase for the P3 but not for the P7 mice. Both the clipped and control pups were weighed daily for three weeks after clipping, and were therefore separated from the home cage and the mother for about 10 min each day. Obviously, these daily manipulations along with the absence of the mother, which nevertheless remained within audible range, affected the P3 more than the P7 pups. This side-effect should always be considered when planning animal experiments with newborns. Of course, this side-effect occurred because of our experimental design and is not a characteristic for the toe clipping methodology itself. When toe clipping is routinely performed, the pups are not weighed and handled daily; this phenomenon will therefore likely not be observed. Finally, the results obtained from the grip and the hotplate tests demonstrated that toe clipping at P7 does neither affect the animal's grip strength nor causes hyperalgesia.
Overall, we conclude that toe clipping is indeed a procedure that induces a short period of stress for the newborns, because of handling and not because of clipping itself. Very likely, this method is not connected with more stress for the animals than for example tattooing of newborns, which requires a comparable or even longer handling time, depending on the individual skills of the person performing the procedure. This technique is however not considered problematic from a welfare point of view in contrast to toe clipping. In our study, we did not observe any significant detrimental short- or long-term effects for the animals, hence we therefore think toe clipping is an acceptable methodology for marking and genotyping pups. This opens the possibility of using toe clipping as an alternative to tail biopsy because it enables, at the same time, permanent marking of the animals as well as the collection of tissue for genotyping. Therefore, toe clipping at P7 allows animals to be genotyped two weeks before the weaning age and the fast selection of the mice needed for experiments or breeding, thus reducing the breeding costs. Last but not least, toe clipping of P7 pups is much easier than tail biopsy sampling from mice at weaning age because the handling of P7 pups that cannot bite is very easy and as a consequence marking of the animals is much less stressful not only for the investigator but also for the animals, which do not need to be restrained anymore.
Footnotes
Acknowledgements
We would like to thank Zsuzsanna Pataki, Fabienne Weber, Martin Mörter and Leonardo Mamaril for competent technical assistance; Hans Sigg, Margarete Arras and Philippe Bugnon for advice and discussions; Hanns Ulrich Zeilhofer for providing the hotplate test equipment; Petra Schwarz for supplying part of the histology and staining and Hans Welzl for advising us on the grip test.
