Abstract
Summary
Herpes B virus infection is almost asymptomatic in macaques (Macaca spp.), which are the natural hosts of this pathogen, but is the cause of high mortality in humans. Reactivation of the latent virus in the trigeminal ganglia (TG) results in the shedding of infectious particles into the oral mucosal membrane. Saliva contaminated with the reactivated virus from the ganglia of the natural host is considered to be important for viral transmission to humans and other monkeys. In the present study, we investigated the prevalence of the herpes B virus genome in the left and right TG of seropositive asymptomatic cynomolgus macaques. The latent virus genome was detected using a polymerase chain reaction and microplate hybridization assay. We found that the virus DNA was present in one or both TG of 12 of the 30 macaques (40%) tested, with the virus being detected from both TG in five of the 12 macaques and from a single TG in the remaining seven.
Herpes B virus disease in humans is caused by infection with the herpes B virus (Cercopithecine herpesvirus 1), which is harboured in the natural hosts, Asian macaques including rhesus (Macaca mulatta) and cynomolgus (Macaca fascicularis) macaques (Keeble et al. 1958, Davenport et al. 1994, CDC 1998). The infection results in severe encephalomyelitis in humans with a mortality of over 50%, whereas almost no symptoms are seen in the natural hosts (Weigler 1992). Herpes B virus is transmitted mainly by contact with contaminated excreta from infected macaques (Cohen et al. 2002). Asian macaques are important laboratory animals because of their physiological and morphological similarity to humans with inevitable frequent contact between animal care workers and macaques. Therefore investigation of the herpes B virus prevalence in Asian macaques is important for effective health and safety controls.
The herpes B virus, like other alphaherpesviruses, such as herpes simplex virus type 1 (HSV-1), HSV-2 and varicella-zoster virus (VZV), has a high neurotropism (Whitley & Hilliard 2001). The virus invading the nerve terminals is transported by a retrograde flow to the sensory ganglion cell bodies and then establishes a latent status in the cells (Espana 1973, Boulter & Grant 1977, Lees 1991, Weigler 1992). As reported in human herpesviruses, reactivation of the herpes B virus leads to the shedding of infectious particles from mucosal membranes following anterograde transport in the sensory neurons (Zwartouw & Boulter 1984, Weigler 1992, Weigler et al. 1993, 1995, Huff et al. 2003). The virus particle is excreted at the peripheral sites controlled by the latently virus-infected ganglion. The reactivated herpes B virus from the trigeminal ganglia (TG) and lumbosacral ganglia is suggested to be propagated and excreted from the oral or ocular, and the genital mucosal membrane, respectively (Boulter 1975, Weigler et al. 1995).
More than 40 cases of herpes B virus infection in humans have been reported since the first report by Sabin and Wight (Sabin & Wright 1934, Engel et al. 2002). It has been suggested that the virus is transmitted mainly by bites and/or by contact with contaminated saliva from virus-infected macaques (Cohen et al. 2002). We investigated the prevalence of the herpes B virus in the TG of the cynomolgus macaque, which is one of the most frequently used Asian macaques. We examined seropositive macaques, which were suggested to be latently infected with the virus in some ganglia. We postulated that the virus was present in one or both TG and was dependent on the site of original virus infection. The left and right TG were removed separately from macaques and DNA was extracted. The genome of the herpes B virus in the TG was detected and identified by a polymerase chain reaction (PCR) and microplate hybridization assay, as reported previously (Oya et al. 2004).
Materials and Methods
Animals
A total of 30 cynomolgus macaques were investigated for the presence of viral DNA. Ten (group A) and 20 (group B) macaques bred in China and imported in 1999 and 2000, respectively were sourced from contract research laboratories in Japan. All macaques were between two and six years old (the majority of macaques were 2–4 years old), and showed no clinical symptoms. Antibody to herpes B virus was detected from all macaque sera by enzyme-linked immunosorbent assay. Table 1 shows some characteristics of the macaques.
Characteristics of cynomolgus macaques used in the present study
M, male
F, female
Sample preparation
The left and right TG were removed separately at necropsy from the macaques following euthanasia using an overdose of intravenous injection of pentobarbital and simultaneously 5 mL of whole blood was collected. The peripheral blood mononuclear cells (PBMC) were collected from the buffy coat after centrifuging at 1850
DNA extraction from TG and PBMC
The TG samples and PBMC samples were digested with 570 μg/mL of proteinase K (Promega, Tokyo, Japan) at 55°C for 5 h in TES buffer. The DNA was extracted and purified with phenol and chloroform followed by precipitation in ethanol. The DNA was dissolved in 200 μL of TE buffer (TES buffer without SDS) and stored at 4°C.
Plasmid as a positive control and a probe
A cloned 2.6 kb SalI-EcoRI fragment containing the US5, US6 and a part of US7 derived from the herpes B virus SMHV strain (SMHV/pBV-DNA) was kindly provided by Dr Akio Yamada of the National Institute of Infectious Diseases of Japan (Bennet et al. 1992).
Polymerase chain reaction
We amplified the C region or E region, which consisted of most of US5 and the sequence between US5 and US6, and the 3′-end of US5 and the sequence between US5 and US6, respectively, as described previously (Oya et al. 2004). Amplification of the C region and E region was carried out with an HB2A and HB2B primer set, and HB3A and HB2B primer set, respectively (Table 2). Polymerase chain reaction was performed essentially as described previously but with some modifications (Oya et al. 2004). We prepared a 100 μL PCR mixture containing each primer at 0.5 μmol/L, reaction buffer, a mixture of dNTPs (0.2 mmol/L each), 2.5 units of Ex Taq polymerase (Takara, Shiga, Japan), 2.5% (v/v) DMSO (Merck, Tokyo, Japan) and 10 μL of template. Non-diluted or 1:10 diluted DNA solution was used as the template. Polymerase chain reaction was carried out as described below; initial denaturation at 94°C for 3 min, 30 cycles of denaturation at 94°C for 2 min, annealing at 65°C for 3 min and extension at 72°C for 4 min and final extension at 72°C for 10 min.
Herpes B virus primers used in the present study

Nucleotide positions referred to the E2490 sequence number described by Smith et al. (1998) (GenBank accession no. AF083210)
Microplate hybridization
The PCR products were processed by extraction with phenol and chloroform, precipitation in ethanol, and dissolved in 100 μL of TE buffer. The DNA solution was further purified using a spin column (SUPREC™-02; Takara, Shiga, Japan) according to the manufacturer's instructions. Microplate hybridization was carried out as described previously (Oya et al. 2004). The C region amplicon derived from the SMHV strain was used as the probe.
Statistical procedures
Fisher's exact test and the χ2-test with Yates’ correction (P < 0.05) were used, where appropriate, for comparison of the distribution of virus-positive macaques.
Results
Detection and identification of herpes B virus DNA in TG
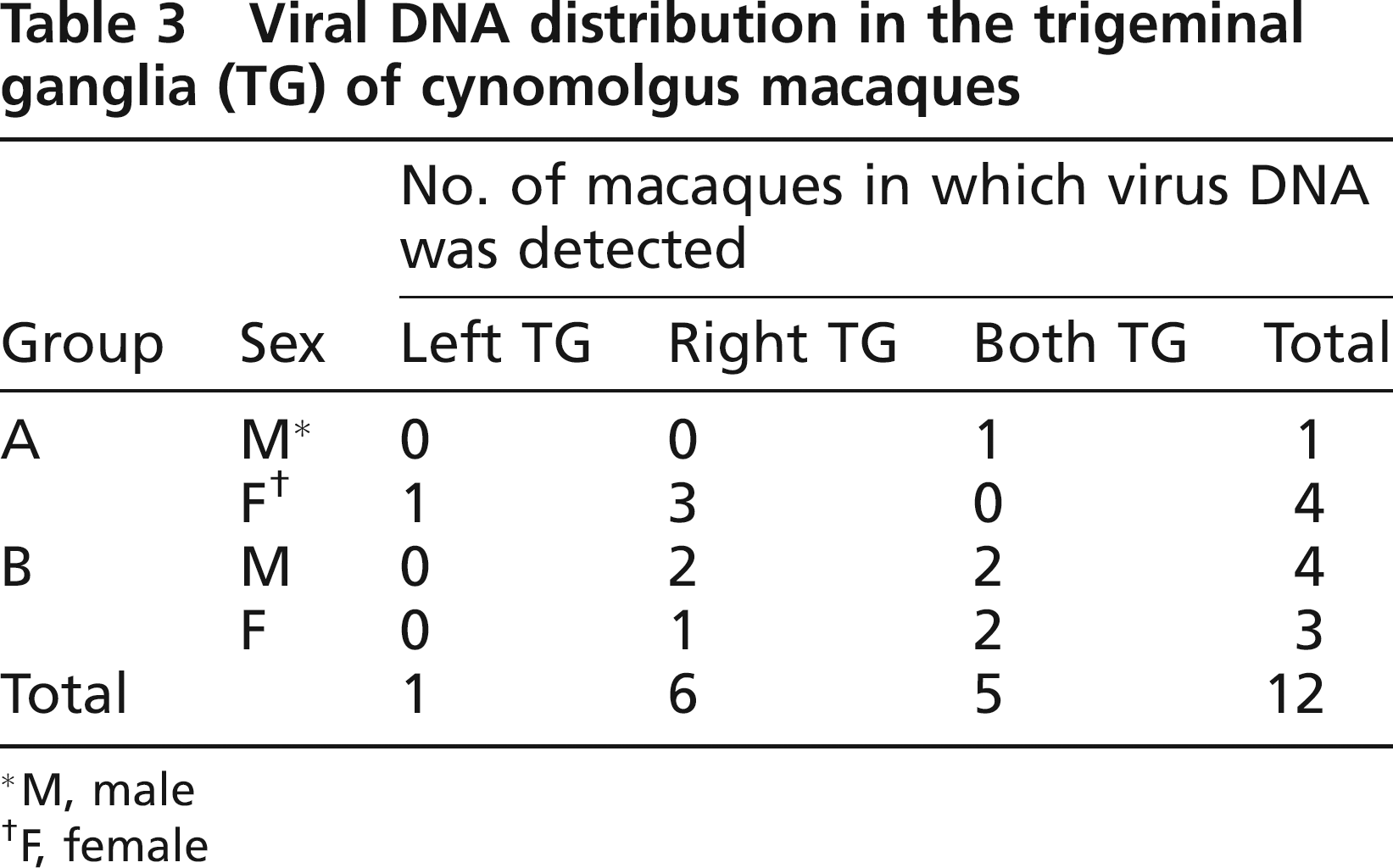
Polymerase chain reaction and microplate hybridization assay were performed to investigate the prevalence of the latent infection of the herpes B virus in the left and right TG of 30 seropositive cynomolgus macaques. Herpes B virus DNA was identified in the TG of 12 of the 30 macaques, five in group A and seven in group B (Table 3). In terms of gender, of the 16 male and 14 female macaques, the virus was detected from the TG of five male and seven female macaques. There was no statistically significant gender difference in the number of virus-positive macaques. It was confirmed that the virus was retained in a latent state in 30 seropositive cynomolgus macaques by detection of no herpes B virus genome in the PBMC of seropositive cynomolgus macaques using PCR (data not shown). The distribution of virus between the left and right TG is also shown in Table 3. There was no statistically significant difference between the number of macaques in which the virus DNA was detected in either one or both TG.
Viral DNA distribution in the trigeminal ganglia (TG) of cynomolgus macaques
M, male
F, female
Hybridization of detected DNA with the SMHV strain probe at 56°C and 42°C
The amplicons from two macaques of group A (A6 and A7) were hybridized with the SMHV probe at both 56°C and 42°C, whereas those from the other three (A2, A9 and A10) were hybridized at only 42°C. However, all PCR products from the TG of the group B macaques (B2, B3, B4, B9, B12, B14 and B16) were hybridized with the probe at both temperatures.
Discussion
We investigated the prevalence of the herpes B virus in the left and right TG of seropositive cynomolgus macaques by a PCR and microplate hybridization assay. In the present study, a total of 30 macaques imported in two batches from China were investigated. The herpes B virus DNA was detected and identified in the TG from 50% and 35% of the group A and group B macaques, respectively, indicating that 40% of the seropositive macaques had virus DNA detectable in their TG. Other reports have shown that the viral DNA was detected by PCR from 27.8% and 10% of seropositive rhesus and Japanese macaque TG, respectively (Weigler et al. 1993, Ohsawa et al. 2002). It is suggested that the macaques with no detectable viral DNA in their TG may have the virus in other ganglia and/or that the quantity of viral DNA used in each PCR would be smaller than the detectable limits of each PCR assay.
Our results suggest that almost 60% of the herpes B virus genome-positive macaques examined in the present study were infected with the herpes B virus in a single TG. In contrast, studies of HSV-1 and VZV have shown that the virus DNA was detected from both TG in most of the patients (Mahalingam et al. 1990, Furuta et al. 1992, Cohrs et al. 2000). Cohrs et al. reported that HSV-1 DNA was detected in the single TG of one (8.3%) out of 12 human subjects in which the virus DNA was detected in TG. It has been reported that VZV DNA was detected in both TG of all (100%) of 17 (Cohrs et al. 2000) and all of seven (Mahalingam et al. 1990) human subjects in which virus DNA was detected. Furuta et al. reported that VZV DNA was detected in a single TG of one (16.7%) of six human subjects in which virus DNA was detected in TG (Furuta et al. 1992). These contrasting results may reflect differences in the region of the body where the hosts were exposed to the virus, and/or the ability of the virus to replicate and spread in the host from the site of primary infection. However, we cannot exclude the possibility that the latent virus in either TG is reactivated, and that the resulting infectious particles then spread to the other TG. Mainly elderly persons with a serious clinical history, which might be induced by immunosuppression, have been investigated in human studies, whereas apparently healthy young macaques (the majority of macaques were 2–4 years old) were used for our examination. In elderly subjects with a compromised immune system, the human herpesviruses may recrudesce more frequently than the herpes B virus in the young animals examined in the present study.
Further investigation of the prevalence of the herpes B virus in various Asian macaque species would lead to a better understanding of virus transmission between host species and conclusions would be facilitated by the determination of viral nucleotide sequences.
Footnotes
Acknowledgements
We thank Akio Yamada for providing recombinant plasmids. This study was supported by a grant-in-aid for the Emerging and Re-emerging Disease project from the Ministry of Health, Labor, and Welfare of Japan.
