Abstract
A Syrian hamster that had been treated with topical antibiotic ointment for 10 days following injuries sustained during fighting was presented moribund. Postmortem and microscopic examinations revealed lesions consistent with clostridial typhlitis and enteritis. Anaerobic culture of caecal contents resulted in the isolation of two Clostridium species, and caecal contents contained Clostridium difficile enterotoxins. Based on these findings, a diagnosis of acute C. difficile enterotoxaemia was made. This report discusses the pathogenesis of C. difficile enterotoxaemia and the potential role of topical antibiotic ointment therapy in initiating the disease.
Keywords
Antibiotic-induced clostridial enterotoxaemia is a well-known syndrome among three common laboratory species – rabbits, guineapigs and hamsters. It is thought that some commensal gastrointestinal flora of these three species is sensitive to certain antibiotics, such as penicillins, tetracyclines, cephalosporins, aminoglycosides, macrolides and lincosamides. 1–3 Disruption of this flora by these antibiotics allows increased proliferation of the bacteria of the genus Clostridium, 4 which are Gram-positive, spore-forming, anaerobic bacteria. Clostridium spiroforme is most commonly isolated from rabbits with enterotoxaemia. 5 Clostridium difficile was first demonstrated, by Koch's postulate, to be the causative agent in clindamycin-induced colitis in hamsters by Bartlett et al. 6 and is now known to be the most common isolate from guineapigs and hamsters. 3,7,8 The pathological changes and clinical signs of enterotoxaemia are caused by proliferation of and toxin release by the Clostridium. C. spiroforme produces an iota-like cytotoxin. 7 Bartlett et al. 9 isolated a clostridia-produced cytotoxin from the caecum of hamsters affected by clindamycin-induced enterocolitis. Further research has shown that C. difficile produces two exotoxins, A and B. 10,11 Toxin A, an enterotoxin, causes the initial mucosal cell damage and inflammation, which results in fluid loss. Toxin B, a cytotoxin, requires toxin A to produce the primary assault in order to gain access to the cells and cause further damage. 12–14
Because of their innate sensitivity to antibiotic-induced clostridial enterotoxaemia, antibiotic therapy in Golden Syrian hamsters (Mesocricetus auratus) is challenging. The pugnacious nature of these commonly used laboratory rodents makes intraspecific wounding an occasional problem when adults are group-housed. Antibiotic treatment of fight wounds may be indicated if wounds are extensive or contaminated. Oral or parenteral administration of the above-mentioned antibiotics can result in overgrowth of C. difficile, resulting in fatal typhlitis and enterotoxaemia. 15 While the effects of orally or parenterally administered antibiotics on laboratory hamsters have been well described, 1,16 the pathological effects associated with the use of topical antibiotic administration have only been described in experimental settings. We describe here a case of C. difficile typhlitis in a hamster that had received prior topical antibiotic therapy. This report reinforces the exquisite sensitivity of hamsters to antibiotic-induced clostridial enterotoxaemia and highlights the need to use antibiotics with caution even when administered topically.
Materials and methods
Ninety-six commercially bred, five-month-old male HsdHan:AURA Golden Syrian hamsters (Harlan Sprague-Dawley, Indianapolis, IN, USA) were obtained and used in an undergraduate biology laboratory to demonstrate the effects of photoperiod on the mammalian reproductive system, in accordance with a University of Missouri Institutional Animal Care and Use Committee approved protocol, and the National Research Council Guide for the Care and Use of Laboratory Animals. The vendor's health surveillance programme included testing hamsters for Sendai virus, lymphocytic choriomeningitis virus, pneumonia virus of mice, reovirus 3, simian virus 5, Mycoplasma sp., Encephalitozoan cuniculi, Clostridium piliforme, Campylobacteria sp., Salmonella sp., Corynebacterium kutscheri, Bordetella bronchiseptica, Streptococcus zooepidemicus, Streptococcus pneumoniae, Group B beta streptococcus, Streptobacillus moniliformis and Pseudomonas aeruginosa. Half of the hamsters were maintained in an 8:16 light:dark cycle, and half in a 14:10 cycle. All hamsters were housed in a facility accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care, International. They were housed conventionally, three per cage, in polycarbonate or polypropylene wire-topped cages containing corncob bedding (Bed o' Cobs, The Anderson's, Maumee, OH, USA). For environmental enrichment, a short section of 3 in. internal diameter polyvinyl chloride pipe was added to each cage. Hamsters were provided commercial rodent diet (Purina Lab Diet 5001, Purina Mills, LLC, St Louis, MO, USA), and tap water ad libitum. Animal rooms were maintained at 68–76°F (20–24°C) and 50–70% relative humidity.
Approximately three months after arrival, five of the hamsters in the 8:16 light cycle group presented with bite wounds due to fighting. The bite wounds of three hamsters were minor, consisting of one to a few small (<5 mm) superficial skin lesions. Two hamsters had more severe lesions, consisting of multiple (6–10) small superficial wounds on their backs. One of the more severely injured hamsters also had an approximately 1 cm diameter wound on his right side near the costovertebral gland. The wound exposed the underlying subcutaneous tissue and muscle, but the peritoneum appeared intact. All hamsters were alert and responsive, and aggressive when handled. There were no behavioural indications of pain.
The injured hamsters were individually housed to prevent further fighting. The fur surrounding the wounds of the two more severely wounded hamsters was clipped, and the wounds were cleaned with a dilute povidine iodine solution. A triple antibiotic ointment containing polymixin B sulphate, neomycin sulphate and bacitracin zinc (Triple Antibiotic Ointment, Invacare, Holliston, MA, USA) was applied topically to the wounds twice daily for five days, followed by application once daily for an additional five days.
Results
All skin wounds were resolved or resolving without signs of infection 10 days after the initial presentation, and the antibiotic therapy was discontinued. Four hamsters were clinically normal. However, the hamster with the right flank wound, which had been clinically normal the previous day, was lethargic, dehydrated, cool to the touch and obtunded. Faeces in the hamster's cage appeared normal. Initial differential diagnoses included septicaemia, due to possible infection of the flank wound, and antibiotic-induced enterotoxaemia. The hamster was euthanized and necropsied at the University of Missouri Research Animal Diagnostic Laboratory.
Postmortem examination confirmed that the cutaneous flank wound did not extend into the peritoneal cavity. The small intestine, caecum and most of the colon were distended and filled with green liquid ingesta, and there was marked congestion of several vessels of the gastrointestinal tract. On histological examination of haematoxylin and eosin-stained sections of the ileum, caecum and colon, there was loss of apical enterocytes with abundant soughed cells in the luminal contents, hyperplasia of crypt (ileum) and gland (caecum and colon) enterocytes, pyogranulomatous inflammation, mucosal haemorrhage, submucosal oedema and venous congestion (Figures 1A–C). Numerous individual or chains of Gram-positive, approximately 1 × 3 μm rods, morphologically consistent with several clostridial species including C. difficile, were noted in the contents of the Gram-stained sections of the caecum and colon (Figure 1D). Sections of ileum, caecum and colon were also stained with a Steiner silver stain and found to be negative for C. piliforme and Lawsonia intracellularis. No significant histological abnormalities were noted in the kidney, liver or lungs.
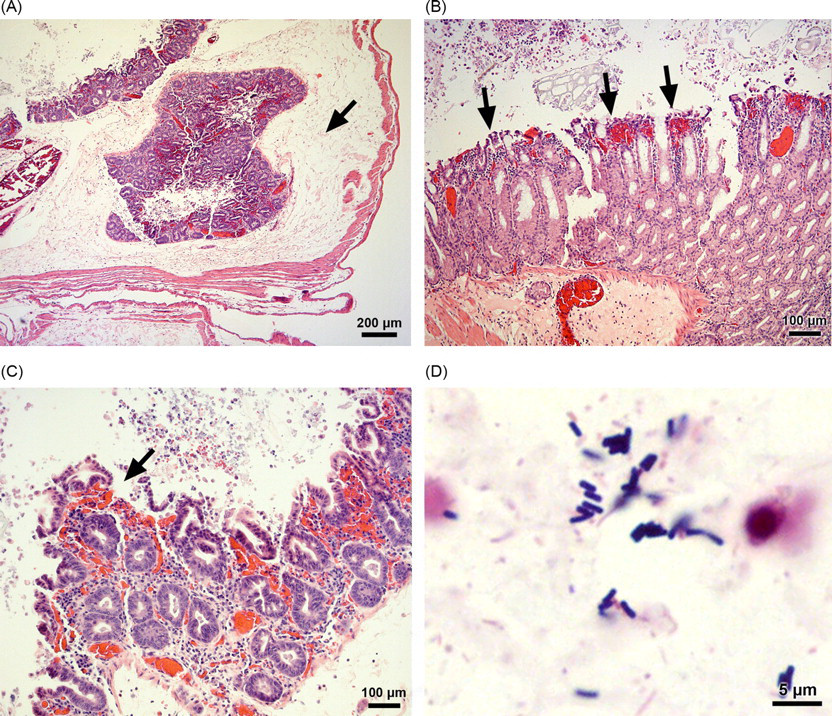
Clostridium difficile-induced typhlitis in a Syrian hamster. (A) Caecal section showing diffuse inflammation and severe submucosal oedema (arrow) (H&E, 40 ×). (B) Caecal section showing haemorrhage (arrows) (H&E, 400 ×). (C) Caecal section showing effacement of the apical enterocytes (arrow) (H&E, 400 ×). (D) Caecal contents showing numerous Gram-positive rods (Gram-stained section, oil immersion, 1000 ×)
Anaerobic culture (blood agar, 35°C for 48 h) of caecal contents resulted in the isolation of two distinct Clostridium colony types which could not be further speciated. Aerobic cultures of the blood and the flank wound (sheep blood agar, 7% CO2, 35°C) were negative for bacterial growth. Aerobic culture of the caecal contents (MacConkey agar, 35°C) was negative for growth of Salmonella spp. Culture of the ileal contents for Campylobacter spp. (Campy blood agar plate, Remel, Lenexa, KS, USA; 42°C for 24 h) was negative. C. difficile enterotoxins A and/or B were demonstrated in the caecal contents by antigen capture enzyme-linked immunosorbent assay (C. difficile TOX A/B Test from TECHLAB, Blacksburg, VA, USA).
Based on the gross and histological lesions, including an abundance of Gram-positive rods morphologically consistent with C. difficile, the isolation of Clostridium spp. from the caecal contents, and the presence of C. difficile enterotoxins A and B in the caecum of the affected hamster, a diagnosis of clostridial typhlitis and enterotoxaemia due to C. difficile was made. Septicaemia, salmonellosis, campylobacteriosis, C. piliforme (Tyzzer's) infection and Lawsonia intracellularis infection were ruled out based on negative enteric and blood cultures, lack of histological evidence of systemic disease, and examination of silver-stained sections of intestinal tract.
Discussion
Hamsters are susceptible to antibiotic-induced enterotoxaemia with typhlitis and colitis caused by C. difficile. Antibiotic therapy only renders the hamster susceptible, and C. difficile must be present in its gastrointestinal tract for antibiotic-induced enterotoxaemia to occur. 17 Only a small percentage of clinically normal hamsters carry C. difficile in their gastrointestinal tracts. 18,19 Thus, most hamsters must be exposed to C. difficile at the time of antibiotic therapy to become ill. Transmission of C. difficile to uninfected hamsters requires ingestion of the organism. Exposure may occur through direct contact or via faecally contaminated fomites, and the small percentage of clinically normal hamsters that carry C. difficile could serve as asymptomatic reservoirs. Spores can survive for at least five months in the environment, 20 and are probably the most common transmission mode. The vegetative form, however, has been shown to be shed in the faeces of humans with C. difficile-associated diarrhoea, survive in the environment on moist surfaces for several hours and survive exposure to gastric contents at a pH of 5 or greater, 21 suggesting the possibility that vegetative forms from symptomatic individuals can also transmit infection. The gastrointestinal bacterial flora of normal hamsters is predominantly composed of Bacteroides and Lactobacillus, 15,22 some of which inhibit the growth of C. difficile in vitro. 23 Disruption of this normal flora allows overgrowth of pathogenic C. difficile. Some antibiotics may also directly enhance C. difficile colonization in the gastrointestinal tract. Recently, clindamycin and ampicillin have been shown to increase expression of C. difficile genes that code for colonization factors and to increase adherence of C. difficle in vitro. 24 An increase in toxins A and B leads to typhlitis and enterotoxaemia and the subsequent clinical signs, including watery or haemorrhagic diarrhoea, dehydration, roughened hair coat, hunched posture and death within 2–10 days. 3,15
A similar condition in humans, known as C. difficile-associated diarrhoea or antibiotic-associated pseudomembranous colitis, is primarily a nosocomial infection and affects approximately three million people in the USA annually. 25 Predisposing factors for disease include antibiotic therapy (clindamycin, penicillins, cephalosporins, aminoglycosides, metronidazole, vancomycin and antipseudomonals); advanced age; underlying disease; faulty immune response to C. difficile toxins; 25 gastrointestinal manipulations and surgery; 26 and autoimmune hepatitis. 27 Over the past few years, a newly emerging group of C. difficile strains, BI, has gained attention due to its increased virulence and increased antibiotic resistance, mainly to fluoroquinolones. Outbreaks of severe disease due to the new strains have been associated with increased incidence, morbidity and mortality. 28–30
In the 1970s, the hamster's susceptibility to antibiotic-induced C. difficile enterocolitis established it as a model for human diarrhoea and pseudomembranous colitis caused by C. difficile. Chang et al. 31 compared faecal samples from a human patient with antibiotic-associated colitis to caecal contents from a hamster treated with clindamycin to induce enterocolitis. From both samples a toxin was isolated, a high concentration of C. difficile was recovered, and the supernatant produced cytotoxic effects which were neutralized by clostridial antitoxins. Thus, a toxin producing clostridial bacterium was implicated as the agent in both conditions. 31 The antibiotic-induced C. difficile enterocolitis hamster model continues to be used for pathogenesis and therapeutic research. 32–39 Several strains of the newly emerging, highly virulent C. difficile BI strains have recently been assessed for their pathogenic potential in hamsters. Upon treatment with clindamycin and inoculation with spores from standard and BI strains, one strain, BI6, was found to be particularly virulent in hamsters, consistently causing death within 48 h of inoculation. 40
In the case reported herein, a fatal clostridial typhlitis occurred in association with topical antibiotic administration. Self-grooming may have resulted in unintentional oral exposure. Fatal enterotoxaemia has been shown to occur after ingestion of topical antibiotic ointment in other species. LaRegina et al. 41 conducted a study to examine possible effects of topically applied antibiotic ointment in the white-tailed rat (Mystromys albicaudatus). Application of a polymixin B sulphate, neomycin sulphate, bacitracin ointment to skin from which fur had been removed by shaving resulted in death with lesions consistent with antibiotic-associated typhlitis and colitis. These animals were observed to groom and lick the treated areas after application, so ingestion was likely the primary means of exposure. Polymixin B, neomycin and bacitracin were the same antibiotic ingredients in the topical ointment applied to the hamster in this case, which supports unintentional ingestion via grooming as a factor in this hamster's disease. Neomycin has been demonstrated to induce antibiotic-associated diarrhoea in hamsters, and is the most likely culprit among the three antibiotics administered here. Fekety et al. 42 found that one large (285 mg/kg) dose of neomycin given orally caused lethal typhlitis, although smaller doses did not reliably cause disease. 42 Michelich et al. 43 found that neomycin, in combination with a powdered, purified diet, caused diarrhoea and death, and C. difficile was isolated from most hamsters with diarrhoea. In that study, diet appeared to be another factor, as hamsters given neomycin in combination with ground commercial chow did not develop diarrhoea or illness. 43 Bacitracin, a polypeptide antimicrobial, is not known to induce diarrhoea or colitis, 44 is inhibitory for C. difficile, 42 and blocks clindamycin-induced mortality in the hamster model. 45 It has been used therapeutically to successfully treat humans with C. difficile infection. 44,46,47 Polymixin B, another polypeptide antimicrobial, is also not known for inducing enterocolitis in humans. 42 To the authors' knowledge, there are no reports of polymixin B-induced colitis in hamsters.
This hamster's fatal clostridial typhlitis may have been affected by factors other than ingestion via self-grooming. The topically applied antibiotics may also have been absorbed through the flank wound. Percutaneous absorption of antibiotics has caused colitis in humans and hamsters. Clindamycin applied topically to human skin is absorbed, 48,49 and many human cases of C. difficile pseudomembranous colitis after therapy with topical clindamycin have been reported. 50,51 Feingold et al. 52 studied the effects of topically applied clindamycin in hamsters by shaving a dorsal area and bonding a nylon mesh around the treated area to prevent oral ingestion. Clindamycin treatment resulted in deaths within three to seven days, and clostridium toxin was recovered from caecal contents. Percutaneous absorption of the clindamycin was implicated as causing the clostridium overgrowth. Polymixin B sulphate, neomycin sulphate and bacitracin zinc are not absorbed across intact skin. 53–55 However, neomycin is absorbed through abraded skin or wounds, 53 and was likely absorbed via the hamster's wounds.
Outbreaks of C. difficile enterotoxaemia without previous antibiotic administration have also been reported in Golden Syrian hamsters. 14,56–60 In these cases, several factors have been suggested to contribute to disease susceptibility including diet, experimental procedure and age. Hamsters fed an atherogenic diet were more susceptible to both spontaneous and experimental C. difficile-induced diarrhoea and death when compared to those animals fed with a control diet. 59 Because the caecal flora from both atherogenic and control diet-fed hamsters inhibited C. difficile growth in vitro, Blankenship-Paris et al. 60 suggested something other than altered caecal flora had caused the increased susceptibility. Boss et al. 58 considered purified powdered diet as a possible contributing factor in an outbreak of C. difficile enteritis among hamsters on a carcinogenicity study. The authors also suggested that the stress of daily handling and oral gavage of the test drug were other possible factors. Rehg and Lu 56 isolated C. difficile from three Millhill Albino hamsters with diarrhoea and typhlitis. They determined that environmental contamination with high concentrations of C. difficile from a previous outbreak in the room likely lead to the illness. Aging has also been considered a possible factor in susceptibility to clostridial disease. C. difficile-induced colitis and typhlitis were diagnosed as the causes of spontaneous diarrhoea and death in hamsters on a prion study in which 90% of the affected hamsters were over 210 days old. 57
In the case reported here, the above factors do not explain the disease. Hamsters were fed only a commercial rodent diet, so non-traditional diet or dietary supplementation with food items was not a factor. There had been no prior outbreaks of C. difficile enterocolitis among hamsters in this colony, which suggests that high environmental contamination was not a significant problem. While age in this eight-month-old hamster is a potential factor, all of the unaffected hamsters were also eight months old, which suggests that age was a less likely determining factor, although it may have been a contributing factor.
The incidence of disease in our study was low with only one of the five antibiotic-treated hamsters developing clinical enterotoxaemia. Stress associated with the initial fighting and the daily handling for treatment may have been factors in this hamster's enterotoxaemia. Because the affected hamster had more severe wounds, it may have suffered a more severe physiological stress than the other hamsters, rendering it more susceptible to C. difficile enterotoxaemia. In addition, the larger wound size and correspondingly larger dose of topical antibiotic applied could have allowed the ingestion of more antibiotics during self-grooming, and/or more absorption of neomycin through the wounds. The affected hamster may also have harboured C. difficile in his gastrointestinal tract before exposure to antibiotics, which could have resulted in more rapid overgrowth of enteropathogenic C. difficile.
To the authors' knowledge, this is the first case report of unintentional clostridial enterocolitis and enterotoxaemia in Syrian hamsters that was associated with topical antibiotic ointment therapy. Although a definitive causal relationship was not established, it is probable that inadvertant exposure to the antibiotic neomycin by oral ingestion or absorption across the non-intact skin resulted in intestinal dysbiosis and subsequent C. difficile enterocolitis. This report reinforces the fact that hamsters are extremely sensitive to antibiotic-associated clostridial enterotoxaemia and highlights the need to use caution when administering antibiotics, even topically, to hamsters. For cutaneous wounds or local, cutaneous infections, topical disinfectants such as dilute povidine iodine may be sufficient. In cases where topical antibiotic treatment is indicated, methods to prevent ingestion of topical antibiotics such as a tightly adhering wound dressing could be considered. In general, penicillins, tetracyclines, cephalosporins and some macrolides and lincosamides should not be used, or used with caution and close attention to recommended dosages. Safer alternatives include trimethoprim sulfonamides, fluoroquinolones and chloramphenicol.
