Abstract
Blood flow is important for the healing of bone fractures. Until now, however, there have been no publications on the daily, continual measurement of intramedullary blood perfusion using laser Doppler flowmetry (LDF) in the conscious animal. In this study, a model for the daily, continual measurement of intramedullary blood perfusion by LDF and the temperature near the cortex both in intact and ostectomized tibiae in the conscious rabbit is described. The probes for blood perfusion and temperature measurement were implanted permanently at three different localizations into the right tibia of 10 adult New Zealand White rabbits. The probes were held in place by a bilateral, single-plane external fixator. In five of these animals, a midshaft tibial ostectomy was created in order to simulate a fracture. Intramedullary blood perfusion and temperature were measured daily over 49 days.
While in intact tibiae no significant (P > 0.05) differences were found in blood perfusion readings taken at various time points, for mean values or for blood perfusion over time, in ostectomized tibiae the differences were significant: various time points (P = 0.0056), mean values (P = 0.0034) and blood perfusion over time (P = 0.0337). Blood perfusion readings at the centre probe were elevated compared with those at the proximal and distal probes.
Thus, a revascularization in the ostectomy gap during the fracture healing was proven by means of the LDF. No influence of the blood perfusion on the temperature in the ostectomy area could be determined during healing of the ostectomy. The described model seems suitable for the continual measurement of intramedullary blood perfusion both in intact and ostectomized tibiae in the conscious rabbit.
Blood flow is not only especially important for the healing of bone fractures (McInnis et al. 1980), it also plays a significant role in the decomposition of degradable implants and in transporting degradation products away (Claes 2002).
Direct measurement of efferent and afferent blood flow at the bone is hardly possible due to low blood flow and the complex capillary system. Moreover, the efferent arteries or afferent veins have to be exposed, which in turn influences the measurement itself (Kunze et al. 1981). As additional invasive methods for determining the blood flow in the bone, the tracer microsphere method (radioactive-labelled microspheres), the determination of radioactive ion uptake or the radioactive gas washout technique are used (Tothill 1984). Disadvantages of these methods are the use of radioactive substances and the necessity of euthanizing the experimental animal or of removing the bone to be investigated for the blood flow measurement (Tothill 1984). Using these methods, continuous measurement of the blood flow in the bone of an experimental animal is not possible over a lengthy period.
A further technique for the measurement of blood flow in soft tissue and bone tissue is laser Doppler flowmetry (LDF), which does not require removal of the tissue under investigation or sacrifice of the laboratory animal (Salerud & Hellem 1992, Lausten et al. 1993, Kregor et al. 1995, Jain et al. 1996, 2000, Herzog et al. 2002). This method is based on the Doppler effect. A monochromatic light (780 nm) from a laser diode is guided to the tissue by optic fibres. The afferent light is scattered in the tissue. The backscattered light is picked up by another optic fibre, which takes the signal back to the photo detector. If light hits moving objects, in this case blood cells, the frequency of the light beam is changed, depending on the velocity of these objects. Reflected light from non-moving objects does not create any change in frequency. The relative number of moving blood cells is derived from the number of photons returned to the photo detector. The signal is processed in the instrument. The output signal is the product of the number of moving blood cells in the measured volume and the average velocity of the cells, which is given in perfusion units (PU).
Laser Doppler imaging (LDI) is based on the same principle. While with LDF measurements can be taken at specific sites, with LDI the blood flow can be visualized in tissue surfaces.
In contrast to the methods described above, the blood flow values determined using the LDF and LDI are only relative. However, they correspond to the values for bone circulation obtained with the tracer microsphere method (Lausten et al. 1993, Shymkiw et al. 2001).
Previous studies using LDF to measure blood perfusion in the bone mainly used single-fibre probes and needle probes or standard probes. However, Salerud and Hellem (1992) also used spacers made up of titanium with optical lenses and hollow needles to receive the needle probes, permanently implanted into the bone. Jain et al. (1996, 2000) described the experimental use of permanently implanted single-fibre probes in dogs for the repeated measurement of the blood flow in the tibia compared with measurement with standard probes during fracture healing after plate osteosynthesis. Although only light sedation was required to take the weekly blood flow measurements with the single-fibre probes over the entire 10 weeks, it was necessary to anaesthetize the animals and expose the plated bone surgically for the four measurements with the standard probes (Jain et al. 1996).
Some publications report of cortical and/or intramedullary blood perfusion measurements in anaesthetized rabbits, which were carried out using LDF or LDI (Salerud & Hellem 1992, McDonald & Pitt Ford 1993, 1994, Chan et al. 1999, Wolf et al. 2000, Shymkiw et al. 2001, Herzog et al. 2002).
Until now, however, there have been no publications on the daily continual measurement of intramedullary blood perfusion using LDF in the conscious animal. Neither are there any studies on the development of temperature near the bone cortex during fracture healing in the accessible literature. Thus, the objective of this study was to first develop an in vivo model for the daily measurement of intramedullary blood perfusion using LDF on unsedated rabbits at the intact bone and then to study the influence of a diaphyseal ostectomy on intramedullary blood perfusion and on temperature near the bone.
Materials and methods
Animals and conditions of husbandry
Ten adult New Zealand White rabbits, with an average body weight of 4.5 ± 0.21 kg were used in this experiment. The animals were supplied by Charles River, Kisslegg, Germany. The experimental design and use of the animals were approved by the responsible agency (Lower Saxony State Authority for Consumer Protection and Food Safety, permit 03/689). The animals were kept in single cages (EC3 rabbit cage system, Scanbur BK, Karlslunde, Denmark) under standardized conditions in compliance with the recommendations of the European Commission (ETS 123, 2003). A 12/12 h day–night cycle was maintained, the room temperature was 18 ± 1°C and relative humidity was about 60%. The rabbits received a daily ration of 150 g complete rabbit diet (K-H, Ered, 4 mm, Sniff Spezialdiäten GmbH, Soest, Germany) as well as autoclaved hay and water (drinking water, untreated) ad libitum. Microbiological status was conventional and pathogen free. The animals were given two months to become accustomed to being handled daily by their animal keepers before the beginning of the experiment. The 10 rabbits were randomly divided into two experimental groups with five animals each. Group A was formed to study intramedullary blood perfusion in intact tibiae, whereas in the animals of group B, a diaphyseal ostectomy was carried out to simulate a transverse fracture.
Experimental procedure
Measurements of intramedullary blood flow and temperature were made using the three laser Doppler units PF5010 LDPM and two temperature units PF5020 respectively, each with two channels (Perimed, Järfälla, Sweden). The probes required for this, which were constructed of polysulphon with a steel reinforcement in the back of the probe shaft, were equipped with a temperature sensor and a plug system for connecting it with the laser Doppler unit and temperature unit (Figure 1). The steel reinforcement of the probe shaft did not extend to the soft tissue surrounding the tibia. The probes were 25 mm long (without cables) and 3.5 mm in diameter. The probe tip was 1.5 mm in diameter and 2.0 mm long with an efferent and afferent optical fibre cable. The distance between the emitter and detector in the probe tip was 250 μm in all of the probes. The beam transmitted from the laser diode had a wavelength of 780 nm and a maximum output of 1 mW. The bandwidth of the measurement limits of the Doppler shift ranged from 20 Hz to 15 kHz. The probes were calibrated according to the manufacturer's instructions prior to implantation.

With the probes used in group A, the temperature sensor was still situated in the probe tip, whereas in the probes in group B, it was located further down in the probe shaft due to the results from group A. Defective probes were sent to the supplier for analysis.
The rabbits were anaesthetized by intramuscular injection of 17 mg/kg S-ketamine (Ketanest®S, Parke-Davis, Karlsruhe, Germany) and 0.25 mg/kg medetomidine (Domitor®, Pfizer, Karlsruhe, Germany). The anaesthesia was maintained with 1.5% to 2.5% (volume) isoflurane (Isoba®, Essex, Munich, Germany) in pure oxygen. The animals were also injected subcutaneously prior to surgery with 10 mg/kg enrofloxacin (Baytril® 2.5%, Bayer, Leverkusen, Germany) and 4 mg/kg carprofen injection fluid (Rimadyl®, Pfizer, Karlsruhe, Germany); during surgery they were given 100 mL Ringer's solution intravenously. In both groups of rabbits, a bilateral, single-plane external fixator (Mini-Fixateur externe, Mathys, Bochum, Germany) was implanted into the right tibia of each rabbit, whereby the fixator in group A was only to hold the probe in position. Four Kirschner drilling wires (Ø 1.6 mm) were attached from medial into the tibia and then connected on both sides with clamp jaws and pins (Ø 3.0 mm). From medial three drillings of 1.5 mm diameter were made between the pins through the compact tissue into the marrow space. Through these drillings, the tips of the probes were then introduced into the marrow space, and the distal end of the probes was attached to the external fixator (Figure 2). In group B, the external fixator and the proximal and distal probes were implanted in the same way. In addition, exactly in the centre of the tibia just below the tibiofibular junction, an ostectomy of 1.5 mm width was carried out with an oscillating saw. The tip of the centre probe was placed in the ostectomy gap and fixated using the external fixator (Figure 3). X-rays of the operated limbs were taken in craniocaudal projection immediately after surgery and once weekly thereafter to monitor the position of the probes and the external fixator. After surgery, the limb was bandaged and changed daily to monitor the wound and to clean the pin contacts. Enrofloxacin and carprofen were administered in the dosages indicated above for 10 more days.
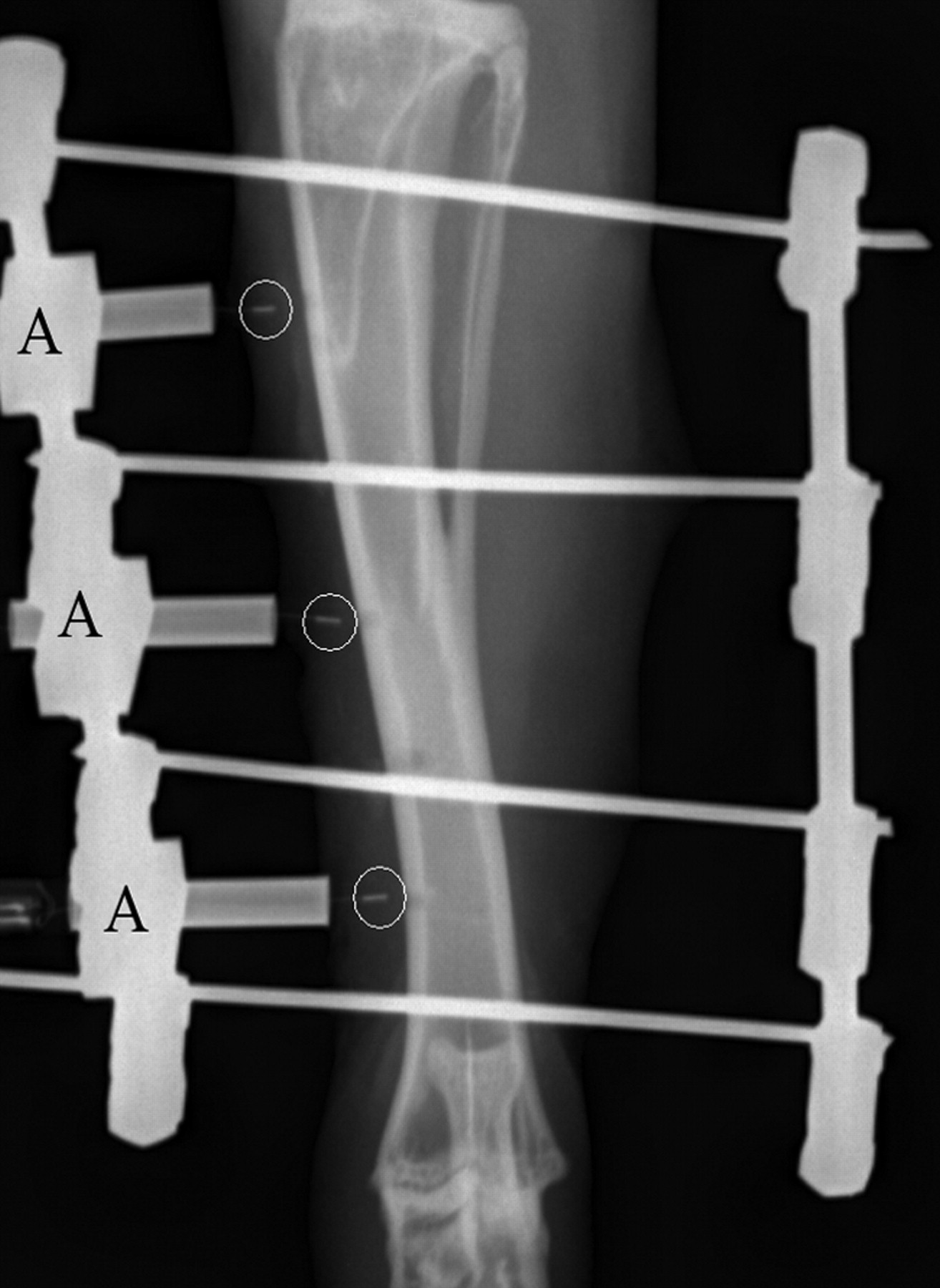
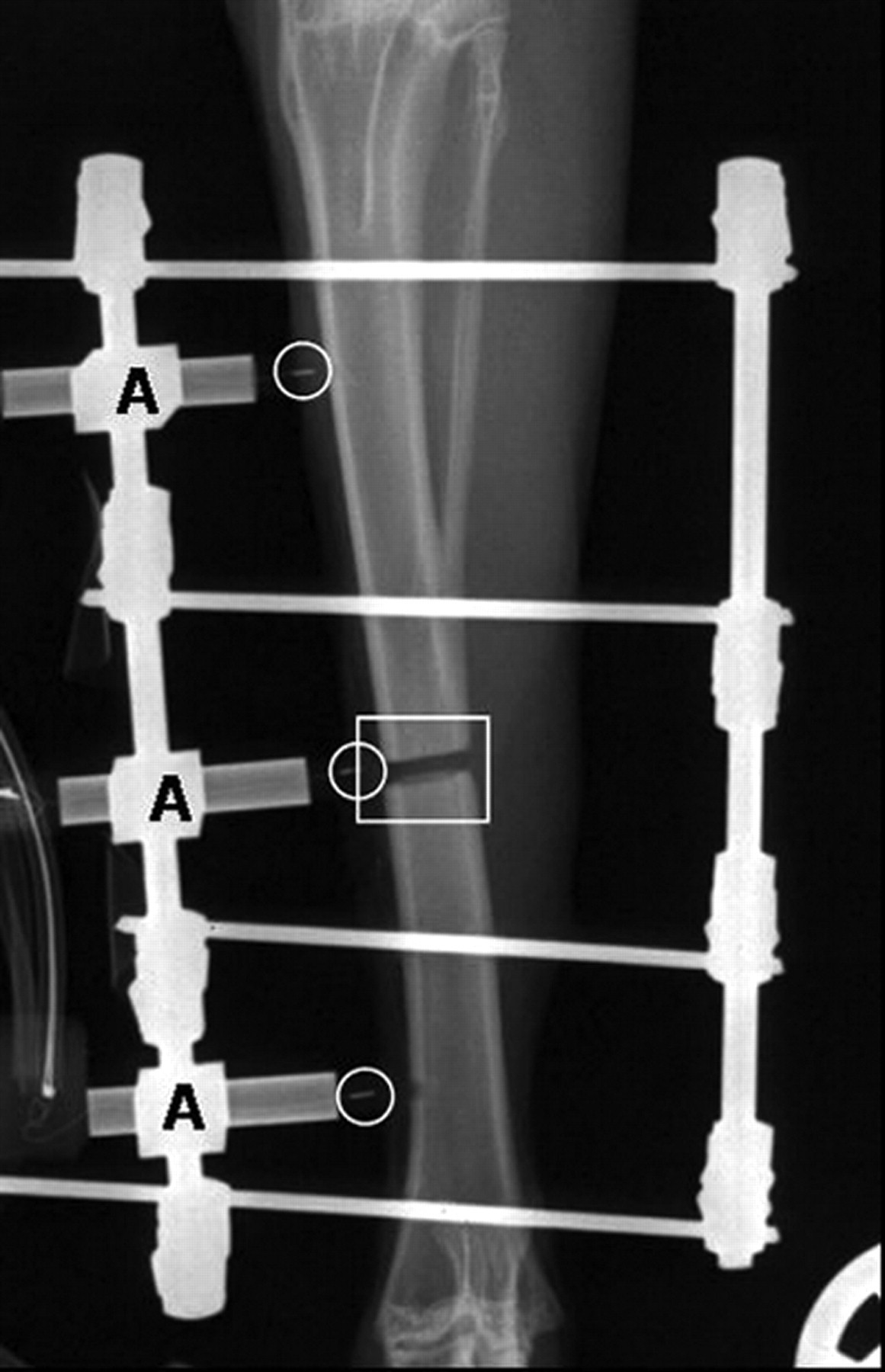
The first measurement of intramedullary blood perfusion and temperature near the cortex was taken immediately after surgery. After that measurements were taken at the same time every morning for 48 days. The bandage was removed for the measurement and the three probes were connected at the same time by jacks with the laser Doppler and temperature units. The animals were held in a semi-upright position by a handler. The measurements for blood perfusion and temperature were taken for a period of 2 min each time, saved on a portable computer and analysed with Perisoft 2.5 software (Perimed). The animals were euthanized after the experiments were completed, and the probes and the external fixator were removed. The right tibiae were explanted for further studies and fixed in 4% formalin solution.
To obtain a reasonable number of time points for the statistical evaluation, 12 time bins were created averaging four consecutive days each. The measurements of the first day (day of surgery) were excluded. For the statistical analysis of blood perfusion, only the time period up to day 29 was taken into account since the curves showed a linear structure over this range. The main statistical analysis was then performed using a mixed linear model for repeated measurements. This method is implemented in the procedure MIXED within SAS software package Release 9. One important advantage of PROC MIXED is that correlations among repeated measures are taken into account; missing values are not a major problem. Significance tests for fixed and random effects are performed within PROC MIXED. Specifically, three tests were conducted for the estimated fixed effects. A ‘group × time’ interaction term provided a test for differences in the slopes of the different curves. The ‘time’ test for the overall growth curve accounted for possible heterogeneous slopes. Finally, ‘group’ tested the null hypothesis of a common intercept (mean) for the curves. For a more detailed description of mixed models, see Brown and Prescott (2006).
The graphic presentation for the blood perfusion values was accomplished using mean value and standard error and for the temperature near the cortex using mean value and standard deviation. The values of defect temperature sensors (temperatures over 50°C or missing temperature readings) were not accepted in the evaluation.
Results
Quantity and localization of defective blood perfusion probes in each group over the seven-week observation period
Quantity and localization of defective temperature sensors in each group over the seven-week observation period
Radiologically, no osteolysis could be detected in the area of the inserted Kirschner drilling wires or probes in any of the animals. At day 48 after surgery, callus tissue had formed in the ostectomy gap in four out of five animals in group B. The probe tips were also surrounded by callus tissue. At the end of the experiment, in one of the five animals in group B, callus tissue was detected in the lateral area of the ostectomy gap, but it did not fill the entire gap. There was no radiographic evidence of osteolysis in this animal either.
Intramedullary blood perfusion
Intramedullary blood perfusion of the intact tibiae
In group A, no significant changes (P > 0.05) were observed until day 29 (days 2–5 to days 26–29) among various time points, mean values of blood perfusion or blood perfusion over time (group A, Figure 4). At the proximal probe site, the blood perfusion rose from initially 14 ± 5 PU to a highest reading of 269 ± 79 PU on day 16 and fluctuated between 66 ± 32 PU and 257 ± 139 PU until the end of the trial. The blood perfusion at the distal probe site behaved similarly. From initially 14 PU it rose to 219 ± 127 PU on day 8 and fluctuated between 90 ± 17 PU and 294 ± 109 PU during the rest of the observation period. From day 10 on to the end of the experiment, the readings at the centre probe site fluctuated between 147 ± 68 PU and 270 ± 95 PU. The highest reading was 444 ± 116 PU on day 29.
Intraosseous blood perfusion (perfusion units) measured during the observation time in non-ostectomized tibiae of five rabbits at the proximal (•), centre (▪) and distal (▴) probes (group A); mean ± standard error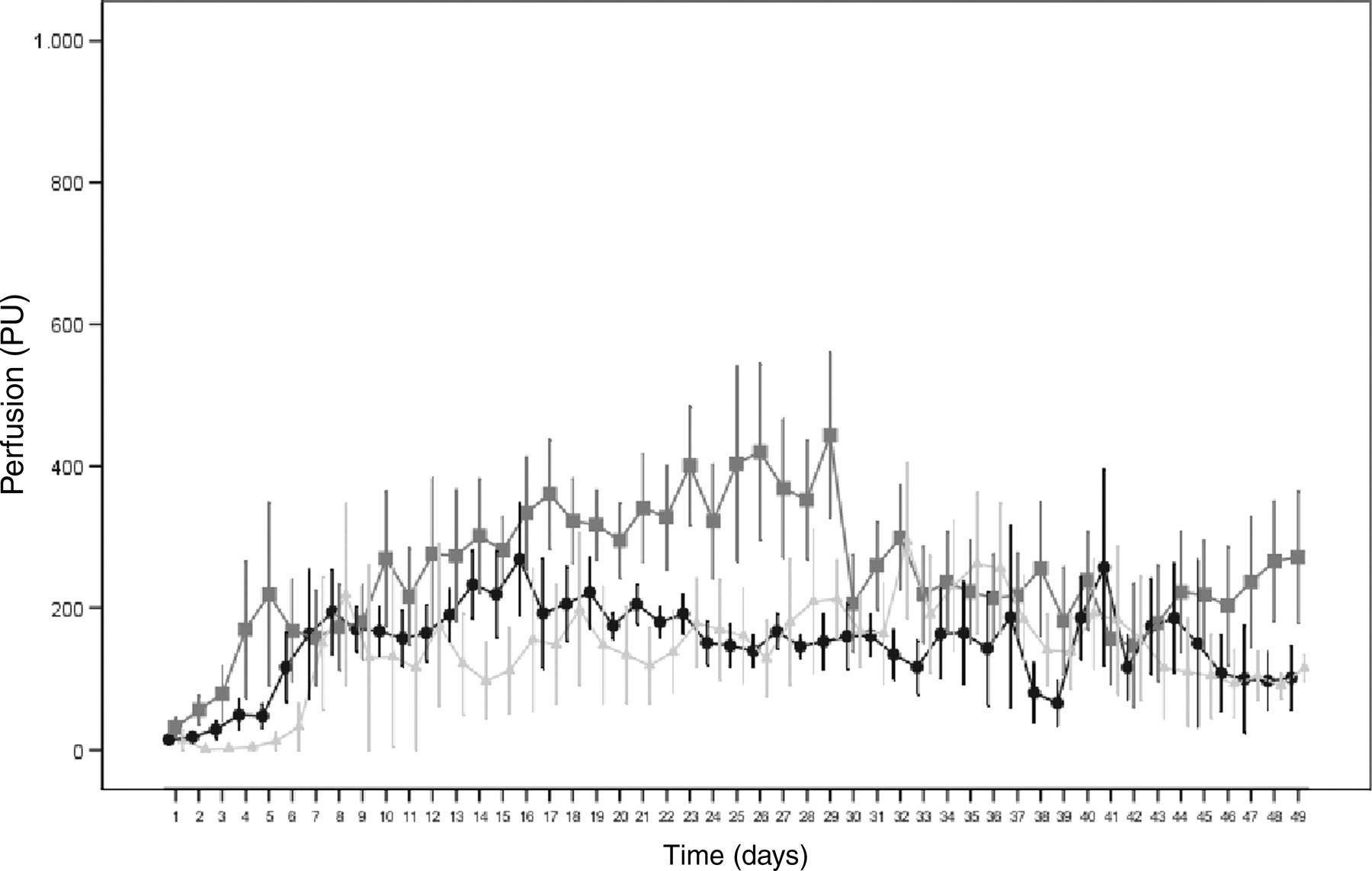
Intramedullary blood perfusion of the ostectomized tibiae
In group B, significant changes were observed until day 29 (days 2–5 to days 26–29) among various time points (P = 0.0056), mean values of blood perfusion (P = 0.0034) and blood perfusion over time (P = 0.0337) (group B, Figure 5). Blood perfusion at the centre probe was elevated compared with blood perfusion at the proximal and distal probes. After an initial rise, the blood perfusion readings given by the proximal and distal probes fluctuated between 93 ± 13 PU and 339 ± 161 PU after day 10. The blood perfusion reading given by the centre probe rose from an initial 11 ± 7 PU continually to 572 ± 203 PU on day 25 and remained on a plateau between 400 and 530 PU through the end of the measurements.
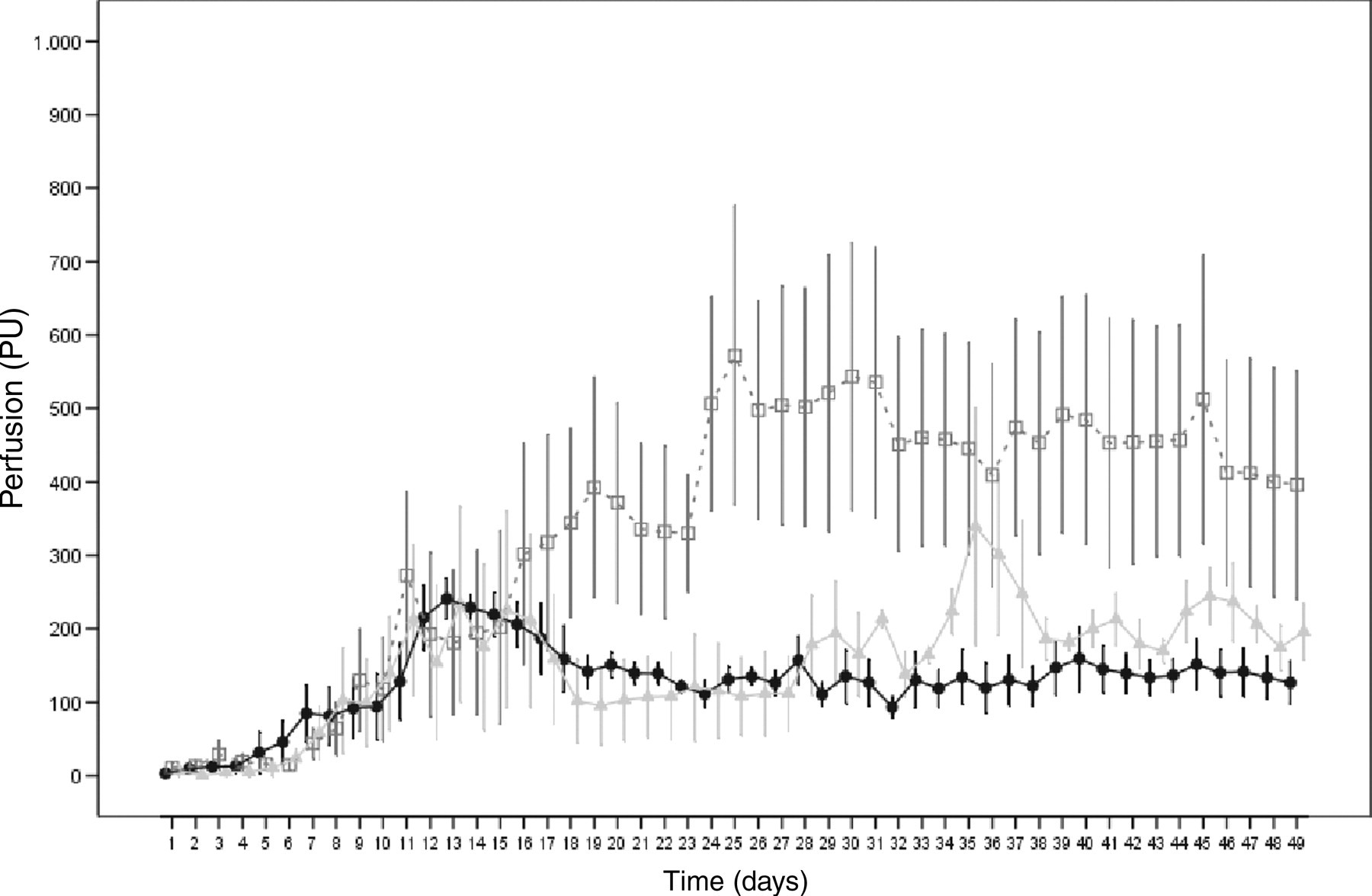
Comparison of the two groups
Until day 29 (days 2–5 to days 26–29), blood perfusion readings taken at the proximal, centre and distal probes of both groups did not vary significantly (P > 0.05), neither with respect to the temporal course nor to the blood perfusion readings (Figure 6). In both groups, the readings at the centre probe site showed high variability. The increase in blood perfusion at the centre probe site of the ostectomized tibiae did not take place until day 7 and thus later than with the intact tibiae (Figure 6). Although the perfusion readings taken at the centre probe of the ostectomized tibiae between day 26 and day 29 were higher than the values of intact tibiae, the differences were still non-significant (P = 0.0857). From day 24 until the end of the measurements, the blood perfusion readings of the ostectomized tibia at the centre probe site were between 77 and 337 PU above the readings given by the centre probe of the intact tibiae (Figure 6).
Intraosseous blood perfusion (perfusion units) during the observation time measured in non-ostectomized tibiae (▪) and ostectomized tibiae (□) at the centre probe; mean ± standard error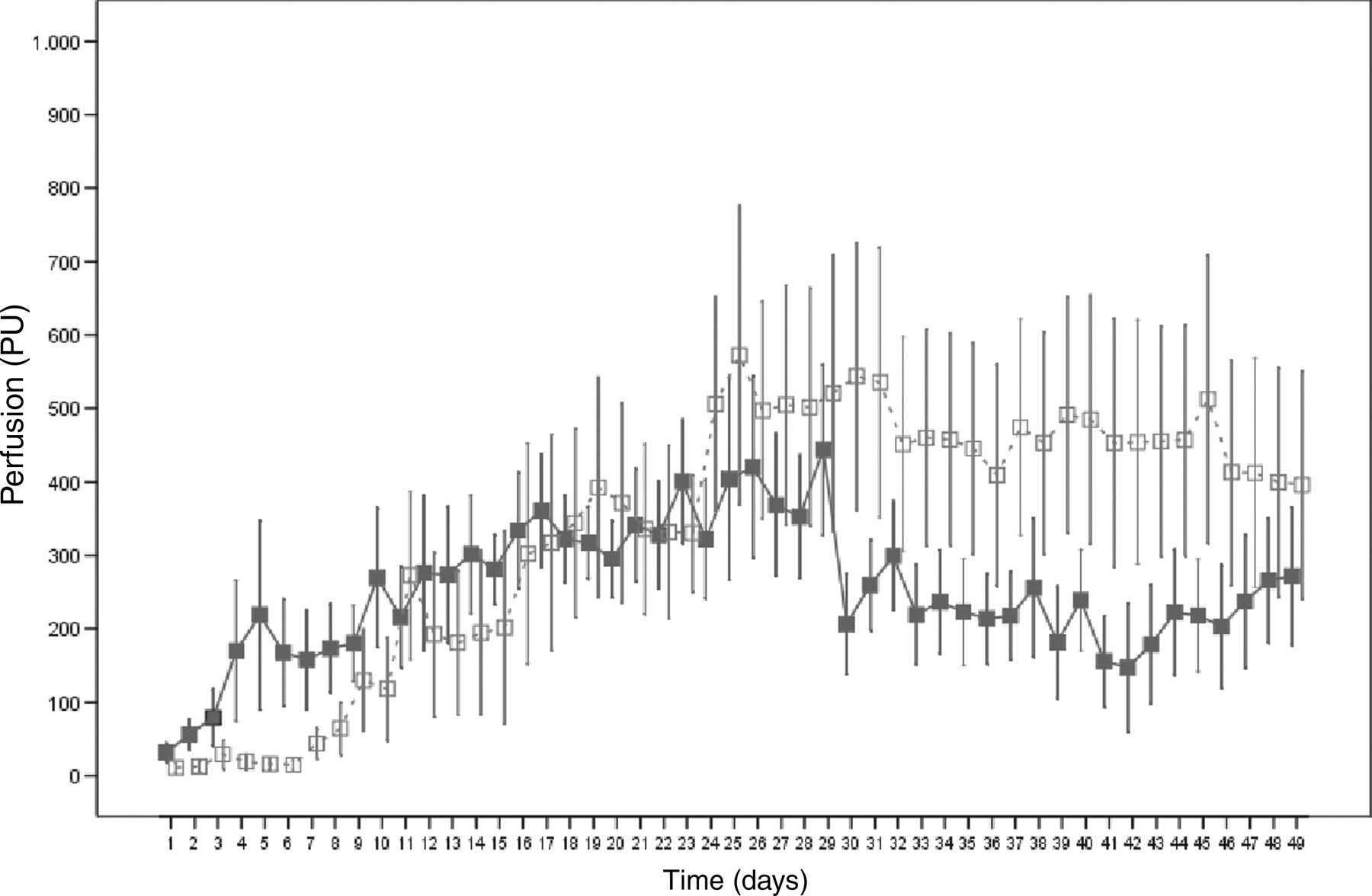
Temperature near the cortex
Temperature near the cortex of the intact tibiae
In group A, no significant (P > 0.05) changes could be observed until day 49 (days 2–5 to days 46–49) among various time points, mean values of temperature near the cortex or temperature readings over time taken at the proximal and centre probes (group A, Figure 7). Immediately following surgery, the temperatures were 24.1 ± 2.4°C at the distal probe, 26.3 ± 1.1°C at the centre probe and 28.0 ± 1.3°C at the proximal probe, and they rose to 37.1 ± 0.8°C, 38.21 ± 0.7°C and 37.9 ± 0.9°C, respectively until the 10th day. From then on they remained nearly stable on this level. The temperatures measured at the distal probe were about 1°C lower (P = 0.033) than those of the two other probes.
Temperature (°C) measured near the tibial cortex during the observation time in non-ostectomized tibiae of five rabbits at the proximal (•), centre (▪) and distal (▴) probes; mean ± standard deviation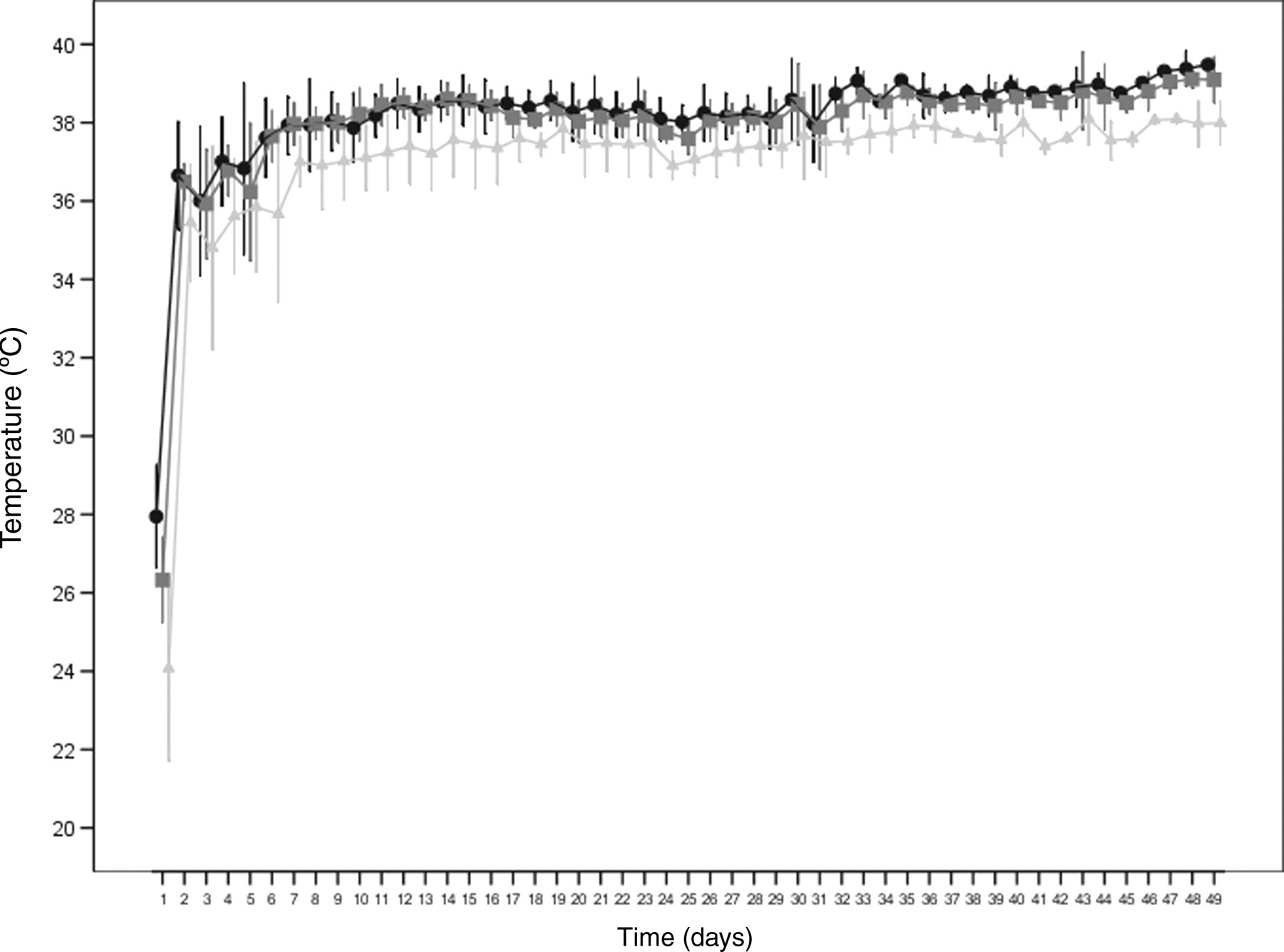
Temperature near the cortex of the ostectomized tibiae
In group B, no significant (P > 0.05) changes could be observed until day 49 (days 2–5 to days 46–49) among various time points, mean values of temperature near the cortex or temperature readings over time taken at the proximal and centre probes (group B, Figure 8). Initially, measurements were made at the distal probe of 24.9 ± 1.6°C, at the centre probe of 25.6 ± 2.1°C and at the proximal probe of 27.53 ± 1.9°C. Until the eighth day, the temperatures rose to 36.2 ± 1.2°C, 37.9 ± 0.9°C and 38.1 ± 0.7°C, respectively and remained nearly stable on this level in the further course of the experiment. The distal probe showed a significantly (P = 0.0392) lower temperature than the proximal and centre probes. The temperature difference between the distal and the two other probes was approximately 1.4°C.
Temperature (°C) measured near the tibial cortex during the observation time in ostectomized tibiae of five rabbits at the proximal (•), centre (□) and distal (▴) probes; mean ± standard deviation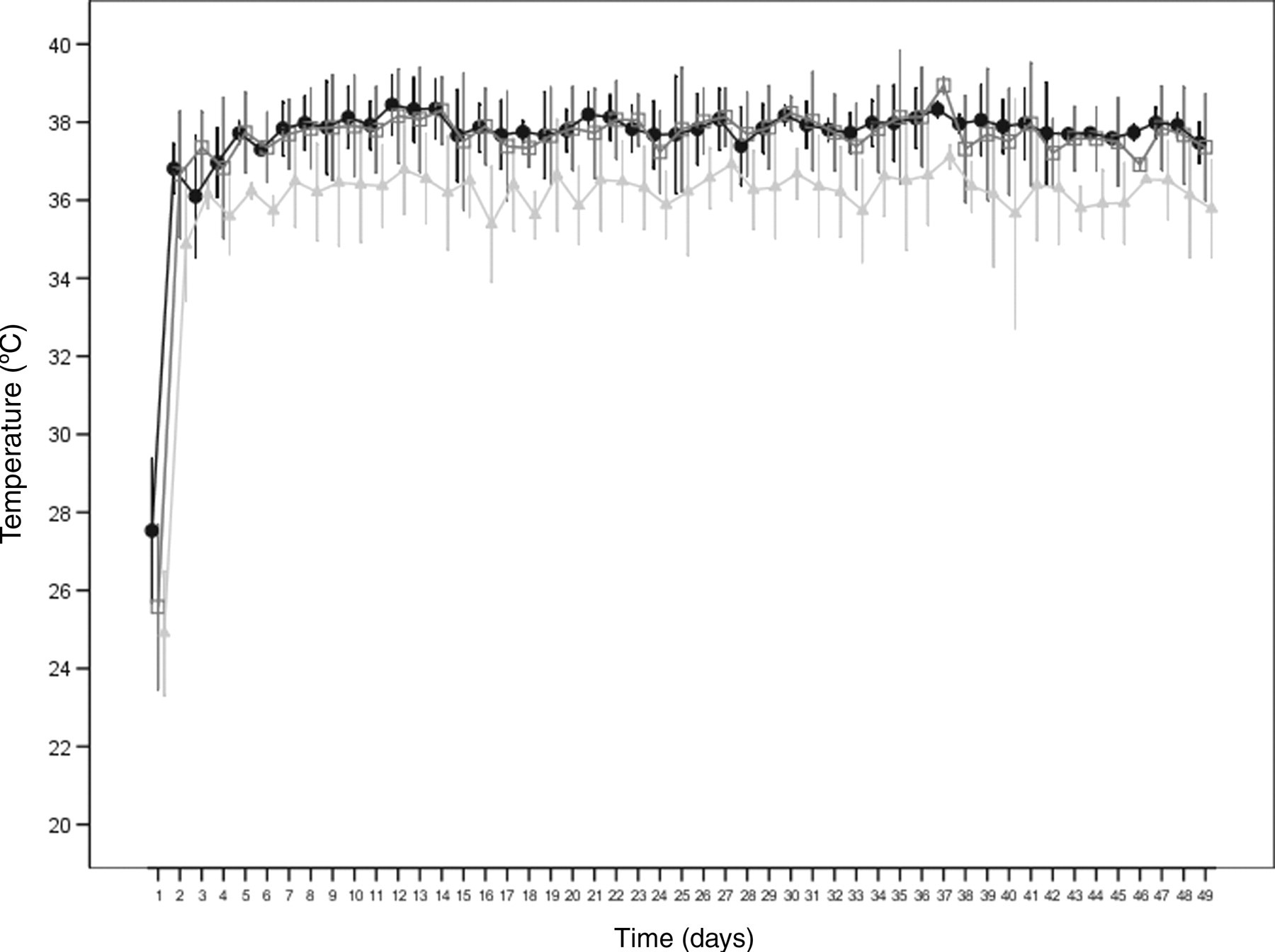
Comparison of the two groups
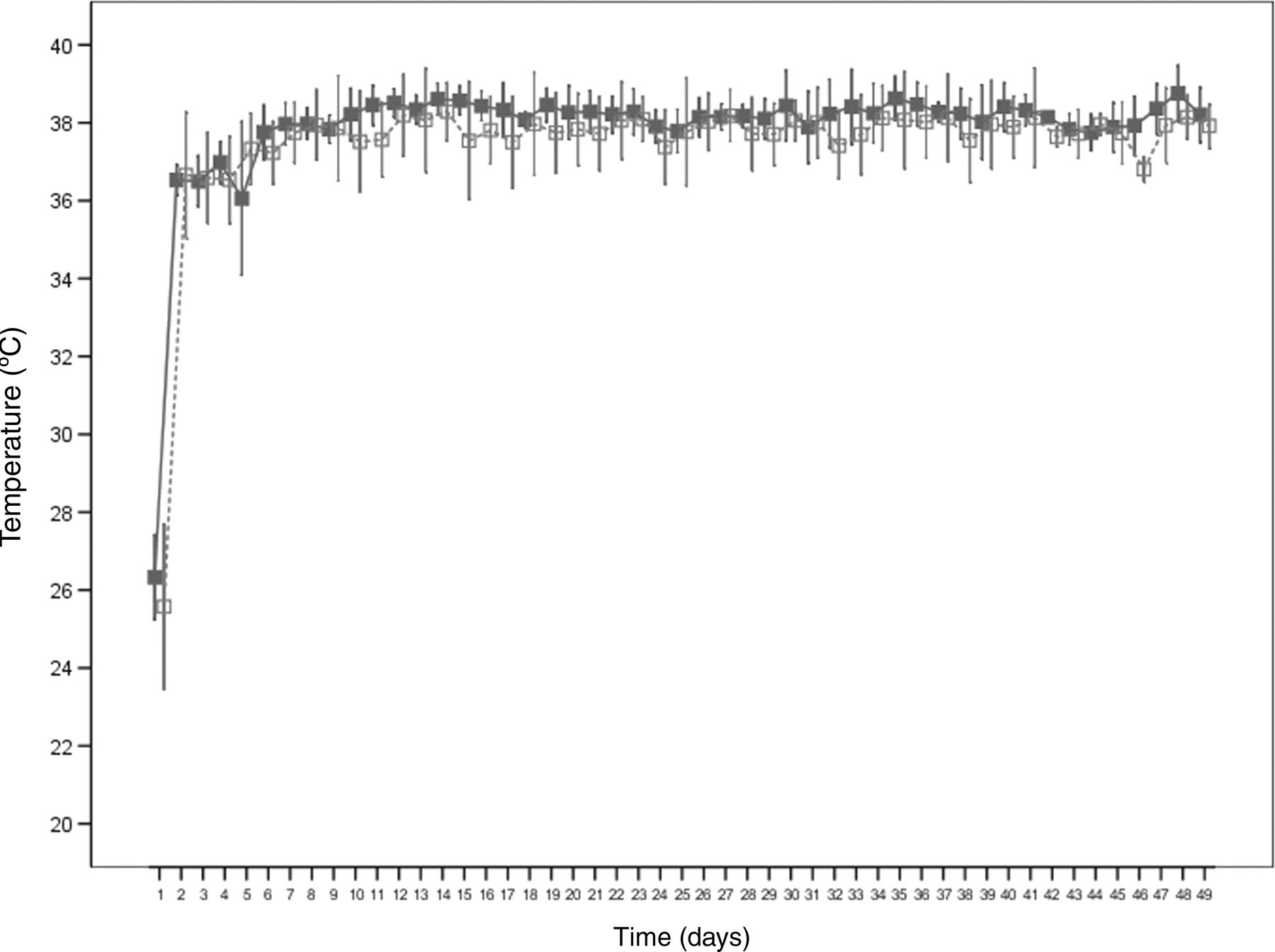
The temporal courses of the temperature curves of both groups were very similar (Figure 9). After an initial rise, the measured temperature changed very little in all probes. In both groups, the proximal and the centre probes had approximately the same temperature values in the course of the observation period, whereas the distal groups in both groups showed lower temperatures. All ostectomized tibia probes, however, showed a lower (P > 0.05) temperature on average than those of the intact tibiae. Figure 9 shows this exemplarily for the centre probes of both groups.
Discussion
The first aim of this study was to develop an in vivo model for the daily measurement of intramedullary blood perfusion using LDF on unsedated rabbits at the intact bone. The second aim was to study the influence of a diaphyseal ostectomy on intramedullary blood perfusion and on temperature near the bone. The rabbit was chosen as the model animal because several studies on cortical and/or intramedullary blood perfusion measurements exist using LDF or LDI in sedated rabbits (Salerud & Hellem 1992, McDonald & Pitt Ford 1993, 1994, Chan et al. 1999, Wolf et al. 2000, Shymkiw et al. 2001, Herzog et al. 2002), thus enabling a comparison of the readings made in this experiment on conscious rabbits.
Discussion of the methods
The result of this experiment showed that the model used with permanently probe implants was suitable for the daily measurement of intramedullary blood perfusion and temperature near the cortex on the conscious animal. The suitability of the probes was tested in a preceding experiment (Hackenbroich et al. in preparation). There it was shown that it was necessary to modify the probes, which were primarily designed for this experiment, with a metal reinforcement. The rate of loss for the probes without metal reinforcement used in the pilot study was altogether about 55% (Hackenbroich et al. in preparation). Similarly, high loss rates were shown in the experiment of Jain et al. (1996): of the probes permanently implanted in dogs to measure blood perfusion in the compact bone, only 15% were still functioning after 10 weeks. At the time of euthanasia only 7% were still functioning.
In the present study, on day 49 of the experiment, about 70% of all used, modified probes for blood perfusion measurement were still intact (Table 1). Of the temperature sensors, 33.3% (Table 2) were still giving reliable readings. These results are comparable with those of a later study by Jain et al. (2000) which had a lesser loss rate of 25% compared with their first experimental design. This was likewise due to a modified probe design and subcutaneously situated connectors of the probes. This shows that pilot studies are indispensable for testing the feasibility of permanent probe implants in animal experiments.
While Jain et al. (1996, 2000) used flexible two-way single conductor cables for repeated measurements in their experiments, in the present study rigid probes each with one afferent and one efferent optical fibre cable were used. The advantage of the latter type of probe over the two-way single conductor cables is that it permits the light rays to penetrate the tissue in question more deeply, thus giving greater volume of measurements (Schemitsch et al. 1994). But even with these probes, blood perfusion can only be measured in a relatively limited area of a few cubic millimetres in front of the probe tip (Schemitsch et al. 1994). This can also explain the high variability of the measurement results in this study: depending on the placing of the probes in the tibia, the readings in the different subject animals can vary greatly due to individual differences in the blood supply. There was also a considerable variance in the results in the in vivo experiments of Jain et al. (1996, 2000) depending on when the measurement was taken. Yet even with the tracer microspheres method, a high variability of the blood flow measurements was reported (Triffit & Pitt Ford 1994).
In contrast to Lausten et al. (1993) and Shymkiw et al. (2001), Swiontkowski et al. (1986, 1988) found no or only little correlation between LDF and the tracer microsphere method. However, LDF was attested a high reproducibility. Several possible reasons were given for the lacking correlation between the two methods, including the different measurement volumes used. While with the laser Doppler unit the blood perfusion was determined in a volume of few cubic millimetres, the tracer microsphere method determined the blood flow in a greater area of the bone in question. Moreover, with LDF the blood perfusion was recorded over a longer period, whereas with the tracer microsphere method the blood flow was only measured at a particular point in time (Swiontkowski et al. 1986, 1988). This makes LDF seem advantageous for the continual assessment of the changes in blood perfusion over the period of the ostectomy healing in comparison with the tracer microsphere method.
Radiographic examinations showed that neither Kirschner wire implantation nor probe implantation led to osteolysis. Also, the extent and speed of the observed formation of callus in the ostectomy gap corresponded to the physiological fracture healing of the rabbit.
Discussion of the continuous measurement of blood flow
Chan et al. (1999) gave a value of 29.8 ± 3.8 PU for the cortical blood perfusion of the rabbit tibia. The measurements were made using LDI in the diaphyseal area of the lateral tibia, after the soft tissue and periosteum had been removed. Similar values are given by McDonald and Pitt Ford (1993, 1994). The cortical blood perfusion was measured medial and lateral diaphyseal at the intact tibia using LDF and was between 20 and 25 PU in adult rabbits, in juvenile animals it was a little higher. Also here the soft tissue and the periosteum were removed beforehand. Shymkiw et al. (2001) reported on measuring the cortical blood perfusion at the exposed rabbit tibia using LDI with two different wavelengths (634 and 810 nm). The readings fluctuated between 16.4 ± 5.6 PU (634 nm) or 28.3 ± 10.1 PU (810 nm) and 22.7 ± 9.8 PU (634 nm) or 36.6 ± 11.3 PU (810 nm). Values of 21.34 PU for the cortical blood perfusion in the rabbit tibia were reported by Herzog et al. (2002). Salerud and Hellem (1992) documented the temporal course of the intramedullary and cortical blood perfusion in rabbit tibiae over a period of 145 days. On four measuring dates (day 21, day 34, day 139 and day 145) the blood perfusion was determined in the compact bone and in the bone marrow. Complete surgical exposure of the tibiae was not necessary to obtain these measurements since spacers with an optical lens or hollow needles of titanium had been implanted previously. While the cortical values fluctuated over the entire period by 3 PU, the bone marrow showed initial values between 30 and 35 PU, dropping to values around 10 PU after day 34.
Wolf et al. (2000) studied the effect of calcitonin gene-related peptide (CGRP) on blood perfusion in the ostectomy gap at the height of the tibiofibular junction of rabbit tibiae after surgery. After the application of CGRP they obtained perfusion readings in the diaphyseal ostectomy gap of 0 PU after five days, 48 ± 8 PU after 10 days and 67 ± 6 PU after 15 days. The value of the preoperative control reading in the intact tibiae was 90 ± 13 PU.
Both the cortical and intramedullary blood perfusion values of these studies are clearly below the results of the study described here. This is possibly due to the fact that in the present study the measurements were carried out on the conscious animal, ruling out the influence of anaesthesia or hypotension-inducing medication. Moreover, impairment of the blood perfusion due to repeated surgical procedures was excluded in the model described here.
Discussion of the results
In both groups, the temporal course and the level of the blood perfusion readings given by the proximal and distal probes were nearly identical. At least with this method, diaphyseal ostectomy therefore does not seem to have any measurable influence on the intramedullary blood circulation of areas distant from the ostectomy. This contradicts the results of Triffitt et al. (1993) as well as Triffitt and Gregg (1994). Using the microsphere method in diaphyseal osteomized rabbit tibiae, they detected an initially reduced flow in the distal part of the tibia, which did not rise until after one or two weeks.
Remarkable are the insignificantly elevated blood perfusion values in the intact tibia of the centre probe in contrast to the proximal and distal probes. A possible explanation for this could be the proximity of the probe tip to the nutritive artery and its branches. The nutritive artery in rabbits extends from lateral directly proximal to the connection between the tibia and fibula in the marrow space and divides into an afferent and efferent main branch (Göthman 1960).
The still insignificant increase in blood perfusion in the ostectomy area from day 7 after surgery reached its maximum on day 24 and lay above the values of the intact tibia until the end of the observation period. This corresponds to the results of other authors using the tracer microsphere method or non-continuous laser Doppler measurements (Paradis & Kelly 1975, Aalto & Slätis 1984, Grundnes & Reikerås 1992, Triffit et al. 1993, Triffit & Gregg 1994, Wolf et al. 2000, Herzog et al. 2002). The explanation of this observation could be the revascularization of the ostectomy area and the elevated blood flow in connection with the bone healing. Rhinelander (1974) could show that in radius fractures attended to with plate osteosynthesis in dogs that after a week the first capillaries already bridged the ostectomy gap.
Wallace et al. (1994) investigated the influence of different external rigid fixators on blood flow during bone healing in the ovine tibia. With a less rigid external fixator, 14 days after surgery blood flow was four times higher in the compact bone and in the bone marrow. Deviating from the present study Wallace et al. (1994) observed the preoperative benchmark readings again after about 42 days both with semi-rigid and rigid fracture fixations. Other authors report elevated blood flow over a longer period after experimental osteotomies in dogs and rats. Even after 112 days, the blood flow after the osteotomy of canine tibiae had not reached the initial values again in the osteotomy area (Paradis & Kelly 1975). In osteotomized femurs of rats, after four weeks, 10 times higher blood flow was observed in the callus, which afterwards slowly diminished again (Grundnes & Reikerås 1992). After 12 weeks, the end of the experimental period, blood flow compared with the control group was still three times higher. Also in the present study blood perfusion readings in the area of the ostectomy were above the control group until the end of the observation period of 49 days.
The advantage of the method for blood perfusion measurement described in this study compared with other studies is the possibility of protocolling blood perfusion changes daily without sedating the animal. Previous studies on blood perfusion measurement at or in the bone using LDF, LDI or other methods had the disadvantage that there were long intervals between the individual tests. Thus, no verified statements could be made about the course of the blood perfusion between the individual tests.
Discussion of the measurement of temperature near the cortex
No publications could be found in the accessible reference literature on the development of temperature in the bone during fracture healing. Thus, this study intended to determine whether an altered intramedullary blood perfusion is also accompanied with a change in the local temperature in the bone. The results of this study show that with the temperature sensors used in this experiment, no influence could be shown of elevated perfusion during the healing of the ostectomy on the temperature in the area of the ostectomy in comparison with the intact tibia. A possible explanation for the similar temperature distribution in both groups between the probes is the varying thickness of the covering of soft tissue along the tibia. In the proximal and centre areas of the tibia, there is more soft tissue medially and laterally than in the distal area of the tibia. Thus, the proximal and centre probes are surrounded by muscle tissue that is well supplied with blood, while the distal probe is only covered by skin that is not well perfused. Since the metal reinforcement did not protrude into the soft tissue of any of the tibia bones, the metal could not have led to increased thermal conductance. The low temperature near the cortex directly following surgery in both groups, in all animals and at all probe sites is presumably due to the influence of anaesthesia.
At all three measuring sites during the observation period, the temperature in the animals with an ostectomy was a few tenths of a degree lower than in the animals without an ostectomy. This can be explained by the changed position of the temperature sensor in the modified probes. Faulty or missing measurement values in the temperature readings in group A, caused by the penetration of body fluids into the probes, led to the transferring of the temperature sensors from the sensor tip to the probe shaft. After that, faulty temperature measurements only occurred sporadically. Therefore, a direct comparison of the temperature values is only possible to a limited extent. But the similarity between the temperature curves and the approximately same temperatures values indicate that the ostectomy did not have any influence on the temperature near the cortex during healing.
A local inflammation reaction with elevated temperature in the implant area of the probes could not be determined in any animal.
Conclusion
The model described here is suitable for the continual measurement of intramedullary blood perfusion both in intact and ostectomized tibiae in the conscious rabbit. A revascularization in the ostectomy gap during the fracture healing was proven by means of LDF. The advantage over previously published LDF measurements is that any influence on blood perfusion caused by repeated anaesthesia and/or surgery could be avoided, and the daily measurement of blood perfusion and temperature near the cortex could take place in the conscious animal. The method seems suitable to investigate dynamic changes of the intramedullary blood perfusion after implanting intramedullary resorbable pins, to calculate the degradation kinetics of resorbable implants and to adjust them to the physiological conditions during the healing of the fracture. However, with the temperature sensors used in this experiment, no influence of elevated blood perfusion on the temperature in the ostectomy area could be determined during healing of the ostectomy.
Footnotes
Acknowledgements
This work was conducted within the framework of the Collaborative Research Center 599 (SFB 599) and supported by the Deutsche Forschungsgemeinschaft (German Research Foundation). Our special thanks to mathematicians Heinz Geerlings and Bernhard Vaske (Institute of Biometry, Medical School Hannover) for conducting the statistical evaluation.
