Abstract
A case of cardiac arrhythmias related to continuous thermodilution cardiac output (CCO) is reported. A sheep anaesthetized for experimental purpose was instrumented with a special Swan-Ganz catheter-type to be used for CCO measurements. One hour after starting the CCO monitoring, isolated ventricular extrasystoles were noticed on the electrocardiogram with an increasing frequency. Subsequently bursts of extrasystoles occurred. Atrioventricular dissociation was also observed. The peaks of temperature of the thermal filament were within the normal range and their presence was noticed when arrhythmias appeared. Mean blood pressure and cardiac output did not change during this episode. When the CCO was switched off, no more arrhythmias were observed. The CCO Swan-Ganz by itself did not generate any arrhythmia. The sheep recovered uneventfully. When arrhythmias occur during anaesthesia where CCO is used, a thermal filament induced origin of the arrhythmia must be considered.
Haemodynamic measurements during surgical experimental procedures are common. These are performed for monitoring purposes or to address a specific hypothesis. In the latter context, cardiac output measurement is often included. Novel methods to measure cardiac output have been introduced which are less invasive and/or automated requiring less manipulation than iced bolus thermodilution (BTD), which is considered the golden standard. Continuous thermodilution cardiac output (CCO) presents the advantage of automation of the measurement and was previously used in sheep without any complications. 1–3
This case report describes a previously unreported cardiac complication related to the use of CCO technology in an anaesthetized sheep.
Materials and methods
A nine-month-old Suffolk mixed breed male sheep of 37 kg body weight was scheduled to be anaesthetized for an experimental laparoscopic cystotomy. This was part of a multidisciplinary study involving a study of the pharmacokinetics of lidocaine and cardiopulmonary effects of different ventilation strategies. This sheep was anaesthetized two weeks previously with a similar anaesthetic protocol and for a similar experimental procedure. The study procedure was discussed and approved by the institutional ethics committee of the University of Veterinary Medicine Vienna and had governmental approval (GZ 68.205/0168-BrGt/2006). The sheep was considered to be in good health according to the results of a physical examination and complete haematological and biochemical blood analyses.
The sheep was fasted for 24 h before anaesthesia, but access to water was allowed.
Anaesthetic management
After local infiltration of the puncture site with 1 mL of procaine hydrochloride 2% (Procaine Hydrochloride® 2%, VMD nv/sa, Arendonk, Belgium), the left jugular vein (Vasocan® Braunüle, 16G [1.7 × 50 mm], B Braun Melsungen AG, Melsungen, Germany) and auricular artery (Vasocan® Braunüle, 20G [1.1 × 33 mm], B Braun Melsungen AG, Melsungen, Germany) were catheterized prior to induction of anaesthesia. The sheep received 2 mg/kg of lidocaine intravenously (Xylanaest purum® 2%, Gebro Pharma, Fieberbrunn, Austria) on three different occasions: prior to anaesthesia, after induction and after onset of capnoperitoneum. Induction of anaesthesia was performed by intravenous injection of 17 mg/kg of thiopental (Thiopental Sandoz®, Sandoz GmbH, Kundl, Austria). The trachea was then intubated (10 mm ID endotracheal tube) and the sheep connected to a circle breathing system (Scandia, Blease Medical Equipment Ltd, Chesham, UK). Anaesthesia was maintained with 2% end tidal isoflurane (Isoba®, Essex Tierarznei Ndl der Essex Pharma GmbH, München, Germany) in oxygen (1 L/min) using a precision vaporizer (Dräger 19.3, Lubeck, Germany). An orogastric tube was put in place. The sheep was positioned in right lateral recumbency and intermittent positive pressure ventilation (IPPV) performed using a volume-controlled mode (minute volume of 12 mL/kg/min, respiratory rate of 20 breaths per minute [bpm]) (Siemens Servo 300, Solna, Sweden) during the experiment. Vital signs and classical anaesthetic parameters, i.e. electrocardiogram (ECG), heart rate, respiratory rate, complete respiratory gas analysis, invasive blood pressure, oesophageal temperature, ventilation volumes and airway pressures and flows, were monitored using an S/5 Datex-Ohmeda multiparameter anaesthesia monitor (General Electrics Healthcare, Helsinki, Finland).
Cardiac output measurement
Cardiac output was measured using a cardiac output monitor (Vigilance, Software release 5.4 E; Edwards Lifesciences, Irvine, CA, USA) and a modified Swan-Ganz catheter featuring a thermal filament (Swan Ganz CCOmbo/VIP Pulmonary Artery Catheter 746F8; Edwards Lifesciences). This combination allows the measurement of cardiac output with both the classic BTD technique and the CCO technique. The CCO technique uses the indicator dilution technique and stochastic system identification enhancing the signal-to-noise ratio; heat is infused continuously on a random on-off pattern. 4
A 9F introducer (IntroFlex Introducer I355BF9H; Edwards Lifesciences) was placed into the left jugular vein. Via this introducer, the Swan-Ganz catheter equipped with a thermal filament was advanced through the right atrium and ventricle and the tip positioned in the pulmonary artery. The advancement and subsequent positioning of the Swan-Ganz was guided by analysis of the blood pressure waveform. The tip was positioned in the pulmonary artery. The thermal filament was positioned from the right atrium to the right ventricle outflow tract. 5 The automated self-test procedure of the computer did not warn about the CCO Swan-Ganz catheter malfunction neither after connection to the cardiac output monitor nor during the measurements.
Results
Arrhythmias
Anaesthesia was induced at 10:40 h. CCO measurements started at 12:05 h and were only interrupted to allow measurement of cardiac output by BTD. Capnoperitoneum was initiated at 13:12 h. The intra-abdominal pressure was 13 mmHg. At 13:26 h, the first isolated ventricular extrasystole (VES) was observed. The incidence of VES slowly increased and the first burst of several VES (2–5 VES) was noticed at 13:55 h. The incidence of bursts also increased slowly. During a CCO interruption of 5 min to allow BTD, cardiac arrhythmias disappeared and a relation between CCO measurement and the apparition of VES was suspected. The position of the Swan-Ganz catheter was verified using pressure trace analysis and found to be unchanged and correct. From this moment on, the temperature of the heating filament was monitored using special settings of the CCO computer. Each time the temperature of the thermal filament reached a peak, this temperature was manually recorded and a marker was produced in the S/5 Datex-Ohmeda multiparameter anaesthesia monitor. Peaks of the filament temperature (40.1–41.3°C) were noticed (Figure 1) at the same time as the VES and/or bursts of VES. To confirm the existence of a causal relationship, the CCO measurement was switched off for 3 min. The arrhythmias disappeared. Then the CCO measurement was again switched on and after 25 s, the arrhythmias reappeared and remained present as long as the CCO measurements were running. Finally, CCO was definitively switched off and no more arrhythmias were observed up to the end of the anaesthesia. Arterial blood pressure was unstable during the episodes with arrhythmias but no hypotension occurred. Cardiac output during the arrhythmias remained unchanged and in physiological range. Recovery was uneventful. The time sequence is summarized in Figure 2.
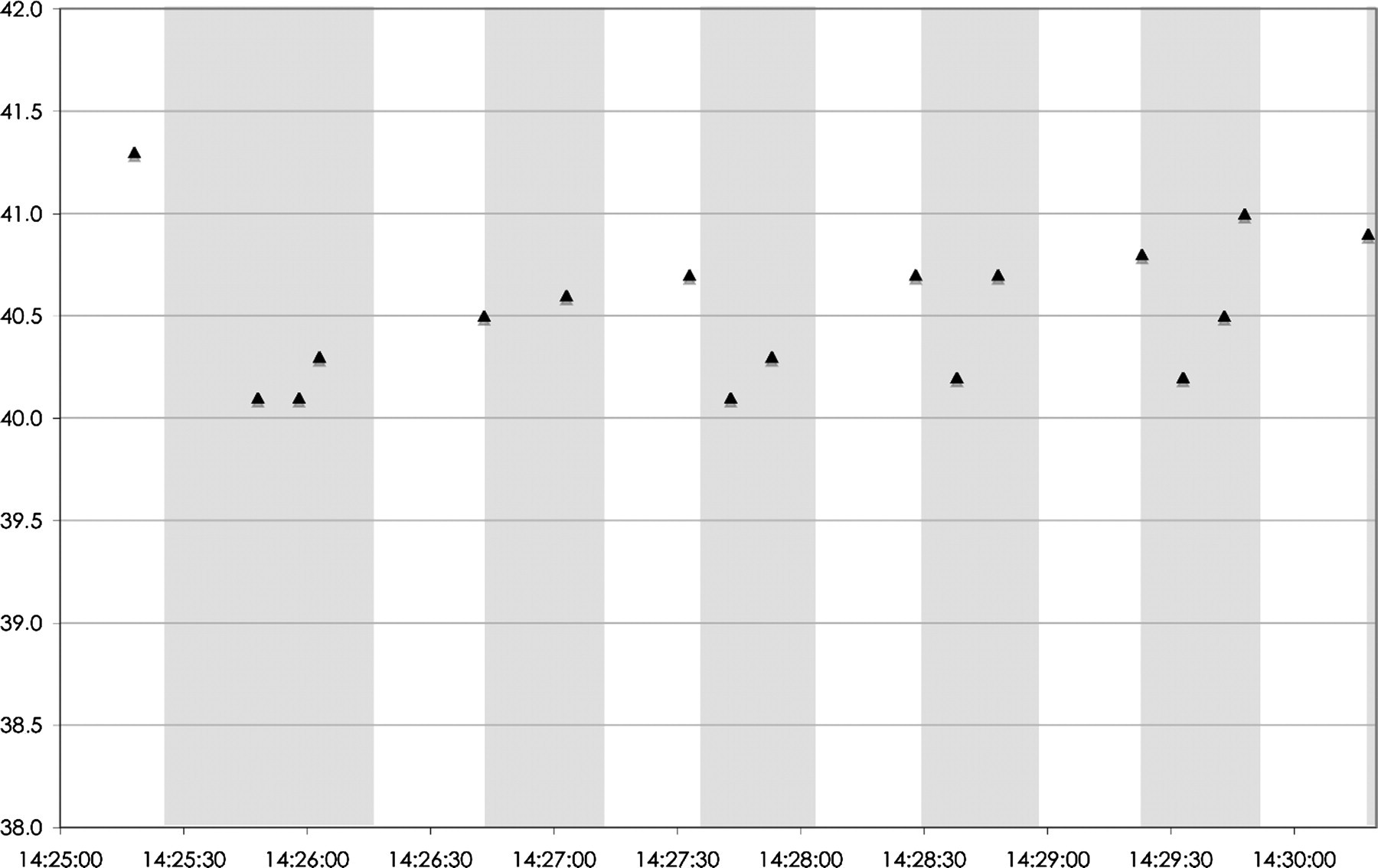
The peak temperature of the thermal filament reported on a timescale. Precision of the time of peak temperature is ±5 s (=precision of the marker on the S/5 Datex-Ohmeda anaesthesia monitor). The grey background represents the periods with arrhythmias
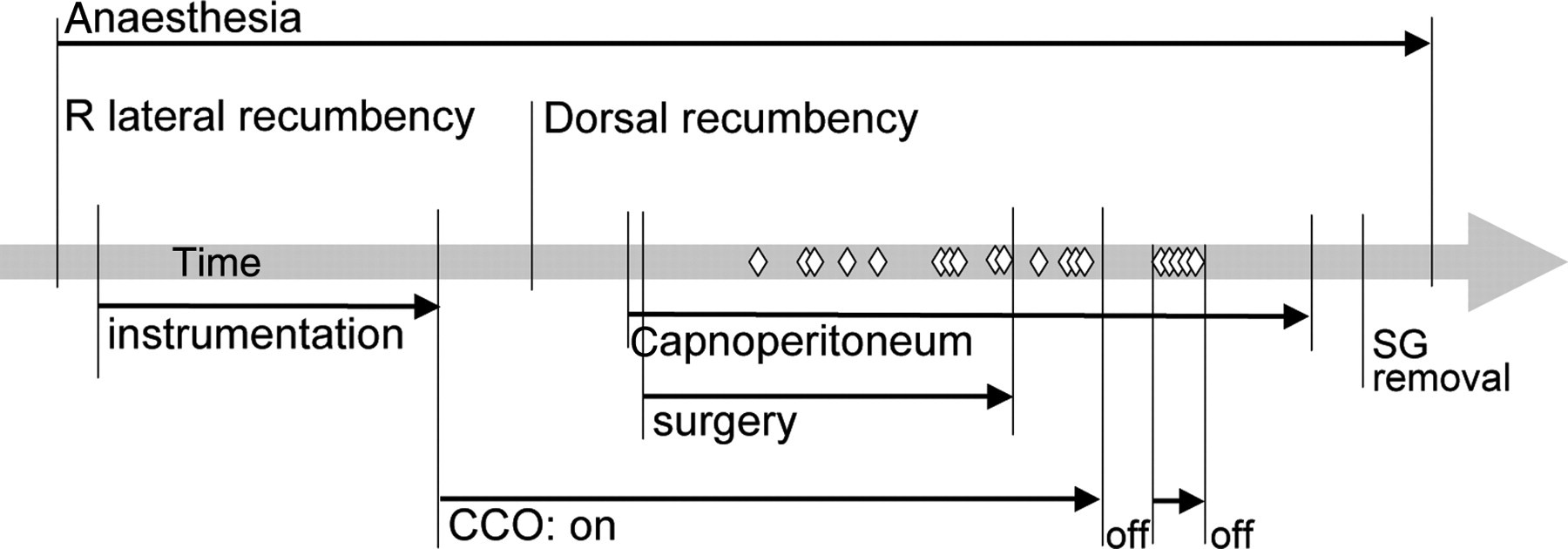
Time sequence of arrhythmia appearance. ◊: arrhythmia; CCO: continuous thermodilution cardiac output; SG: Swan-Ganz catheter
The analysis of the ECG confirmed that the arrhythmias were right VES with a very consistent morphology associated with atrioventricular dissociation (Figure 3).
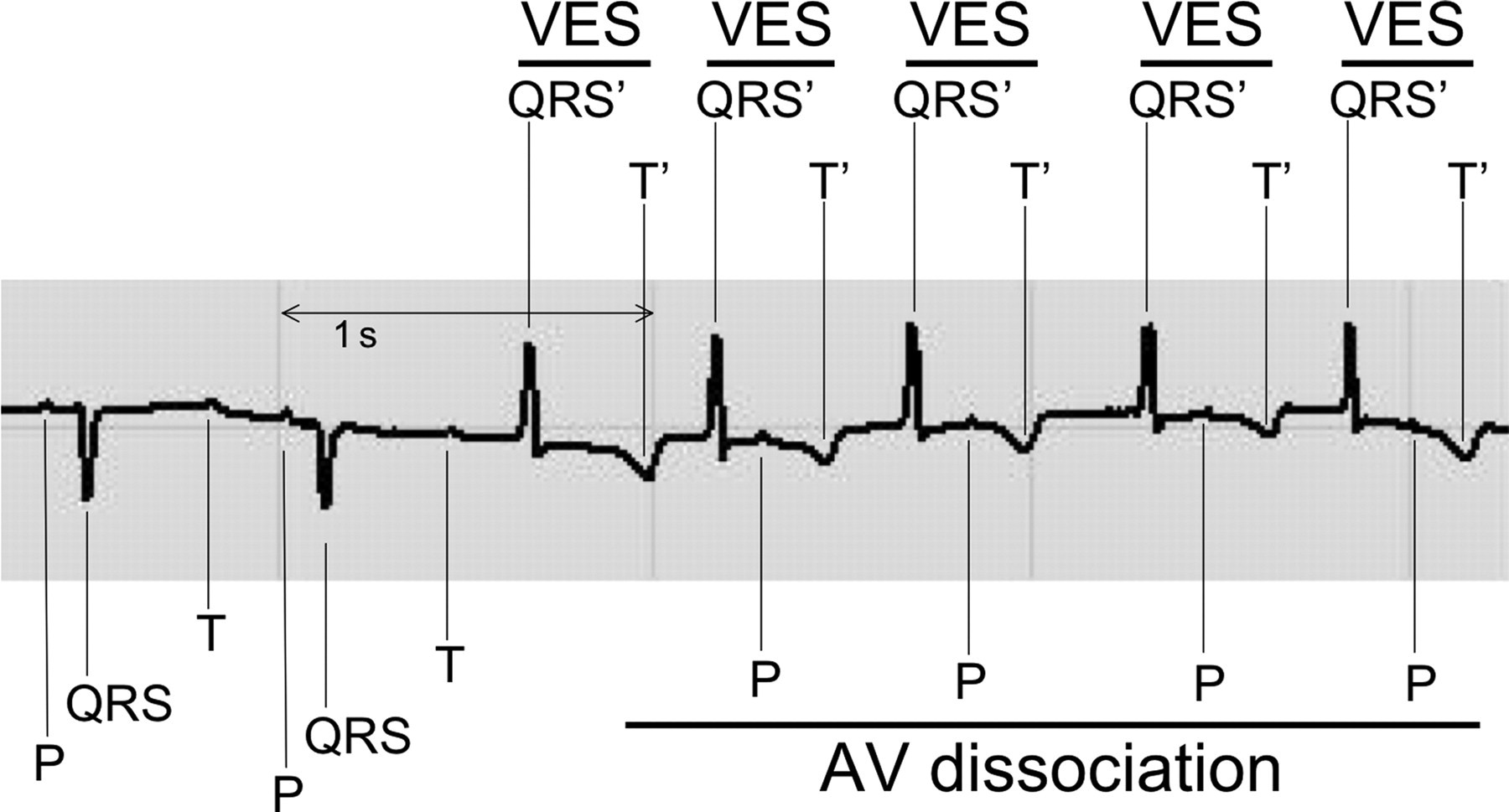
Electrocardiogram (lead II) during continuous thermodilution cardiac output measurement. AV dissociation: atrioventricular dissociation; VES: ventricular extrasystole
Discussion
In general, the possible complications of pulmonary artery catheterization are associated with the establishment of venous access and the insertion, presence and removal of the Swan-Ganz catheter. Complications reported include arterial puncture, pneumothorax, nerve damage, vascular rupture, irritation or damage of the endocardium or intracardiac structure, thrombosis, embolism, pulmonary artery rupture, infection and subsequent septicaemia. 6 Mechanical problems were also reported such as interluminal leak 7 and with the use of the special CCO Swan-Ganz catheter, the peeling of the plastic protection of the thermal filament. 8,9
Ventricular arrhythmias are more commonly encountered during insertion 6,10,11 or removal 11,12 of the Swan-Ganz catheter, presumably caused by direct irritation of the endocardium. Arrhythmia related to the sole presence of the pulmonary artery catheter is less common. Critical arrhythmias encountered in a parturient woman with severe pulmonary hypertension were solved by the total removal of the catheter. 13 In the sheep presented in this article, cardiac arrhythmias occurred that were not linked to the presence of the Swan-Ganz catheter by itself but specifically related to intermittent heating of the thermal filament of the catheter. Indeed, arrhythmias stopped when CCO measurement was switched off despite the fact that the position of the catheter did not change.
The use of CCO in sheep was first described in 1992. 3 The thermal safety of the CCO Swan-Ganz catheter was tested and considered adequate. 2 Continuous use of the CCO technique using CCO Swan-Ganz catheter for up to seven days in sheep did not create additional risk over a standard pulmonary artery catheter. 1,14 The sheep in the present case report did not present any arrhythmic problem during the first anaesthesia performed 14 days earlier. Baseline ECG was normal. The peaks of temperature of the thermal filament were within the normal range. Some irritation of the endocardium due to the first use of the CCO technique could have elicited the problem during the second anaesthesia. However, the VES appeared more than 1 h after the CCO technique was initiated and the position of the Swan-Ganz catheter was not modified during this period. It is possible that cardiac arrhythmias in this case were caused by the CCO catheter being in permanent contact with the endocardium at the level of the thermal filament. The repeated heating and subsequent irritation of the endocardium then presumably provoked the VES and atrioventricular dissociation. The degree of irritation seemed to increase progressively. First, isolated VES appeared with increasing frequency. This was followed by bursts of two to five VES and finally, longer bursts associated with atrioventricular dissociation. The morphology of the VES was very consistent suggesting that the ectopic ventricular depolarization originated from the same place, possibly an area of irritated myocardium, meaning that the irritation of the endocardium was precisely localized.
The use of the CCO technique in anaesthetized sheep can be associated with arrhythmias most likely due to thermal irritation of the right endocardium.
This can be confirmed when switching off the CCO measurements causes the arrhythmias to disappear. To the best of the author's knowledge, this complication has not been reported in humans. These arrhythmias could be potentially dangerous especially in the critically ill patient. Therefore, if this is the case, stopping CCO and continuing the cardiac output monitoring by BTD using the same equipment is recommended.
