Abstract
Summary
A small-scale, economical isolator system was adapted to hatch and raise chicks in a bacteria-free environment as a means to observe bacterial interactions with the intestinal mucosa during early development. The design and construction of flexible plastic isolators for incubation and brooding are described along with methodologies for preparation of eggs for entry into the isolators, incubation and hatching. Two trials were conducted, the first in August 2005 and the second in March 2006. Results from both trials showed no differences in body weights of chicks raised in isolation when compared with those raised conventionally. Growth of bacteria was detected from rectal swabs at day 2 post-hatch, with both trials, showing a light growth of Bacillus sp., coagulase-negative staphylococci and haemolytic streptococcus in trial 1, and a light growth of Bacillus cereus only in trial 2. Although not germfree, the growth of bacteria in chicks raised in isolation was decreased or absent when compared with chicks raised conventionally. Feed was negative for contamination and surface swabs of equipment were also negative until day 3 post-hatch, suggesting possible contamination within the eggs themselves. Despite the presence of bacterial species, the isolator system was successful in producing low bacterial load chicks for comparison studies with conventionally raised chicks.
Flexible film isolators are widely employed for housing animals under germfree, gnotobiotic and specific pathogen-free (SPF) conditions (Phillips et al. 1962, Dennet & Bagust 1979). Such housing is useful as it allows the study of mechanisms underlying the pathology caused by bacterial infection. Isolation systems are usually part of large-scale SPF or physical containment level 2 (PC2) animal housing facilities, which are costly both in terms of construction and management (Cooper 1970). As facilities of this nature were not available on site, the aim was to establish a convenient, cost-effective clean room environment to produce and house chicks with a minimal load of intestinal microflora to facilitate the study of mucosal–bacterial interactions.
Many isolator systems for rearing chicks have placed eggs in the isolator during the hatching period or even at one day of age (Phillips et al. 1962, Dennet & Bagust 1979, Drew et al. 2003). The feasibility of incubation and hatching chicken eggs within an isolator system was examined in an attempt to minimize exposure to the outside environment. The technical details of the flexible plastic isolators used for the incubation, hatching and rearing of low bacterial load (LBL) broiler chicks, along with microbiology results, and growth rate comparisons with conventionally raised chicks are described.
Materials and methods
Isolator construction
A small animal, non-SPF housing facility (∼49.48 m3) situated at the University of Adelaide's Roseworthy Campus was selected for the experiment. Two isolators, previously used for quality control testing of poultry vaccines, were prepared for the incubation, hatching and rearing of LBL chicks (University of New England, Armidale, NSW, Australia). One isolator was designated for incubation and hatching (isolator type B, 500 × 1000 × 1300 mm) (Figure 1) and the other for brooding (isolator type A, 500 × 500 × 1300 mm) (Figure 2). The isolators consisted of a metal frame with two solid metal ends, which accommodated the air inlet (Figure 2b), and outlet (Figure 1d), the access box (Figure 2a), temperature probe inlet (Figure 2f) and drinker line (Figure 2e). The pressed metal floor was elevated above the outer plastic of the isolator in order to allow passage of faeces. The isolator frame was covered in disposable polyvinyl chloride (PVC) flexible plastic (0.2 mL Dry Slip, 1.36 × 100 m, Halifax Vogel Group, Turrella, NSW, Australia), which was replaced after each trial (Figure 2c).


The access box was used as a means of transporting items into and out of the isolator; it consisted of two sections (one with a glued extension) of 150 mm diameter PVC pipe and two sealed lids. Access into the isolator was achieved by inserting glove ports into the plastic walls. Gauntlet rings constructed from 200 mm internal diameter (i.d.) PVC pipe (Virginia Irrigation Service Pty Ltd, Virginia, South Australia) were used to attach a pair of 81.3 cm long, 20.3 cm port neoprene dry box gloves (Mohawk Industry and Nuclear Supply, Inc, CT, USA). Two glove ports were attached on opposite sides of the type B isolator (Figure 1b) and one on the type A isolator (Figure 2d).
A portable air filter (Model LR2, 0.2 µm, 5 m2 high-efficiency filter media area, Ozone Pollution Technology, Silverwater, NSW, Australia) was used to supply microbe-free air at the correct positive air pressure and flow to both isolators via a 50 mm diameter dual spigot (type A isolator, 6.5 m3 air required/h, minimum back pressure 250 Pa; type B isolator, 13 m3 air required/h, minimum back pressure 250 Pa) (Figure 3d). An in-line silencer was attached to minimize noise (Figure 3b). The air filter was connected to the isolator's air inlet connection via a 50 mm diameter duty hose and air valve (Figure 3d). The presence of the air valve provided an effective means of blocking the passage of air during fumigation.

The air relief line (Figure 1d) consisted of a long section of 25 mm diameter PVC pipe that hung parallel to the isolator end to exhaust waste air from the isolator. At the bottom of the pipe there was a fitting to accommodate a cylindrical mesh filter to prevent insects from entering. To avoid dust and other contaminants from the isolator becoming airborne in the room, the mesh filter was submerged in a dilute insecticide solution (Coopex®, 1 g/100 mL, Intensive Farming Supplies, Cavan, South Australia).
Testing for leaks
All external air access to the isolator was sealed. The air filter was turned on and the air valve was opened to distend the isolator. Air leaks were detected by applying a detergent solution to areas for detection of air bubbles. Any leaks were repaired, residual pressure released and all air lines resealed.
Sterilization of isolators
All equipment used for the construction and set-up of the isolator was first thoroughly cleansed with warm 6% v/v sodium hypochlorite solution. Once assembled, the air inlet and outlet were closed, and the isolators fumigated with formaldehyde gas for 24 h (isolator type A, 9 mL formaldehyde solution [37–40%] added to 5 g of potassium permanganate; isolator type B, 18 mL formaldehyde solution [37–40%] added to 11 g of potassium permanganate; Sigma, Sydney, Australia) and then ventilated for another 24 h. After ventilation, the isolators were fogged with Virkon S® (10 g/L) (Independent Veterinary Supplies, Melrose Park, South Australia) disinfectant as a secondary treatment. Insecticide treatment (Coopex®, 1 g/100 mL) was also applied to possible entry points for insects.
Isolator type B: incubation and hatching
Two Maino 50-egg incubators (Model ME3, type A with automatic eggs turning; Intensive Farming Supplies) were used for incubating and hatching under isolation (Figure 1c). The incubators were lightweight and portable, consisting of a plastic tub and lid with a metal egg tray that held 50 chicken eggs. Although the incubators had automatic temperature and egg-turning control, the humidity needed to be manually maintained using the two small rectangular basins provided with the incubators and a large shallow basin that sat underneath the egg tray. Humidity was measured using a small digital humidity metre, which could be attached to the inside of each incubator (Incubators and More, Adelaide, South Australia). The isolator was stocked with sterile milliQ water for use in the basins. All equipment necessary for incubation, candling and hatching was placed in the isolator before securing the flexible plastic.
Isolator type A: brooding
A drinker was constructed based on the Iberline Poultry Nipple System (Intensive Farming Supplies), using square pipe and fittings, three nipples, three orange water collection cups (Figure 2e) and a sight tube to flush out water. Drinking water was gravity-fed from an autoclaved plastic 10 L water drum via a flexible water line that passed into the drinker through an inlet located on the metal isolator wall (Figure 2e). The height of the drinker could be adjusted using cable ties as the chicks grew. Brooding temperature was achieved by the attachment of a brooding lamp and a 150 W ceramic heating bulb (Intensive Farming Supplies). This was secured to the top of the isolator frame via a metal plate (Figure 2g). After the plastic film had been fitted, the lamp power cord was pulled through the plastic film and sealed. Brooding lamp temperature was controlled by way of a universal temperature monitor (Model PM4-RT 240; Amalgamated Instruments Co Pty Ltd, Hornsby, NSW, Australia) and an industrial temperature probe (Model Pt100 RTD, P-RTQ12SA-1; Amalgamated Instruments). The brooding lamp power cord was plugged into the monitor, with temperature set at 32 ± 1°C. The probe was connected to the isolator via a designated access port (Figure 2f).
Sterilization of feed and drinking water
Chicks received high-energy broiler starter crumble (Ridley brand, Gawler Stock Feeds, Gawler, South Australia), which was separated into 500 g portions and vacuum-sealed. The bags were sterilized by gamma irradiation at 25 kGy (Steritech, Dandenong, Victoria, Australia). Feed was administered by transferring a bag of feed through the access box via a soak in Virkon S®. Once in the isolator, the bag was opened and feed dispensed into a standard plastic feeder (Intensive Farming Supplies). The bag was removed through the access box and discarded. MilliQ water was autoclaved in 1 L glass bottles at 121°C for 20 min and transferred into the sterile water drum. After each trial, excess water was removed and the water drum sterilized.
Procedure for low bacterial load chicks
Eggs (n = 100) of similar weight (∼50 g) were collected from a commercial Cobb 500 fast-feathering flock from Hi-Chick Hatchery, Bethel, South Australia. Two trials were conducted, one in August 2005 and the other in March 2006. The eggs collected were therefore from different flocks. Because eggs were sourced from a commercial flock and not from an SPF or germfree source, eggs were selected from hens that had commenced egg production three weeks prior to collection. This was to reduce bacterial transfer from the oviduct and/or cloaca to the egg, which is greatly increased in older hens (Dennet & Bagust 1979). All eggs were laid within a 2 h time period before collection (09:00–11:00 h). Eggs were dipped in Ambicide® (Independent Veterinary Supplies) at 42.2°C and air-dried (42°C). Once dry, they were placed in sterile plastic boxes fitted with high efficiency particulate air (HEPA) venting filters (Whatman HEPA-Cap 75, Singapore). They were then transported to Roseworthy and pre-incubated in the clean room at 17°C for three days and 26°C for 12 h.
After pre-incubation, the eggs were washed prior to entry into the isolator using modified methods (Wang & Slavik 1998, Drew et al. 2003). All equipment was cleaned and sterilized either by autoclave or by overnight soak in Virkon S®. Eggs (10–15) were removed from the sterile boxes and placed into a large aluminium foil tray containing 1% sodium hypochlorite (44°C). Eggs were gently scrubbed using a hard-bristled toothbrush to remove any organic matter attached to the shell. Once all were washed, they were placed into a plastic egg tray and then immediately into a drying oven for 2 min at 42°C.
The inside of the access box was saturated using Virkon S® spray. Ensuring that the inner lid was securely attached, the egg tray was placed in the box. Once inside the isolator, each egg was weighed, assigned a number and placed in the incubator. The process was repeated for the remaining eggs until all were washed and set in the incubators. The incubators were set at 37.7°C with humidity of 50–55% from day 0 to day 18. Incubators were checked morning and evening, the water basins were refilled with sterile MilliQ water and the egg-turning device monitored to ensure even turning was being accomplished. Eggs were candled on day 5 of incubation to observe development. Non-developing eggs were removed and yolks swabbed for detection of bacterial contamination.
On the 18th day of incubation, the turning tray containing the eggs and the large shallow basin was removed and the eggs placed on the incubator floor. Four more small basins were placed against the edge of the incubator. Mesh screens were constructed to guard the basins and prevent the newly hatched chicks from drinking and/or spilling the water. Incubators were set at 36.7°C with a humidity of 60–65%.
At hatch, 21 chicks were transferred into the brooding isolator via an autoclaved cylindrical polypropylene container that was further soaked in Virkon S®. After each transfer, the container was sprayed with Virkon S® and wiped using a disposable sterile cloth. Another 21 chicks were removed from the isolator and placed into conventional brooding pens at the campus poultry unit. Both groups were allowed free access to irradiated feed.
Cultures
Rectal swabs were collected daily from three chicks per group for seven days to observe bacterial growth. Surface swabs were also collected to monitor the sterility of the equipment. Gribbles Veterinary Pathology, Glenside SA performed all swab analysis, as well as the bacterial status of the irradiated feed. Swabs were set up on the following plates: Blood agar (to grow both Gram-negative [GN] and Gram-positive [GP] organisms), Maconkey agar (to grow GN organisms), colistin-nalidixic acid agar (to grow GP organisms), Selenite broth (pre-enrichment for Salmonella) and xylose lysine deoxycholate agar (selective for Salmonella sp.). Plates were incubated at 37°C and read at 24 and 48 h. A smear was made using the swab and Gram-stained to detect GN and GP organisms.
Animal welfare
Animals were monitored daily for loss of condition, from the three chicks that were swabbed, body weights were also recorded to observe growth in each environment over the seven-day period. All experimental work was approved by the Animal Ethics Committees of the University of Adelaide and the Department of Primary Industries and Resources of South Australia.
Statistical analyses
Statistical analyses were performed using the SPSS software package V11.5 (SPSS Inc, Chicago, IL, USA). Student's t-tests were used to compare body weights between conventionally raised and LBL animals. Significance was determined as P < 0.05.
Results
Hatchability
Hatchability using the incubators in an isolator gave a hatch of viable chicks of 60%, i.e. 60 chicks from 100 eggs. Chicks with poor condition, characterized by the presence of mottled feathers and/or irregular leg development, usually observed in chicks that hatch late, were excluded from the trial and humanely euthanized via cervical dislocation. The trial required a total of 42 chicks, hence the chicks used in the trial were those obtained on the first day of hatch. There were no losses of those 42 chicks during both trials.
Microbial analysis
Trial 1
Swab analysis of yolk sacs from removed candled eggs (n = 10) showed that all were negative for contamination except one in which the yolk sac had a heavy growth of coagulase-negative staphylococci. Surface swabs showed that both isolators and incubators were bacteria-free before the addition of eggs and chicks and remained so until day 2 post-hatch when a light growth of coagulase-negative staphylococci and Streptococcus sp. was detected in isolator A. Chicks remained bacteria-free until 48 h post-hatch when a light growth of Bacillus sp. and coagulase-negative staphylococci was detected in rectal swab samples. One chick from this group had a high growth of Streptococcus sp. (Table 1). Conventionally raised chicks possessed the same bacterial species as LBL chicks with the addition of Escherichia coli and Proteus mirabilis. Growth of these species was heavier when grown in culture when compared with the LBL chicks.
0: no growth, +: none to light, ++ : light to moderate, +++ : moderate to heavy, ++++: heavy (light growth: 10–50 colonies; moderate growth: 50–100 colonies; heavy growth: >100 colonies)
Trial 2
Similar to trial 1, culture and microscopy of swabs collected from non-developing eggs removed at candling detected no bacterial growth in the yolk (n = 10). Surface swabs of isolators and incubators showed that all were bacteria-free before the addition of eggs and chicks and remained so until day 2 post-hatch when a light growth of B. cereus was observed. Rectal swabs of chicks in both groups also remained bacteria-free until 48 h post-hatch, as observed in trial 1, but the presence of bacterial species in LBL chicks was remarkably different with the growth of only B. cereus. The conventionally raised group, however, showed no growth of this species, but a heavy growth of E. coli and Enterococcus faecalis was seen from day 2 to day 7 post-hatch (Table 2). Analysis of irradiated feed and drinking water samples showed no growth of bacteria after 48 h of incubation in culture.
0: no growth; +: none to light; ++ : light to moderate; +++ : moderate to heavy; ++++: heavy (light growth: 10–50 colonies; moderate growth: 50–100 colonies; heavy growth: >100 colonies)
Body weights
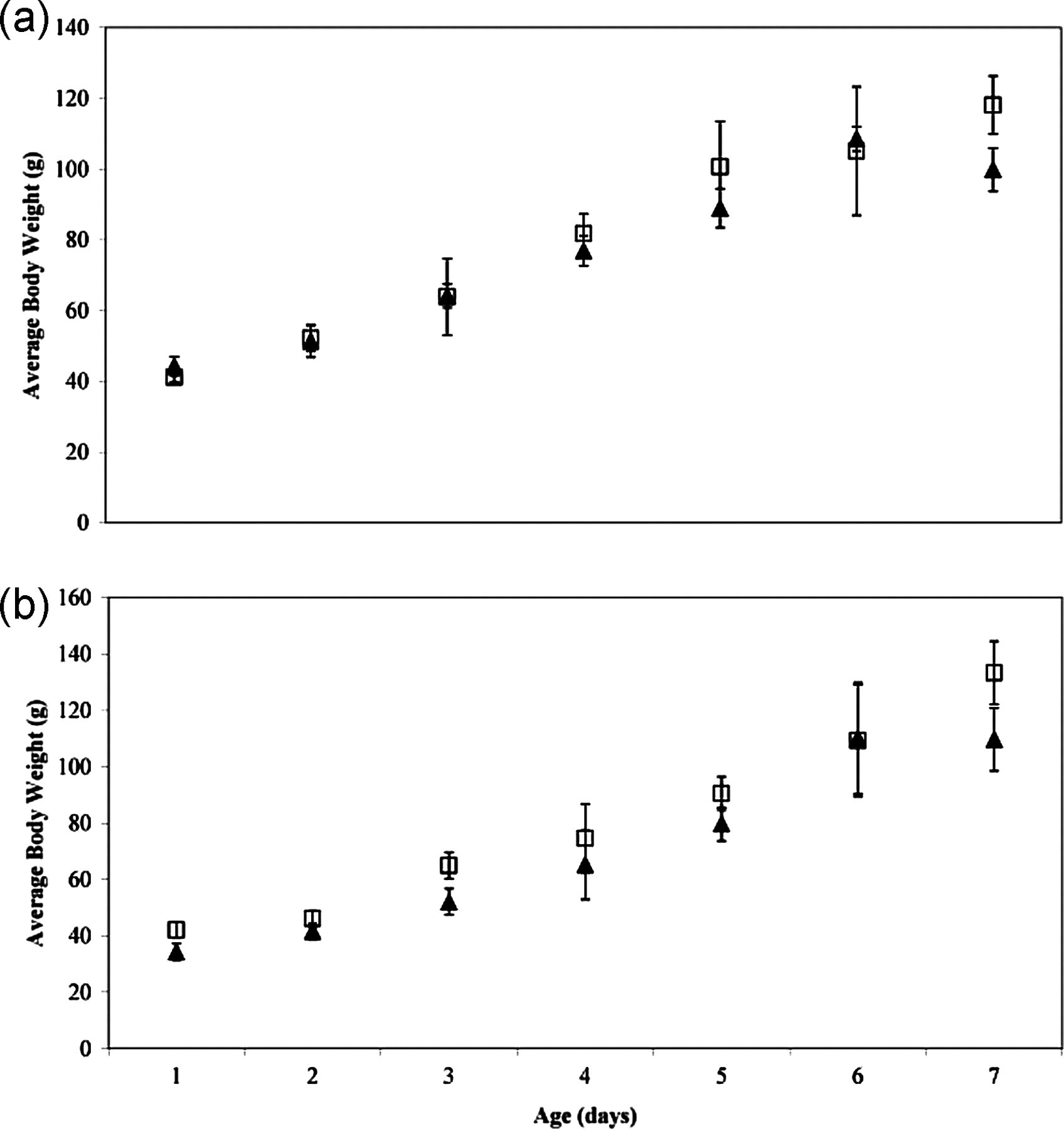
Figure 4 shows the comparative average body weights of LBL and conventionally raised chicks for both trials. There were no significant differences in body weight between the two groups over the seven-day time period in either trial (P > 0.05).
Discussion
Flexible film isolators were designed and assembled as a means for studying microbial–mucosal interaction in early posthatch chicks. The isolator system with all parts provided was established in less than one week including cleaning and fumigation with minimal labour, for under AUS$6000. Ideally, this set-up would benefit its location in a HEPA-filtered barriered animal housing facility, in which traffic of people and materials could be controlled more effectively. Although meticulous in maintaining cleanliness and avoiding possible contamination, appropriate wash-in/wash-out facilities would have been preferred.
The presence of bacteria in conventionally raised and LBL groups was first observed at day 2 post-hatch indicating that it may have taken at least 48 h for bacteria to establish themselves in the gastrointestinal tract, either through environmental exposures or as a result of digestion of the yolk (Coloe et al. 1984, van der Wielen et al. 2002, Deeming 2005). The presence of bacterial species in LBL chicks from rectal swabs during both trials indicated possible contamination.
Microbial analysis of feed and drinking water were tested negative for bacterial growth. All equipment showed no bacterial growth before chicks were transferred into isolator A and remained so until day 2 posthatch. The majority of eggs removed at candling were negative for bacterial growth; however, the detection of staphylococci in the yolk sac of a removed egg, suggested a possible bacterial contamination within certain eggs before entry into the isolator. Subsequent chick development would allow the spread of bacteria within the isolator during hatching. In the current study, late-hatch chicks which were of poor condition were removed from isolator B and excluded from the trial. Whether these chicks contained pathogens warrants further investigation in future studies to determine the exact source of contamination. The differences in bacterial profiles observed between trials suggested that the species of bacteria contaminating the eggs may have been defined by the microbial population of the source flock, indicating the importance of flock and egg selection for isolation experiments.
Coagulase-negative staphylococcus, alpha haemolytic streptococcus and Enterococcus sp. have been found to exist naturally in the gut of poultry (Salanitro et al. 1978, Devriese et al. 1991). B. cereus has also been found in broiler faecal samples (Barbosa et al. 2005) and thus suggests that all are resident commensals of the chicken intestinal microflora. The observed normal growth rates and no loss of condition in LBL chicks indicated no pathogenic effect of these species. A study on B. cereus and its ability to persist in the intestine of rats showed no detection of endotoxin, suggesting that the rat gut physiology may not allow the bacterial spores to germinate and produce endotoxins (Wilcks et al. 2006). This may also be true for the gut of chickens.
It is interesting to note that in trial 2 only the LBL group had B. cereus present. Competition between species whereby the Bacillus sp. cannot successfully colonize in the presence of certain bacteria is a possible explanation. As the LBL environment appeared to be free of other bacterial species, the Bacillus sp. had the potential to thrive, which would explain the increase in growth over the seven-day period.
Previous studies using germfree chickens have used a series of culture methods to detect contamination (Wagner 1959, Phillips et al. 1962, Drew et al. 2003), and have reported negative bacterial growth in these animals and claimed them to be germfree. Consistent with this study, the eggs used in the trials of these investigators were obtained from commercial layer flocks, where all hens would have been exposed to a variety of bacterial species. In the current study, it was very difficult to avoid contamination in both trials. The cleaning methods for entry into the isolator may have been more successful in other studies than the methods presented here, or the use of a smaller number of eggs may have reduced the chances of contamination.
Despite the presence of bacterial species in both trials, the isolator design was able to produce chicks with a LBL than conventionally raised chicks without compromising body weight. The isolator design was also economical in terms of costs and labour, providing a suitable means to study host–microbial interactions in experimental systems. Future studies using this system could assist in the development and efficacy testing of new dietary supplements such as probitotics and prebiotics to promote the growth of poultry through the manipulation of gut microflora. Moreover, it provides an effective, alternative model system for testing novel therapeutic options for human intestinal disorders such as inflammatory bowel disease.
Footnotes
Acknowledgements
This work was supported by funding from the Australian Poultry CRC, established and supported under the Australian Government's Cooperative Research Centres Program. We would like to thank Gribbles Veterinary Laboratory, Glenside SA for conducting all microbial analysis. We thank Jorge Ruiz and all the staff from HiChick Breeding Company, Bethel SA for generously providing the eggs and advice on incubation and hatching.
