Abstract
Summary
Buprenorphine is a partial μ, κ agonist that has been shown to influence spontaneous behaviour in animals. Previously, we have demonstrated significant differences in the analgesic response to buprenorphine between the August Copenhagen Irish (ACI)/SegHsd and the Brown Norway (BN)/RijHsd inbred rat strains. The purpose of this study was to determine whether these strains also differed in their behavioural response to buprenorphine in order to provide an additional parameter for the genetic analysis and localization of genes involved in this response. Male and female rats of both strains were used (n = 6/strain/sex) for this study. Each rat was subjected, respectively, to three treatment regimens at 15:00 h: (A) unchallenged; (B) intravenous saline; (C) intravenous buprenorphine (0.05 mg/kg) according to a crossover design. The relative duration (s/h) of locomotion, grooming, drinking and eating behaviour was subsequently determined from 15:30 to 07:00 h using the automatic registration system, Laboratory Animal Behaviour Registration and Analysis System™. Significant strain differences were observed in unchallenged behaviour between the ACI and the BN rats. ACI rats, but not BN rats, responded to buprenorphine treatment with decreased levels of locomotion, drinking and eating behaviour. The same treatment resulted in an increased grooming behaviour in both strains. Slight but significant sex differences were observed for locomotion and eating in the analysis of variance procedure, but did not reach the level of statistical significance in the multiple comparison procedure. The results of this study emphasize the possibility that strain-specific effects must be taken into account when using behavioural parameters for the assessment of the analgesic effects of buprenorphine in rats.
Buprenorphine is a highly lipophilic oripavine analgesic. Its analgesic effects are generally considered to be mediated through both mu (μ)- and kappa (κ)-opioid receptors (see Cowan 1995, Rothman et al. 1995 for review). It is applied extensively to alleviate clinical and postoperative pain in a variety of animal species and man (see Roughan & Flecknell 2002 for review). Apart from its analgesic properties, buprenorphine has been shown to influence the spontaneous behaviour of animals. In male albino mice (MFI/Ola), the subcutaneous (s.c.) administration of buprenorphine (0.10, 0.30 or 1.0 mg/kg) resulted in an increase in spontaneous locomotor activity when compared with control animals (Cowan et al. 1977b). In the same study, male Sprague-Dawley rats were also injected with s.c. buprenorphine (0.10–3.0 mg/kg) with the overall locomotor activity being increased as well, although the rats remained immobile and showed a typical hunchbacked (‘hedgehog’) posture initially. After 4–5 h, repetitive licking and biting of the limbs and cage bars occurred.
In male outbred Wistar rats, an increase in locomotor activity was also preceded by an initial reduction of activity following a single s.c. dose of 0.05 mg/kg buprenorphine (Liles & Flecknell 1992). Food intake was significantly reduced following a single s.c. dose of 0.05 or 0.1 mg/kg buprenorphine, whereas water intake remained unaffected. Male and female outbred Wistars displayed an increase in locomotor activity as well as a significant reduction in ventral grooming behaviour following 0.05 mg/kg buprenorphine s.c. (Roughan & Flecknell 2000). The intrinsic behavioural effect of buprenorphine can lead to misinterpretation when measuring the analgesic potency of this drug with the use of behavioural parameters, e.g. locomotor activity and grooming (Roughan & Flecknell 2000, 2002). Previously, we observed significant differences in the response to buprenorphine (0.05 mg/kg intravenous [i.v.]) as measured by the tail-flick test in inbred rat strains (Avsaroglu et al. 2007). The August Copenhagen Irish (ACI)/SegHsd strain responded strongly to the analgesic effects of buprenorphine while the Brown Norway (BN)/RijHsd strain had a weak response (n = 6 females/strain). To rule out the sex differences in analgesic response (Cook et al. 2000, Barrett et al. 2002, Terner et al. 2003), a subsequent study was performed where male and female members of both strains were subjected to the same experimental design. Although the response of the female ACI/RijHsd rats was weaker than the males, the strain differences remained significant (unpublished results).
The aim of the present study was to analyse the strain-specific effects of buprenorphine on behaviour in the two, differently responding, rat inbred strains, in order to provide an additional parameter for future genetic studies and localization of genes involved in the response to buprenorphine. For behavioural analysis, Laboratory Animal Behaviour Registration and Analysis System (LABORAS™) was used – a system that automatically registers six distinct behavioural categories in the rat (immobility, locomotion, rearing, grooming, drinking and eating). As this system is entirely automated, continuous long-term behavioural measurements can be performed in the animal's home cage (Bulthuis et al. 1997, Van de Weerd et al. 2001).
Materials and methods
Animals
Twelve rats of the ACI/SegHsd (ACI) strain and 12 rats of the BN/RijHsd strain (BN) (n = 6/sex/strain) were used for this study. The ACI is a black agouti-coloured inbred rat strain with a white belly and feet. The substrain SegHsd is derived from a nucleus colony obtained from Dr A Segaloff's colony at the Ochsner Medical Center, Jefferson, LA, USA. The strain is a model for congenital genitourinary anomalies, hepatic disorders and locomotor activity (Greenhouse et al. 1990). The ACI strain displays a unimodal wheel running activity pattern with a high amplitude of activity (Klante et al. 1999), while significant ultradian components are absent (Wollnik 1991). The BN is a non-agouti brown inbred rat strain. In 1963, the Radiobiological Institute, Nederlandse Organisatie voor toegepast-natuurwetenschappelijk onderzoek (TNO), Rijswijk, The Netherlands started inbreeding the substrain RijHsd. The strain is a model for myelocytic leukaemia, kidney disorders and sleep behaviour (Greenhouse et al. 1990). The BN rat exhibits low levels of corticosterone release after restraint stress or exposure to a novel environment when compared with other rat strains (Sarrieau et al. 1998). This is thought to be caused by differences in corticosteroid receptor efficiencies and regulation (Marissal-Arvy et al. 1999). The weight of the male rats ranged from 195 to 230 g and that of the females from 140 to 170 g at the start of the experiment. Both strains were purchased from Harlan Netherlands BV (Horst, The Netherlands). The rats' health status report indicated them to be free from the microorganisms monitored, based on the FELASA recommendations. Testing of the animals started when they were 12 weeks of age, after an acclimatization period of two weeks. The protocols of the experiments were approved by the Animal Experiments Committee of the Academic Biomedical Centre of Utrecht University.
Husbandry and study design
The animals were housed in a room adjacent to the room where the LABORAS™ equipment was located. Rats were housed in groups of four same-sex individuals of the same strain in Makrolon IV-S cages (Tecniplast®, Milan, Italy) with a woodchip bedding (Abedd®, Dominik Mayr KEG, Köflach, Austria). Surplus rats of the same sex and strain were added to the cages where just two experimental animals were present. The room temperature was maintained at 20–22°C with a relative humidity of 40–55%. The artificial light–dark cycle of the room was 12:12 h with lights on at 05:00 h at approximately 100 lux shelf level. The rats were fed a pelleted maintenance diet (CRM [P]®, SDS, Witham, UK) and had an access to tap water through drinking bottles ad libitum.
Each rat was subjected to three treatment regimens, respectively: (A) unchallenged; (B) i.v. saline; (C) i.v. buprenorphine, according to a crossover design (the animals undergo the different treatments consecutively: A–B–C). Immediately after each treatment, the rat was individually placed on the LABORAS™ platform for the collection of behavioural data. At the end of the LABORAS™ session, the animals were returned to their respective home cages and mates. No subsequent signs of aggression were observed among the cage mates.
Treatment
All rats were handled daily for two weeks, to reduce non-specific stress during the experiments. The unchallenged animals (treatment A) were placed directly on the LABORAS™ platforms without giving an i.v. injection. For treatments (B) and (C), the rats were restrained in a small towel and either saline (0.9% NaCl; B); or buprenorphine (Temgesic® 0.05 mg/kg, Schering-Plough, Amstelveen, The Netherlands; C) was injected into the tail vein using a 25G needle. Both treatments were administered at 0.2 mL/100 g body weight (BW) as this is considered an appropriate volume for i.v. dosing in the rat tail vein (Baumans et al. 2001). As the commercial solution of buprenorphine is too concentrated to be administered at the required volume, the compound was diluted in a laminar airflow cabinet with sterile saline. Neither infections of the injection site nor loss of appetite were observed during the experimental period.
Behavioural assessment
Four rats were tested at the same time using four different platforms (1 rat/strain/sex). At 15:00 h, the animals received the injection after which they were placed individually in a test cage. The unchallenged rats (treatment A) were placed in the test cage at 15:00 h as well. The collection of behavioural data started at 15:30 h and was terminated at 07:00 h the following morning. Each rat was thus tested on three consecutive days with treatments (A), (B) and (C), respectively. The cages were cleaned, disinfected (alcohol 70%) and supplied with fresh bedding before a new rat was introduced in the test cage.
The LABORAS™ system (Metris BV, Hoofddorp, The Netherlands) consisted of three parts: (i) sensor platforms; (ii) electronics (e.g. amplifiers and control unit); (iii) software (Windows-based). The triangular-shaped sensor platform (carbon fibre plate 700 × 700 × 1000 × 30 mm, Metris BV) was positioned on two orthogonally placed force transducers (single point small [SPS] load cells) and on a third fixed point attached to a heavy bottom plate (Corian Plate 695 × 695 × 980 × 48 mm, Metris BV). The whole construction stood on three spikes that were adjustable in height and absorbed external vibrations. The rats were housed in Makrolon type IIIH cages (UNO Roestvaststaal, Zevenaar, The Netherlands; Hopper and Bottle: LabProducts Inc, Seaford, USA) with a woodchip-covered floor. One cage was placed directly onto the sensing platform, the upper part of which (including the top, food hopper and drinking bottle) was suspended in a high adjustable frame and was free from the sensing platform. The vibrations evoked by the movements of the animal were picked up by the carbon fibre measurement plate and passed to the force transducers below it. The transducers were connected to a pre-amplifier (and signal conditioning unit) that was mounted on to the measurement platform. The gain and offset of the pre-amplifier were adjusted by the software, based on the weight of the laboratory animal that was entered by the experimenter, through a calibration routine. The pre-amplifier also filtered out the noise from the signals. The output signals of the amplifiers were sent to the LABORAS™ Control Unit (LCU) which converted the analogue signals into a digital format. The LCU sent the data over a serial line to the PC for further processing. The PC then processed the stored data using several signal analysis techniques to classify the signals into behavioural categories, such as locomotion, grooming, drinking and eating. The behaviour that dominated was scored. Movement of the animal with both forepaws and hindlimbs was classified as locomotion. Grooming included all categories of body and head grooming, penile grooming and scratching (for details see Van de Weerd et al. 2001).
Data processing and statistical analyses
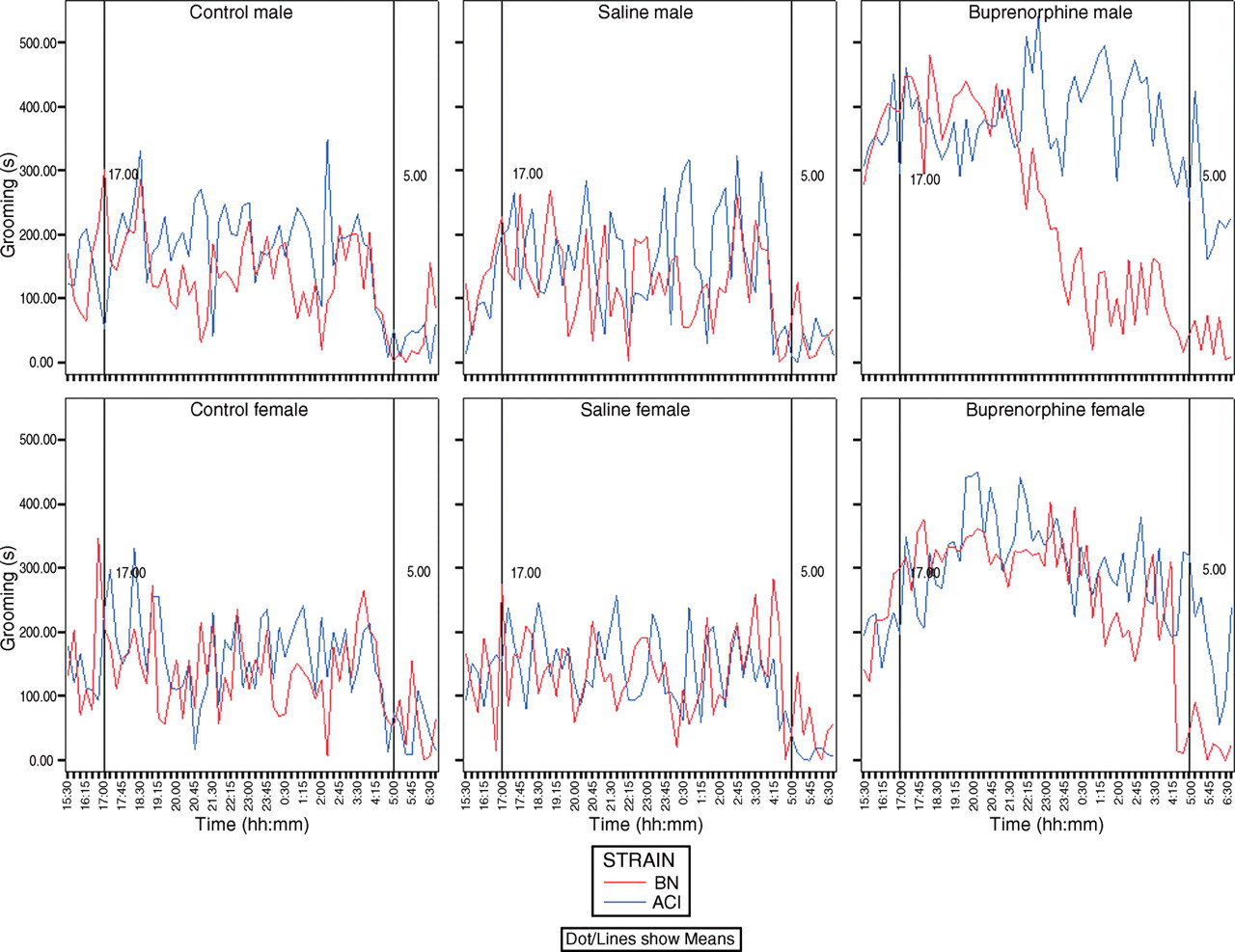
The behaviours: locomotion, grooming, drinking and eating that were recorded during the 15.5 h observation period were quantified as relative duration (s/h) both over the total observation period as well as in four time blocks of 4, 4, 4 and 3.5 h, respectively (Tables 1–4). An activity profile of grooming behaviour was constructed and expressed as duration in seconds/15-minute intervals. (Figure 1 is a typical example illustrating the behavioural pattern over the observed time period). In Tables 1–4, the results are presented as means ± SD.
*Block 1 = 15:30–19:30 h; Block 2 = 19:30–23:30 h; Block 3 = 23:30–03:30 h; Block 4 = 03:30–07:00 h; 17:00 h = lights out; 05:00 h = lights on
†0.2 mL/100 g BW
‡0.2 mL/100 g BW; 0.05 mg/kg buprenorphine
§Significance (P < 0.05) based on repeated measures analysis of variance (ANOVA) with between-subject factors strain (S) and gender (G) and within-subject factor treatment (T). S × T, interaction; S × G, interaction; T × G, interaction; S × T × G, interaction
¶Contrast significance (mean ± SD). For treatment comparison the paired Student's t-test, for strain and gender comparison the unpaired Students's t-test were used. Within one row, values bearing the same superscript letter are significantly different. Underlined values represent significant differences between genders (P < 0.0125, Bonferroni's correction). Bold values represent P < 0.05. Note that a P value of 0.000 does not mean that it is zero, only that it is less than 0.0005
ACI = August Copenhagen Irish; BN = Brown Norway; LABORAS = Laboratory Animal Behaviour Registration and Analysis System
All statistical analyses were carried out according to Petrie and Watson (1999), using a SPSS computer program (SPSS Inc, Chicago, IL, USA, 2004). Two-side probabilities were estimated throughout. The Kolmogorov-Smirnov one-sample test was used to check the normality of these data. All results within groups were normally distributed. The significance of the differences between groups was calculated by a repeated measures analysis of variance (ANOVA) with strain and gender as the main between-subject factors, and treatment as the main within-subject factor. If the repeated measures ANOVA showed significant effects, the group means were further compared with the unpaired and/or paired Student's t-test. The unpaired tests were performed with pooled (for equal variances) or separate (for unequal variances) variance estimates. The equality of variances was tested using an F-test. To take into account the greater probability of a type I error due to multiple comparisons, the level of significance for the Student's t-tests was pre-set at P < 0.05/times a group was used for a comparison (i.e. P < 0.05/4 = 0.0125) instead of P < 0.05, according to Bonferroni's adaptation. In all other cases, the probability of a type I error <0.05 was taken as the criterion of significance.
Results
An overview of the statistical results is shown in Tables 1–4.
Locomotion
Male ACI rats displayed a shorter duration of locomotion when treated with saline (15.5 h period, 15:30–07:00 h; fourth block, 03:30–07:00 h) and buprenorphine (15.5 h period, 15:30–07:00 h; first block, 15:30–19:30 h) when compared with unchallenged levels (Table 1). Although the ACI female rats also had a shorter duration of locomotion around the start of the dark period (first block, 15:30–19:30 h) when given saline or buprenorphine, this did not reach statistical significance over the total time period (15.5 h period, 15:30–07:00 h) measured.
Female but not male BN rats displayed an overall (15.5 h period, 15:30–07:00 h) significantly shorter duration of locomotion when treated with saline compared with unchallenged treatment. This was particularly apparent immediately after the injection (first block, 15:30–19:30 h).
ACI rats displayed an overall significantly longer duration of locomotion when compared with BN rats after both unchallenged treatment and saline administration. This was particularly apparent for females during the largest part of the dark period (first to third block, 15:30–03:30 h). In males, the differences between the strains became apparent later (control: second to fourth block, 19:30–07:00 h; saline: second and third block, 19:30–03:30 h).
Grooming
Rats of both strains and sexes responded to buprenorphine by grooming significantly longer over the total time period measured (15.5 h period, 15:30–07:00 h) when compared with no treatment or saline administration (Table 2). The grooming response of the male BN rats to buprenorphine, however, declined to unchallenged and saline levels in the second half of the dark period (third and fourth block; 23:30–07:00 h; Figure 1). Except for female ACI rats during the third block (23:30–03:30 h), no significant difference in duration of grooming was observed between the unchallenged rats and the saline-treated rats.
*Block 1 = 15:30–19:30 h; Block 2 = 19:30–23:30 h; Block 3 = 23:30–03:30 h; Block 4 = 03:30–07:00 h; 17:00 h = lights out; 05:00 h = lights on
†0.2 mL/100 g BW
‡0.2 mL/100 g BW; 0.05 mg/kg buprenorphine
§Significance (P < 0.05) based on repeated measures analysis of variance (ANOVA) with between-subject factors strain (S) and gender (G) and within-subject factor treatment (T). S × T, interaction; S × G, interaction; T × G, interaction; S × T × G, interaction
¶Contrast significance (mean ± SD). For treatment comparison the paired Student's t-test, for strain and gender comparison the unpaired Students's t-test were used. Within one row, values bearing the same superscript letter are significantly different. Underlined values represent significant differences between genders (P < 0.0125, Bonferroni's correction). Bold values represent P < 0.05. Note that a P value of 0.000 does not mean that it is zero, only that it is less than 0.0005
ACI = August Copenhagen Irish; BN = Brown Norway; LABORAS = Laboratory Animal Behaviour Registration and Analysis System
Over the total time period (15.5 h period, 15:30–07:00 h), no strain differences in the relative duration of grooming were observed in unchallenged and saline-treated rats. However, during the third block (23:30–03:30 h) for males, there were significant strain differences for all treatments. Furthermore, in this block, there were also strain differences for unchallenged female rats. The increased duration of grooming was observed immediately after the administration of buprenorphine (first block, 15:30–19:30 h) in male rats of both strains, whereas in females this became apparent only after the start of the dark period (second block, 19:30–23:30 h; Figure 1).
Drinking
A clear treatment effect was observed (Table 3). ACI rats of both sexes displayed a significantly shorter duration of drinking after buprenorphine administration when compared with unchallenged treatment (males and females: 15.5 h period, 15:30–07:00 h; females: first block, 15:30–19:30 h and third block, 23:30–03:30 h; males: second and third block, 19:30–03:30 h).
*Block 1 = 15:30–19:30 h; Block 2 = 19:30–23:30 h; Block 3 = 23:30–03:30 h; Block 4 = 03:30–07:00 h; 17:00 h = lights out; 05:00 h = lights on
†0.2 mL/100 g BW
‡0.2 mL/100 g BW; 0.05 mg/kg buprenorphine
§Significance (P < 0.05) based on repeated measures analysis of variance (ANOVA) with between-subject factors strain (S) and gender (G) and within-subject factor treatment (T). S × T, interaction; S × G, interaction; T × G, interaction; S × T × G, interaction
¶Contrast significance (mean ± SD). For treatment comparison the paired Student's t-test, for strain and gender comparison the unpaired Students's t-test were used. Within one row, values bearing the same superscript letter are significantly different. Bold values represent P < 0.05. Note that a P value of 0.000 does not mean that it is zero, only that it is less than 0.0005
ACI = August Copenhagen Irish; BN = Brown Norway; LABORAS = Laboratory Animal Behaviour Registration and Analysis System
A clear strain effect was also found with ACI rats of both sexes displaying a significantly longer duration of drinking after unchallenged and saline treatment when compared with BN rats (15.5 h period, 15:30–07:00 h). This effect remained only significant for unchallenged treatments at the end of the dark period.
Overall, drinking behaviour occurred more in the dark (first, second and third block, 15:30–03:30 h) than in the light period (fourth block, 03:30–07:00 h).
Eating
A treatment effect was observed similar to that of drinking (Table 4). ACI rats of both sexes displayed a significantly shorter duration of eating after buprenorphine administration when compared with both unchallenged and saline treatment (males and females: 15.5 h period, 15:30–07:00 h; females: first block, 15:30–19:30 h and third block, 23:30–03:30 h; males: second and third block, 19:30–03:30 h). In contrast, the duration of eating behaviour was not disturbed in the BN rats despite the administration of buprenorphine. This resulted in a significant strain difference in the relative duration of eating throughout the measured time interval when buprenorphine was administered (15.5 h period, 15:30–07:00 h; first to third block, 15:30–03:30 h).
*Block 1 = 15:30–19:30 h; Block 2 = 19:30–23:30 h; Block 3 = 23:30–03:30 h; Block 4 = 03:30–07:00 h; 17:00 h = lights out; 05:00 h = lights on
†0.2 mL/100 g BW
‡0.2 mL/100 g BW; 0.05 mg/kg buprenorphine
§Significance (P < 0.05) based on repeated measures analysis of variance (ANOVA) with between-subject factors strain (S) and gender (G) and within-subject factor treatment (T). S × T, interaction; S × G, interaction; T × G, interaction; S × T × G, interaction
¶Contrast significance (mean ± SD). For treatment comparison the paired Student's t-test, for strain and gender comparison the unpaired Students's t-test were used. Within one row, values bearing the same superscript letter are significantly different. Bold values represent P < 0.05. Note that a P value of 0.000 does not mean that it is zero, only that it is less than 0.0005
ACI = August Copenhagen Irish; BN = Brown Norway; LABORAS = Laboratory Animal Behaviour Registration and Analysis System
As with drinking, most of the eating behaviour occurred in the dark period (first to third block, 15:30–03:30 h).
Discussion
After administration of buprenorphine, strain-dependent differences were found for locomotor activity and for drinking and eating behaviours. Previous studies have reported an initial depression followed by an increase in locomotor activity after the administration of buprenorphine in rats (Cowan et al. 1977b, Liles & Flecknell 1992, Bartoletti et al. 1999, Roughan & Flecknell 2000). In the present study, we observed a significant decrease in relative duration of locomotion in the male ACI rats. Although only the male BN rats showed an initial depression, this was not followed by an increase in duration of locomotion (Table 1). This discrepancy found between previous studies and the present study can be attributed to strain-related differences. Roughan and Flecknell (2004) observed no difference in locomotor activity response after administration of buprenorphine between the outbred Wistar and inbred F344 strains using behavioural data recorded on video, thus suggesting that, in their study, strain does not influence the effect of buprenorphine on this parameter. However, the present findings indicate that there is indeed a strain effect. Liles and Flecknell (1992) observed a lower level of locomotor activity when compared with pretreatment levels immediately after (1–3 h postinjection), but also at the end of the measurement period (15–17 h postinjection). In the present study, a significant decrease was observed in the total duration of locomotion measured over 15.5 h in male ACI rats. The duration of analgesic action of buprenorphine which is said to be 6–12 h (Roughan & Flecknell 2002), appears to be outlasted by its behavioural effects. It is hypothesized that buprenorphine causes a disruption of circadian (Liles & Flecknell 1992) and ultradian (Roughan & Flecknell 2000) rhythmicity, thereby altering the activity patterns of the animals over a prolonged period of time. The opioid analogues, such as morphine and fentanyl, have been shown to induce a phase shift in locomotor activity. Recent findings indicate that direct involvement of opioid receptors in altering the electrical activity of the circadian pacemaker and regulation of clock genes. Future experiments will focus on elucidating the subtypes of opioid receptors involved in this regulation and as a consequence, the possible influence of buprenorphine (Vansteensel et al. 2005).
Buprenorphine has been described to produce cataleptic states when administered in rats (Cowan et al. 1977a). It is suggested that buprenorphine interacts with the central dopaminergic systems, thereby depressing the central nervous system (Cowan et al. 1977b, Bartoletti et al. 1999, Smith et al. 2003). Rat strain differences in dopamine (DA) receptor levels and resulting differences in behaviour have been described (Zamudio et al. 2005). In addition, Baumann et al. (2000) found that opiate modulation of the hypothalamic-pituitary-adrenal axis (HPA) and mesolimbic DA function can be strain-dependent as well. Thus, the observed strain differences in locomotor response to buprenorphine could be related to the differences in opiate-induced HPA and DA reactivity.
With respect to drinking behaviour, the ACI strain responded immediately to the administration of buprenorphine by exhibiting less drinking behaviour up until the end of the dark period. Liles and Flecknell (1992) found little or no effect on water intake after the administration of clinical doses (0.01 and 0.05 mg/kg) of buprenorphine to male outbred Wistar rats. Their conclusion was that water consumption is likely to be a more reliable parameter for assessing postoperative pain and the efficacy of analgesics in rats than food intake or locomotor activity. In light of the results of the present study, this conclusion does not seem to be valid for all rat strains. Similar to the results for drinking, ACI rats also displayed significantly less eating behaviour during the entire period measured. This was also found by Liles and Flecknell (1992) in Wistar outbred rats. The BN strain however did not alter its eating behaviour after the administration of buprenorphine. As both drinking and eating were affected negatively in the ACI strain after buprenorphine, one might assume that this was because of the overall lowered level of activity in this strain.
An evident effect of buprenorphine on grooming activity was found, but no strain difference was observed for this parameter. Rats of both strains and sexes exhibited an increase in grooming activity during the dark period. Although a number of stressors can elicit grooming behaviour in rats, it has been postulated that grooming rather reflects the process of de-arousal due to the termination of or habituation to a stressful situation (Spruijt et al. 1992). In the present study, only the administration of buprenorphine resulted in an increased grooming activity, whereas the i.v. administration of saline did not. In contrast, Roughan and Flecknell (2000) found a decrease of ventral grooming behaviour after administration of buprenorphine to outbred Wistar rats.
Unchallenged behaviour and influence of intravenous saline injection
During most of the dark period, ACI rats have a longer duration of locomotor activity when compared with BN rats. In addition, the ACI strain displays a longer duration of locomotion when compared with the BN strain during the dark period after tail vein injection. Overall, ACI rats of both sexes responded to the administration of saline with a decrease in the duration of locomotion immediately after the injection until the end of the dark period. This effect was far less visible in the BN strain. Van Herck et al. (2000) found that orbital puncture depressed the relative duration and the frequency of locomotion during the dark period. It has been suggested that rats respond to injury with a reduced level of activity or even immobility. When judged on the basis of these behavioural changes, the degree of discomfort caused by orbital puncture was found to be similar to tail vein puncture (Van Herck et al. 2001). Tail vein puncture can thus be construed as causing discomfort to the animal in the strains tested. ACI rats may experience more discomfort from tail vein injection than the BN rats. It was shown in previous studies that genotype can influence the HPA activity and reactivity to stress (Sarrieau et al. 1998). Strain comparison studies on stress-induced HPA reactivity using both the ACI and BN rats were not found in a literature search, although the BN strain appears to be an overall hyporeactive strain to stress-induced corticosterone response (Sarrieau et al. 1998, Marissal-Arvy et al. 1999). In future studies, it would be interesting to correlate the locomotor response to saline injection of the two strains with circulating corticosterone levels.
During the total period, ACI rats had a longer duration of drinking behaviour when compared with BN rats, but the time for food intake did not differ. We did not measure the amount of food and water intake, but Walsh (1980) has found that animals of the ACI strain have the highest relative food intake (g/100 g BW) and one of the highest relative water intakes (mL/100 g BW) out of the 16 rat strains tested. Overall, it can be concluded that the ACI strain displays a higher level of activity of locomotion and drinking (especially during the dark period) when compared with the BN strain. Several studies have demonstrated uninterrupted activity patterns with a high level of activity in the ACI rat strain when compared with other inbred strains (Büttner & Wollnik 1984, Klante et al. 1999). The results from this study confirm these findings.
Although an acclimatization period of 30 min in the test cage was incorporated prior to each behavioural measurement, slow intra-session habituation of the rats to the novel environment might have influenced the results. In addition, it has been shown that arousal can significantly influence the time needed for habituation (Leussis & Bolivar 2006). The tail vein injection from treatments (B) and (C) could therefore have prolonged the habituation period as well. In a previous study, significant strain and sex differences in habituation period were established between the SHR and WKY rat strains (Hendley et al. 1985). The SHR strain even failed to habituate to the test cage irrespective of age and sex, possibly related to the characteristic hyperarousal behaviour of this strain. The obtained strain differences from the present study might thus have partly been due to habituation differences between the two strains.
In summary, the ACI rats responded to buprenorphine by an overall lowered level of locomotion, eating and drinking behaviours, whereas the effects of buprenorphine on the BN rats were much less pronounced. Grooming activity however was increased in both strains and sexes under the influence of buprenorphine. Considerable strain differences have been found in unchallenged behaviour between the ACI and the BN rats, whereas only the ACI strain seemed to respond to the effects of tail vein injections. Future genetic analysis to localize quantitative trait loci (QTLs) involved in the strain-specific analgesic and behavioural responses may lead to the detection of candidate genes and may thus contribute to detecting which mechanism is responsible for the strain-specific response. Mouse chromosome 10 contains the Oprm1 gene encoding the mouse μ-opioid receptor type (Belknap et al. 1995, Bergeson et al. 2001). Oprm1 is an obvious candidate gene for analgesic sensitivity to μ-opioid agonists, as polymorphisms have been shown to reduce morphine potency (Mogil 1999). A number of other μ-opioid-induced traits, e.g. alterations in locomotory behaviour, have QTLs that map to the same proximal region of chromosome 10 as does the μ-opioid receptor locus (Bergeson et al. 2001). It stands to reason that genetic analysis of the analgesic and behavioural response to buprenorphine of F2-intercross (ACI × BN/F1 × F1) progeny could localize QTL's mapping to the same region as the Oprm1 gene on rat chromosome 1 (Watanabe et al. 1999). The interstrain variability in the response to buprenorphine may also be due to polymorphisms related to pharmacokinetics rather than pharmacodynamics. Specifically, polymorphisms in genes encoding for cytochrome P450 enzymes involved in the biotransformation of buprenorphine could account for the phenotypical differences observed (Iribarne et al. 1997, Mogil 1999).
Footnotes
Acknowledgements
The authors wish to thank Anja van der Sar for her assistance during the experiments and Professor Frauke Ohl for critical reading of the manuscript.
