Abstract
The aim of the present study was to evaluate the effects of prenatal and postnatal protein deprivation on the morphology and density of vasopressin (VP) and vasoactive intestinal polypeptide (VIP) immunoreactive neurons in the suprachiasmatic nucleus (SCN) of young rats. Female Wistar rats were fed either 6% (malnourished group) or 25% (control group) casein diet five weeks before conception, during gestation and lactation. After weaning, the pups were maintained on the same diet until sacrificed at 30 days of age. The major and minor axes, somatic area and the density of VP- and VIP-immunoreactive neurons were evaluated in the middle sections of the SCN. The present study shows that chronic protein malnutrition (ChPM) in VP neurons induces a significant decrease in number of cells (–31%,) and a significant increase in major and minor axes and somatic area (+12.2%, +21.1% and +15.0%, respectively). The VIP cells showed a significant decrease in cellular density (–41.5%) and a significant increase in minor axis (+13.5%) and somatic area (+10.1%). Our findings suggest that ChPM induces abnormalities in the density and morphology of the soma of VP and VIP neurons. These alterations may be a morphological substrate underlying circadian alterations previously observed in malnourished rats.
Keywords
The importance of nutrition on the developing brain has been studied mainly because of the widespread incidence of infantile malnutrition and the evidence showing that many of its effects are permanent. The severity of the effects depends on the timing, intensity and duration of the nutritional disturbance (Morgane et al. 1993). The effects of malnutrition on the central nervous system during the prenatal and postnatal periods in experimental animals include a decrease in cellular (Katz et al. 1982) and synaptic (Ahmed et al. 1987) density, soma size, synaptic spines and dendritic branching density (García-Ruiz et al. 1993). Several studies have shown alterations in the regulation of several neurochemical systems and anomalies in many other aspects of neuronal metabolism, morphology, spatial memory, learning and behaviour (Morgane et al. 1978, Cintra et al. 1988, Aguilar-Roblero et al. 1997, Cintra et al. 2002).
Physiological phenomena such as the release of hormones, sleep–wake cycle or activity of different brain neurotransmitters all exhibit circadian rhythms (Meijer & Rietveld 1989). In mammals, the mechanism for the generation and entrainment of circadian rhythms resides in the suprachiasmatic nucleus (SCN) of the anterior hypothalamus. The molecular mechanism underlying rhythm generation has been described as interlocked positive and negative transcription-translation-based feedback loops (Reppert & Weaver 2001). The circadian pacemaker is regarded as a multioscillatory system, in such a way that neurons are considered independent oscillators, whose coupling may determine the output of the circadian system (Welsh et al. 1995). On the other hand, astrocytes have been considered as participants in the coupling of the oscillators that form the circadian pacemaker (Morin et al. 1989, Prosser et al. 1994). The SCN is entrained to the 24 h day by the light–dark cycle, mostly by the light period of the day (Pittendrigh 1981). Photic information is conveyed to the SCN by a direct retinal projection, the retinohypothalamic tract, and uses glutamate and pituitary adenylate cyclase-activating polypeptide as the main neurotransmitters (Ebling 1996, Hannibal et al. 1997) and indirectly from the thalamic intergeniculate leaflet via the geniculohypothalamic tract and uses neuropeptide Y as a neurotransmitter (Card & Moore 1989). In rats, these retinohypothalamic and geniculohypothalamic terminals are distributed in the ventral region of the SCN, and this region also receives a dense serotonergic input from the midbrain raphe nuclei (Moore et al. 1978). Functionally and morphologically, the SCN has been subdivided into a ventrolateral core and a dorsomedial shell (Moore et al. 2002). The ventrolateral subdivision of the SCN receives direct or indirect photic information and contains neurons immunoreactive for vasoactive intestinal polypeptide (VIP). The dorsomedial region of the SCN receives mostly non-photic input (Leak et al. 1999) and contains arginine-vasopressin (VP) immunoreactive neurons (van den Pol & Tsujimoto 1985).
Studies carried out to assess the effects of chronic protein malnutrition (ChPM) on the circadian timing system have revealed alterations of circadian rhythms in malnourished rats. Hall et al. (1978) found a delay in the rhythm of food and water intake. Herbert and Reiter (1981) reported a decrease in the amplitude and a delay in the plasma melatonin rhythm. Cipolla-Neto et al. (1987) described a decrease in the amplitude of different behavioural patterns and Cintra et al. (1988) found a decrease in the total time of rapid-eye-movement sleep during the light phase and an increase during the dark phase, which may reflect an alteration in the adjustment to the light–dark cycle. Aguilar-Roblero et al. (1997) reported that ChPM induces alterations in the circadian organization of drinking behaviour at 30, 90 and 120 days of age under the light–dark cycle, and a decrease in the number and density of neurons immunoreactive to VIP and VP in the SCN of rats of 150 days of age. Furthermore, Cintra et al. (1994) reported a decrease in the size of three cell types in the SCN in Nissl-stained histological sections and alterations in the rapid-eye-movement sleep (Cintra et al. 2002) induced by ChPM in rats at the age of 30 days. As no morphological investigations of VIP- and VP-immunoreactive cells of the SCN of malnourished rats at 30 days of age have been reported so far, the present study was designed to establish whether the functional alterations described in the circadian rhythms of rats of 30 days of age subjected to ChPM (Aguilar-Roblero et al. 1997, Cintra et al. 2002), might be accompanied by changes in the structure of the SCN. Since the structure and number of cells can be an indication of neuronal activity and function of a cerebral region, the effects of ChPM on the morphology and density of VIP- and VP-immunoreactive cells were evaluated in the middle sections of the SCN. The current study focuses on neurons containing VIP and VP because they are the most representative subpopulations of the ventrolateral and dorsomedial regions of the SCN, respectively (van den Pol & Tsujimoto 1985). Moreover, the neuronal content of these peptides in the SCN is crucial for the maintenance of rhythmic functions (van Esseveldt et al. 2000).
Materials and methods
Animal model
This study was conducted in compliance with our national legislation and was ethically reviewed. Primiparous female and male Wistar rats, bred at the Instituto Nacional de Pediatría, were used for breeding. All animals were maintained in an experimental room of the same facility designated as a partial barrier unit. The room temperature was maintained at 21 ± 2°C, and the room air was exchanged 10 times per hour. The room lighting was set to a 12 h light–dark cycle (lights on at 07:00). Relative humidity was maintained at 52 ± 10%. Bedding material (wood shavings) was sterilized and changed three times per week. Food and water were provided ad libitum. Water was changed daily.
Twenty female rats (250–275 g) were separated into two groups, five weeks prior to mating and fed isocaloric diets containing either 6% casein (malnourished group) or 25% casein (control group), (rat chow cubes, Teklad Wisconsin, USA). Details of the composition of these diets have been reported previously (Galler & Tonkiss 1991). For breeding, one male rat was placed in a cage with solid bottoms (508 × 408 × 204 mm; Laboratory Products, USA), containing two females in the evening. Vaginal smears were obtained each morning to detect when mating had occurred, which is indicated by the presence of sperm in the smear. After mating, the females continued to receive their respective diets. One week prior to the expected delivery date, all sperm-positive females were placed in polycarbonate nesting cages (480 × 270 × 160 mm; Laboratory Products, USA). The proportion of females giving birth (80%) was similar between groups. After delivery, litters born on the same day from each diet group were randomly fostered. Random fostering was accomplished by gathering all pups born on a particular day in a container and mixing them gently prior to drawing from the combined group for distribution to females. The purpose of random fostering of neonates is to increase the homogeneity of the populations. Litter size was adjusted to eight pups (4 females and 4 males) per litter to assure adequate and standardized nutrition until weaning. The dams continued with their diets during the lactation period after which the pups were weaned (21 days of age), and then maintained on the same diets as those fed to their mothers during gestation and lactation. They were housed in groups of four in same-sex colony cages and given ad libitum access to the diet cubes. All animals were treated humanely to minimize discomfort in accordance with the ethical principles and regulations specified by the Animal Care and Use Committee of our institution and the standards of the National Institutes of Health of México (NOM-062-ZOO-1999: Technical specifications for production, care and use of laboratory animals, DOF 22 August 2001).
Animal perfusion and tissue processing
To determine the cytoarchitectural distribution of VP- and VIP-immunoreactive neurons in the SCN, on postnatal day 30, male rats from each group were randomly selected and anaesthetized with sodium pentobarbital (40 mg/kg, intraperitoneal; Pfizer, Toluca, Estado de México, México) between 12:00 and 13:00 h to prevent circadian fluctuations. To control for possible litter-effects, only one male from each litter was used for the experiments in this study. Three animals from each group were perfused transcardially with 0.9% saline for vascular rinse, followed by 4% cold paraformaldehyde in 0.1 mmol/L phosphate-buffered saline (PBS) at pH 7.4, with a continuous infusion pump (Masterflex, USA) through a cannula inserted into the left ventricle. Perfusion rates were 3.8 and 7.6 mL/min for malnourished and control rats, respectively (Mendonca et al. 2004). After perfusion, the brains were removed from the skulls and postfixed in fresh fixative for 3 h at 4°C and rinsed in PBS. After removal of the occipital and frontal lobes, the blocks of tissue containing the anterior hypothalamus were cryoprotected in solutions of 10, 20 and 30% sucrose in 0.1 mmol/L PBS, until they sank and were then frozen in optimal cutting temperature (OCT) embedding medium (Tissue-Tek, San Francisco, CA, USA). Coronal sections of 30 µm thickness were made with a cryostat (Leica CM1850, Germany). Alternate sections were separately collected in PBS, in order to obtain two independent sets of sections from each brain. Each set was then processed for VP or VIP immunostaining. Free-floating sections from each animal were rinsed with PBS and then treated with 0.3% hydrogen peroxide solution for 10 min to inhibit endogenous peroxidase activity (Merck, Darmstadt, Germany). The sections were rinsed with PBS and then non-specific binding sites were blocked by incubation in 5% bovine serum albumin (BSA, Amersham Biosciences, Buckinghamshire, UK) and 1% Triton X-100 (Sigma, St Louis, MO, USA) in PBS (BSA-TX-PBS) for 2 h at room temperature. The floating sections were rinsed in PBS and incubated with a rabbit-polyclonal antibody against VP or VIP (Biomeda, Foster City, CA, USA), at a dilution of 1:500 in BSA-TX-PBS, for 48 h at 4°C. After incubation with primary antibody, the sections were rinsed in PBS and then incubated for 1 h at room temperature with biotinylated anti-rabbit IgG (DAKO, Carpinteria, CA, USA). The sections were subsequently rinsed in PBS and incubated with a streptavidin–horseradish peroxidase (HRP) (DAKO, Carpinteria) for 1 h at room temperature. HRP label was visualized using a solution of diaminobenzidine (DAKO, Carpinteria). The sections were mounted on glass microscope slides coated with poly-
To assess the morphometric changes in VP- and VIP-immunoreactive neurons in the SCN, another series of five animals from each of two groups was perfused intracardially with physiological saline followed by 10% neutral buffered formalin at pH 7.4, with a perfusion pump (Masterflex). Each brain was dissected carefully from the cranial cavity and postfixed for 5 h in fresh fixative. Small blocks of tissue containing the anterior hypothalamus were dehydrated through a graded series of alcohols and embedded in paraffin (Leica, Nussloch, Hedelberger, Germany). These tissue samples were subsequently sectioned in the coronal plane at 5 µm thickness with a microtome (Leica RM 2155, Germany). The middle sections of the SCN were mounted on slides coated with poly-L-lysine and deparaffinized. The sections were treated with 0.3% hydrogen peroxide for 10 min and later with 1% Triton X-100 in PBS for 5 min. After blocking with BSA-TX-PBS for 2 h, the sections were incubated for 2 h in a humid chamber with rabbit-polyclonal antibodies against VIP or VP (Ready-to-use, Biogenex, San Ramon, CA, USA). This was followed by incubation with secondary antibodies for 1 h (biotinylated anti-rabbit IgG). The sections were subsequently incubated with streptavidin–HRP for 1 h. Immunoreactivity was visualized with the diaminobenzidine reaction. The sections were air-dried, cleared with xylene and coverslips were applied with Entellan, and were subjected to morphometric evaluations. All procedures were performed at room temperature. Three 10 min washes in 0.1 mmol/L PBS were performed between steps. All tissue sections, from control and malnourished animals, were processed at the same time parallelly to minimize any potential variance in staining procedure. As controls of the staining specificity, some sections were processed through all the incubation steps except that the primary antiserum was omitted from the incubation solutions (Mendonca et al. 2004). No specific immunostaining was observed in these sections.
Morphometric analysis
Quantitative histological evaluations were performed using an image analysing system (Metamorph, Universal Imaging Corporation, PA, USA) connected to a light microscope (Leica DMLS, Germany). Two representative sections from the middle level of the SCN were selected from each rat, corresponding approximately to anteroposterior –1.3 mm from the bregma (Paxinos & Watson 1982). Slides from two groups were randomized and coded such that all subsequent analyses were conducted blind. All histological examinations were performed by a single observer. The anatomical borders of the SCN were delimited with phase-contrast microscopy. The VP (dorsomedial subdivision) and VIP (ventrolateral region) immunoreactive neurons were manually counted at × 60, covering an area of 25,000 µm2. In addition, 15 well-delineated neuronal cell bodies with prominent nuclei were randomly chosen in each animal for measurement of VP- or VIP-immunoreactive neurons. On each of these cells, the somatic area (cell body area was measured delimiting the outline of neuronal soma), major axis (length of the longest chord through the neuronal soma) and minor axis (maximum width of the neuronal soma perpendicular to the longest chord) were evaluated at × 100. A one-way ANOVA was performed on data for each parameter from the malnourished and control groups. Differences were considered to be significant at P < 0.05.
Results
At birth, malnourished pups weighed significantly less (5.5 ± 0.3 g) than pups from dams fed with 25% casein diet (6.4 ± 0.2 g). At 30 days of age, malnourished animals weighed significantly less (22.1 ± 1.6 g) than control animals (67.7 ± 3.1 g). The brain weights of the malnourished animals (1.19 ± 0.12 g) were significantly lower than those of the corresponding control group (1.45 ± 0.14 g). A significant reduction in the SCN mean area was observed in the malnourished group (59749 ± 4396.1), compared with the control group (72320.6 ± 3645.7).
VP and VIP immunolabelling were localized to the perikaryon of neurons. Differences in the cytoarchitectural distribution of characteristic SCN cellular groups between control and malnourished animals were not evident by visual inspection. VIP neurons were observed to be located in the area of the SCN immediately adjacent to the optic chiasm. A few immunoreactive neurons were also scattered in the dorsal portion of the nucleus. In contrast, the greatest concentration of VP neurons was observed in the region dorsomedial of the SCN. However, the soma of some of the neurons were scattered in the ventral region of the SCN. Some VP and VIP neurons were entirely embedded within the optic chiasm (Figures 1 and 2), as previously reported (van den Pol & Tsujimoto 1985).
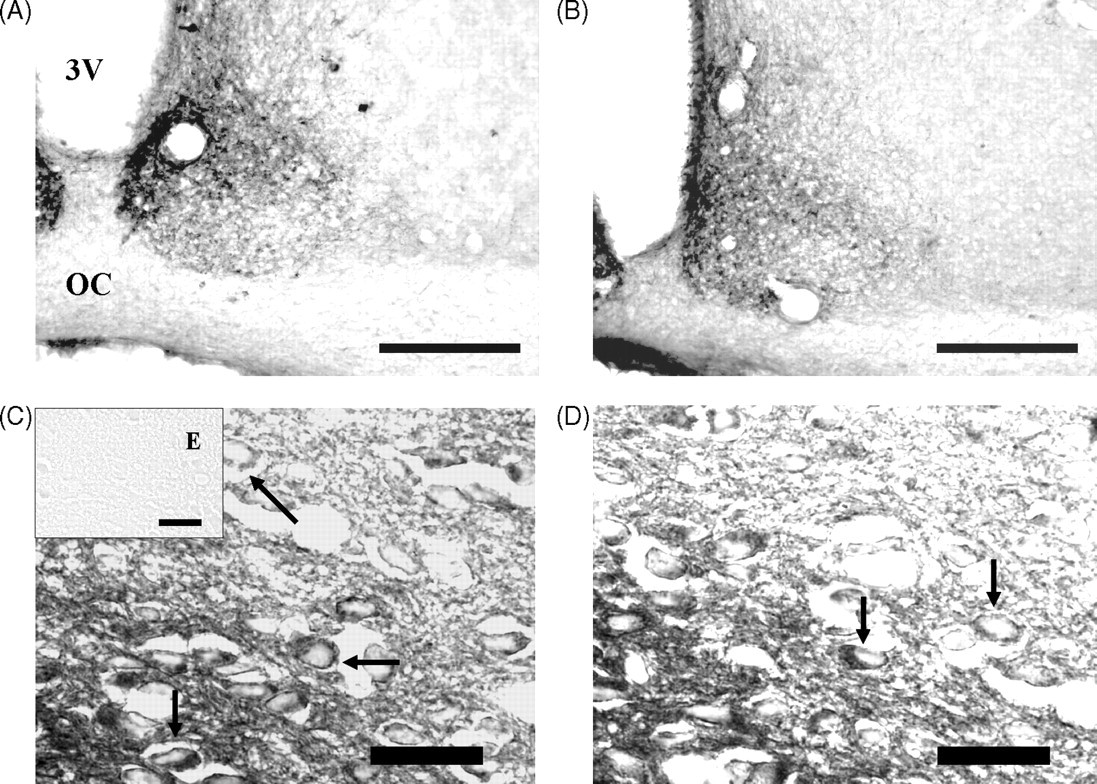
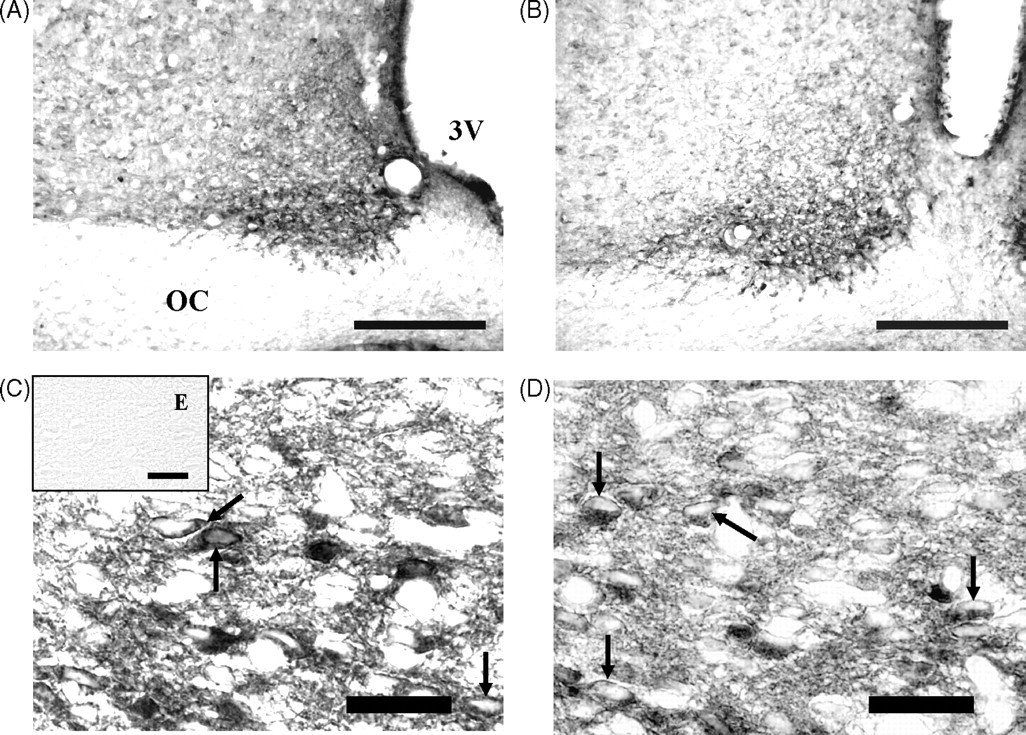
The morphometric analysis of density of VP- and VIP-immunoreactive neurons in SCN showed a significant decrease in the animals exposed to ChPM compared with the control group (–31.1 and –41.5%, respectively). On the other hand, ChPM induced significant increases in the major (+12.2%) and minor (+21.1%) axes and somatic area (+15.0%) in the VP-immunoreactive cells compared with the control group. VIP-immunoreactive cells showed significant increases only in the minor axis (+13.5%) and somatic area (+10.1%) when compared with the control group. No statistical difference was found in the major axis between animals exposed to ChPM (+3.4%) and the control group (Table 1).
Effects of chronic protein malnutrition on morphometric parameters of vasopressin (VP) and vasoactive intestinal polypeptide (VIP) immunoreactive neurons in the suprachiasmatic nucleus of rats at 30 days of age
Data were obtained from five animals per group and two coronal sections per animal at the middle level of the suprachiasmatic nucleus and represent mean ± standard deviation
*Statistically significant difference compared with control group
Discussion
Several studies have shown that ChPM induces structural and functional alterations in the central nervous system that ultimately alters the ability of the organism to interact and cope with its environment. The mean body weight of the malnourished group was reduced (68%) as compared with the control group, similar to previous studies (Aguilar-Roblero et al. 1997).
Furthermore, the mean brain weight showed a significant decrease of 17.9% in the malnourished group, as compared with the control group. A reduction of 17.4% in the SCN area was observed in malnourished animals, as compared with controls. These results suggest that the observed alterations in the SCN area were proportional to the reduction in the brain weight of these malnourished animals. The changes detected in the body weight of the malnourished animals seem to be coherent with a significant reduction in the brain weight as well as the number of cells in the SCN. Morphological studies investigating the effects of chronic low-protein malnutrition (6% casein) starting five weeks prior to mating, on suprachiasmatic nuclei, are rare. Such studies show that ChPM reduces the major and minor axes of three cell types in the SCN, in Nissl-stained SCN slices, especially in young rats at the age of 30 days (Cintra et al. 1994), but these studies did not analyse the neurochemical nature of the cells. In our study, we analysed only VIP and VP neurons; however, the SCN also includes neurons producing calretinin, gastrin-releasing peptide, neurotensin, enkephalin, somatostatin or substance P (Moore et al. 2002). Aguilar-Roblero et al. (1997) described a decrease in the density of cells immunoreactive to VP and VIP in the SCN of adult rats at 150 days of age. However, there are no reports on the short-term effects of ChPM on the immunoreactivity of VP- and VIP-immunoreactive cells, and there are no morphological studies suggesting which cell type is affected. Our study of the VP and VIP neurons of the SCN in protein-deprived rats revealed morphological alterations in the parameters evaluated. Minor (+21.1%) and major (+12.2%) axes and somatic area (+15.0%) were significantly increased in VP cells. VIP neurons showed a significant increase in the major axis (+13.5%) and soma size (+10.1%), while there was a tendency to an increase in the major axis (+3.4%). These data show that the effects of ChPM are not uniform throughout the SCN, as reported for other environmental factors that affect the developing VP and VIP neurons in the SCN (Rojas et al. 1999, Rojas-Castañeda et al. 2000). We have previously observed that ChPM produces alterations on the minor (–4.6%, –2.1%) and major (+5.0%, –0.3%) axes and somatic area (+3.6%, –1.9%) of VP and VIP cells (respectively), compared with the control group, in rats of 200 days of age (Rojas-Castañeda J, unpublished observations). This suggests that ChPM alters the normal developmental pattern observed in control animals. This can be explained by the existing neuronal heterochrony, i.e. the VP and VIP neurons in the SCN have different neurogenesis and differentiation periods (Altman & Bayer 1978a,b, Moore 1991). Moreover, the phenotypic expression of VIP neurons occurs in two developmental stages (Ban et al. 1997); therefore, ChPM at any given point of time is affecting cells of the SCN at different stages of their development. Each brain region or subregion has different metabolic activity, and the changes observed in neuronal elements may be secondary to metabolic changes initiated by dietary protein levels (Brock & Prasad 1992). On the other hand, our results could indicate an effect of neural plasticity mechanisms such as the increase in cell size in malnourished rats.
The results of the present investigation indicate that ChPM induces alterations in the development of the VP and VIP neurons of the SCN. More specifically, ChPM induces a decrease in the density of cells immunoreactive to VP (–31.1%) and VIP (–41.5%) in the SCN, compared with the control group. We have reported that ChPM produces a decrease in density of VP (–15%) and VIP (–25%) cells of the SCN in adult rats (Aguilar-Roblero et al. 1997). This suggests that the effect of ChPM on the number of VP and VIP cells is permanent and may indicate a higher vulnerability for VIP cells in the SCN of rats of 30 days of age. It may also be associated with alterations in the sleep–wake cycle and drinking behaviour in this age (Cintra et al. 1988, Aguilar-Roblero et al. 1997). The decrease in the density of VP and VIP cells, in the current study, may be due to direct loss of neurons or a reduction in the intracellular level of VP or VIP in SCN neurons that would preclude detection by immunostaining due to subthreshold amounts of antigen/epitope, as a consequence of changes in the metabolism.
Malnourished rats show several alterations of circadian rhythms at 30 days of age, which could partly be due to the morphometric alterations observed in the present study, since the size and density of cells could be morphological substrates of functionality. On the other hand, other mechanisms may also be involved in these circadian alterations such as coupling among oscillators, entrainment to light–dark cycles or transmission of rhythmicity to the effectors of the circadian timing system.
Further studies are needed to investigate the volume and stereological characteristics of the several types of cells in the SCN and the concentration of peptides or mRNA in several lighting conditions.
Footnotes
Acknowledgements
The authors are grateful to Pedro Medina for animal care and Dr Sultan Habeebu for his useful comments and suggestions.
