Abstract
This experimental animal study aimed at evaluating a new prosthesis to replace the ossicular chain; we developed a new technique for surgical implantation into the middle ear of rabbits. The rabbit middle ear is, owing to the relative anatomical dimensions involved, an ideal environment for implantation procedures involving the ossicles, as the surgical conditions are similar to those of the human middle ear. This study included a total of 34 approximately six-month-old female white rabbits (New Zealand) weighing between 3.2 and 4.4 kg. The implants used were constructed of ceramic materials (titania, TiO2) of various pore sizes. Directly prior to implanting the total ossicular reconstruction prostheses (TORPs), as well as at 28, 84 and 300 days after implantation, electric response audiometry was used to determine the hearing thresholds of the animals (bone conduction; click stimulus nHL). An erbium:YAG laser was used to excise the original ossicular chain. Following implantation, we were unable to detect any stenosis of the outer ear canal or perforation of the tympanic membrane. The conductive hearing threshold was in the range of 4.21 ± 6.68 dB nHL (n = 131). The hearing level showed no significant difference before and after surgery (P < 0.05).
One aim of tympanoplasty is the reconstruction of the ossicular chain to improve hearing. 1,2 Over the past few decades various materials, including ceramics and metals, have been recommended as high-quality implant materials. 3–9 Although alloplastic materials are suitable for implantation, they do have certain drawbacks such as the dissolution of the materials in long-term implantation, impaired sound conduction, reduced biocompatibility and a lack of biointegration resulting in extrusion. Recently, commercially pure titanium has gained significant acceptance as an alloplastic material used in reconstructive ossicular chain tympanoplasty. Its effectiveness has been documented in experimental studies and in clinical trials. 10–13 Oxide ceramics are also among the biocompatible substances of choice currently. For instance, there has been a marked preference for titanium dioxide (TiO2), as studies show that it is associated with reduced rates of ion release on the implant surface, especially in 0.1 mmol/L hydrochloric acid at 37°C. 14,15 The hydrolyzed surface of the material adsorbs proteins, 16,17 which allows for direct interaction with cells and the extracellular matrix. Ceramics as a whole are also unique because their surface compositions can be varied. An implant material with a porous surface structure can therefore be produced, which influences the interaction between the surrounding tissue and the implanted substance. In addition, porosity suppresses the development of fibrous capsules and inflammatory reactions, a key factor in promoting long-term stability and integrity. 18
The purpose of this interdisciplinary experimental study was the functional characterization of a newly developed ossicular chain replacement prosthesis. The suitability of this prosthesis, which is composed of TiO2 ceramic, was investigated by performing reconstructive surgery of the ossicular chain in the middle ear of rabbits. The rabbit model has been widely used for surgical and experimental approaches. 19 Known surgical techniques have been used to insert various prostheses into the middle ear for material-testing purposes, although no functional replacement for the ossicular chain has been created. The present study is based on a surgical technique derived from laser surgery of the human middle ear, designed to allow a low-impact procedure by means of the contact-free use of the erbium:yttrium-aluminium-garnet (er:YAG) laser. Brainstem audiometry investigations were carried out in order to verify that the implantation material had no toxic effects and that the use of lasers had no deleterious impact on inner ear function.
Materials and methods
Animals
For this experimental animal study (no. 509c-42502-97/990), approved by the administrative district council of Hannover in accordance with paragraph 8 of the German Animal Protection Act (TierSchG) dated 25 May 1998, a total of 34 approximately six-month-old New Zealand White female rabbits originating from the Charles River animal breeding farm (Sulzfeld, Germany) were used in the examinations. Species selection was based on its similarity to human middle ear anatomy and the standardized, microsurgical approach to the tympanic cavity that is favoured. 19 The animals were divided into three groups and euthanized after periods of 28, 84 and 300 days postsurgery.
The animals weighed between 3.2 and 4.4 kg prior to surgery. They were housed in individual cages in a climate-controlled room at the Medical University of Hannover's Central Animal Laboratory. The cages measured approximately 50 × 60 × 45 cm in size, with a floor consisting of a perforated metal plate. They were equipped with a nipple drinking system providing the animals with water ad libitum. The rabbits received 100 g of a pelleted, all-in-one food (SSNIFF, Soest, Germany) and some 150 g of autoclaved straw daily. The temperature in the room was around 20 ± 2°C, with humidity at 55 ± 5%. The animals were kept under an artificially controlled, 12 h light/dark cycle.
Anaesthetic management
All measurements were performed under inhalation anaesthetic. Anaesthesia by intubation has crucial advantages over anaesthesia by barbiturates. The depth of anaesthesia can be better controlled, enabling mortality to be considerably reduced. 20
At the beginning of the study, a total of 34 animals were involved in the experiment. Five rabbits died the first time anaesthesia was induced. In these individuals, bradycardia (i.e. heart frequency <100/min) was observed immediately after the tube was put in place, with the animals not responding to subsequent treatment (i.e. repeated intravenous [i.v.] administration of Atropin® [0.05–2 mg/kg body weight]). In these rabbits, cardiac arrest occurred 3–4 min after the onset of bradycardia. Following these anaesthetic incidents, which occurred at the start of the experimental phase, the anaesthesia regimen was altered. The regimen was augmented by additional administration of the anticholinergic agent Robinul® (Riemser, Riems Island, Germany). Two further rabbits died during the last measurements due to uncontrollable bradycardia.
Prior to surgery, each animal was examined by a veterinarian as to its general condition in order to eliminate disease and to minimize risks of anaesthetic complications. Body weight was determined in order to allow precise individual dosage of the medication. Two premedication drugs were administered intramuscularly (i.m.) 25 mg/kg of ketamine (Ketanest®, Albrecht GmbH & Co KG, Aulendorf/Württemberg, Germany) and 5 mg/kg of midazolam hydrochloride (Midazolam®, CuraMed Pharma GmbH, Hameln, Germany). Anaesthesia was induced by administering the short-acting anaesthetic agent Propofol-Lipuro 1% (Propofol®, B Braun Melsungen AG, Melsungen, Germany) at a dosage of 1 mg/kg, into the auricular vein of the left ear via an indwelling i.v. catheter (0.9 mm × 25 mm; B Braun Melsungen AG). Five milligrams of glycopyrrolate (Robinul®) were then administered i.m., with 5 mg of buprenorphine hydrochloride (Temgesic®, Essex Pharma GmbH, Munich, Germany) administered subcutaneously. Following endotracheal intubation, anaesthesia was maintained with 1.5% isoflurane (Forene®, Abbot, Wiesbaden, Germany). An infusion of 10 mL/kg/h of Sterofundin-HEG-5 (B Braun Melsungen AG) was used to stabilize the animals' circulatory system. In order to ensure that decreasing body temperature did not influence the data, each animal's body temperature (taken per rectum) was stabilized at 39±0.5°C using a heating mat on which the rabbits were placed in prone position. To avoid temperature variations, an additional red-light heating lamp was activated when the temperature fell by >0.5°C. Temperature readings were taken continuously throughout the experimental period. Anaesthesia was performed using a semi-open anaesthetic machine (Dräger Medical AG & Co KGaA, Lübeck, Germany) and maintained using a mix of isoflurane (Forene®) and oxygen (1.5%/1.5 L/min on average). Once the measurements were completed, the animals were immediately euthanized using 3 mL of pentobarbital administered i.v. (Eutha 77®, Pitmann-Moore, Inc, Washington, USA) while still under anaesthetic.
Surgical procedure
The implants used for the surgical procedures were constructed of the ceramic material TiO2. Each prosthesis consisted of a round and flattened head (diameter 2.7 mm; height 0.5 mm) with a centrally placed and elongated hollow shaft (diameter 0.6 mm; length 3.7 mm). The individual weight of a single prosthesis averaged 0.10 g. The shafts of the prostheses were densely sintered and thus virtually poreless, irrespective of the material used. The total ossicular replacement prostheses (TORPs) were implanted in the right middle ear cavity. The left middle ear of each animal remained intact and served as a control.
The operational site was disinfected prior to surgery. In this region, povidone iodine was applied repeatedly on a large area behind the ear up to the apex of the head using a sterile swab (Barunol®, B Braun Melsungen AG). The animal was completely covered with sterile cloths. The surgical instruments were cleaned of blood and residue with a brush and autoclaved after each procedure. The surgical microscope was covered with a sterile disposable plastic cover before each operation.
Gaining access to the middle ear is more difficult in rabbits than in humans owing to the very small dimensions involved, 21 thus necessitating the use of a surgical microscope (OPMI Twin ER Zeiss, Oberkochen, Germany). First, an incision of around 2 cm in length was made behind the ear (Figure 1), and the muscles were cleanly transected. The bony initial portion of the ear canal could be located by palpation. This exposed the cartilaginous ear canal up to the junction with the bony region over a length of 0.5 cm. Where the two zones meet, the cartilage was removed and a retractor inserted. The ear canal was opened and the eardrum was now visible. The mucosa of the ear canal was carefully dissected off, with the eardrum partly removed, and the middle ear opened. A small House spoon was used to widen the rim of the ear canal where it met the eardrum, until the head of the malleus and the incudostapedial joint were fully exposed. The ossicular chain is very fragile and can be destroyed by even minimal manipulation.
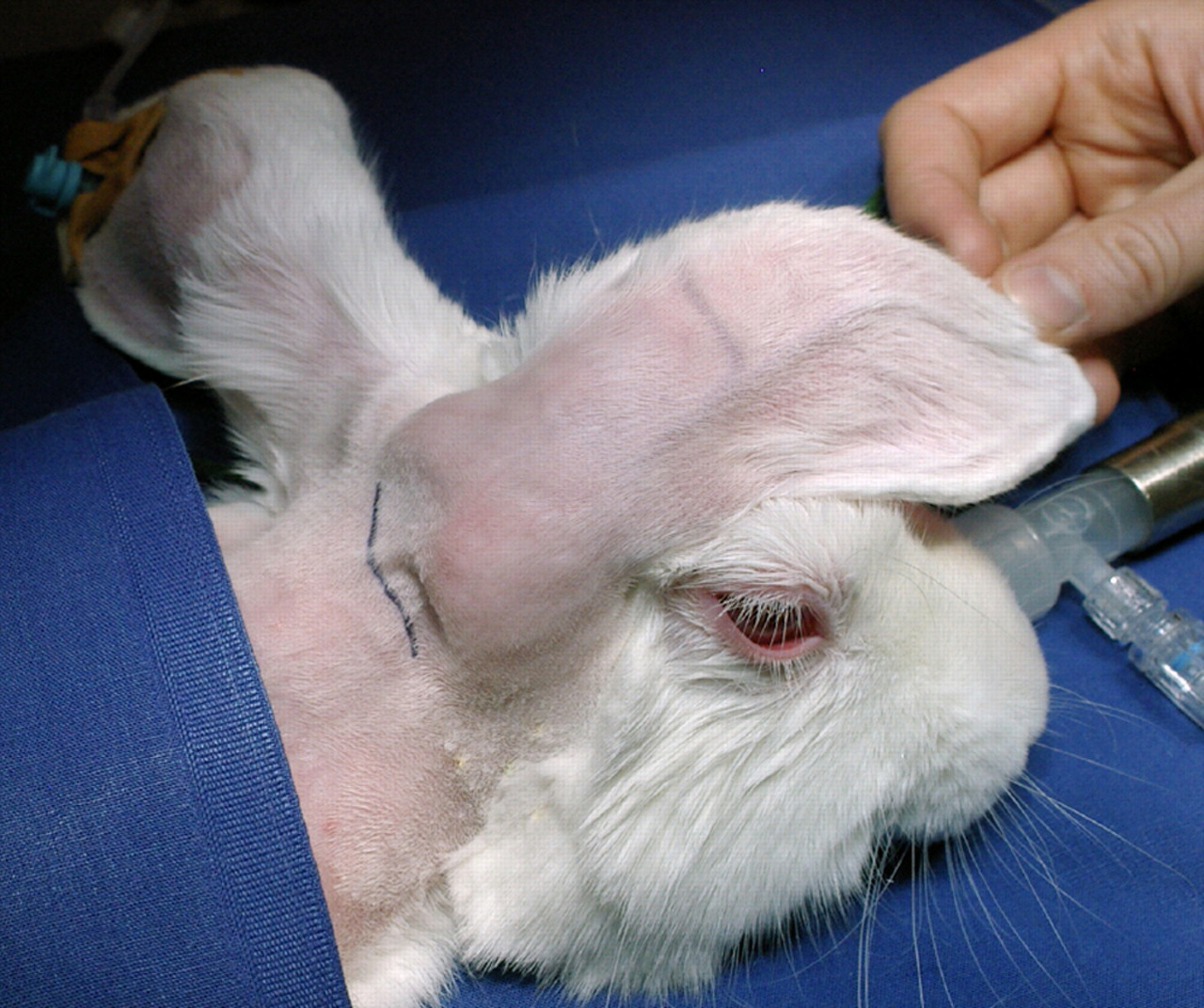
Retroauricular skin incision (approximately 2 cm in length)
The transection of the incudostapedial joint was performed using an er:YAG laser with a total of 20 impulses at 30 mJ. The incus was separated from the head of the malleus using 30 impulses at 50 mJ. The final step involved separating the stapes superstructure from the crura of the stapes using 20 impulses at 15 mJ. Cut-off material generated by the removal of the ossicles was removed from the middle ear using a small suction device. Material fragments broken off during the excision of the ossicular chain were removed from the middle ear using small suction devices. Unhindered placement of the implant between the tympanic membrane, the remaining manubrium of the malleus, and the stapes footplate was performed by individually adapting the size of each prosthesis. In five cases the overhanging bony canal over the facial nerve had to be removed in order to obtain a good view of the footplate, thus enabling the prosthesis to be properly positioned. It proved unnecessary to displace the facial nerve.
To avoid excessive formation of granulation tissue at the cartilage/bone interface of the auricular canal, silicone foils and tamponades (Gilitta; Braun) soaked in doxycycline (Ratiopharm, Ulm, Germany) were deposited at this site. The wound margins were adapted using Vicryl suture material (4/0 1.5 metric, SH-1 plus; Ethicon GmbH, Norderstedt, Germany) and closed with Mersilene (2/0 3.0 metric; Ethicon) button sutures. After surgery, 10 mg/kg of the antibiotic Baytril® (Bayer Vital GmbH, Leverkusen, Germany) were given in tablet form for oral intake over a 10-day period. The tablets were dissolved in water and administered by mouth using a small syringe.
Postoperative course
Overall, very few complications were experienced either intra- or postoperatively. Three animals showed postoperative vertigo with nystagmus, although this improved over time without treatment. No wound infections occurred. Facial paralysis was not observed in any cases. Owing to wound dehiscence, the wound of one animal had to be resutured.
Brainstem audiometry investigations
Functional measurement of acoustically evoked potentials via bone conduction was performed using the Nicolet Viking IV P System (Nicolet Biomedical, Inc, WI, USA). The device used was a bone conduction earphone manufactured by Oticon, Inc (Nashville, USA), which was manually positioned during the measurements (Figure 2). In each case, the lowest stimulus intensity at which evoked potentials could still be detected was determined in order to establish the potential threshold; the recording was made using subdermal needle electrodes. A click stimulus was then applied (dBnHL).
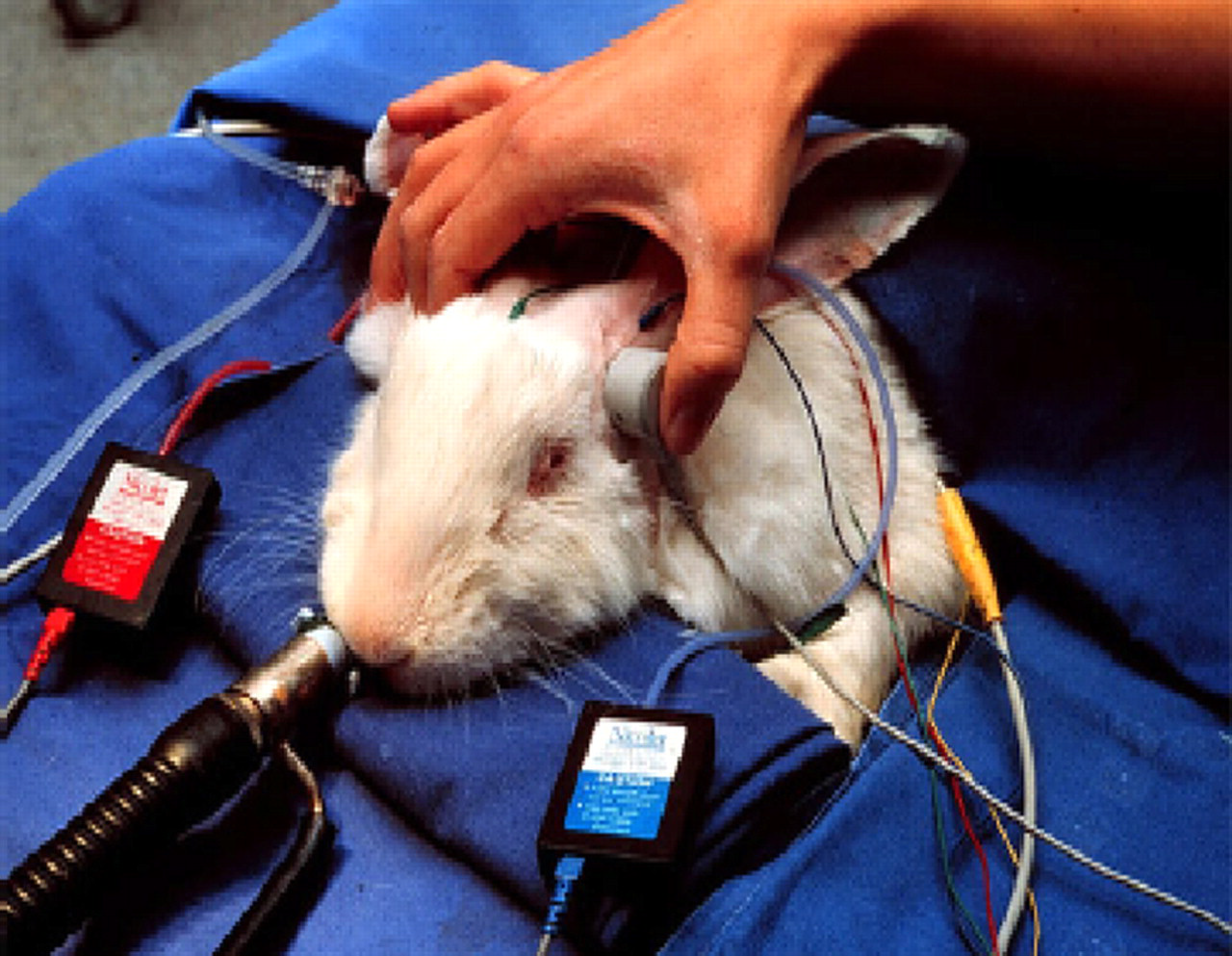
Position of the bone conduction earphones at the crown of the rabbit's head
Statistical analysis
For all recorded data, the arithmetic mean, standard deviation, median and minimum and maximum values were calculated. Data were tested for normal distribution using the Kolgomorov-Smirnov and Anderson-Darling goodness-of-fit tests. The Wilcoxon signed-rank test was used to calculate the significance levels (P < 0.05) of the measured data obtained. The statistical program used was SAS version 9.1 (SAS Institute, Inc, Cary, USA).
Macroscopic assessment
Assessment under the surgical microscope was carried out immediately following euthanasia and preparation of the temporal bones (with opening of the tympanic bulla). The eardrum and the surrounding tissues were microscopically examined for increased vascularization and new tissue formation. The position of the prosthesis was also assessed in terms of its contact with the eardrum and stapes. Forceps were used to move the eardrum slightly so that, in the functional test, the coupling of the prosthesis to the surrounding structures could be assessed.
Results
Macroscopic findings
Evaluation of surrounding soft tissue
The analysis of macroscopic findings, as the postoperative period progressed, showed that the cushion of tissue around the prosthesis had regressed (Figure 3). Whereas, on day 28 postoperatively, such tissue was particularly in evidence on the head of the prosthesis, it receded almost fully thereafter (Figure 4). On day 300 only isolated connective tissue cicatrices were visible. The head of the implant was lined with a fine epithelial layer (Figure 5).
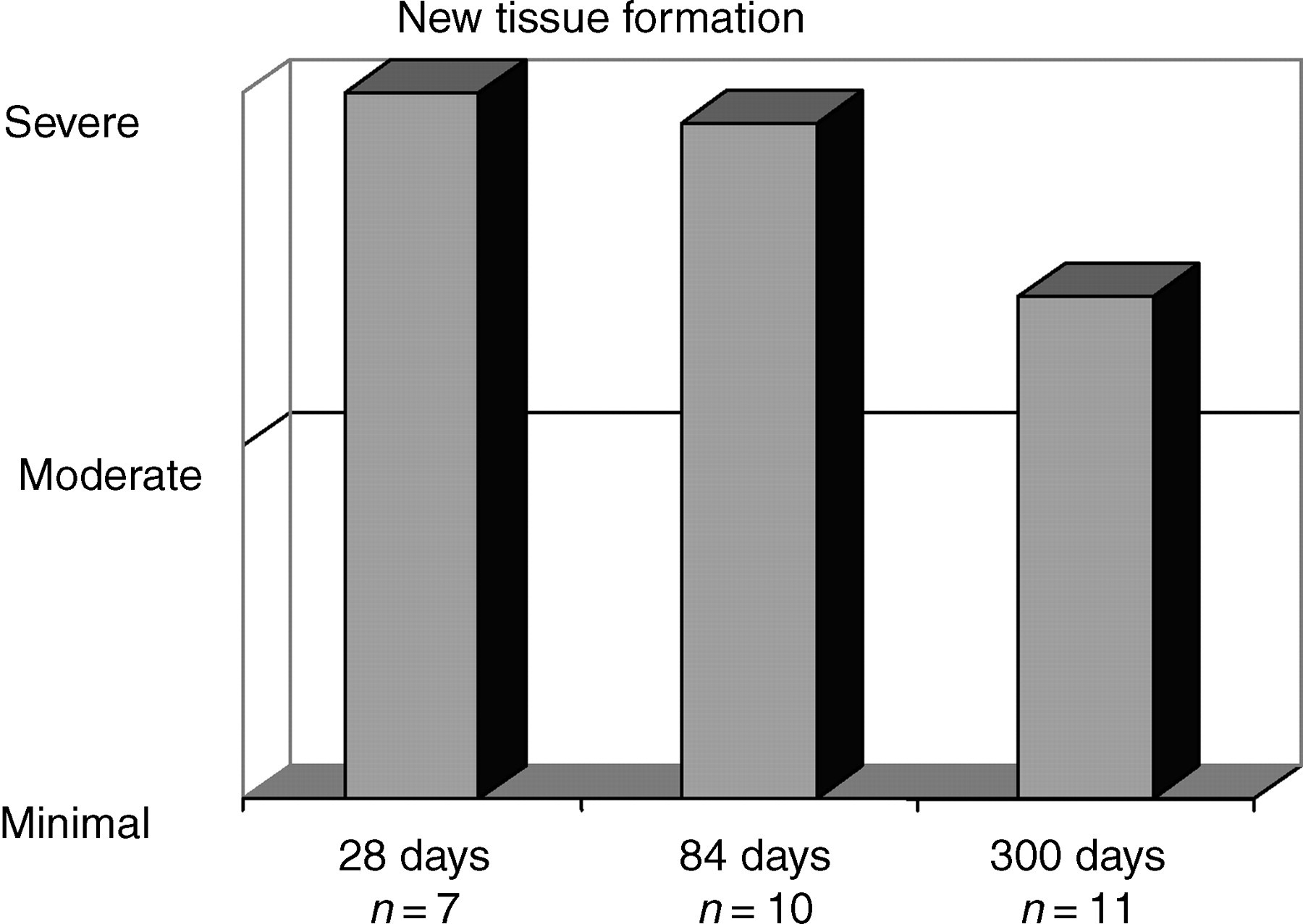
Macroscopic assessment over the course of the experiment. New-tissue formation = formation of new tissue in the vicinity of the eardrum, the prosthesis and the ear canal
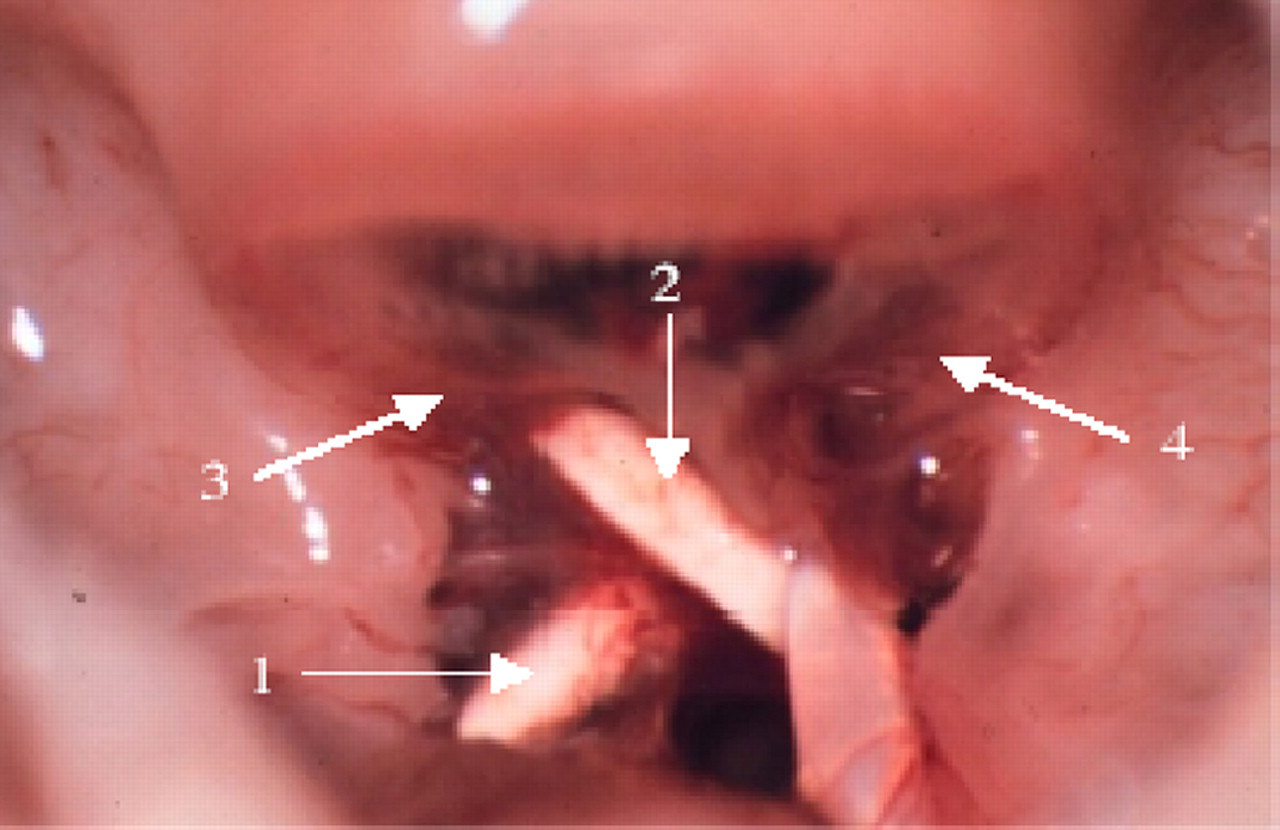
Prosthesis in the middle ear cavity, 28 days postoperatively. 1: prosthesis shaft; 2: prosthesis head; 3: tympanic membrane; 4: connective tissue. The prosthesis shows good contact with the rudimentary remnant of the stapes and the eardrum. Overall, there is considerable tissue growth around the prosthesis
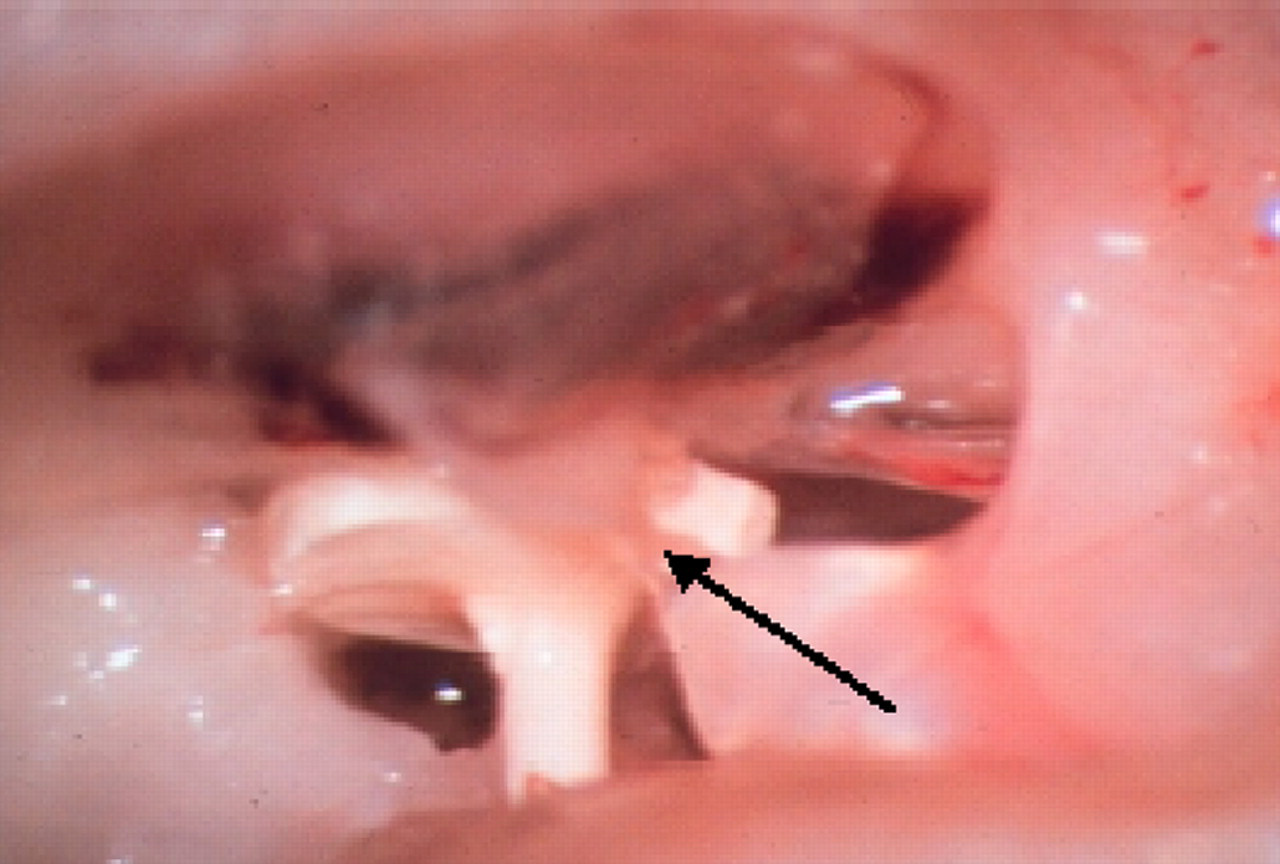
Prosthesis in the middle ear cavity, 300 days postoperatively. Arrow: less connective tissue after a period of 300 days. The prosthesis shows good contact between the head of the malleus and the eardrum. The process of the malleus is securely in contact with the rudimentary crura of the stapes. Only minimal formation of new tissue can be observed around the prosthesis. The black arrow indicates the tendon of insertion of the tensor tympani muscle
Coupling of the prosthesis to the eardrum
In almost half of all animals (48.6%), the prosthesis was observed to be very well coupled to the eardrum. In 51.4% of the rabbits, contact with the eardrum was good – ‘good’ here meaning that there was partial contact between the prosthesis (specifically, only parts of the prosthesis head) and the eardrum; where contact existed over the entire area of the prosthesis head, this was designated ‘very good’ (Figure 6).
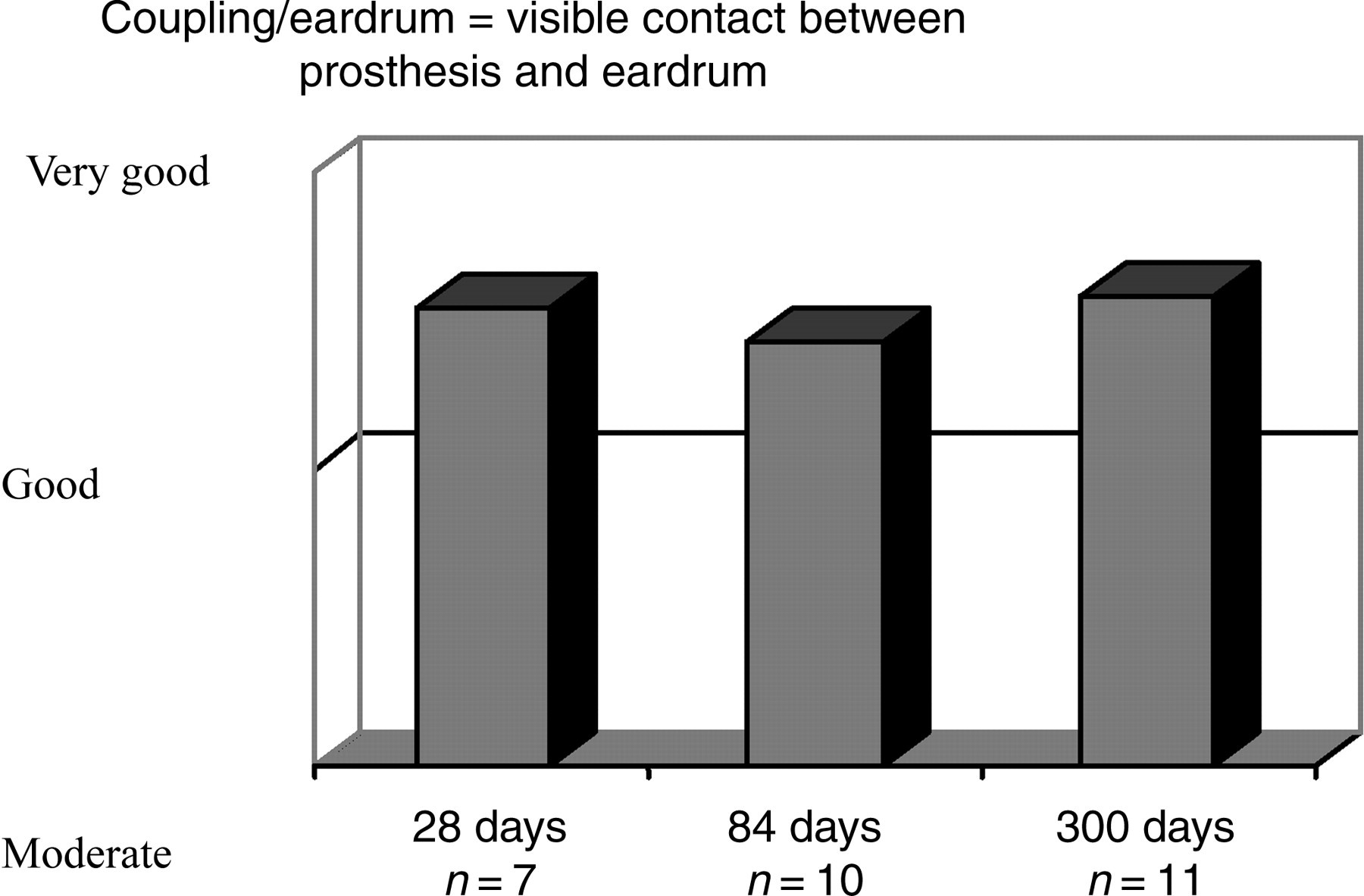
Macroscopic assessment of prosthesis/eardrum contact. Coupling/eardrum = visible contact between prosthesis and eardrum
Contact between the prosthesis and the stapes footplate
In 42.9% of the rabbits examined, very good contact was observed between the shaft of the prosthesis and the stapes footplate. Twenty animals (57.1%) showed good coupling with the stapes footplate, indicating that the shaft was not (as with ‘very good’ contact) positioned centrally between the rudimentary crura of the stapes, but lay next to them at a slight angle (Figure 7).
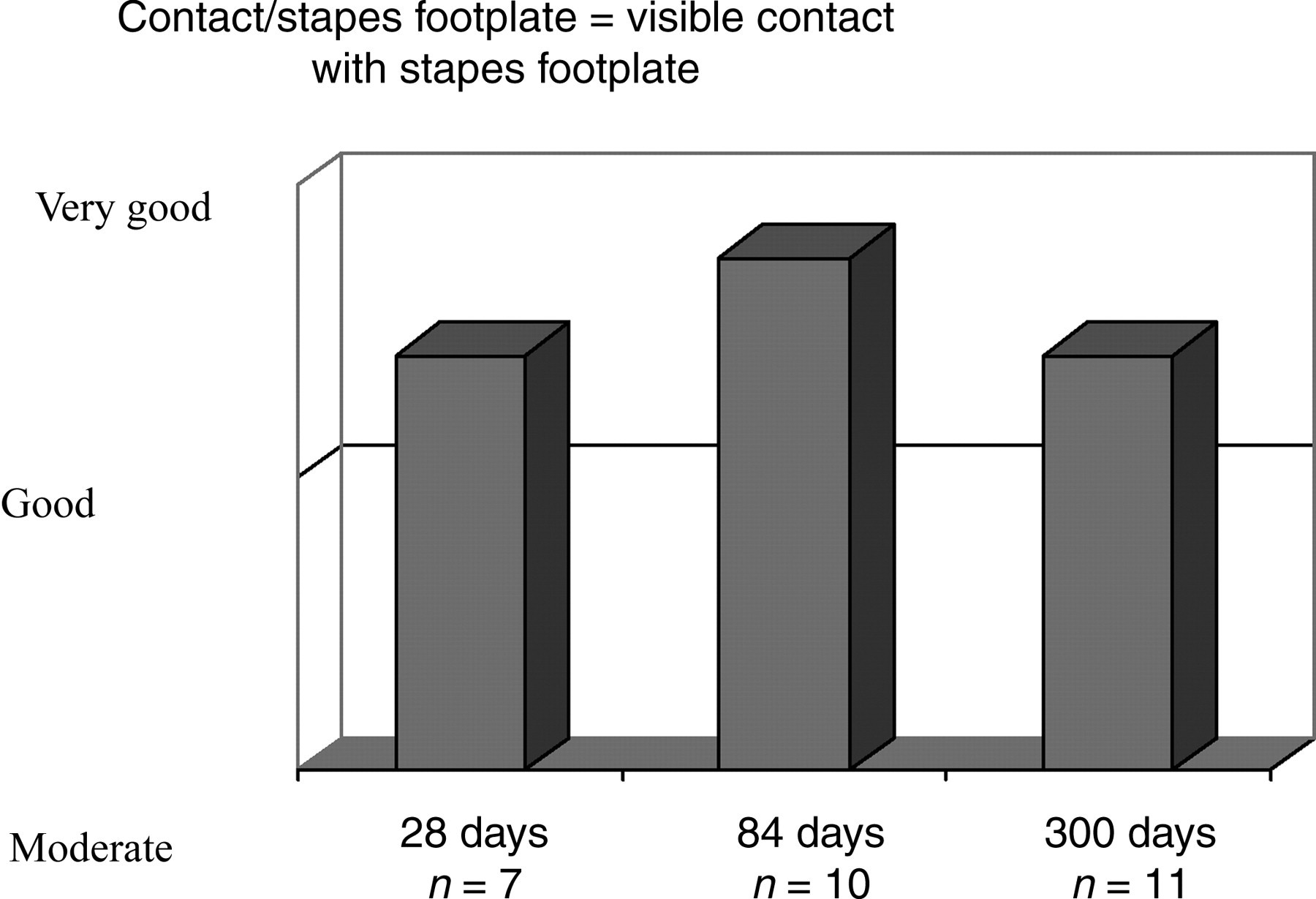
Macroscopic assessment of prosthesis/stapes footplate contact. Contact/stapes footplate = visible contact with stapes footplate
Brainstem audiometry investigations
In order to determine comparative values of potential thresholds for bone conduction, all data from all non-operated ears obtained over the entire experimental period were averaged, as were data from all operated ears at the time of preoperative measurement (Table 1 and Figure 3). Application of the click stimulus yielded a mean potential threshold of 4.21±6.68 nHL (n = 131) for bone conduction (Table 1). There was no significant difference between pre- and postoperative potential thresholds (P < 0.05).
Potential thresholds: bone conduction (dBnHL), non-implanted ear
P means: significantly different to the non-implanted ear
Discussion
The surgical outcome of ossiculoplasty depends largely on the material used and on the surgical technique employed. In contrast to other investigations, 6,19 our study revealed no eardrum perforations or stenoses of the ear canal. Compared with the level observed 28 days postoperatively, new tissue formation showed a marked decline at the later follow-up checks, so that the incorporation of the prosthesis into the tissue during the healing process occurred without inflammation. Auditory brainstem response (ABR) testing revealed no audiometrically detectable damage to the inner ear. The application of lasers in experimental middle ear surgery can therefore be recommended, especially given the fragile anatomy of the rabbit middle ear. The effects on the middle ear of the use of different laser systems have been investigated in numerous studies. 22,23 Elevated temperature and pressure are regarded as the chief causes of inner ear damage. Stubig et al. 24 were unable to detect any cochlear damage upon irradiation with the Ho:YAG laser, applying single-pulse energies between 30 mJ and 70 mJ at an impulse width of 250 μs. The laser was applied directly to the footplate at up to 52 impulses per second. The accompanying temperature increase was only 9.7°C. There are very few studies in the literature dealing with the problem of the thermal stress to which the inner ear is subjected following stapedectomy with the pulsed er:YAG laser used in the present study. 25,26 Experimental animal studies revealed the pulsed er:YSGG laser to be highly reliable in terms of compatibility when radiation is applied to the basal coil of the cochlea in the guineapig. In other studies, 22 application of the Ho:YAG laser to the guineapig cochlea has been shown to affect the measured compound action potential in over 40% of animals treated.
In our study, no changes in the recorded brainstem potentials were registered following removal of the stapes suprastructure using the er:YAG laser. This system can therefore be regarded as an effective and safe method for experimental ear surgery in the rabbit.
The contactless treatment of the fragile ossicular chain using the er:YAG laser permits a low-impact procedure for implanting newly developed ossicular prostheses in the rabbit middle ear. As part of the preoperative investigations, reference values for the physiological bone conduction auditory threshold of the rabbit were successfully determined, and it was verified that the er:YAG laser and the implantation material do not produce any detrimental impact or toxic effects, respectively (Tables 1–3).
Bone conduction auditory threshold and measurement time-point for the individual animals (dBnHL) over the course of the experiment
X = animal died from untreatable bradycardia before the experiment was completed
*Animal was euthanized after the measurement
Potential thresholds: bone conduction (dBnHL), implanted ear
P means: significantly different to the non-implanted ear
