Abstract
The NOD/Shi-scid, IL-2Rγ null (NOG) mouse is a severely immunodeficient mouse used for the engraftment of human tissues and cells. In this study, 2406 mice (8–62 weeks old, 503 males and 1903 females) were subcutaneously engrafted with human tissues. In 16 mice (12–26 weeks old, 1 male and 15 females), a mass was seen in the anteroventralis of the thorax on gross examination with an incidence of 0.7%. Histologically, the masses were composed of sheets of lymphoblastic cells. A ‘starry sky’ pattern was observed with numerous mitoses. Immunohistochemically the lymphoblastic cells were positive for Thy 1. The lymphoblastic cells were also seen in the spleen, lung, liver, kidney and heart. The gross and histopathological findings led to the diagnosis of spontaneous thymic lymphoma in NOG mice.
The NOD.Cg-Prkdcscid Il2rgtm1Sug /Jic mouse (NOG mouse) is a severely immunodeficient mouse derived from the non-obese diabetic/severe combined immunodeficient mouse, NOD.CB17-Prkdcscid mouse (NOD-scid mouse). In addition to lacking T and B lymphocytes, the NOG mouse has multifunctional defects in non-killer (NK) cell activity, macrophage function, complement activity and dendritic cell function. The strain is relatively new, compared with more conventional strains such as CB17/Icr-Prkdcscid mouse (CB17-scid mouse) or NOD-scid mouse, and is known to be an excellent recipient for engraftment of human haematopoietic cells and solid tumours. 1,2 The immunodeficient state and its implications for xenografts have been well studied, but the spontaneous lesions of this strain are not well known.
Spontaneous thymic lymphoma is a common finding in CB17-scid and NOD-scid mice, while it has not been reported in NOG mice. 3–5
NOG mice are routinely used as recipients for human tissues in our facility. Lesions similar to the spontaneous thymic lymphoma seen in CB17-scid and NOD-scid mice are occasionally observed. In this report, the incidence and pathological characteristics of spontaneous thymic lymphoma in the NOG mouse were investigated.
The NOG mice used in this study were acquired from the breeding facility of the Central Institute for Experimental Animals (CIEA, Kanagawa, Japan). The mice were imported at the age of 5–6 weeks from CIEA to PharmaLogicals Research Pte Ltd (PLR, Singapore). All animals were housed in plastic cages within a bioBubble® system (bioBubble, Fort Collins, CO, USA) of a pathogen-free state. Mice were fed pelleted chow (CE-2; Clea Japan Inc, Tokyo, Japan) and tap water ad libitum. All fresh surgical specimens of human tissues were obtained at PLR. The present animal experiments and use of human tissues for engraftment and further research were approved by the Institutional Animal Care and Use Committee and Ethics Review Committee at PLR, respectively. These guidelines meet the generally accepted international criteria on humane treatment that spare the animal needless pain and suffering, and require confirmation that the experiments conducted are of actual scientific benefit to humankind.
Most of the NOG mice were engrafted with human tumour tissues at 6–12 weeks of age. The engrafted tissues were mainly from the mammary gland (22%), large intestine (20%), stomach (7%), ovary (7%), kidney (7%) and other tumour cases (37%). They were necropsied between 3 and 120 days after the engraftment. Animals were euthanized at the time of engrafted tumour passage or as when a humane endpoint was reached (i.e. exhibiting any clinical signs of cachexia, weakness, dyspnoea or airway obstruction). All animals including animals that were found dead were necropsied and were evaluated macroscopically. A total of 2406 mice (503 males and 1903 females) were examined. The mice were 8–62 weeks old. Gross examination revealed a white to pale yellow, lobulated mass in the thorax in 16 of 2406 animals examined. The mass from eight mice along with the liver, kidney, heart, lung and spleen were collected and fixed in 4% paraformaldehyde in 0.1 mmol/L phosphate buffer (pH 7. 4) for 16–24 h at 4°C and embedded into paraffin using the AMeX method or routine method. 6,7 For histopathological examination, tissues were sectioned, stained with haematoxylin and eosin and read under light microscopy.
The tissues were sectioned and immunohistochemically stained for Thy 1. Thy 1 is known as thymus cell antigen 1, theta and is expressed on thymocytes, peripheral/intraepithelial T lymphocytes and neurons of all mouse strains. 8 Immunohistochemical staining was performed to confirm that the tumour cells had originated from mouse T-cells. 8 Rabbit sera raised against anti-mouse Thy 1 (CL2001, Cederlane Laboratories Limited, Burlington, Ontario, Canada) was applied as the primary antibody. The labelled streptavidin–biotin method using a Ventana HX Discovery System (Ventana Medical Systems, Inc, Tucson, AZ, USA) was used for staining. All slides were counterstained with hematoxylin, dehydrated and coverslipped for analysis under light microscope.
Masses in the thorax were seen in one out of 503 male and 15 out of 1903 female animals (0.7%, Table 1). They occurred mainly in mice 12–26 weeks old (Table 1). Grossly, the masses were located in the anteroventralis of the thorax. They were 0.4–2 cm in diameter, whitish with a lobular structure and without contiguity with the lung (Figure 1a). In six animals, large spleens were noted. No significant changes were found in the other organs. Clinical signs included weakness and dyspnoea and were thought to be a result of airway obstruction caused by the growth of the thoracic mass. The thoracic mass was not observed in mice over 27 weeks old (Table 1).
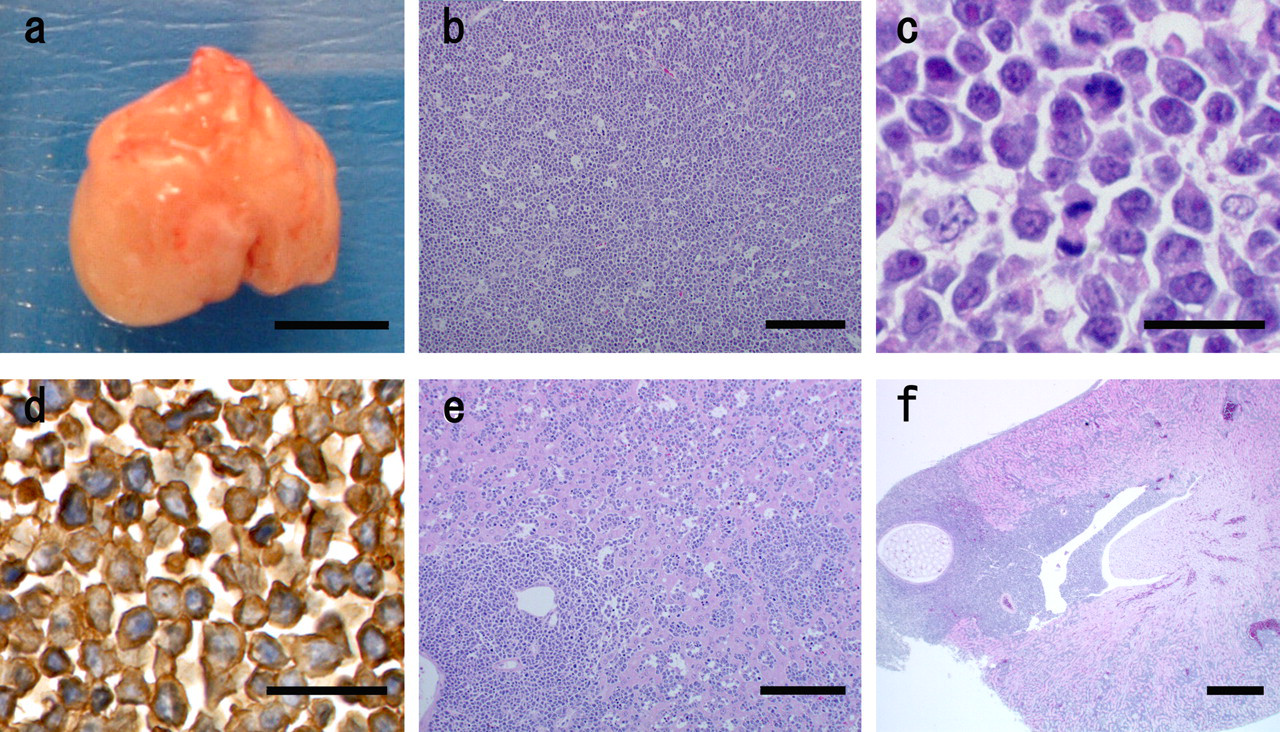
The gross and histopathological findings in the thoracic mass and various organs in the NOG mouse. (a) Grossly the mass was seen in the thorax (bar, 5 mm). (b) Histopathologically, sheets of lymphoblastic cells were observed in the thoracic mass, producing a ‘starry sky’ pattern (bar, 150 µm). (c) The lymphoblastic cells were small to medium and round to slightly irregular shape with scant cytoplasm. Nuclei were rounded to oval with condensed chromatin. Mitotic figures were numerous (bar, 20 µm). (d) Immunohistochemistry with anti-Thy 1 antibody. The lymphoblastic cells showed positive with Thy 1 (bar, 20 µm). (e) In the liver, lymphoblastic cells were seen mainly in perivascular areas and infiltrated the sinusoids (bar, 100 µm). (f) In the kidney lymphoblastic cells infiltrated stemming from the reniportal area (bar, 500 µm)
Incidence of the thoracic mass in NOG mice
Histopathologically, sheets of lymphoblastic cells were observed with a ‘starry sky’ pattern in the thoracic mass (Figure 1b). Lymphoblastic cells were small to medium in size and round to slightly irregular in shape with scant cytoplasm. The nuclei were rounded to oval with condensed chromatin. Mitotic figures were numerous (Figure 1c). The lymphoblastic cells showed positive for immunohistochemical staining with Thy 1 (Figure 1d). In some cases, epithelial tissue was observed in the mass. This was considered to be thymic epithelial tissue.
Lymphoblastic cells were also seen in the spleen, lung, liver, kidney and heart. In the spleen, lymphoblastic cells were mainly seen in the white pulp, extending to the red pulp. In the lung, lymphoblastic cells were observed along peribronchial and perivasuclar lymphatic channels. In the liver, lymphoblastic cells were seen mainly in perivascular areas and infiltrated the sinusoids in some cases (Figure 1e). Lymphoblastic cells infiltrated the kidney, stemming from the reniportal area (Figure 1f). In the heart, lymphoblastic cells were seen in the epicardium. Lymphoma was seen in non-treated mice and in mice engrafted with various tumour types. Therefore, no relationship was seen between lymphoma occurrence and engraftment of human tumour tissues.
All animals with the thoracic mass showed the same clinical signs. Histopathologically, the mass mainly consisted of lymphoblastic cells. The same cells were seen in the spleen, lung, liver, kidney and heart. The lymphoblastic cells were positive for Thy 1, indicating that they were of T-cell origin. 8 Considering the clinical, gross and histological findings, the thoracic mass was diagnosed as spontaneous thymic lymphoma. 9
In CB17-scid mice and NOD-scid mice, thymic lymphoma is seen frequently. 3–5 The morphology of the thymic lymphoma in NOG mice was similar to the spontaneous thymic lymphoma in CB17-scid mice and NOD-scid mice in that lymphoblastic cells were seen in the thoracic mass, spleen, lung, liver, kidney and heart and that the cells had a T-cell character.
Spontaneous thymic lymphoma has not been previously reported in NOG mice, whereas a high incidence of thymic lymphoma was seen in NOD-scid mouse (24 out of 36 mice) and CB17-scid mouse (41 out of 269 mice). 3–5 Irradiation of 450 cGy for eight days induced thymic lymphomas in NOD-scid mice (2 out of 25 mice), whereas none of the NOD.Cg-Prkdcscid Il2rgtm1Wjl /SzJ, a similar strain to NOG mice, showed lymphoma development. 5
The incidence of thymic lymphoma in NOG mice was markedly lower, with only 16 of 2406 animals in comparison with the other immunodeficient mouse strains. 3–5 The age of onset was earlier in NOG mice at 12–26 weeks compared with 20–40 weeks in NOD-scid mice and 10–65 weeks in CB17-scid mice. 3,4 The sex difference in NOG mice was similar to NOD-scid mice, where females showed a higher incidence than males, as opposed to CB17-scid mice where males show a higher incidence. 3,4
The mechanism of lymphomagenesis in mice is not clear. Since there are differences in incidence, age of onset and sexual distinction between the three strains, there is presumably some genetic difference that may affect the incidence of lymphomagenesis. In NOD-scid mice the phenotype indicates that T-cell differentiation is blocked at an early stage of maturation in which immature T-cells are in a stage of rapid proliferation. 4 Compared with NOD-scid and NOG mice, the development of thymic lymphoma in NOD-scid mice may be dependent on cytokine signalling mediated through the IL-2Rγ-chain. In NOD-scid mice association with endogenous ecotropic murine leukaemia provirus locus, Emv30, is reported. 4,10 Studying the genetic difference between the strains may elucidate the mechanisms of lymphomagenesis in immunodeficient mice.
