Abstract
Large numbers of Atlantic salmon (
The need for standardization of research fish
Standardization of the variables in a research protocol is a vital prerequisite for the generation of reliable and reproducible data. This process must include the test system (the animals and the conditions under which they are kept) as well as the treatment to which they are subjected. The correct reporting of studies involving animals is also an essential part of ethically acceptable research,
1
as it contributes to the implementation of the three Rs (
Hundreds of genetically well-defined rodent lines, including mutants and transgenic animals, are available for mammalian researchers. The situation is very different for scientists studying salmonids, despite the fact that large numbers of these animals are used in research worldwide. In Norway alone, 3.3 million Atlantic salmon (
Analysis of the Norwegian statistics shows that the majority of fish are used for potency testing of fish vaccines.
7
One of the main reasons for the large numbers of fish used is their genetic variation. Research fish are normally acquired from local breeders or from national breeding companies. These suppliers may be able to provide some basic information on breeding and production traits, but they have usually no knowledge of the genetic constitution of the animals. Furthermore, the health monitoring of fish populations is often inadequate compared with the standards that are now commonplace in mammalian research, making even access to healthy fish difficult for research institutions.
8
There are currently no FELASA guidelines for health monitoring of fish (
As stated by Festing, 11 the best strategy to obtain reliable results with high statistical power from animals with genetic heterogenicity is to use small numbers of animals from several inbred strains. Producing such inbred lines for a species with a four-year generation interval would take 80 years using the definition of inbreeding for mice (20 generations) and thus is not an option. In the early 1980s, the production of homozygous salmonids was started using the techniques of androgenesis and gynogenesis. 12,13 These methods have since been adopted for a wide variety of fish species. 14 However, interest gradually waned due to the emergence of inbreeding depression, the physical space required to house these animals, and the long generation interval (3–4 years). Today, only a few rainbow trout lines are available (G Thorgaard, Washington State University, USA; E Quillet, INRA, France) and none of these produce at a commercial level. There are no clonal lines for Atlantic salmon or brown trout. Furthermore, significant differences in the susceptibility of trout and salmon to pathogens have been identified. 15,16 Extrapolation of results from one species to the other may therefore not be valid, increasing the need for standardized salmon.
Consequences of genetic variation in fish
Genetic differences between families form the basis for selective breeding, which has resulted in improved growth, feed uptake and disease resistance. 17 Since most research fish in Norway are used for vaccine testing, 7 their disease resistance is of crucial importance. Heritability of disease resistance in fish has been shown for parasites, 18 viruses, 9 fungi 19 and bacteria. 20
Identification of the genes that influence disease resistance in fish is the subject of intensive research worldwide and provides excellent examples of how the genetic constitution of fish influences experimental results. One of the first candidates to be tested was the major histocompatibility complex (MHC). The MHC is an essential part of the immune system and has been found to be closely linked to resistance to virally-induced disease in chickens. 21,22 In humans, the MHC is largely linked to autoimmune disease, 23 although links to hepatitis C virus resistance and the progression of HIV infections have also been identified. 24,25 The MHC is highly polymorphic in fish as in other species and consists of two unlinked classes of molecules (classes I and II) that are encoded by one major locus per class in salmonids. 26–29 Several studies have shown that MHC genes also affect resistance towards infectious diseases in fish. 30–35 Thus, the results from any challenge or vaccination trial will depend upon the MHC content of the salmonids used, in addition to other genes of immunity which the animals possess.
In animals with identical MHC classes, other unknown genes may also affect disease resistance. Mouse models have elucidated some of the genetic complexity regulating autoimmune diseases in humans. 23 In a study on Atlantic salmon, three half-sibling families with identical MHC were challenged with three different pathogens. 36 Mortality rates were 8–27% for infectious pancreatic necrosis (IPN), 73–84% for infectious salmon anaemia (ISA) and 83–96% for furunculosis. Similar studies in which two half-sibling families with identical MHC were challenged with 11 different ISA viral isolates 37 revealed differences between and within families (Figure 1). These studies suggest that both the MHC as well as background genes affect resistance, and that different genes affect resistance to different isolates and different pathogens. Variations in the genetic constitution of the experimental animals will, therefore, have a great influence on the results of experiments on disease resistance or in vaccination trials.
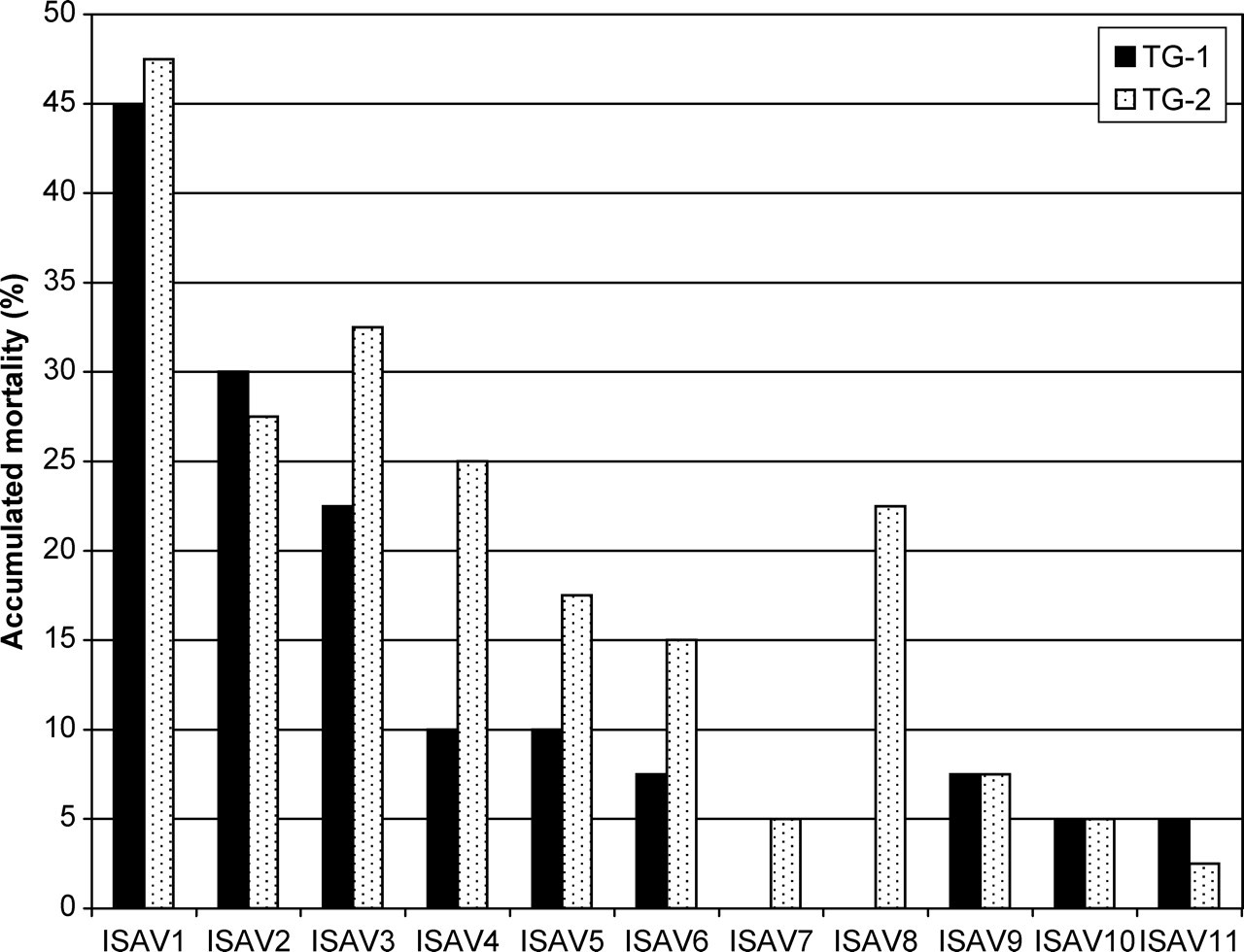
Accumulated mortality in two half-sibling groups (TG-1, TG-2) of Atlantic salmon,
A challenge study performed on a random population of breeding salmon with families containing seven MHC class II alleles (
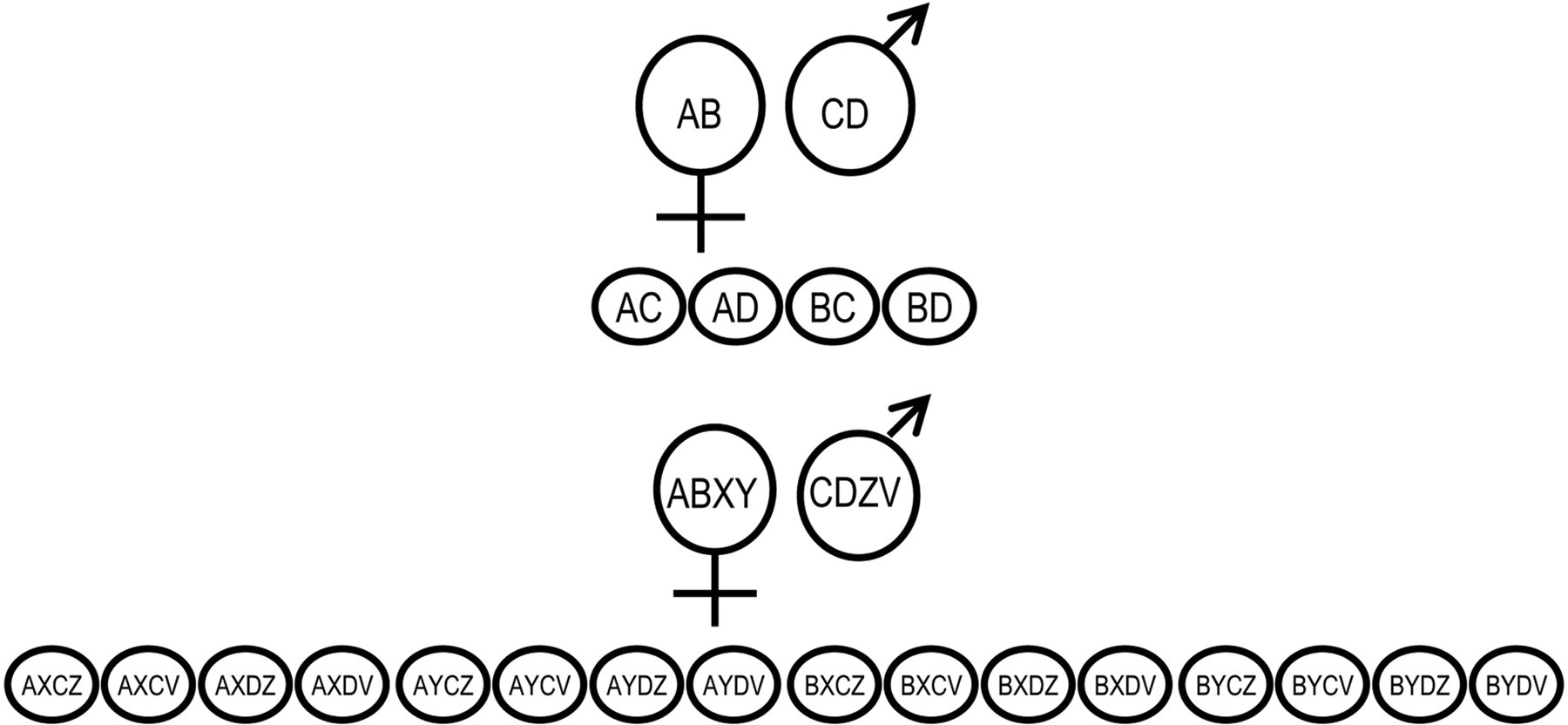
One polymorphic gene in the mother and father gives rise to four possible haplotypes in the offspring, while two affecting genes give rise to 16 haplotypes. Number of possible haplotypes with multiple affecting genes is given by the formula 4
Recently, the duplicated MHC regions in Atlantic salmon have been sequenced. 39 When comparing these regions to those of rainbow trout, 40 there are apparent differences in the three functional non-classical MHC class I genes residing in the rainbow trout region (UCA, UDA, UEA), but only in one MHC class I pseudogene (ψUCA) in the Atlantic salmon IB region. In rainbow trout, the IB region has been associated with resistance to both IPN virus and infectious haematopoietic virus. 30,34 Studies are in progress to evaluate how these duplicated regions affect IPN resistance in Atlantic salmon (unpublished data), but it may be assumed that, since only one non-classical class I pseudogene resides in this region, it will have a different effect on resistance.
The above data show that although closely related, gene content and function vary in salmonids and extrapolation of results between studies may not be possible for some traits.
Methods for producing genetically standardized salmonids for research
Inbreeding or full-sibling crosses
Traditional inbreeding is not an option for the production of genetically homozygous salmonids as they have a generation interval of three to four years. An alternative to full inbreeding, although not optimal, would be to cross siblings, reducing much of the genomic variability compared with outbred animals. Such crosses are surprisingly viable in salmonids (Storset A, personal communication) although inbreeding depression, including the appearance of lethal mutations, may arise. These crosses are easily produced by major breeding companies, but have not yet been adopted on any scale by the research community. An additional problem is that, assuming both parents are heterozygous for two to six genes affecting the outcome of the study, extensive genotyping of the offspring would be needed to offer animals with maximum homogenicity.
Androgenesis or gynogenesis
Androgenesis or gynogenesis has been attempted by several research groups for a wide variety of fish species. 14 Androgenesis involves the treatment of eggs with a strong radioactive source that cuts the maternal genome into pieces, followed by fertilization with normal sperm and then inhibition of the first mitotic division using either heat or pressure shock just after the chromosomes have been duplicated but before the cell has had time to divide (Figure 3). The resultant animals are completely homozygous, bear only male chromosomes but produce both male and female offspring. Gynogenesis involves irradiation of sperm, fertilization of normal eggs and then inhibition of the first mitotic cell division (Figure 3). The resultant gynogens are all females and carry a duplicate set of the maternal chromosomes. Each androgen or gynogen is unique, with different combinations of maternal or paternal chromosomes. Once they reach sexual maturity, they may either be crossed, producing identical heterozygous offspring, or used for a second round of androgenesis or gynogenesis to produce unique clonal lines (Figure 4).
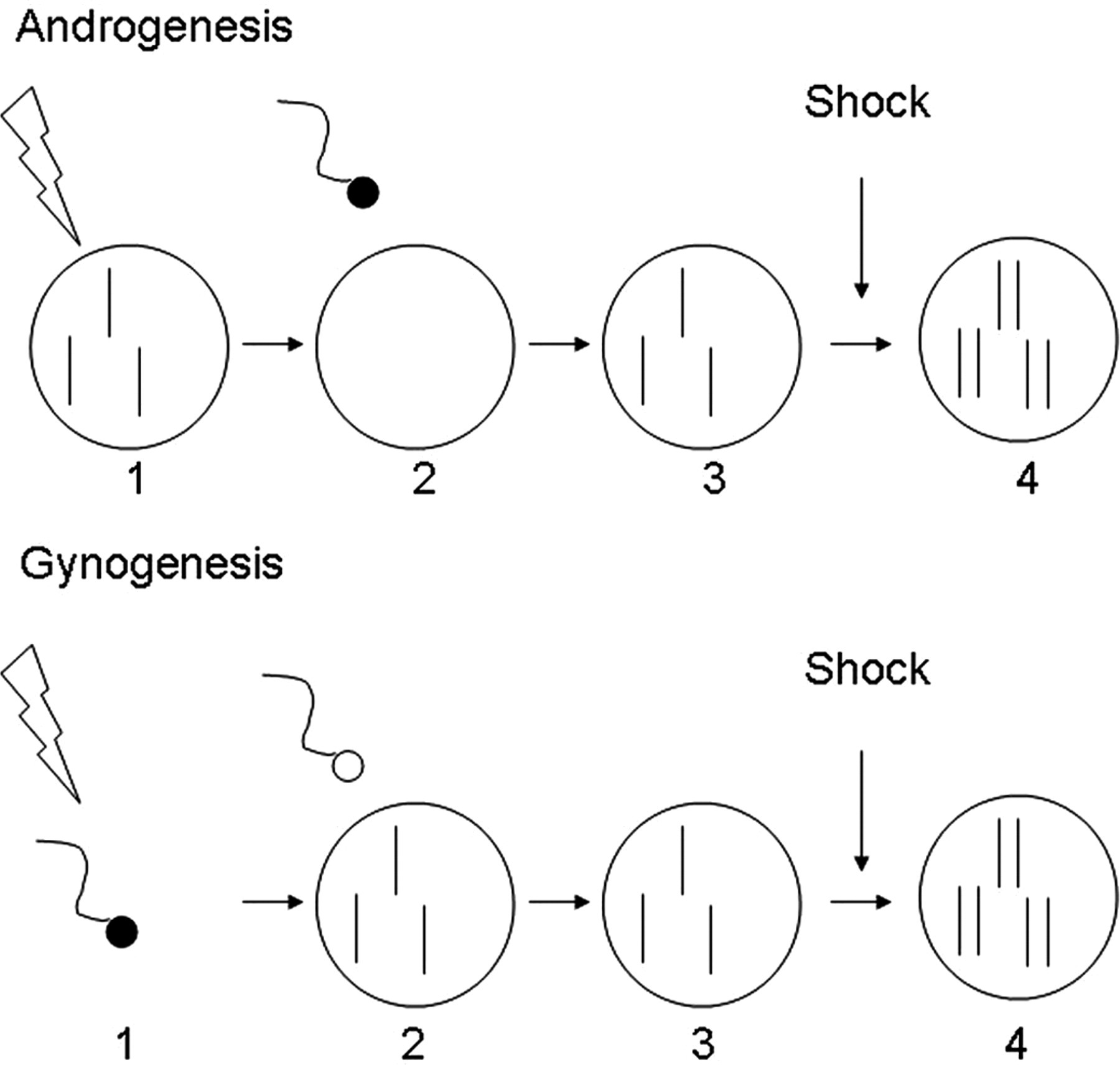
Schematic presentation of androgenesis and gynogenesis. For androgenesis, eggs are irradiated with a strong radioactive source that destroys the maternal genome. The eggs are then fertilized with normal milt, thereby introducing a haploid paternal genome into the egg. Finally, heat or pressure shock is used to prevent the first cell division, thus doubling the paternal genome. A similar process is used for gynogenesis, but in this case the milt is irradiated and then used to fertilize normal eggs
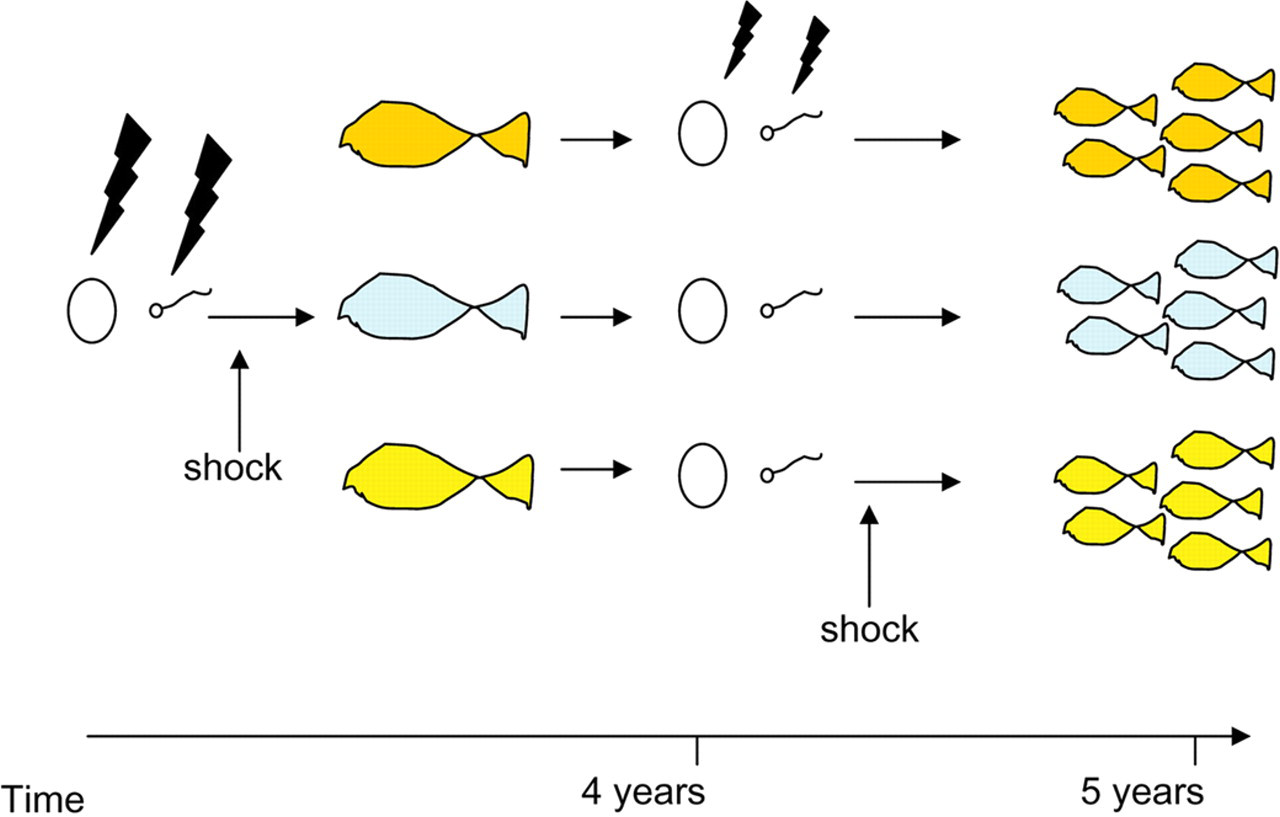
The first round of androgenesis or gynogenesis produces animals with different genetic makeup even though they originated from one parent. To produce clonal lines of genetically identical animals, a second round of androgenesis or gynogenesis is needed
In general, the survival rate after androgenesis and gynogenesis is low due to unsuccessful doubling of the parental genome, homozygous detrimental mutations or poor egg quality (Thorgaard G, personal communication). Survival is usually assessed at the eyed-stage, at hatching or at start-feeding, with little data available on survival to reproductive age. Scheerer
Gamete quality is one of the factors known to influence the success rate of androgenesis and gynogenesis. 41,42 To improve survival, much attention has been paid to improving the methods involved, including the optimal radiation source, dose, type of shock and application time. 43 Other factors such as the time of stripping have also been investigated. 44 Morphological abnormalities such as tail or spine deformities are frequently observed. 45 These may be attributable to detrimental homozygous mutations carried by the parent or to the damage caused by irradiation of components of the egg that are essential for viable development. Although animals with severe deformities die before start-feeding (personal observation), the welfare aspects of androgen or gynogen survivors must be assessed and compared with those that arise when large numbers of outbred animals are used.
Clonal lines
A second round of androgenesis or gynogenesis (depending on the sex) must be undertaken on the isogenic salmonids that survive to a fertile age in order to produce clonal lines (Figure 4). These clonal lines may then either be further maintained through new cycles of androgenesis or gynogenesis, or crossed within or between lines to produce identical homozygous or heterozygous animals. Manipulation techniques also exist for fish with, for example, hormonal sex reversal, 46 enabling a cross within or between clonal line siblings, producing identical homozygous or heterozygous animals, respectively. Shock inhibition of the second meiotic division, doubling the homozygous egg genome, followed by fertilization with irradiated milt is another alternative for preserving these lines. 47
The production and maintenance of homozygous clones are not without problems. Inbreeding depression is considerable and many fish are lost during maturation. Gonadal defects and poor egg quality further reduce the number of animals that can be reproduced. 41,48 Access to a suitable aquarium facility for keeping 5–20 kg animals, in addition to the challenges of a two to four year generation interval, may also discourage scientists from choosing these techniques. Thus, most clonal lines have been generated for a specific scientific purpose and have then been terminated.
As each androgen or gynogen animal gives rise to a unique clonal line, each of these represents a unique biological tool for identifying parameters that are important for the fish industry, such as feed uptake, growth rate, deformities and disease resistance. Thorgaard and co-workers have generated multiple clonal lines of rainbow trout using gynogenesis and androgenesis,
49
allowing analysis and genetic dissection of traits as the differences are identified
50,51
and genetically characterized.
52
Robinson
Crosses between different clonal lines with subsequent androgenesis of the F1 population have been invaluable tools for gene mapping and for detection of QTLs. These double-haploid F2 progeny are genetically different from one another due to recombination events occurring during the production of F1 gametes. Young
Many research groups have demonstrated that the production of clonal fish lines is feasible, but the maintenance of these lines is fraught with problems due to husbandry costs and inbreeding depression. However, if the scientific community itself demands standardized research fish, these problems could be overcome. Inbreeding depression will always be a challenge, but the existence of clonal rainbow trout lines 53,57 demonstrates that these are surmountable.
The issue of who should assume responsibility for maintaining and supplying the scientific community with genetically standardized fish remains to be addressed. The expense of maintaining clonal lines of salmonids with a generation interval of three to four years is likely to be considerably higher than that for laboratory rodents or zebrafish.
Cryopreservation of milt and eggs
An ideal situation would be to establish a gene bank with genetically defined eggs and milt from androgen or gynogen animals, allowing the production of animals of known genotype whenever required. The problems involved in the cryopreservation of milt have been solved and this procedure is routinely performed with high fertility rates. 58 Cryopreservation of eggs and embryos is more complicated due to their large size and water content, in contrast to the situation in mice and humans. 59 All oocytes and embryos are subjected to morphological and functional damage during cryopreservation, but the extent of the injury, as well as differences in survival and developmental rates, may vary considerably between species, developmental stage and origin. 60 The technology is, however, improving. It is thus likely that these technical problems will be solved in due course, facilitating a continuous supply of genetically defined animals and conservation of clonal lines.
Laboratory fish models
Rodent models are rapidly being replaced by laboratory fish models in some areas of toxicology and basic research,
61,62
but there has been very little focus on replacing farmed fish with smaller or more standardized laboratory animals. Our basic knowledge of the health, genomics and embryology of the most common laboratory fish species, such as zebrafish (
Conclusions
There are good ethical and scientific reasons for improving the availability of standardized Atlantic salmon for use in research. The production of standardized animals, although more complicated than in mammals, is technically possible. If gynogenesis or androgenesis proves to be too problematic or time-consuming, other approaches should be sought. Branch organizations and breeders should be encouraged to make these available for more general use in the research community. Research on laboratory fish species should be intensified to increase their usefulness as models for salmonids. In parallel, greater attention should be paid to the principles of modern laboratory animal science that are already possible to implement in fish research, including standardization of the animals' environment, health-monitoring programmes and good statistical design.
