Abstract
Summary
Individual differences and a rather long-lasting reproductive cycle, as well as the relatively small number of oocytes that mature during one reproductive cycle makes it difficult to establish a cryopreserved stock of preimplantation embryos of the guineapig (Cavia porcellus) when compared with other laboratory rodents. Only a few data for superovulation protocols that can be used for routine laboratory use in guineapigs are available. However, a huge number of different strains exist for many purposes and the establishment of a frozen repository makes sense. Here, we describe the successful freezing of preimplatation embryos of the strain 2BS with a two-step freezing protocol in a freezing medium containing 1,2-propanediol as cryoprotectant. Human menopausal gonodotrophin induced superovulation in the embryo donors.
The guineapig (Cavia porcellus) is used as a laboratory animal since the late 18th century (Terrill & Clemons 1998). Despite the fact that this species is still playing an important role in laboratory animal science, the number of scientific experiments for which guineapigs are used is decreasing. In 2004, guineapigs were used in about 27,000 scientific experiments in the UK, representing <1% of total animal research. Over half of these were studies of the respiratory, nervous and immune systems. The use of guineapigs has fallen by over three-quarters since 1988, mostly because of a reduction in their use in safety testing. A recent contribution to this reduction has been the introduction of a milder test for the potential of chemicals to cause allergic skin reactions (skin sensitization), which uses mice instead of guineapigs. However, guineapigs remain essential in many areas of research.
Because of similarities of the immune system to that of man, it serves as an excellent model in this field of research (Liang et al. 2005, Orme 2005, Bowick et al. 2006).
Because of a long gestation period, spontaneous ovulation and active corpora lutea, the guineapig is also an excellent animal model for the study of reproduction in humans (Suzuki et al. 2003).
The most common strains in use are outbred animals of various stocks or strains. A common outbred stock is the Albino short-haired Duncan-Hartley English guineapig. Several inbred strains are also available. The strain 2, and strain 13 guineapigs are the most widely known and used strains. New strains for various purposes have been established during the last decades according to Festing (1993). The Central Laboratory Animal Facility of the Hannover Medical School maintains complement-deficient strains as described by Bitter-Suermann and Burger (1986, 1990) as well as the standard inbred strain 2BS, that was derived from the NIH strain 2 in 1980. The exponential increase of genetically-modified mouse strains necessitates the amplification of maintaining capacities for this species. Furthermore, because of the decrease in the use of guineapigs for scientific experiments, solutions for the preservation of the genetic pool of these scientifically valuable guineapig strains must be found. The establishment of a ‘Cryobank’ might solve this problem.
However, compared with mice and rats, guineapigs have small- to medium-sized litters and only few preimplantation embryos can be obtained from one female. Induction of superovulation by inhibin vaccine as described by Shi et al. (2000a,b), or injections of the pregnant mare's serum gonadotrophin after long-term implantation of progesterone tubing (Kosaka & Takahashi 1989) are not suitable for routine laboratory use, especially because these methods are time-consuming and quite expensive. For the purpose of establishing a frozen repository, another protocol using human menopausal gonadotrophin (hMG) administered subcutaneously on three consecutive days of the oestrus cycle seems more practicable (Suzuki et al. 2000). However, this method needs to be optimized for routine use. Great individual variations in the duration of the oestrus cycle (15-19 days) (Terril & Clemons 1998) further complicate the situation.
Here, we describe the superovulation of the strain 2BS using a modified protocol of Suzuki et al. (2000) and the successful cryopreservation by a two-step freezing protocol. The suitability of 1,2-propanediol (PROH) and dimethyl sulphoxide (DMSO) as cryoprotectants is compared.
Material and methods
Animals and husbandry
Guineapigs of inbred strain 2BS were maintained at the Central Animal Facility of the Hannover Medical School. Maintenance and use of the animals were in accord with the German Animal Welfare Legislation (Tierschutzgesetz 1998). All experiments were approved by the local Institutional Animal Care and Research Advisory Committee and permitted by the local government. The guineapigs for this study were housed in a controlled environment with 20 ± 2°C, 55 ± 5% humidity and a day-night cycle of 12 h each (from 18:00 h to 06:00 h light; 18:00 h to 06:00 h dark). Animals in experiment were caged in wire-topped Makrolon type IV cages (Tecniplast, Italy) on autoclaved softwood granulate bedding (Altromin, Lage, Germany) and autoclaved hay. They received a commercial diet (Ssniff, Soest, Germany) and tap water ad libitum. The females used for this study were 16 ± 1 weeks old and had a body weight of about 550 g. They were all nulliparous. For timed mating, females with an open vagina were placed into the cage of a male overnight. Successful mating was determined by vaginal smears taken the next morning. For this, loose cellular material was removed from the vagina by gently scraping the dorsal vaginal wall with a sterile loop, dispersed in a drop of saline on a slide, air-dried, stained with methylene blue solution (Löffleŕŕs methylene blue solution, Merck, Darmstadt, Germany) and examined microscopically. Sperms could be identified as dark blue thread-like structures. Vaginal plug controls were not performed. According to our study, the plug fell out of the vagina or was pulled out by the female. The next morning the plug could hardly be found in the bedding.
Superovulation
The protocol for superovulation was adapted from Suzuki et al. (2000). Females received an intraperitoneal injection of 5 IU hMG (Sigma-Aldrich; www.sigmaaldrich.com) on days 14, 15 and 16 after the last vaginal opening, i.e. oestrus. The females were mated as soon as the vagina opened. Vaginal smears were taken the next morning to assure successful mating. When sperms were present, the female was considered pregnant. For comparison, untreated females with open vaginas were also mated.
Collecting preimplantation embryos
A total of 53 females with sperm-positive vaginal smears were killed on days 2.5, 4.5 or 6.5 postcoitum (p.c.). The oviducts and part of the consecutive uterus horns were excised. Preimplantation embryos were flushed from the oviduct with phosphate-buffered saline (PB1, Whittingham 1971) through the infundibular opening using a blunt end 33 gauge needle, attached to a syringe filled with PB1 medium. The embryos were washed three times in fresh PB1. Only morphological intact embryos were kept for further use.
Cryopreservation and thawing
Preimplantation embryos were frozen by a modified two-step method using either 1.5 mmol/L PROH or 1.5 mmol/L DMSO as cryoprotectant in PB1 as described by Hedrich and Reetz (1990) for the rat. The freezing medium was precooled to 4°C. Plastic straws (Minitüb®; www.minitube.de), with one end closed using a metal bulb, were loaded with 250 μL freezing medium. The embryos were deposited in the centre of the medium and the other end of the straw was sealed with a glass bulb. The straws were then transferred to a programmable automatic ethanol cooling bath (Thermo-Haake P2-C75P; Thermo, Karlsruhe, Germany) equilibrated at 0°C. After 5 min, cooling was started with a velocity of 1°C/min to -6°C. Seeding was induced manually and cooling was continued at 0.4°C/min to -32°C. The same temperature was held for 5 min and then the straws were directly transferred to liquid nitrogen for storage.
Thawing at a rate of about 300°C/min was achieved by warming the straws at room temperature for about 40 s. After melting, the freezing medium with the embryos was flushed into a 30 mm Petri dish and an equal volume of fresh PB1 supplemented with 0.1 mmol/L sucrose (Merck) was added immediately to dilute the cryoprotectant. After 10 min, another volume of PB1 (without sucrose) was added. This step was repeated twice. Embryo viability was verified by staining with fluorescein diacetate (FDA, Sigma) (Mohr & Trounson 1980, Niemann et al. 1981). Viable blastomeres showed green fluorescence (Zeiss-Axiovert 135, UV-filter for green fluorescence at 546 nm; Carl Zeiss AG, Germany).
Statistical analysis
For descriptive statistics, we used the Stat View 5.0 program (2002).
Results
Effect of superovulation
The epithelial structure sealing the vagina ruptured the day after the last hormone injection, indicating that all treated females were in rut (oestrus). On day 2.5 p.c. more embryos were collected than on day 4.5 p.c. from untreated as well as from superovulated females. The total number of embryos on both days of collection was higher in the superovulated females. On day 6.5 p.c. morulae and blastocysts were found in untreated females, no embryos could be flushed from the oviducts of superovulated females.
Different developmental stages were found on the same day of pregnancy. On day 2.5 of pregnancy, we found one- to eight-cell stages in both groups. On day 4.5 of pregnancy four-cell to morula stages were found in untreated females and eight-cell to blastula-stages in superovulated females. On day 6.5 p.c. mainly early blastocyst stages were collected from untreated females, preimplantation stages were never found in the oviducts of superovulated animals. Upon microscopic examination all embryos seemed to be morphologically intact. Table 1 summarizes the results.
Mean number ± standard error of preimplantation embryos collected 2.5, 4.5 and 6.5 days postcoitum (p.c.) from untreated and superovulated females
The strain 2BS used in this study, normally gives birth to two pups, occasionally to three. This is in contrast to the number of embryos that can be collected from untreated females.
Cryopreservation
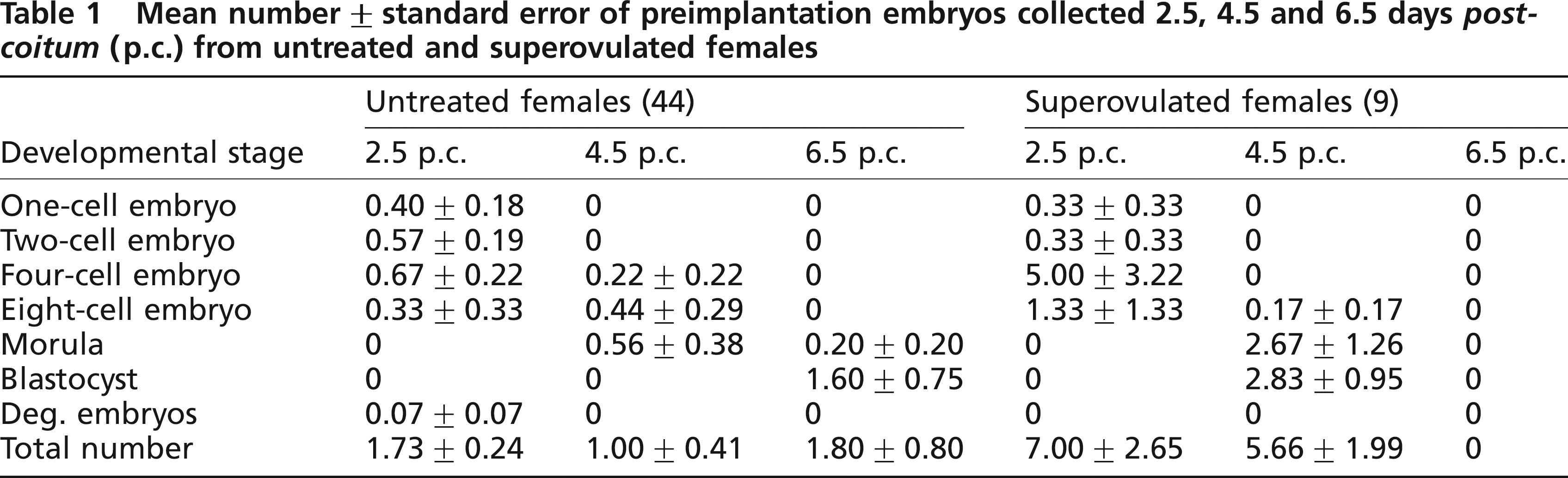
We cryopreserved four- to eight-cell embryos collected on day 2.5 of pregnancy of untreated and superovulated females. Most of the embryos appeared to be morphologically intact upon thawing when normal light was used for microscopy. FDA staining however, showed that most of the blastomeres were not viable when 1.5 mmol/L DMSO was used (Figure 1). Nearly all blastomeres fluoresced after FDA staining when 1.5 mmol/L PROH served as the cryoprotectant (Figure 2). There was no difference whether embryo donors had been superovulated or not. Table 2 compares the results of the freezing methods used.
Frozen-thawed guineapig embryos at magnification x80. Cryoprotectant used was 1.5 mmol/L dimethyl sulphoxide. Embryos have been collected on day 2.5 of pregnancy after superovulation. (a) Normal light illumination, (b) Same embryos as in Figure 1a after fluorescien diacetate A staining. An ultraviolet filter for green fluorescence at 546 nm was used. Viable blastomers show green fluorescence Frozen-thawed guineapig embryos at magnification ×80. Cryoprotectant used was 1.5 mmol/L propanediol. Embryos have been collected on day 2.5 of pregnancy after superovulation. (a) Normal light illumination, (b) Same embryos as in Figure 2a after fluorescein diacetate staining. An ultraviolet filter for green fluorescence at 546 nm was used. Viable blastomers show green fluorescence
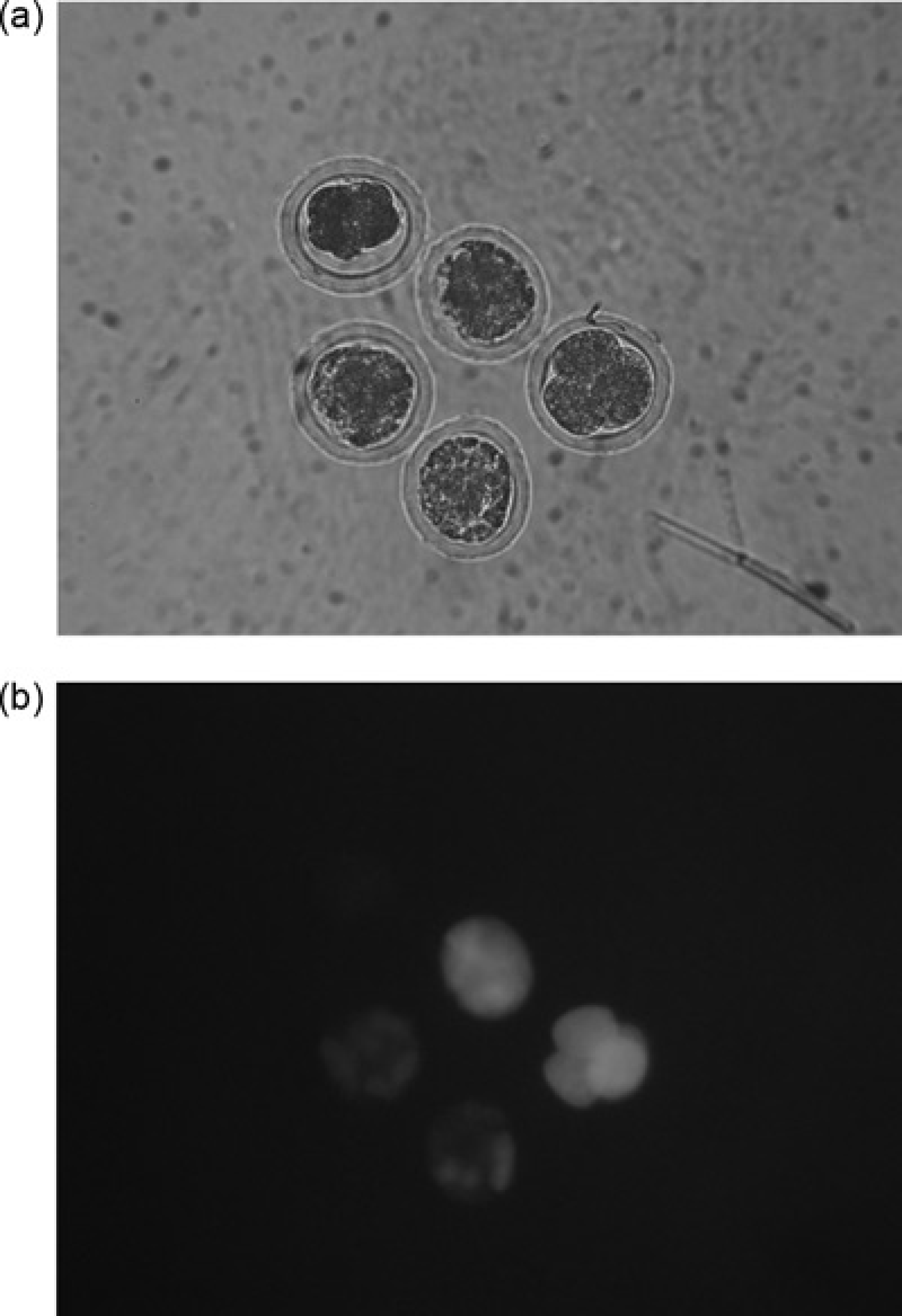
Comparison of dimethyl sulphoxide (DMSO) and propanediol (PROH) as cryoprotectant in a 1.5 mmol/L concentration
Viability was confirmed by fluorescein diacetate staining after thawing
Discussion
Effect of superovulation
The hormone treatment considerably increased the number of oocytes that matured during one ovulatory cycle. Moreover, it seemed to variably accelerate the embryo development. Although morulae and blastocysts were still in the oviducts on day 6.5 in untreated females, these developmental stages were already found on day 4.5 in the treated group. We never found preimplantation embryos on day 6.5 in treated females, suggesting that implantation has occurred before. These findings are in accordance with the findings of Ueda et al. (1994) who found preimplantation embryos from day 1 to day 5 of pregnancy, but not on day 6 after treatment with gonadotrophins.
The best day for collecting preimplantation embryos for cryopreservation from superovulated guineapigs was day 2.5 of pregnancy according to our study.
Different developmental stages were found in the same individuals after superovulation. There are still too few results for statistical analysis.
The modified protocol of Suzuki et al. (2000) for superovulation was successful in inducing oestrus and vaginal opening. Superovulated females also produced considerably more embryos than untreated animals. This protocol, therefore, is a reliable, fast and inexpensive method suitable for routine laboratory use.
Cryopreservation
Our results show that preimplantation embryos of the guineapig can be successfully cryopreserved. PROH was superior to DMSO. These findings are in accordance with the findings for the cryopreservation of mouse and rat embryos, where PROH enabled higher revitalization rates than DMSO (Stein et al. 1993, Nowshari et al. 1995, and our unpublished data). In contrast to cryopreservation of mouse embryos, it seemed to be necessary to evaluate the viability by FDA staining, as it was difficult to judge the morphological integrity by normal light microscopy. The cytoplasm of the blastomeres in guineapig embryos has a granulated appearance, because it contains many lipid droplets. Therefore, preimplantation embryos of the guineapig are more like swine embryos than mouse or rat embryos, despite the evolutionary relatedness.
For swine, it was shown that the high lipid content was responsible for hypothermic sensitivity (Dobrinsky 1997, 2000), and freezing the embryos of this species is still difficult. Surprisingly, viability was maintained in guineapig embryos although a method was used that was originally adapted for mouse embryos which contain nearly no lipid.
For successful cryopreservation, it is important that most of the intracellular water is removed while at the same time an osmotic shock is avoided. We conclude that the guineapig embryos dehydrate nearly completely during the first phase of our freezing protocol. Thus, there is no intracellular water that can form deleterious ice crystals during the fast thawing process (300°C/min) and cause freezing injury.
Till now, there is no report of a successful embryo transfer of frozen-thawed embryos and only one of the transfer of freshly collected embryos in guineapigs (Suzuki et al. 2000). To demonstrate that viability of guineapig embryos can be preserved during a whole cycle of freezing and thawing, frozen-thawed embryos have developed to term when transferred to surrogate dams. This will be our next challenge.
